Immediate perception of a reward is distinct from the reward’s long-term salience
Figures
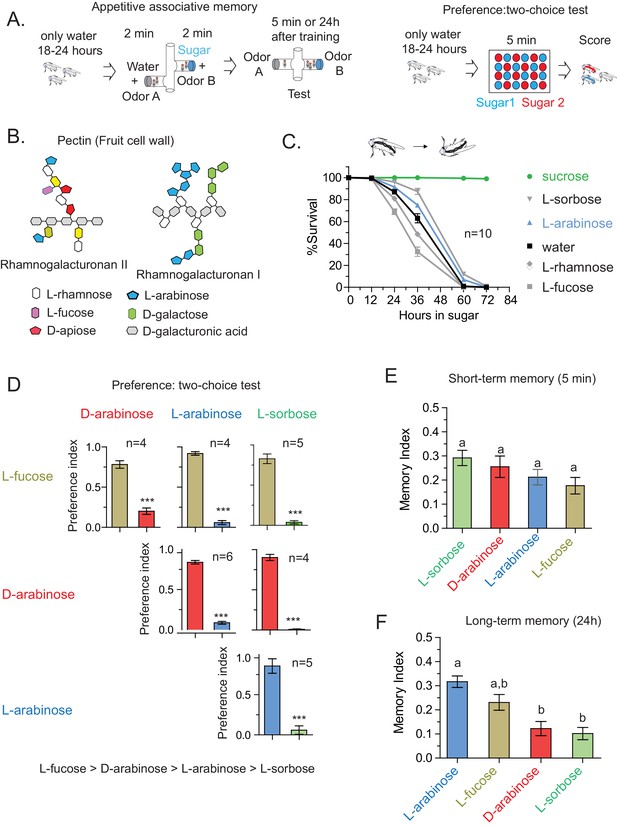
Flies’ immediate preference for a sugar is not predictive of their long-term memory: various sugars from fruits.
(A) Schematic of behavioral assays. In the appetitive associative memory paradigm, hungry flies are trained for 2 min with the sugar-odor pair and memory is assayed by subsequently giving a choice between the two odors. In the preference assay, hungry flies are given a choice between two sugars mixed with different colors; after 5 min color of the abdomen is used to assess consumption. (B) Schematic of selected pectic polysaccharides present in fruits’ cell walls, adapted from Harholt et al. (Harholt et al., 2010). (C) Survival percentages for flies given solely 1 M sugar solutions. n = 10 (50 flies per n) for each time point. (D) Two-choice tests comparing flies’ preference for each of four sugars when both sugars are presented side-by-side for 5 min (50 flies per n). (E) Short-term (5 min) associative memory scores for the sugars. (n = 7–11) (F) Long-term (24 hr) associative memory scores for the sugars. L-fucose is a component of pectin as well, although the amount is low compared to L-arabinose. (n = 20–24) Memory scores labeled a are significantly different (<0.05) from bars labeled b, analyzed by one way ANOVA with Tukey’s multiple comparisons test. Detailed explanations of what constitutes a single n is found in Materials and methods. Results with error bars are means ± s.e.m.
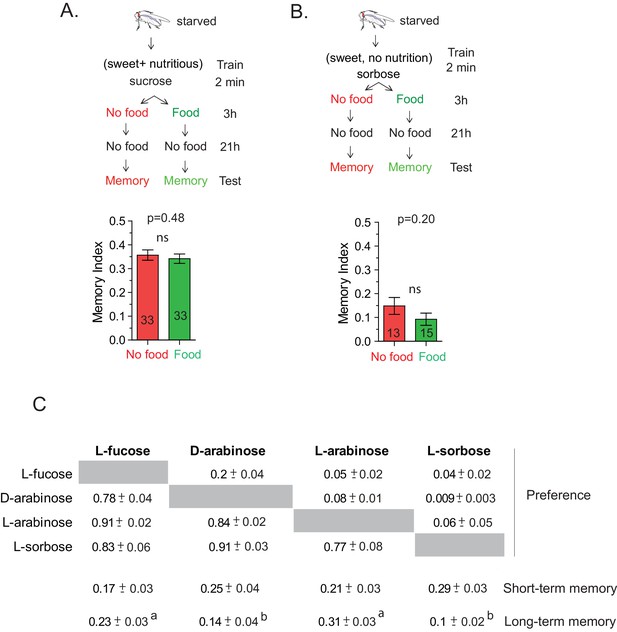
The CS-US association occurs during the two-minute training.
