New fossil remains of Homo naledi from the Lesedi Chamber, South Africa
Figures
Geographical location of the Rising Star cave in the Cradle of Humankind UNESCO World Heritage Site.
https://doi.org/10.7554/eLife.24232.003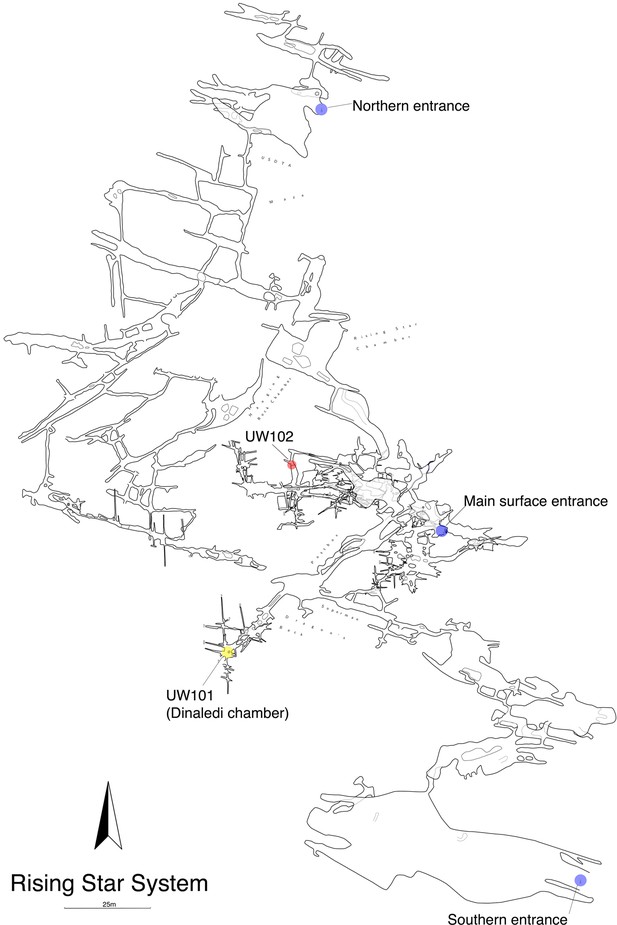
Location of the Lesedi Chamber (U.W.102) in the Rising Star system (red circle).
The Dinaledi Chamber (U.W. 101) is marked by a yellow circle, while three surface entrances into the system are marked by blue circles.
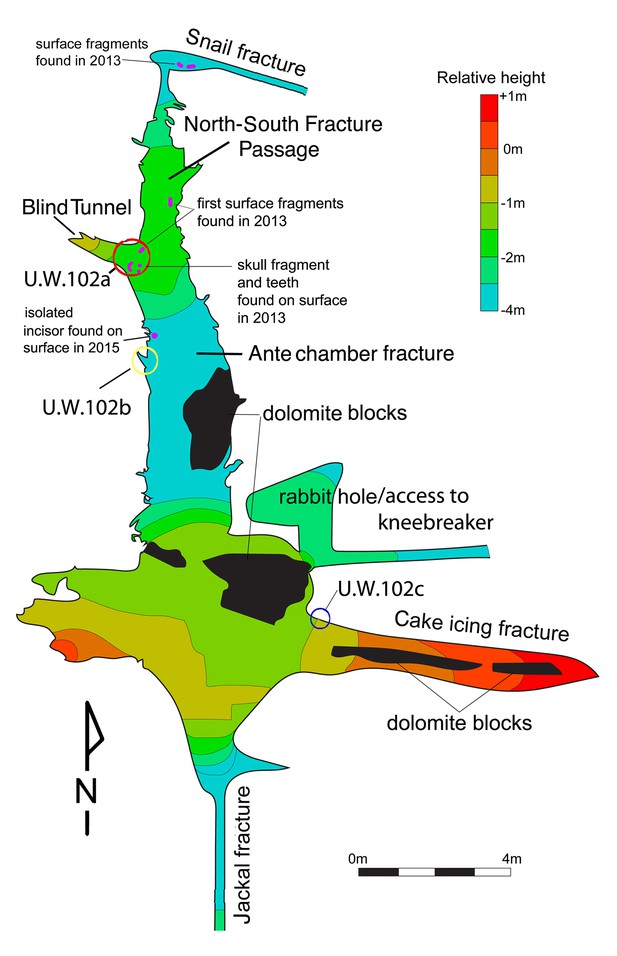
Schematic of the Lesedi Chamber, showing the three hominin-bearing collection areas: U.W.102a, 102b, and 102c.
