Curvature-induced expulsion of actomyosin bundles during cytokinetic ring contraction
Figures
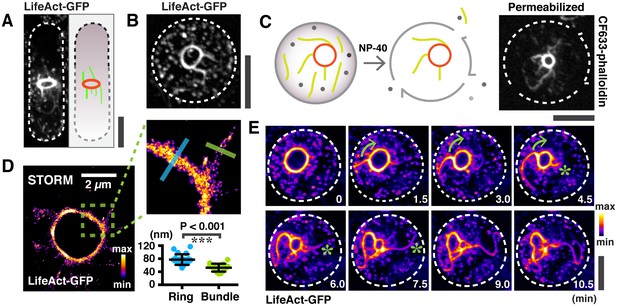
Large actin bundles are expelled during actomyosin ring contraction.
(A) S. japonicus cells expressing LifeAct-GFP. (B) Spheroplasts expressing LifeAct-GFP. (C) Permeabilized spheroplasts (by 0.5% NP-40) stained with CF633-phalloidin. (D) Quantification of the widths of actin rings and bundles by STORM. n = 16 random bundles and 21 random positions of 5 rings. The full-width-half-maximum (FWHM) value of a Gaussian-fitted line was measured to represent the widths. (E) Time-lapse micrographs of spheroplasts expressing LifeAct-GFP. Asterisks: expelling actin bundles. Arrows: rotations of an actin bundle. All scale bars are 5 µm unless specified. Error bars: s.d.
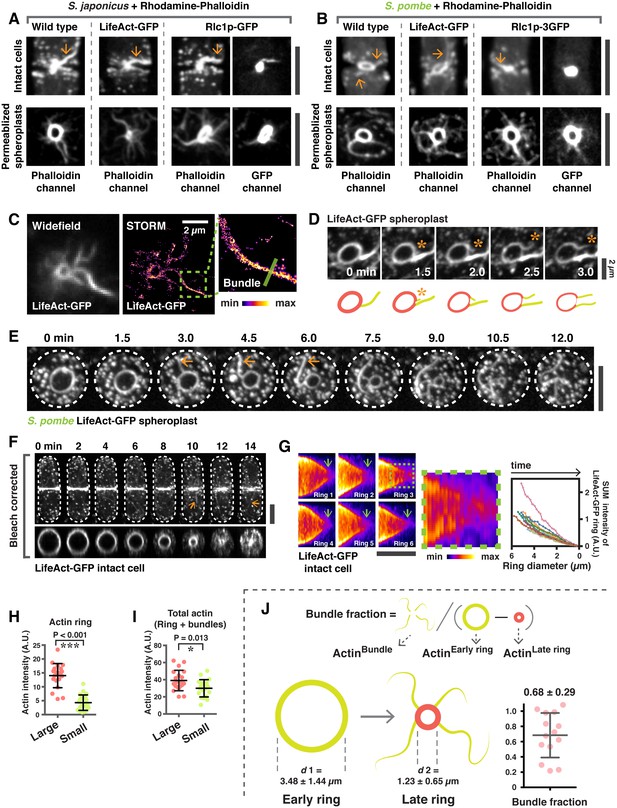
Analysis of actin bundles during actomyosin ring contraction and disassembly in S. japonicus and S. pombe.