(A) Flies trained with sucrose, which produces robust 24 hr memory, were immediately provided either rich nutritious food or water. If sucrose’s nutritional value is assessed beyond the 2-min training, nutritious food immediately after training may interfere with the fly’s ability to attribute its nutritional status to the 2-min training, and thus interfere with memory formation. However, flies trained with 1 M sucrose and given food or no food for 3 hr post-training showed similar memory, unpaired t-test, p=0.480. (B) Flies trained with L-sorbose—a sweet but non-nutritious sugar that produces short- but not long-term memory—were immediately fed sucrose. If the nutritional evaluation occurred after the 2-min training, immediate feeding on sucrose may substitute as a nutritional cue, resulting in enhanced long-term memory. However, flies trained with 1 M L-sorbose and given sucrose immediately after training showed similar memory to flies not given sucrose, suggesting that the critical association period was confined to the 2-min training, unpaired t-test, p=0.207. (C) Matrix of flies’ preference for one sugar (top) when paired side-by-side with another sugar (side) for 5 min. Each comparison was tested with four independent trials, 50 flies per trial. Numbers are the proportion eating the sugar listed at top. (Proportions may not sum to 1.0; often several flies would not eat either sugar.) Short-term memory and long-term memory averages shown below with ± s.e.m. Memory scores labeled a are significantly different (p<0.05) from bars labeled b, analyzed by one-way ANOVA with Tukey’s multiple comparisons test.
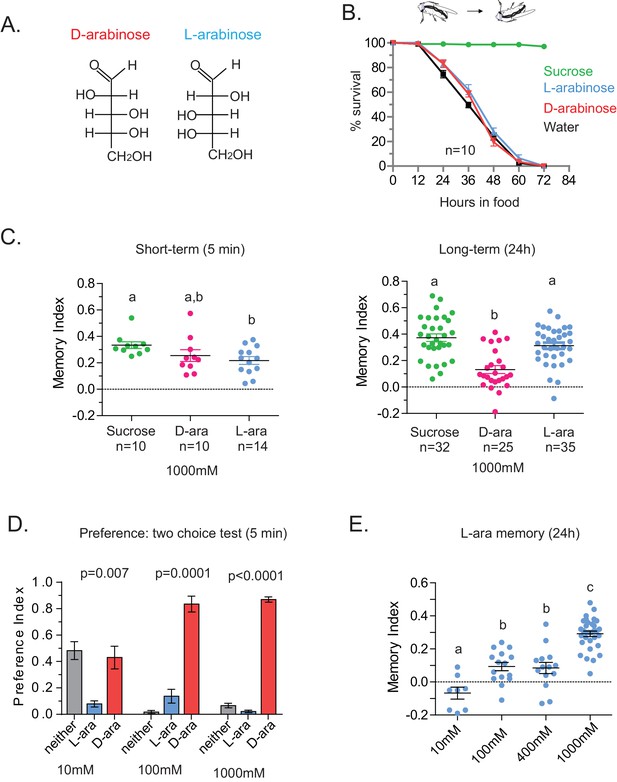
Flies’ immediate preference for a sugar is not predictive of their long-term memory: L- vs D-arabinose.
(A) Structures of D- and L-arabinose. (B) Survival percentages for flies given solely 1 M sugar solutions. n = 10 (50 flies per n) for each time point. (C) Short- and long-term memory of sucrose and D- and L-arabinose. (D) Two-choice tests comparing flies’ preference for D- and L-arabinose when both sugars are presented side-by-side for 5 mins. n = 4 (50 flies per n). (E) Long-term (24 hr) memory scores for increasing concentrations of L-arabinose. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001. The significant differences (p<0.05) between conditions in Figure 2C and E were analyzed by one-way ANOVA with Tukey’s multiple comparisons test and differences are denoted by different letters. Detailed explanations of what constitutes a single n is found in Materials and methods.
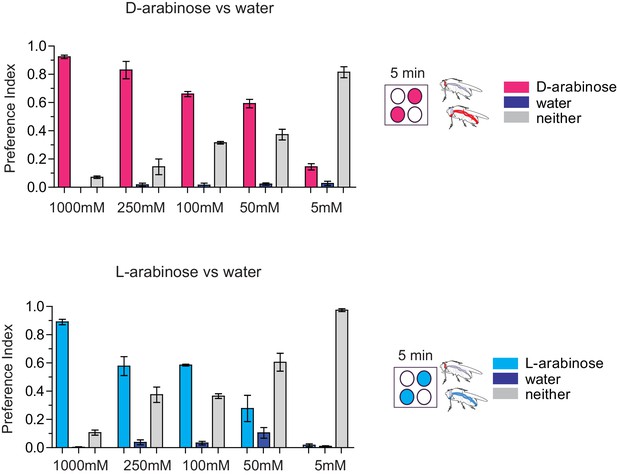
Palatability of D- and L-arabinose over a concentration range.
Fifty male flies per trial were given water alone for 24–36 hr, then put on a microtiter plate checkered with food-dye-labeled water and either D-arabinose or L-arabinose at various concentrations. After 5 min, flies were removed and the color visible in the abdomen was scored.