https://doi.org/10.7554/eLife.24232.006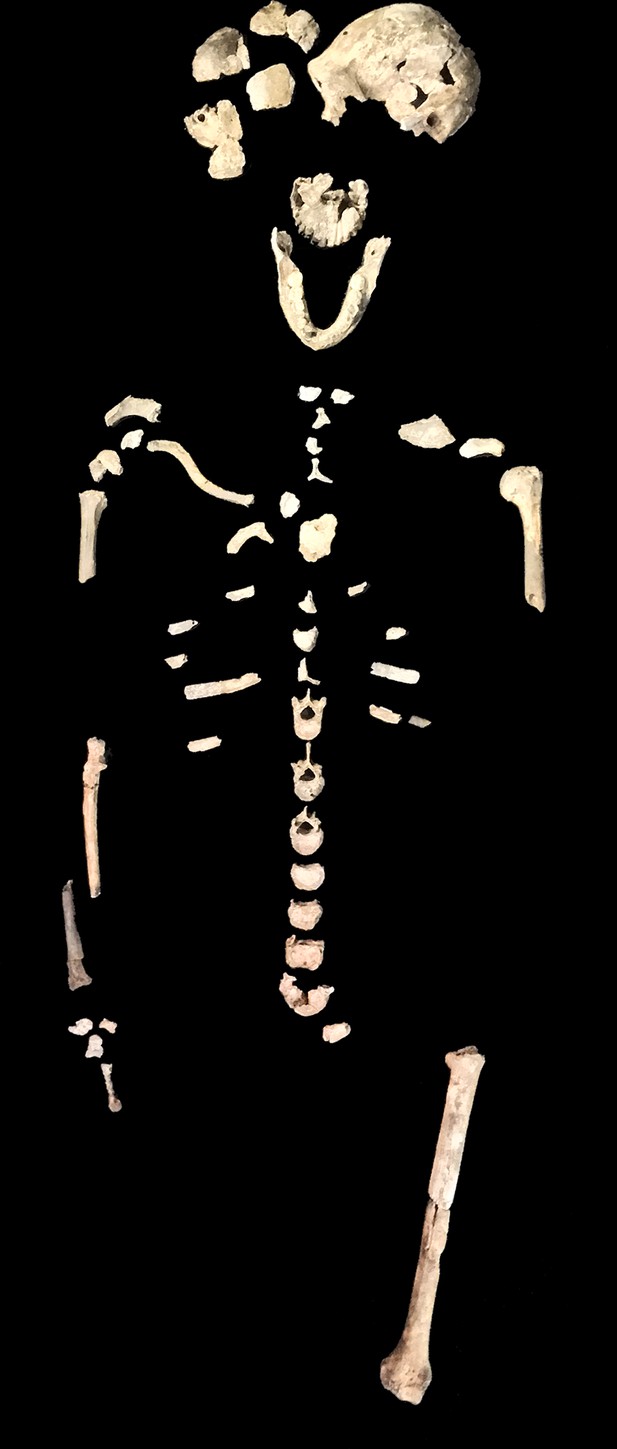
Skeletal material from locality 102a provisionally assigned to the LES1 skeleton.
The adult cranial material from 102a all belongs to a single cranium; most of the adult postcranial material probably belongs to the same individual. The adult cranial and postcranial material is shown here, except for the U.W. 102a-001 femur. The possibility that the femora represent two adult individuals makes it unclear which femur may be attributable to the skeleton; for the purposes of illustration, the U.W. 102a-003/U.W. 102a-004 femur is included in this photograph.