(A) Intact cells and spheroplasts of S. japonicus wild type and wild type expressing LifeAct-GFP or Rlc1p-GFP were permeabilized and stained with Rhodamine-conjugated phalloidin. Arrows indicate ring-associated bundles. (B) Intact cells and spheroplasts of S. pombe wild type and wild type expressing LifeAct-GFP or Rlc1p-GFP were permeabilized and stained with Rhodamine-conjugated phalloidin. Arrows indicate ring-associated bundles. (C) Visualization of actin rings and bundles by STORM. (D) Actin rings and bundles during ring contraction of a spheroplast expressing LifeAct-GFP. Asterisks indicate expulsion of actin bundles. Scale bar, 2 µm. (E) Time-lapse micrographs of S. pombe spheroplasts expressing LifeAct-GFP. Arrows: expelling actin bundles. (F) Time-lapse micrographs of an intact cell expressing LifeAct-GFP. Arrows indicate actin bundles associated with the contracting ring. The orthogonal view of the ring in the same cell is shown in the bottom panel. (G) Kymographs of contracting rings labeled with LifeAct-GFP. A zoom-in panel shows disintegration of actin structures near the end of cytokinesis. The sum fluorescence intensity of LifeAct-GFP was quantitated and plotted as a function of ring diameter (n = 10 rings). Photobleaching correction was not applied in this analysis. (H) Quantification of the actin ring intensity in permeabilized spheroplasts stained with CF633-phalloidin. The sum intensity of rings was measured by drawing a line along the ring circumference without including ring-associated bundles. The intensity of CF633-phallodin served as the proxy for actin intensity. Large: ring diameter ≥ 3 µm; small: ring diameter < 3 µm. ***p< 0.001. n = 20 rings for each group. (I) Quantification of total actin sum intensities in permeabilized spheroplasts stained with CF633-phalloidin. The sum intensity of total protein was measured by drawing a square to cover the actomyosin ring and the associated bundles. The sum intensity of CF633-phallodin served as the proxy for actin intensity. Large: ring diameter ≥ 3 µm; small: ring diameter < 3 µm, n = 20 rings for each group. *p=0.013. (J) Quantification of the actin filaments that are lost from the ring during contraction via bundle expulsion. Spheroplasts expressing LifeAct-GFP (as a proxy for actin) were used to estimate the amount of actin ring disassembly via bundle expulsion. To minimize the effects of photobleaching, two consecutive time points, 12 minutes apart of the same spheroplast was imaged. The actin intensity in the ring-associated bundle (ActinBundle) and the actin intensity of rings at early (ActinEarly ring) and later stages (ActinLate ring) were quantitated. The fraction of ring actin that is lost via bundle expulsion is represented by ActinBundle / (ActinEarly ring – ActinLate ring). The d1 and d2 represent the average ring diameters used to quantitate ActinEarly ring and ActinLate ring, respectively. n = 14 spheroplasts quantitated. All scale bars are 5 µm unless specified. Error bars represent s.d.
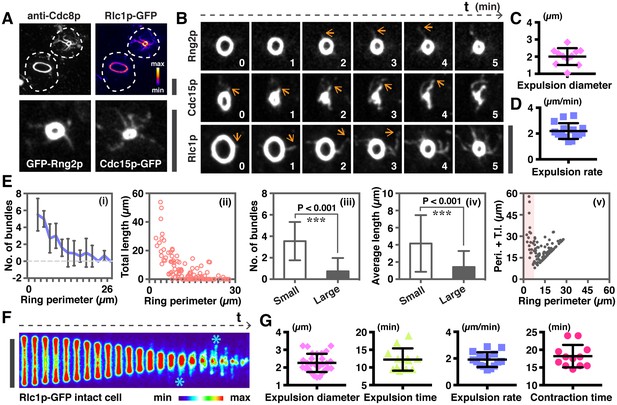
Quantitative analysis of actomyosin bundle expulsion.
