Single synapse evaluation of the postsynaptic NMDA receptors targeted by evoked and spontaneous neurotransmission
Figures
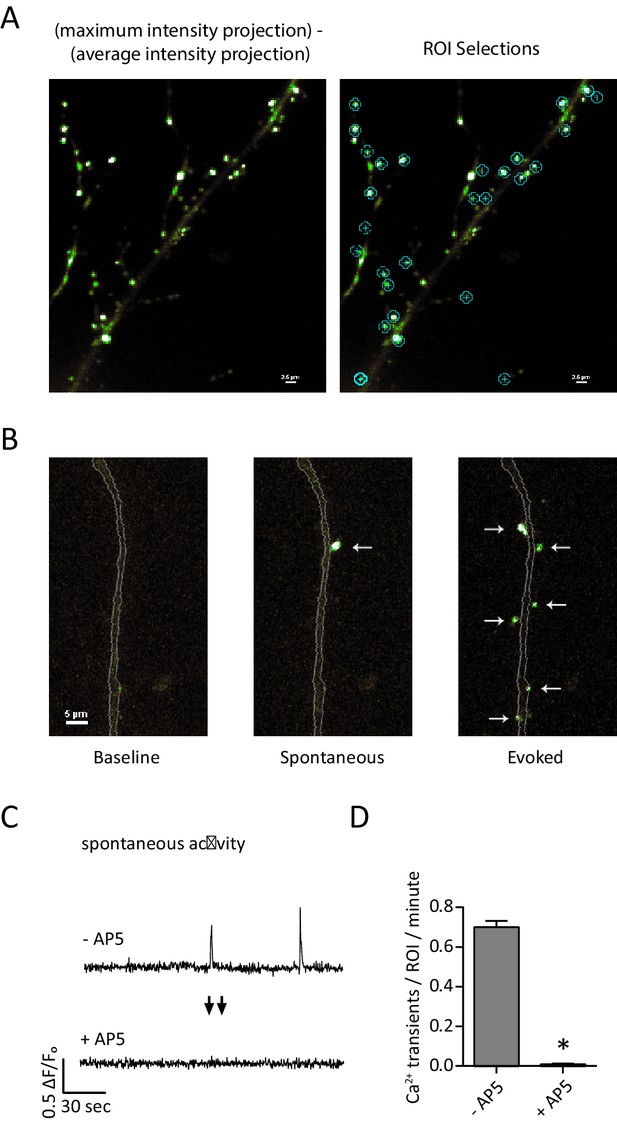
Detection of postsynaptic Ca2+ transients with GCaMP6f-PSD95.
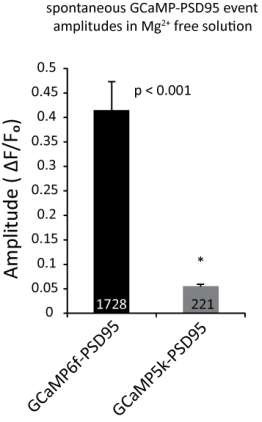
(A) Example images and ROI selection. Each image is a maximum intensity projection – average intensity projection for an 8 min recording both with (right) and without (left) the ROI selections. (B) Example images showing a section of dendrite (gray outline) during baseline fluorescence, peak fluorescence of a spontaneous event, and peak fluorescence of stimulus evoked events. Arrows indicate postsynaptic signals. (C) Example traces from a GCamp6f-PSD95 puncta before and after AP5 treatment. (D) Quantification showing Ca2+ transients detected in TTX before and after the addition of AP5. N = 600 ROIs from six experiments, two cultures, p<0.001 via students paired T-test.
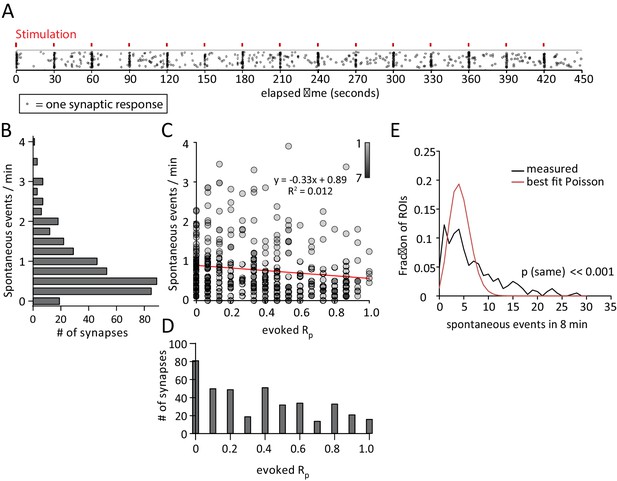
Spontaneous response frequency and evoked response probability show little correlation within the synapse.