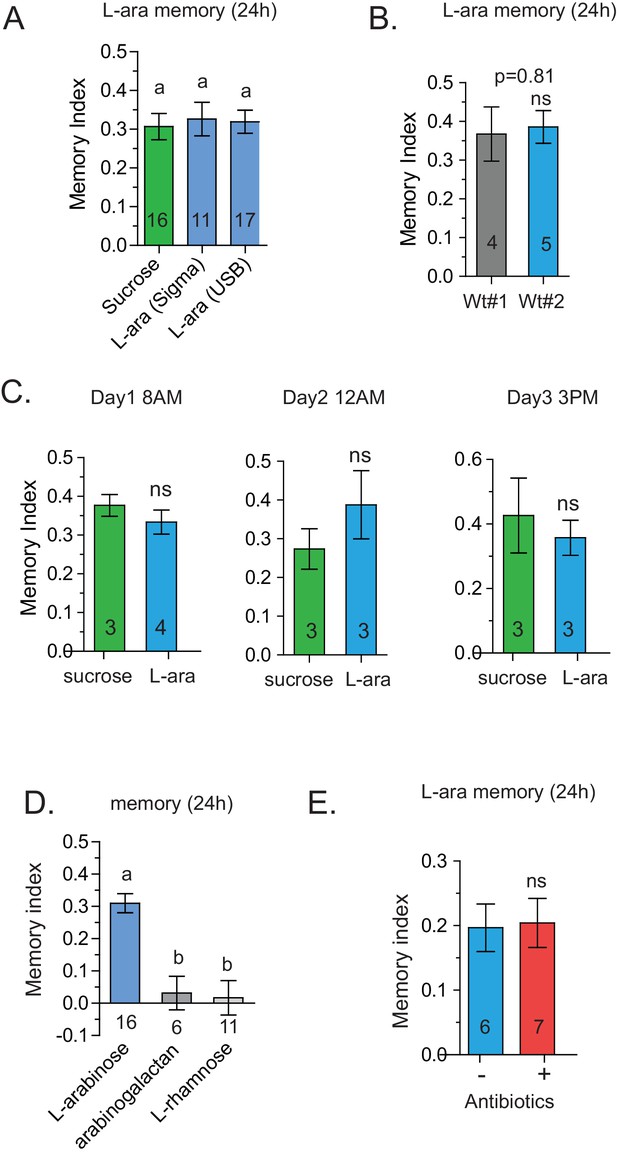
Specificity of L-arabinose memory.
(A) L-arabinose from different sources (USB and Sigma) generated equivalent long-term memory, controlling for other contaminating sugars. (B) Canton-S flies obtained from two different labs show similar long-term memory when trained with L-arabinose. (C) Since flies’ feeding behavior can be influenced by the time of day, flies were tested at different times on consecutive days by different experimenters. Under all testing conditions, flies formed long-term memories of L-arabinose. (D) While L-arabinose forms memory, an L-arabinose-galactose polymer does not; neither does another natural L sugar, L-rhamnose, demonstrating the selectivity of L-arabinose memory. (E) Flies fed a cocktail of three broad-spectrum antibiotics (kanamycin, ampicillin, and tetracycline) for the 48 hr before training show memory indistinguishable from untreated control flies. For two samples unpaired two tailed t-tests and for multiple samples one-way ANOVA with Tukey’s multiple comparisons test were performed, and significant differences (p<0.05) are denoted by different letters. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.
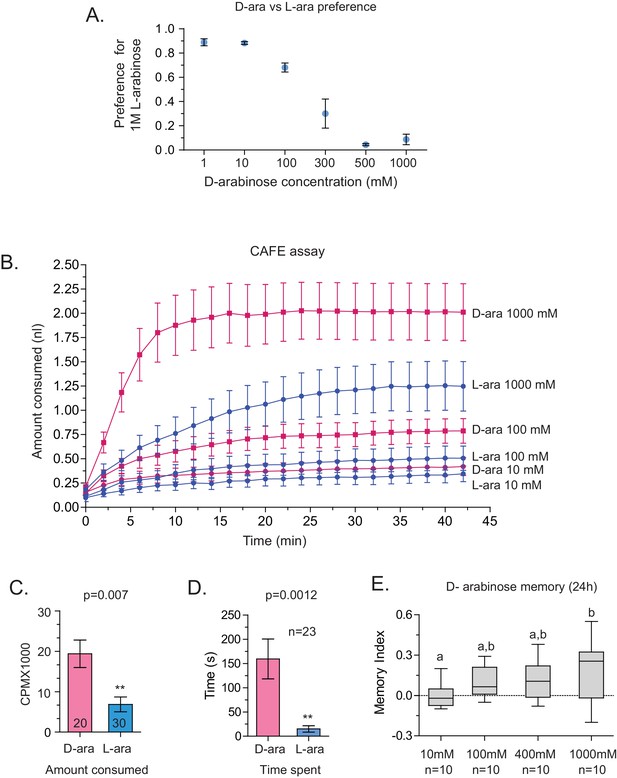
Although both are sweet, D-arabinose is preferred over L-arabinose.