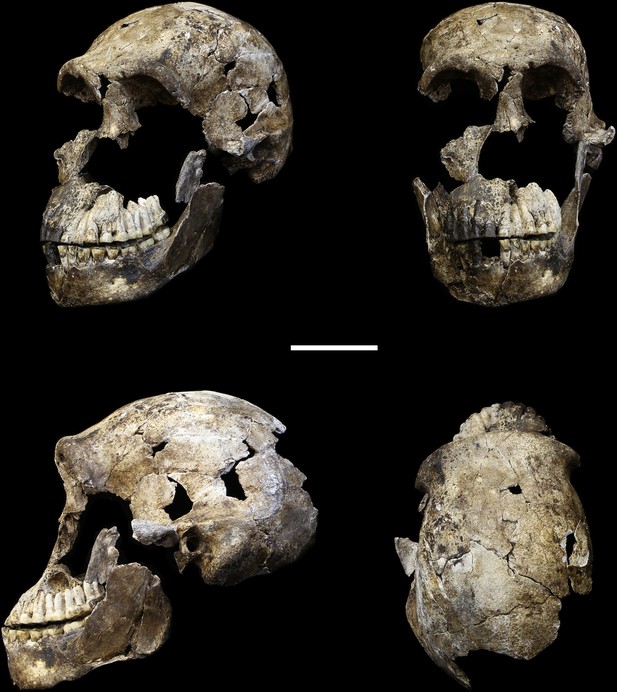
LES1 cranium.
Clockwise from upper left: three-quarter, frontal, superior and left lateral views. Fragments of the right temporal, the parietal and the occipital have also been recovered (not pictured), but without conjoins to the reconstructed vault or face. Scale bar = 5 cm.
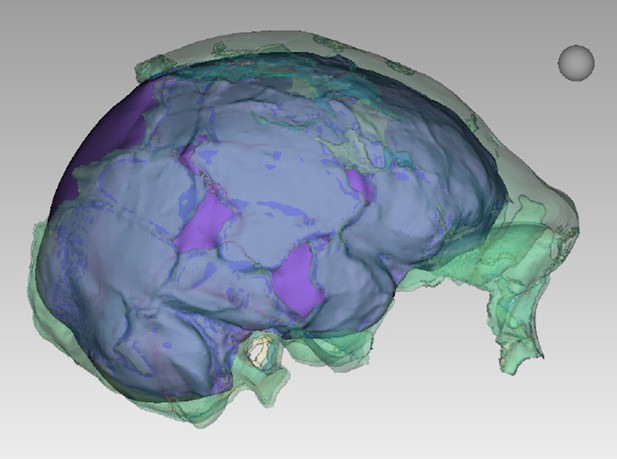
Digital reconstruction of endocranial volume in LES1.
The refitted calvaria was mirrored and filled, resulting in a volume estimate of 610 ml. Scale sphere = 10 mm.

Frontal and vault morphology in H. naledi compared to that in other hominin species.