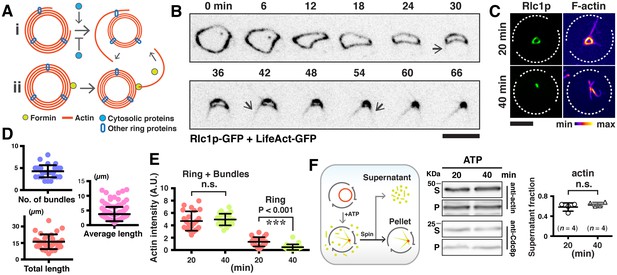
(A) Top panel: localizations of tropomyosin Cdc8p and Rlc1p-GFP in permeabilized spheroplasts. Both proteins were detected by immunofluorescence microscopy. Bottom panel: spheroplasts expressing GFP-Rng2p, and Cdc15p-GFP were imaged to show the localization of these proteins on ring-associated bundles. (B) Time-lapse micrographs of spheroplasts expressing GFP-Rng2p, Cdc15p-GFP, and Rlc1p-GFP, respectively. The time is indicated at bottom right corner in minutes. (C) Quantification of the Rlc1p-GFP ring diameter during initial bundle expulsion (n = 11 rings) in spheroplasts. The ring diameters were calculated by fitting the measured perimeters in Figure 2—figure supplement 1B with Diameter = Perimeter / π. (D) Quantification of the Rlc1p-GFP bundle-released rate (n = 15 bundles) in spheroplasts. (E) Quantitative analysis of bundle expulsion in permeabilized spheroplasts. i: quantification of the number of bundles as a function of ring perimeter; ii: total bundle lengths as a function of ring perimeter; iii: number of bundles associated with small or large rings; iv: average bundle length associated with small or large rings; v: sum of total bundle length (T.l.) and ring perimeter (Peri.) as a function of ring perimeter. Large: ring perimeter ≥ 11 µm; Small: ring perimeter < 11 µm; n = 75 (Large), 46 (Small), respectively. Graphs ii and v: each data point represents one permeabilized spheroplast, n = 121. (F) Kymographs of contracting rings at 1 min interval in intact cells expressing Rlc1p-GFP. Asterisks: actomyosin bundles. (G) Quantifications of the ring diameter during initial bundle expulsion (n = 28 rings), time of bundle expulsion (n = 12 rings), traveling rate of expelled bundles (n = 15 bundles), and total ring contraction time (n = 12 rings) in intact cells. Scale bar: 5 µm. Error bars: s.d.
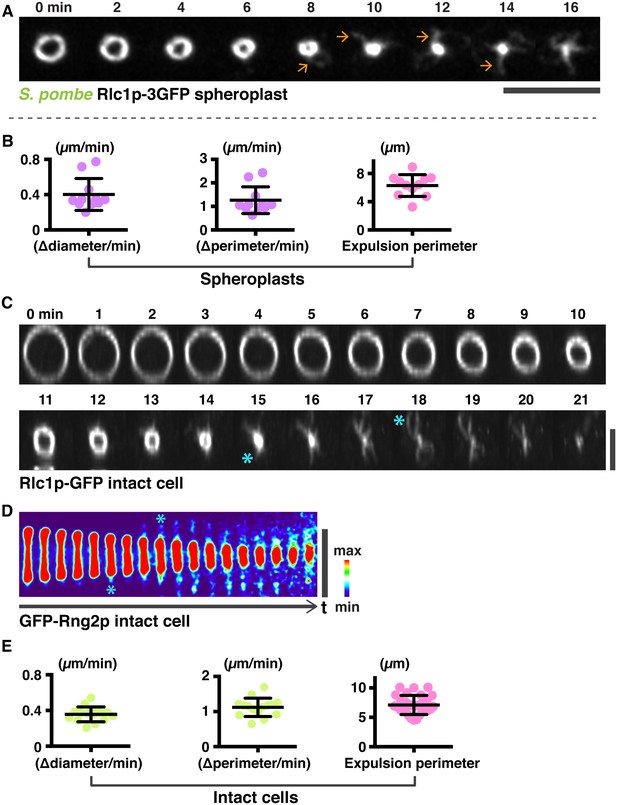
Analysis of actomyosin ring components during ring contraction and disassembly.
(A) S. pombe spheroplasts expressing Rlc1p-GFP shows expulsion of actomyosin bundles. (B) Quantification of the Rlc1p-GFP ring contraction rate (diameter and perimeter change over time) and ring perimeters during initial bundle expulsion in spheroplasts (n = 11 rings). The ring diameters were calculated by fitting the measured perimeters (right panels) with Diameter = Perimeter / π. (c) Time-lapse micrographs of the same contracting ring shown in Figure 2F. The Rlc1p-GFP ring was tilted to the axial plane for visualization. Asterisks indicate expelled bundles. (D) Kymographs of GFP-Rng2p in a contracting ring in an intact cell taken at 1 min interval. Asterisks indicate expelled bundles. (E) Quantifications of the Rlc1p-GFP ring contraction rate (diameter and perimeter changes over time) and ring perimeters during initial bundle expulsion in intact cells (n = 15 rings). The ring perimeters were calculated by fitting the measured diameters in Figure 2G with Perimeter = πd, d: diameter. Scale bar: 5 µm. Error bars represent s.d.
Expulsion of actomyosin bundles during contraction of isolated rings.