(A) At equal concentrations, flies overwhelmingly prefer D-arabinose to L-arabinose; the preference begins to shift when L-arabinose is ≥3 times more concentrated than D-arabinose. (B) CAFÉ assay quantifying flies’ intake of D- and L-arabinose across a range of concetrations. (C) In 5 min, flies consume more radioactive [32]P-mixed D-arabinose than radioactive [32]P-mixed L-arabinose during two-choice tests when either sugar is tested separately against water. (D) Single flies monitored by video spend more time on 1 M D-arabinose than 1 M L-arabinose when the two are presented side-by-side. (E) The greater the concentration of D-arabinose, the better the memory score. This suggests that the flies are not eating so much D-arabinose that they become sick, and would otherwise remember if not for eating large amounts of sugar. Memory does not improve at lower concentrations. For multiple samples, one way ANOVA with Tukey’s multiple comparisons test was performed, and significant differences (p<0.05) are denoted by different letters. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.
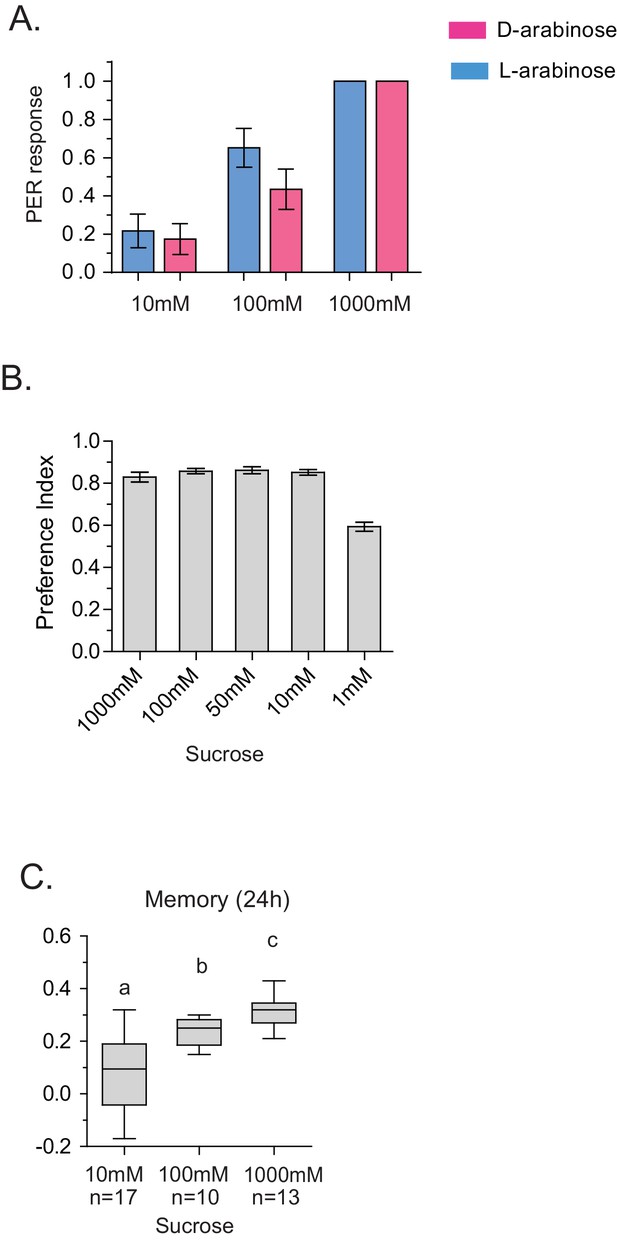
Detection and memory.
(A) Proboscis extension reflex response to D- and L-arabinose. Flies were given a drop of water, then 10 mM D-ara, then 100 mM D-ara, then 1 M D-ara, water, followed by 10 mM L-ara, 100 mM L-ara, and 1 M L-ara. Alternate flies were given D-ara or L-ara first. (n = 26 for each concentration). (B) Flies only begin to have trouble detecting sucrose in the two-choice test (paired with water) when sucrose is ≤1 mM. (C) Flies form better memories with increasing concentrations of sucrose. 10 mM sucrose (the maximum possible contamination given L-arabinose purity of ≥99%) does not produce robust long-term memory. For multiple samples, one-way ANOVA with Tukey’s multiple comparisons test was performed, and significant differences (p<0.05) are denoted by different letters. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.
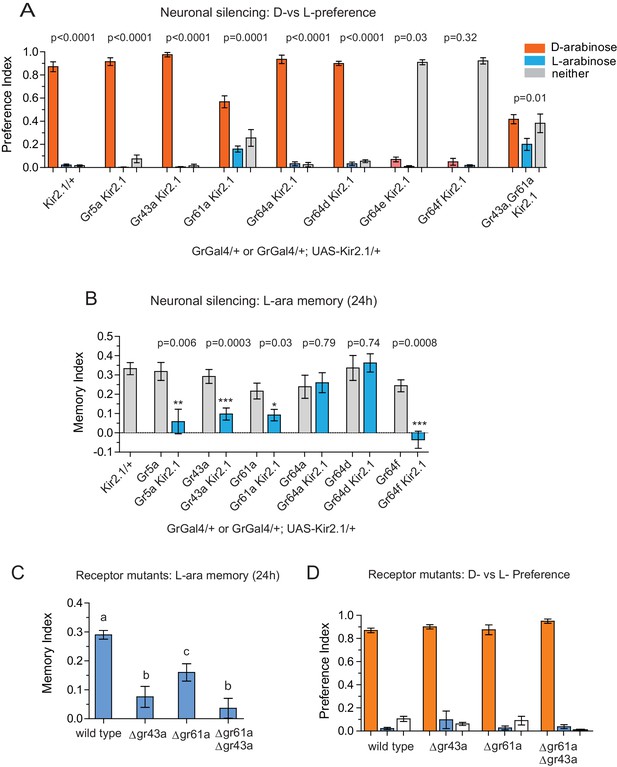
Gr43a- and Gr61a-expressing neurons are involved in D vs L preference and in L-arabinose memory.