Several of the crania pictured here are similar to H. naledi in endocranial volume, including Sts 5, MH1, KNM-ER 1813, and D2282, representing four different species. However, these skulls contrast strongly in other features. H. erectus is highly variable in size, as illustrated here by D2282 from Dmanisi, Georgia, one of the smallest and earliest H. erectus crania, and the L2 cranium from Zhoukoudian, China, one of the largest and latest H. erectus specimens. The relatively early KNM-ER 3733 has a size and endocranial volume close to the mean for H. erectus. Cranial remains that are attributed to H. erectus share a combination of anatomical features despite their diversity in size. Many such features of H. erectus are also shared with H. naledi, H. habilis, or Au. sediba, and notably, the differences in the frontal and vault between KNM-ER 1813 (H. habilis) and KNM-ER 1470 (H. rudolfensis) are mostly features that the smaller KNM-ER 1813 shares with H. naledi, H. erectus, and Au. sediba. The H. naledi skulls share some aspects of frontal morphology with Au. sediba, H. habilis and H. erectus that are not found in Au. africanus or H. rudolfensis, including frontal bossing and a supratoral sulcus. Two additional traits of the H. naledi anterior vault are shared with Au. sediba and H. erectus:slight postorbital construction and a posterior position of the temporal crest on the supraorbital torus. More posteriorly on the vault, H. naledi further shares an angular torus with H. erectus, and some individuals also have sagittal keeling. Both of these traits are also present in some archaic humans. Some H. naledi crania, such as DH3, are substantially smaller than any H. erectus cranium, and the small size and thin vault bone of even the largest H. naledi skull, LES1, are outliers compared to H. erectus, matched only by some Dmanisi crania. The facial morphology of H. naledi is more distinct from those of H. erectus and H. habilis. The nasal bones of LES1 do not project markedly anteriorly, although like many specimens of H. erectus, LES1 has a projecting nasal spine. LES1 has a relatively flat lower face, with a transversely concave clivus and incisors that project only slightly past the canines. This morphology is similar but less extreme than that found in KNM-ER 1470 of H. rudolfensis, and is not shared with the other species pictured here. H. naledi has several distinctive features of the temporal bone that are absent from or found in only a few specimens of the other species pictured, including a laterally inflated mastoid process (comparable to some specimens of Au. afarensis), a weak or absent crista petrosa (comparable to Au. afarensis), and a small external auditory meatus (comparable to KNM-WT 40000 of Kenyanthropus platyops [Leakey et al., 2001]). In this illustration, KNM-ER 1813, KNM-ER 1470, KNM-ER 3733, and ZKD L2 are represented by casts. Because these images are in a nonstandard orientation, scale is approximate.
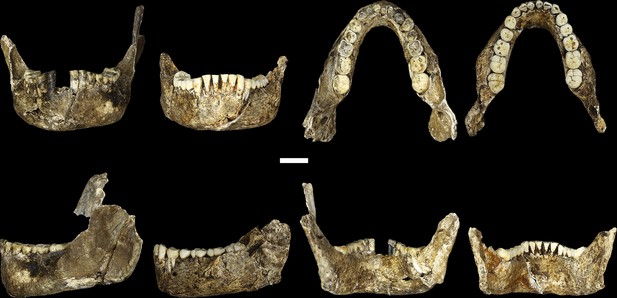
LES1 mandible compared to the DH1 holotype mandible of H. naledi.
In each pair, LES1 is on the left and DH1 on the right. Top left: anterior view. Top right: occlusal view. Bottom left: left lateral view. Bottom right: posterior view. Scale bar = 2 cm.
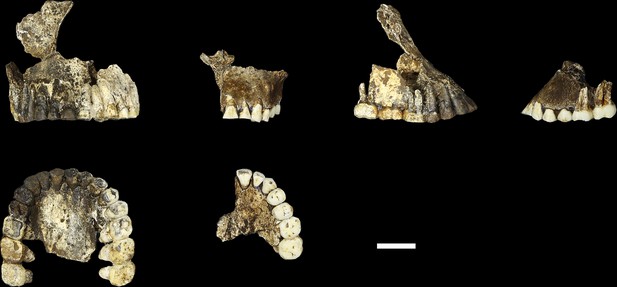
Comparison of LES1 maxilla to the DH1 holotype maxilla of H. naledi.
In each pair, LES1 is on the left and DH1 on the right. Top left: anterior view. Top right: right (LES1) and left (DH1) lateral view. Bottom: occlusal view. Scale bar = 2 cm.
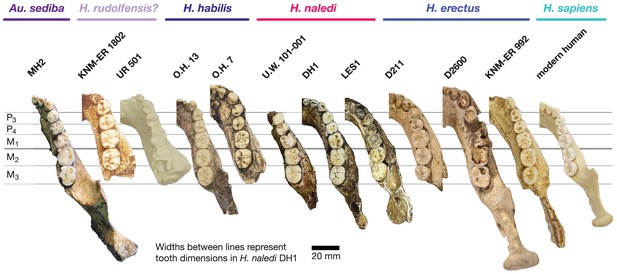
Mandibular and dental anatomy in H. naledi compared to other species of Homo.