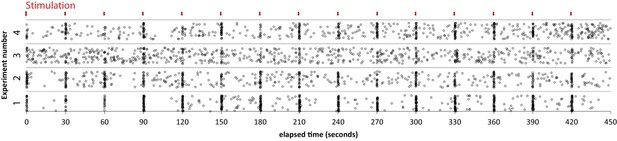
(A) Example results from an experiment where each mark is a single response from one of the 50 ROI’s. Marks are scattered randomly in the y axis to facilitate visualization of responses that overlap in time. (B) Histogram showing spontaneous event rates per minute for 400 ROIs from 8 cells and four cultures. (C) Scatter plot of evoked release probability (Rp) vs spontaneous response rate for the same 400 ROIs. (D) Histogram showing the distribution of evoked response probabilities. (A) Distribution of ROIs by events per 8 min plotted with Poisson theoretical best fit. χ2 test indicates these are not equivalent distributions.
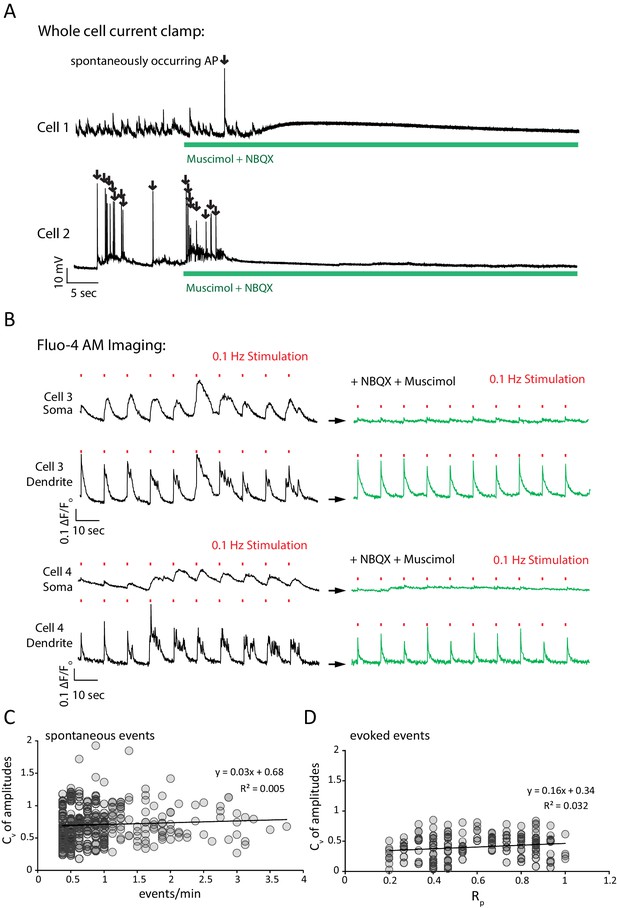
NBXQ and muscimol control network activity while allowing field stimulation; Cv analysis evidence that each ROI isolates a single synapse.
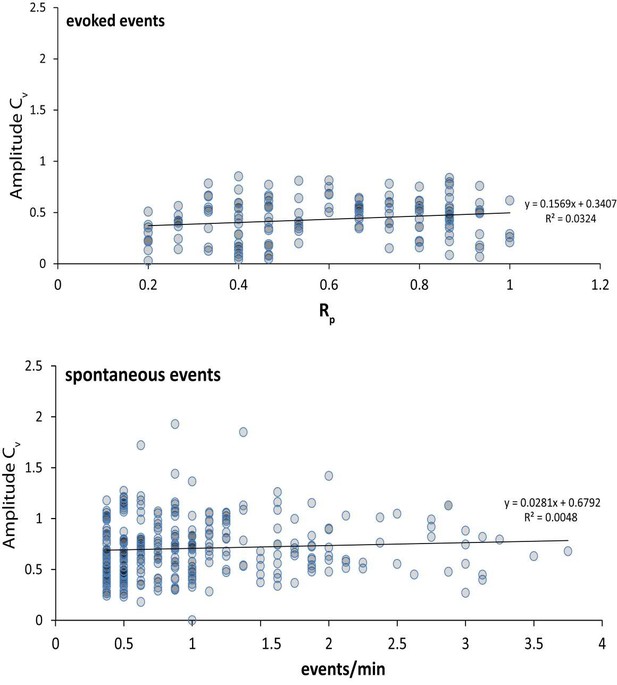
(A) Application of NBQX and muscimol silence network activity in a neuronal culture. Example traces show current clamp recordings of two cultured pyramidal cells first in Mg2+ free Tyrode’s solution and then in Mg2+ free Tyrode’s solution containing 5 µM NBQX plus 5 µM muscimol (green bar). Upon perfusion of NBQX and muscimol, spontaneously arising action potentials are silenced and the voltage fluctuations of each cell driven by excitatory synaptic network activity are silenced. (B) NBQX and muscimol allow reliable stimulation, and prevent reverberatory excitatory network activity by taking advantage of the predominant somatodendritic expression of GABAA receptors. Example traces show Fluo-4 AM fluorescence (reporting bulk Ca2+ changes, rather than synapse specific Ca2+ as monitored by GCaMP6f-PSD95) from 2.5 µm circular ROIs placed on the cell soma or a proximal, isolated dendritic process for two cells. Cells are first imaged in Mg2+ free Tyrode’s solution and stimulated 10 times with 30 mA from a 3 mm spaced parallel bipolar electrode at 0.1 Hz (red marks denote stimulus). During the first train of stimulations, network activity generates multi AP responses that are visible as complex, multi-peak signals in the dendrite which are consistent with network bursting activity (black traces). At the cell soma, a smoothed version of the same signal is visible where each stimulus generates a longer Ca2+ signal. Cells are washed for 1 min with Mg2+ free Tyrode’s solution plus NBQX and muscimol and again stimulated 10 times at 0.1 Hz (green traces). During the second stimulation, each stimulus generates a unitary response with a single peak in the dendrite. At the cell soma, only small Ca2+ signals are visible in the presence of NBQX and muscimol which is consistent with GABAA currents shunting excitatory inputs from the dendrite. The lack of signal at the cell soma compared to the dendrite confirms that Ca2+ signals seen after NBQX and muscimol application are driven by synaptic activity elicited by the stimulus rather than a stimulus artifact which would be visible in both the somatic and dendritic compartments. (C) Spontaneous event amplitude coefficient of variation does not show a population of ROIs with multiple