(A) Silencing of Gr61a-expressing neurons with Kir2.1 impaired D > L discrimination and preference; silencing Gr64e- and Gr64f-expressing neurons nearly eliminated detection of both sugars. (B) Silencing Gr43a- and Gr61a-expressing neurons impaired L-arabinose memory. Gr64aGAL4 and Gr64dGAL4, whose expression is restricted to LSO and VCSO neurons did not impair L-arabinose memory. Silencing Gr64f- and Gr5a-neurons reduce L-arabinose memory, but they also impair L-arabinose detection. (see Figure 3—figure supplement 1). (C) Gr43a and Gr61a receptors are important for L-arabinose memory. (D) No single receptor mutant impaired D > L preference. For multiple samples, one-way ANOVA with Tukey’s multiple comparisons test was performed, and significant differences (p<0.05) are denoted by different letters. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.
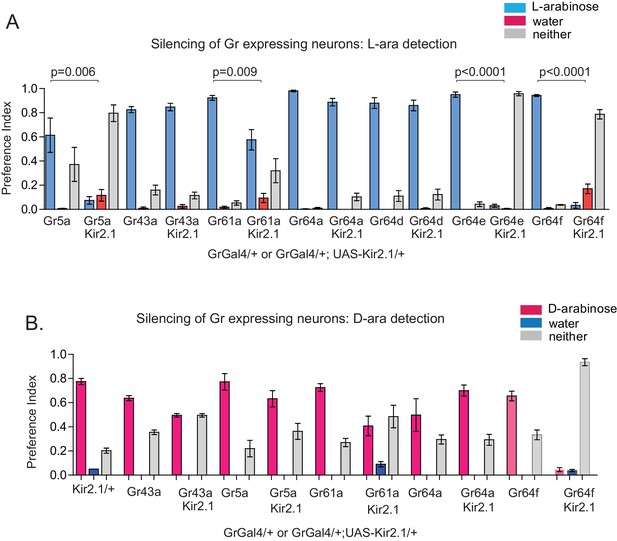
Gr expressing neurons involved in D- and L-arabinose detection.
(A) Fifty male flies per trial were given water alone for 24–36 hr, then put on a microtiter plate checkered with food-dye-labeled water and 1 M L-arabinose. After 5 min, flies were removed and the color visible in the abdomen was scored. n = 4 for all silencing and mutant two-choice experiments, 50 flies per n. Silencing Gr5a-, Gr64e-, and Gr64f-expressing neurons nearly eliminated L-arabinose detection; silencing Gr43aGAL4 neurons had no detectable effect on L-arabinose detection. (B) Silencing Gr64f-expressing neurons virtually eliminates D-arabinose detection; silencing of Gr61a- and Gr43a-expressing neurons reduces but does not eliminate D-arabinose detection compared to the corresponding GAL4/+ control.
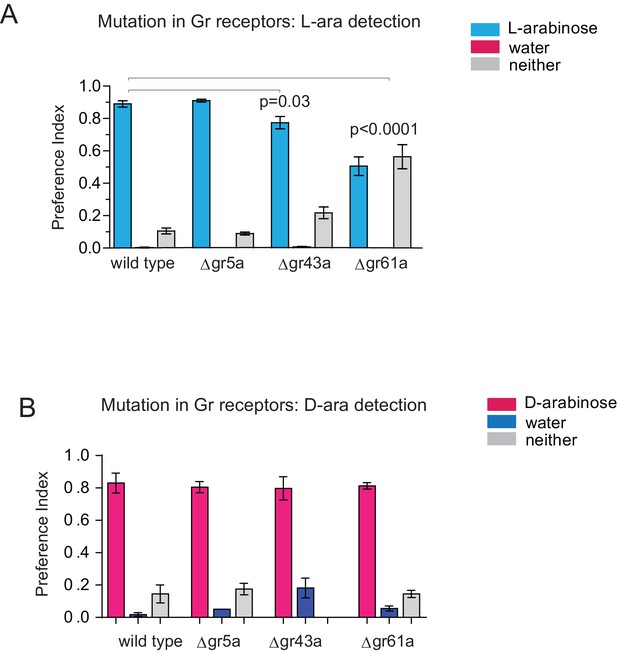
L- and D-arabinose detection do not rely on any single receptor.