Right demi-mandibles attributed to H. rudolfensis, H. habilis, H. naledi, H. erectus, and H. sapiens are pictured. All mandibles are aligned using the line marking the distal edge of the first molar. Each of the six horizontal lines corresponds to the edges of teeth in the DH1 mandible, the holotype specimen of H. naledi, with corresponding teeth labeled to the left. Using these lines, it is apparent which specimens have longer premolars and first molars, and which have longer second and third molars compared to DH1. The dentition of the LES1 mandible has been affected by interproximal wear, resulting in shorter mesiodistal measurements. Mandibular morphology and dental proportions vary slightly among most species of Homo, particularly in comparison with the large differences in dental proportions among species of Australopithecus and Paranthropus. Still, H. naledi is clearly distinguishable from other species of Homo (Berger et al., 2015; Laird et al., 2017). Fossils of H. rudolfensis, H. habilis, and H. erectus differ from H. naledi in the proportions of different parts of the postcanine tooth row and in features of the mandibular corpus. H. erectus. While fossils attributed to H. erectus vary in dental proportions, the early African and Georgian fossil specimens (here represented by KNM-ER 992, D211 and D2600) have larger first molars than H. naledi, comparable premolar sizes, and highly variable second and third molar sizes. The mandibles attributed to H. erectus mostly have greater corpus height than H. naledi mandibles and are highly diverse in corpus breadth, symphyseal thickness, and robusticity. Many have a strong post-incisive planum, most obvious in D2600 (shown). All three also differ from H. naledi in the crown complexity of their molars and premolar morphology, as illustrated in more detail in Figure 12. Some specialists would attribute these three mandibles of H. erectus to three different species. H. habilis. The two Olduvai mandibles of H. habilis are themselves quite different from each other in size. Both have similar dental proportions to H. naledi with bigger teeth across the postcanine dentition. Tobias (1967) viewed O.H. 13 as being similar to H. erectus and described it as an ‘evolved H. habilis’. Its occlusal morphology and dental proportions do resemble KNM-ER 992 (Wood, 1991), although the mandibular corpus is thinner and shallower, with a curved base in lateral profile. A strong post-incisive planum is evident in both mandibles. H. rudolfensis. The KNM-ER 1802 and Uraha (UR 501) mandibles have often been attributed to H. rudolfensis, although both attributions may be doubtful (Leakey et al., 2012). However, both lack any special similarities with contemporary australopiths and represent a megadont early Homo morphology with corpus size and robusticity much greater than those of H. naledi. Au. sediba. Molar sizes in the MH2 mandible are around 1 mm larger than the average for H. naledi, but the proportions are very similar to those of H. naledi, and like H. naledi, MH2 has a weak post-incisive planum and a small symphysis area. H. sapiens. The modern human mandible shown here, from a recent South African individual, has similar first molar size to the H. naledi mandibles, but much smaller premolars and second and third molars. The crown complexity in this individual, which is not unusual for African population samples, is substantially greater than evidenced in H. naledi. The mandibular corpus is smaller and much less robust than H. naledi. KNM-ER 1802, UR 501, O.H. 13, O.H. 7, and KNM-ER 992 are illustrated here with casts; the remainder are original specimens. The left side of O.H. 7 is shown here mirrored.