(A) i, bundle expulsion mediated by cytosolic proteins; ii, newly polymerized actin filaments that are unable to incorporate into a highly curved actomyosin ring. (B) Time-lapse micrograph shows contraction of an actomyosin ring in a cell ghost after addition of LifeAct-GFP (concentration: ~4 ng/µl) and 0.5 mM ATP. Arrows: expulsion of actomyosin bundles. (C) Cell ghosts containing Rlc1p-GFP were treated with 0.5 mM ATP for 20 min or 40 min respectively, and stained with CF633-phalloidin to visualize F-actin structures. (D) Quantification of the number of expelled bundles, total bundle length, and average bundle length in each cell ghost after 40 min ATP treatment (n = 32 cell ghosts, 137 bundles). (E) Quantification of the sum actin intensity in cell ghosts (ring + bundles) or ring (ring) after treatment with ATP. To measure the total actin intensity in a cell ghost stained with CF633-phalloidin, a square to cover the actomyosin ring and the associated bundles as the region-of-interest was selected, and the sum intensity of the region-of-interest was measured. For the actin intensity of the ring in cell ghosts, a line along the ring circumference to cover the ring area was selected as the region-of-interest for sum intensity measurement. n = 23, 24 cell ghosts for 20 min, 40 min time point, respectively. ns: no significant difference. ***p<0.001. (F) Immunoblots of actin and Cdc8p from cell ghosts treated with ATP for 20 min and 40 min. S, Supernatant: soluble G-actin or short F-actin. P, Pellet: actomyosin rings and associated bundles. Four blots were used for quantifications. Scale bar: 5 µm. Error bars: s.d.
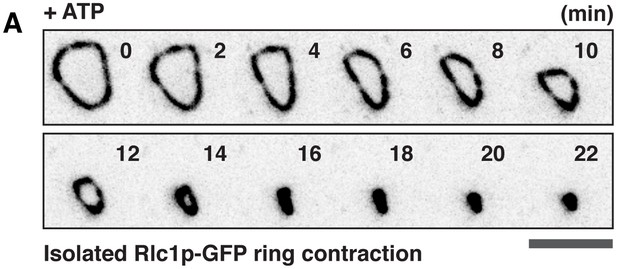
Contraction of the isolated Rlc1p-GFP ring in cell ghosts upon ATP treatment.
(A) Contraction of an isolated actomyosin ring in a cell ghost expressing Rlc1p-GFP treated with 0.5 mM ATP. No obvious Rlc1p-GFP bundle was visualized. The time is indicated at the top right corner in minutes. Scale bar: 5 µm.
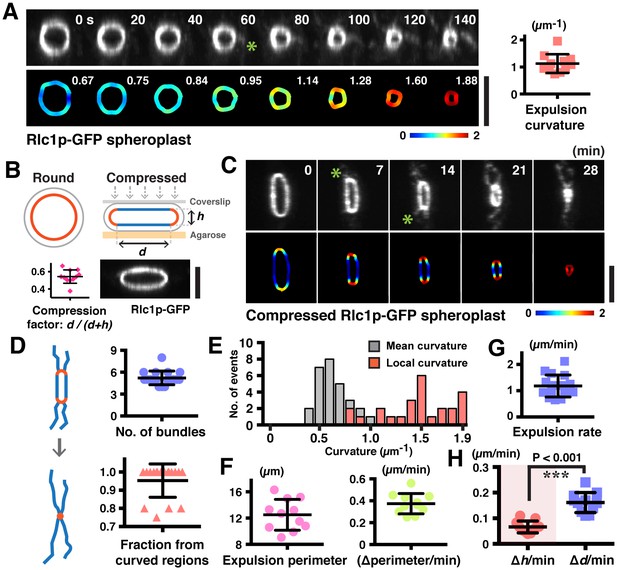
Increased ring curvature promotes actomyosin bundle expulsion.