(A) Gr43a mutants have a small L-arabinose detection deficit while Gr61a mutants have a moderate detection deficit. (B) No single receptor removal impairs D-arabinose detection. Results with error bars are means ± s.e.m.
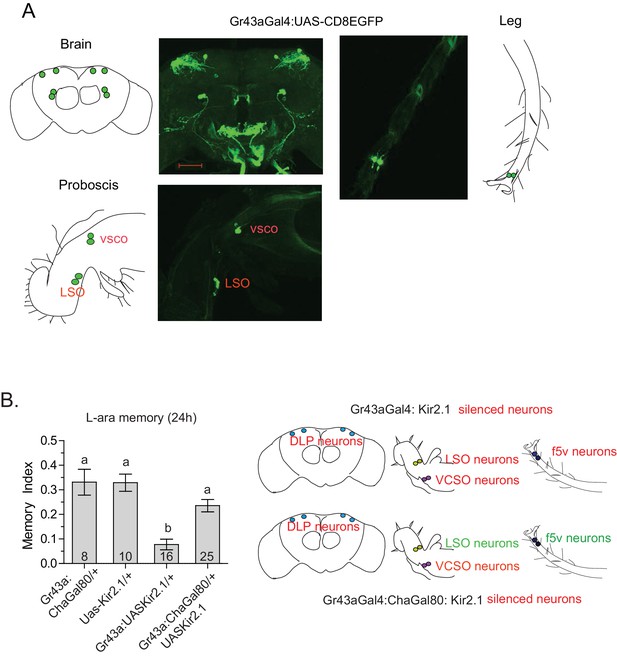
Tarsal Gr43a neurons are critical for L-arabinose memory.
(A) Gr43aGAL4 neurons are observed in the dorsolateral protocerebrum, central brain, proboscis, leg, and gut (not shown). (B) Silencing only the dorsal protocerebral (DLP) and VCSO neurons does not impair L-arabinose memory. Left panel: memory score in various genetic backgrounds. Right panel, top: in Gr43aGAL4/+; Kir2.1/+ flies, all indicated neurons are silenced (including proventricular neurons, not pictured). Bottom: in Gr43aGAL4:ChaGal80/+; UAS-Kir2.1/+ flies, only the neurons indicated in red type are silenced. For multiple samples, one-way ANOVA with Tukey’s multiple comparisons test was performed, and significant differences (p<0.05) are denoted by different letters. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.
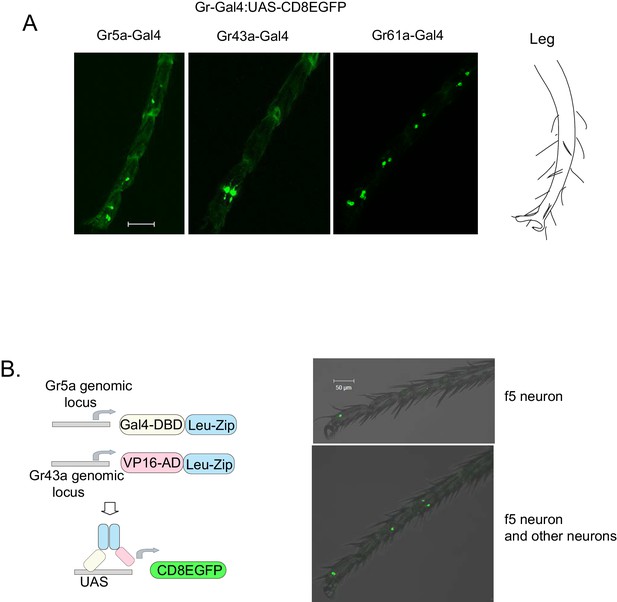
Expression patterns of Gr-GAL4s.
(A) Distal tarsi expression patterns of Gr5aGAL4 (3 neurons), Gr43aGAL4 (2 neurons), and Gr61aGAL4 (6 neurons). Scale bar 50 μm. (B) Gr5a-Gr43a splitGal4 labeling (Pfeiffer et al., 2010). GAL4 DNA-binding domain (DBD) and VP16 transcription activation domain (AD) fused to leucine zipper dimerization domains (Leu-Zip) were inserted into the Gr5a (DBD) and Gr43a (AD) genomic locus using CRISPR-Cas9. The neurons coexpressing both receptors drive mCD8eGFP from the UAS promoter. f5 neurons in the distal tarsi are marked by this technique. In some animals, an f5 neuron is the only neuron marked (top panel). However, in other animals, additional neurons were also marked in the leg (bottom panel) and proboscis. Scale bar 50 μm.