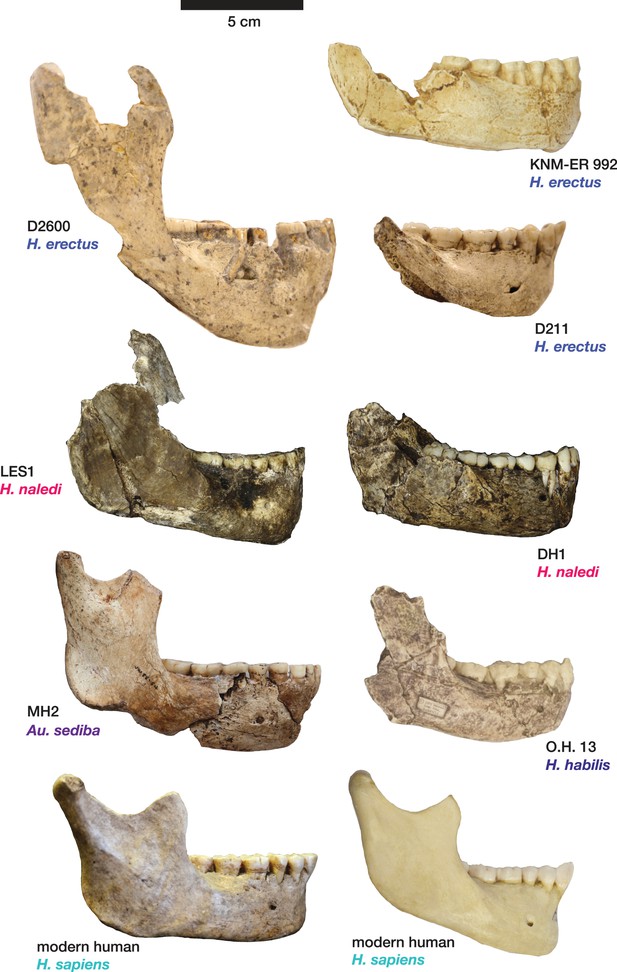
Comparison of H. naledi mandibles to other hominin species, from lateral view.
The DH1 holotype mandible and the LES1 mandible of H. naledi have a moderately deep mandibular corpus compared to other species of Homo; the LES1 mandible has a slightly greater corpus height anteriorly (at P3) than posteriorly (at M2). LES1 has rather a high coronoid process; the height of the condyle was probably lower than this. The mental foramen is at the midpoint or slightly higher in both H. naledi mandibles, and in both, the symphysis is nearly vertical. These features vary substantially within Homo. Modern humans (bottom) typically have a chin, but otherwise vary substantially in corpus height, whether the base of the corpus is parallel with the alveolar portion or with the occlusal surfaces of the teeth. Here that variability is illustrated with two modern human mandibles of male individuals, one from island Melanesia (left), and one from southern Africa (right). H. erectus exhibits very extensive variation in corpus height and thickness. D2600 (shown) is extremely thick and robust, but is not an outlier; other H. erectus mandibles approach or equal its corpus dimensions. The position of the mental foramen also varies, as does the relative anterior versus posterior corpus height and the symphyseal profile, from more sloping to near vertical (as illustrated by KNM-ER 992, although this specimen is damaged at the symphysis). MH2 (Au. sediba) has comparable corpus height and robusticity to the H. naledi mandibles, with a more sloping symphysis. O.H. 13 is a more gracile mandible than the H. naledi specimens in many respects. It has a curved base and a sloping symphysis. The more complete left side of LES1 is shown here and mirrored for comparison to other specimens. KNM-ER 992 and O.H. 13 are represented here by casts.
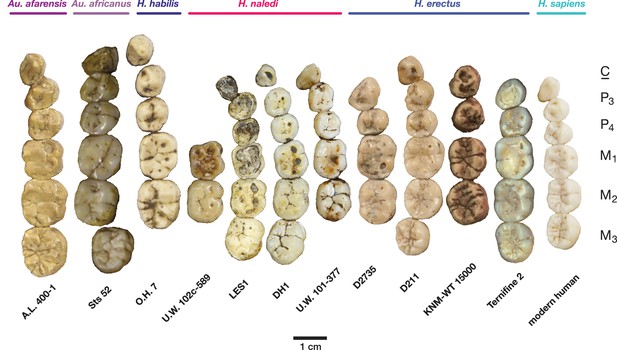
Occlusal view of H. naledi mandibular teeth compared to those of other hominins.