synaptic inputs. Coefficient of variation was calculated for each ROI with two or more spontaneous events during the experiment in Figure 2. Each mark represents a single ROI. If a non-negligible population of ROIs received fluorescent signals from multiple synapses we would expect to see a population of ROIs with both high spontaneous response rate and a high coefficient of variation among amplitudes. We do not detect a population of ROIs where this is the case. (D) The analysis in C. was repeated for evoked signals, and again we did not detect a population of high variance and high Rp ROIs that would indicate multiple inputs per ROI.
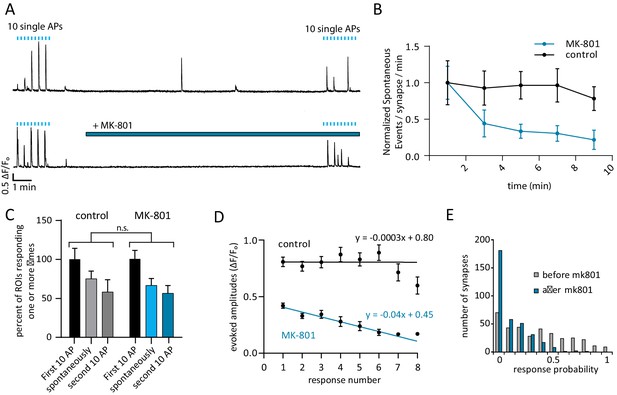
Use-dependent NMDAR blocker MK-801 can selectively silence spontaneous signals without effecting evoked responses within the same synapse.
(A) Example traces outlining the experiment with (top) and without (bottom) MK-801 treatment. 10 action potentials were evoked, 10 s apart (blue tick marks). 4 min later, perfusion was changed to Tyrode’s solution containing 10 µM MK-801 or vehicle. After 10 min in this solution, 10 additional action potentials are evoked, 10 s. Please note that sample traces were selected among examples that are on the larger the side of the average values (especially those after MK-801 application) for better visualization of the impact of MK-801 block on single events (B) Plot shows normalized spontaneous event frequency over the stimulation-free 10 min treatment with MK-801 or control solution. For MK-801 N = 350 ROIs from 7 cells and three cultures. For the control condition, N = 300 ROIs from 6 cells and five cultures. (C) Bars show the percentage of ROIs that respond one or more times to either the first round of 10 stimulations, the second round of 10 stimulations, or spontaneously respond during the intervening time. Control cells (vehicle treatment) and MK-801 treated cells are compared and show no significant differences. (D) Average amplitudes as ∆F/Fo for each nth response within an ROI (per response not per stimulation, failures to respond are skipped so ‘zeros’ are not averaged). For both groups, fit line generated via linear least squares method. (E) Histogram showing distribution of evoked response probabilities in cells before and after MK-801 treatment.
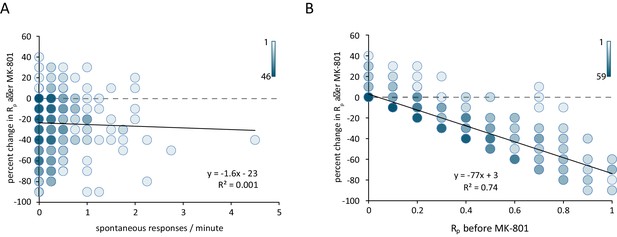
Reduction in postsynaptic responses by MK-801 correlates with evoked response probability but not spontaneous response rate.
(A) Scatter plot of change in Rp (before – after MK-801) vs spontaneous response rate before MK-801 was added. Shading indicates overlapping data points. For both plots N = 350 ROIs from 7 cells and five cultures. (B) Scatter plot of the change in Rp after MK-801 treatment vs initial evoked response rate. Linear fit lines generated via least squares method.
Videos
Monitoring spontaneous and evoked neurotransmission on to single hippocampal neurons.
To visualize spontaneous and evoked neurotransmission GCaMP6f-PSD95 transfected cells were recorded for 8 min during which single action potentials were stimulated every 30 s for a total of 15 action potentials.