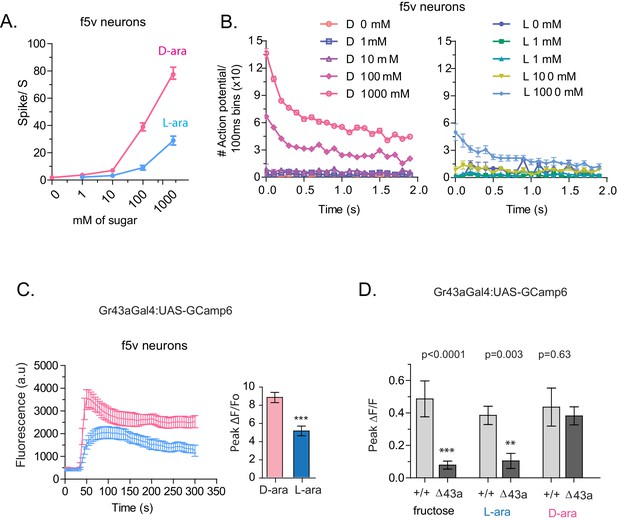
Tarsal Gr43a neurons respond differentially to D- and L-arabinose.
(A) Spikes per second of recorded f5V tarsal neuron in response to D- or L-arabinose at various concentrations. (B) Spikes per second binned by 100 ms over the first two seconds of response. (C) The evoked-calcium activity of Gr43aGAL4 neurons in the distal tarsi. (D) Removal of Gr43a receptor impairs fructose and L-arabinose activation of Gr43aGAL4 neurons. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.
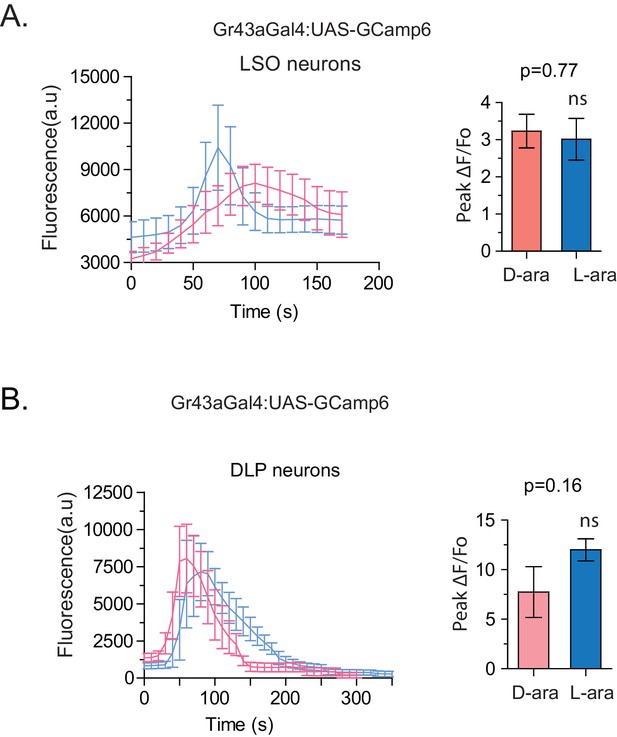
Evoked-calcium activity of Gr43a neurons in response to D- and L-arabinose.
(A) Imaging of calcium responses of LSO neurons in proboscis. The left panel shows curve-aligned fluorescence to compare the shape of the responses, and the right panel plots peak magnitudes as ∆F/Fo. (B) Imaging of calcium responses of dorsal protocerebral neurons. The left panel shows curve-aligned fluorescence to compare the shape of the responses, and the right panel plots peak magnitudes as ∆F/Fo. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.
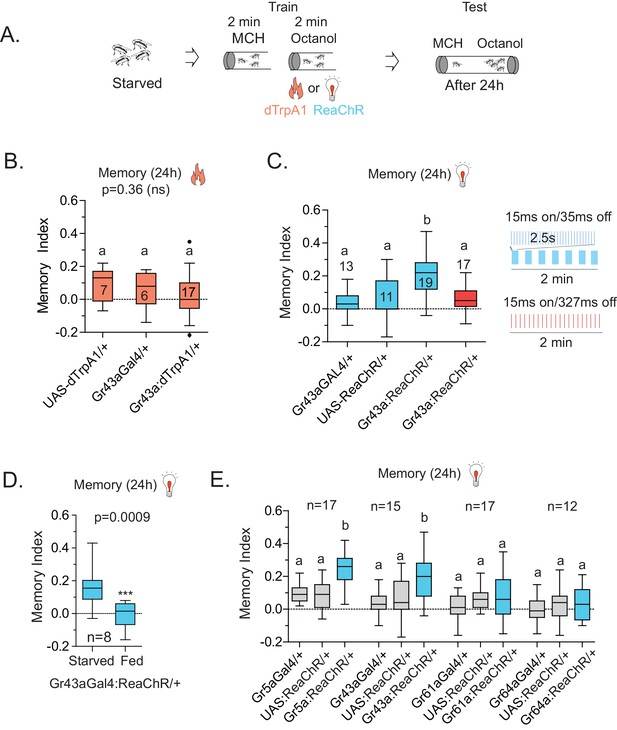
Activation of Gr43a neurons is sufficient to form rewarding associative memory.