Teeth from the canine to the third molar are shown, if present, in the orientation in which they are found within the mandible. All individuals are aligned vertically by the distal margin of the first molar. Mandibles from the Lesedi Chamber, U.W. 102 c-589 and LES1 are shown next to DH1 and U.W. 101–377 (Berger et al., 2015). The mandibles illustrated from H. erectus have relatively little occlusal wear, so their morphology can be seen more clearly than that of worn mandibles. The immature U.W. 101–377 (H. naledi) is comparable in developmental age and wear to O.H. 7 (H. habilis), as well as to D2735 and KNM-WT 15000 (H. erectus). When compared to H. habilis, H. erectus, and australopiths, H. naledi is notable for its relatively small first molars, its relatively small canines, and its lack of supernumerary cusps and crenulation on the molars. The complexity of molar cusp and groove patterns is especially evident in the chronologically early H. erectus specimens from Africa and Georgia shown here. For example, the unworn M2 of the immature U.W. 101–377 mandible of H. naledi has a relatively simple crown anatomy with very little wrinkling or crenulation. By comparison, the M2 of D2735, D211, and KNM-WT 15000, all with minimal occlusal wear, show extensive crenulation and supernumerary cusps. Canine size and molar crown complexity vary substantially among modern human populations, but the southern African individual illustrated on the right is not atypical for its population, and has greater molar crown complexity and larger canine dimensions than any of the H. naledi mandibular dentitions. The morphology of the third premolar varies extensively among these hominin species and within H. erectus. The H. naledi P3 anatomy can be seen clearly in the immature U.W. 101–377 individual. It is characterized by roughly equally prominent lingual and buccal cusps and an expanded talonid. In H. naledi, this tooth is broadly similar in morphology and size to the P4. This configuration of the P3 is not present in the other species, with only KNM-WT 15000 exhibiting some expansion of the lingual cusp in what remains an asymmetrical and rounded P3. A.L. 400–1, O.H. 7 and KNM-WT 15000 are represented by casts; The left dentition of U.W. 102 c-589 and O.H. 7 have been mirrored to compare to right mandibles. Images have been scaled by measured first molar dimensions.
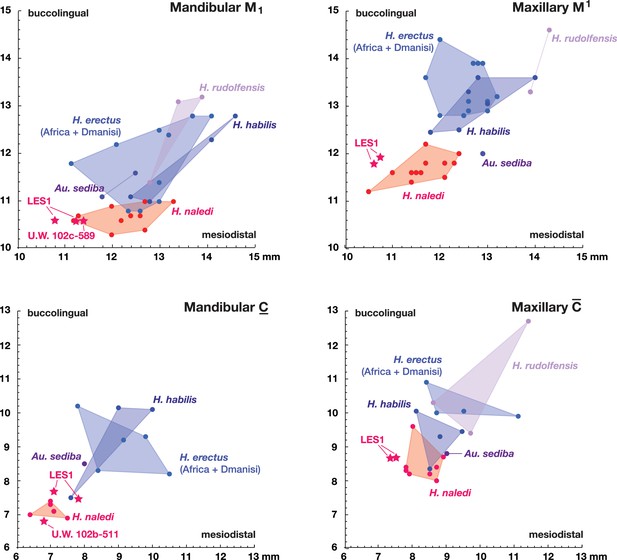
Metric comparisons of the Lesedi Chamber dental material.
H. naledi is clearly differentiated in first molar and canine dimensions from other species with broadly similar cranial and dental morphology, including Au. sediba, H. habilis, H. rudolfensis, and early H. erectus samples from Africa and Georgia. The material from the Lesedi Chamber is within the range of or similar to H. naledi in these dimensions and well differentiated from the other samples. Top left: mandibular first molar dimensions. Top right: maxillary first molar dimensions. Bottom left: mandibular canine dimensions. Bottom right: maxillary canine dimensions. The LES1 first molars and maxillary canines have a substantial degree of interproximal wear, and the values plotted here are not corrected for this wear, which shortened the mesiodistal dimension by as much as a millimeter. The values plotted here should thus be regarded as minimum values. The H. erectus sample here includes specimens from the Lake Turkana area, Konso, Tighenif (Ternifine), Thomas Quarry, and Dmanisi; Asian H. erectus specimens are omitted. Attributions of H. habilis and H. rudolfensis specimens are indicated in the Materials and methods.