(A) Analysis of the curvature of sliding rings in spheroplasts expressing Rlc1p-GFP (n = 11 rings). The ring curvatures were color-coded, and mean curvatures of each stage of contraction are shown in the lower panel. The mean ring curvature during initial bundle expulsion was plotted in the right graph. Asterisk: an expelled bundle. (B) Distorted actomyosin rings after spheroplasts were compressed mechanically (n = 11 rings). d: flat; h: curved regions. The degree of compression was quantified by compression factors. (C) Micrographs of bundle expulsion from the curved regions of rings. The ring curvatures are color-coded. Asterisks: expelled bundles. (D) Quantification of the number of ring-associated bundles, and fraction of bundles expelled from curved regions (n = 94 bundles from 18 rings). (E) Quantification of the local curvatures at the initial sites of bundle expulsion and the mean ring curvatures during bundle expulsion (n = 25 bundles from 11 rings). (F) Quantification of the ring perimeter during initial bundle expulsion and the rate of perimeter change in contracting rings (n = 11 rings). (G) Quantification of the traveling rate of expelled bundles (n = 16 bundles) (H) Rates of contraction in flat (△d) and curved (△h) regions (n = 11 rings). Scale bar: 5 µm. Error bars: s.d.
Curvature analysis of actin rings in cell ghosts and Rlc1p rings after compression of spheroplasts.
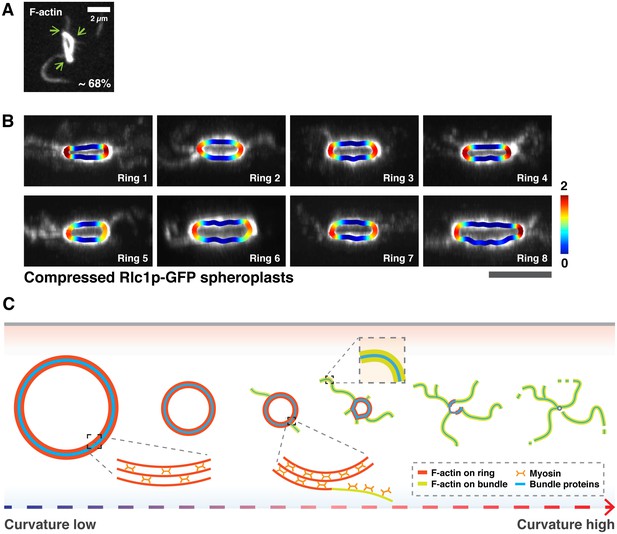
(A) Association of actin bundles with curved edges in isolated rings after 20 min ATP addition. Cell ghosts were stained with CF633-phalloidin, n = 220 bundles from 75 cell ghosts. Green arrows indicate curved edges. (B) Shown are eight representative images from 8 independent time-lapse videos of bundle expulsion at the sites of high local curvature. Local curvatures are color-coded. (C) Schematic representation of curvature-dependent expulsion of actomyosin bundles. Actomyosin ring-based cytokinesis progresses in a curvature dependent manner. Actomyosin rings compact until the ring attains a smaller diameter and hence a high curvature. Large actomyosin bundles composed of F-actin and other ring components are expelled from regions of high ring curvature until the ring disassembles. All scale bars are 5 µm unless specified. Error bars represent s.d.
Videos
Expulsion of F-actin bundles during ring contraction in a spheroplast expressing LifeAct-GFP.
Asterisks mark the elongation of an actin bundle. Left panel is presented in 'Fire' lookup table and its calibration bar. Spheroplasts are outlined by dotted lines. Time zero indicates the start of the video.
Expelled filaments/bundles contain GFP-Rng2p, Cdc15p-GFP, and Rlc1p-GFP.
Asterisks indicate expelling bundles. Time zero indicates the start of the video.
Ring contraction in intact S. japonicus cells expressing Rlc1p-GFP.
Asterisks indicate actomyosin bundles are expelled from the contracting rings. Time zero indicates the start of the video.
Expulsion of actin bundles in 0.5 mM ATP-treated isolated rings in cell ghosts.
Asterisks indicate expelled actomyosin bundles during contraction. Time zero indicates the start of the video.
Curvature-induced expulsion of actomyosin bundles in distorted rings.
Curvature of distorted rings is color-coded. Ring contraction is shown in three different angles of projections. Time zero indicates the start of the video.
Additional files
-
Supplementary file 1
Table shows fission yeasts S. japonicus and S. pombe strains used in this study.
- https://doi.org/10.7554/eLife.21383.015