(A) Schematic of heat and light-activated associative olfactory training. (B) Activation of Gr43aGAL4 neurons by dTrpA1 (at 31 °C) does not induce long-term memory. (C) A 20 Hz, 15 ms pulse-width activation for 2.5 s, repeated every 20 s, induces long-term memory in flies expressing ReaChR in Gr43aGAL4 neurons; genetic controls do not show significant memory, and the same amount of light using the same pulse-width but distributed uniformly over the 2 min generates no memory (red). Schematics of light patterns are not to scale. (D) Optogenetic activation of Gr43aGAL4 neurons induces memory only in hungry flies, not in flies fed ad libitum. (E) Optogenetic activation of Gr43a- and Gr5a-expressing neurons leads to substantial 24 hr memory; activation of Gr61a- or Gr64a-expressing neurons does not. For multiple samples, one-way ANOVA with Tukey’s multiple comparisons test was performed, and significant differences (p<0.05) are denoted by different letters. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.
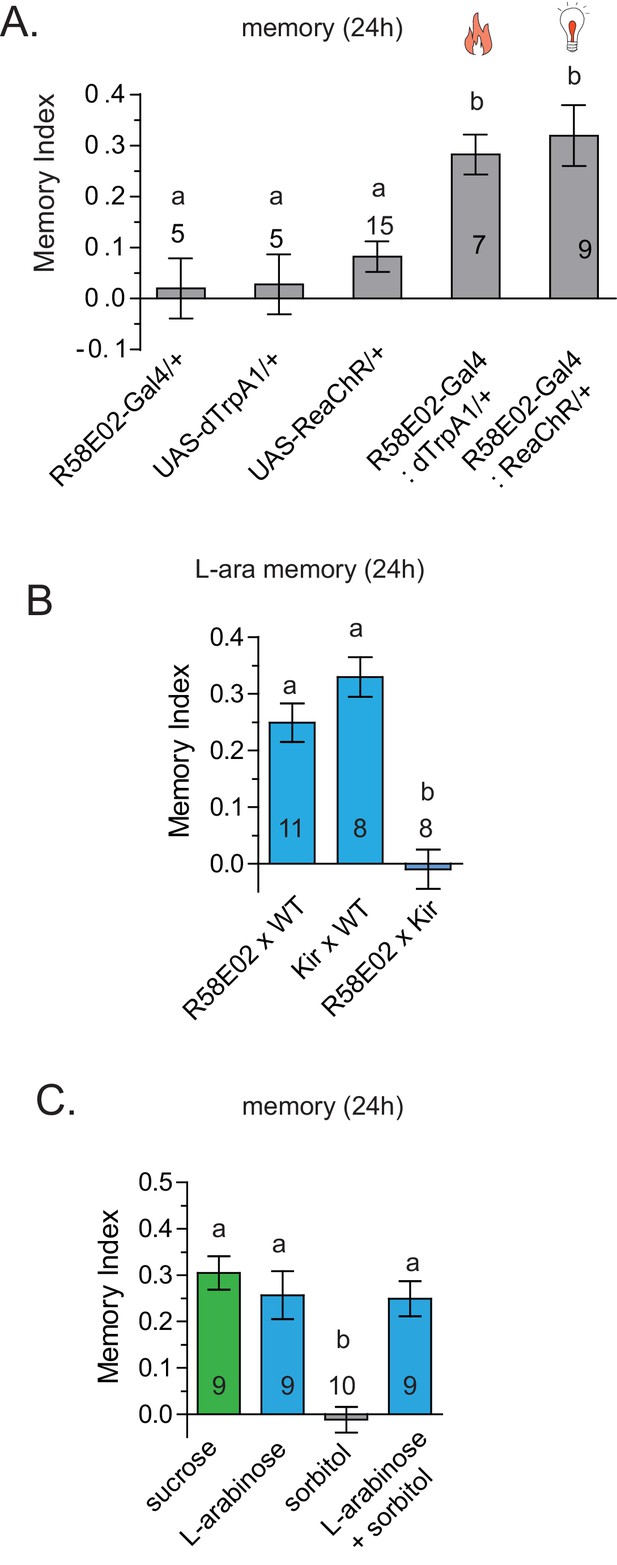
dTrpA1 activation of R58E02 neurons does not produce long-term memory.
(A) Activation of R58E02GAL4 dopaminergic neurons, either with light (ReaChR; 1 Hz, 500 ms pulse-width for 20 s, repeated three times over 2 min, or 10 Hz, 10 ms pulse-width, repeated continuously for 2 min; data combined) or with heat (dTrpA1; 31 °C for two minutes) is sufficient to generate robust 24 hr memory. (B) Silencing of R58E02GAL4 neurons eliminates 24 hr L-arabinose memory. (C) Supplementing L-arabinose with 1 M sorbitol does not increase L-arabinose memory. For multiple samples, one-way ANOVA with Tukey’s multiple comparisons test was performed, and significant differences (p<0.05) are denoted by different letters. Results with error bars are means ± s.e.m. ns, not significant. *≤0.01, **≤0.001 and ***≤0.0001.