U.W. 102a-021 right clavicle from the Lesedi Chamber.
Left, from top: superior, anterior, inferior, posterior views. Right, from top: medial and lateral views. Scale bar = 2 cm.

U.W. 102a-002 right humerus fragment.
From left: posterior, medial, anterior and lateral views. Right, from top: Scale bar = 5 cm.
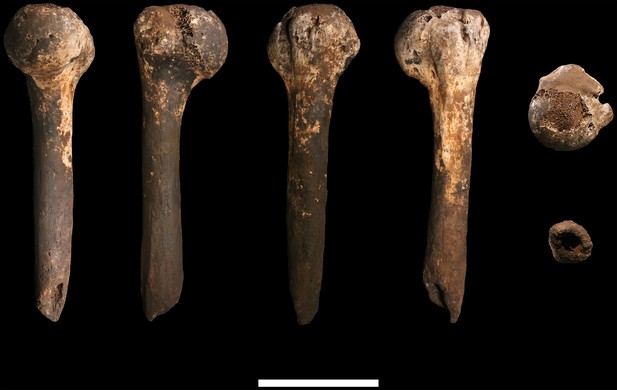
U.W. 102a-257 left proximal humerus fragment.
From left: posterior, medial, anterior and lateral views. Top: proximal view. Bottom: distal view. Scale bar = 5 cm.

U.W. 102a-015 ulna fragment.
From left: anterior, medial, posterior and lateral views. Right from top: proximal and distal views. Scale bar = 2 cm.
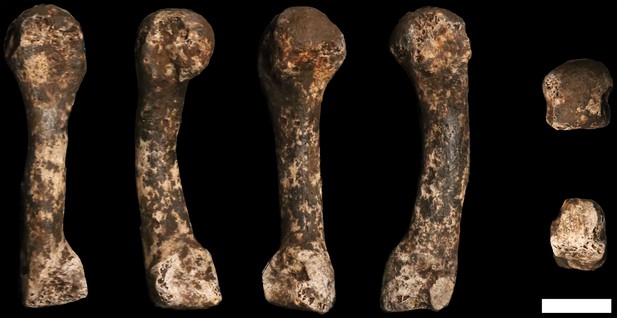
U.W. 102a-028 right fourth metacarpal.
From left: dorsal, ulnar, palmar and radial views. Right from top: distal and proximal views. Scale bar = 1 cm.
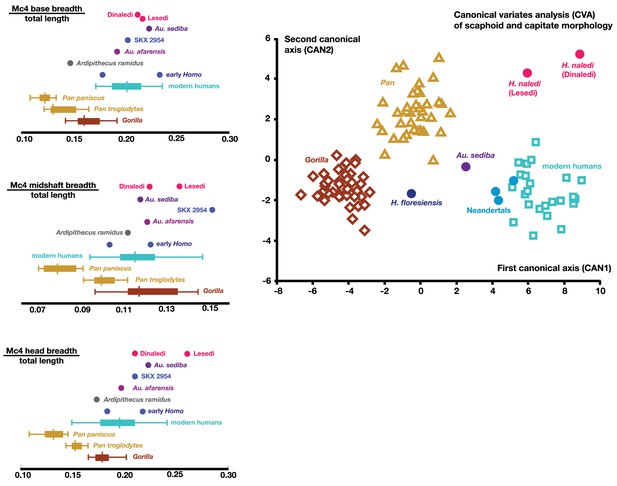
Quantitative comparisons of hand and wrist material from the Lesedi Chamber.
Left: ratios of fourth metacarpal dimensions in H. naledi compared to those in other hominin and great ape samples. Right: canonical variates analysis of capitate and scaphoid morphology in humans, chimpanzees, gorillas, and fossil hominins. H. naledi from the Dinaledi Chamber occupies a unique position in scaphoid and capitate joint configurations, which is closely matched by the capitate and scaphoid from the Lesedi Chamber. In this analysis, no a priori groups are assumed; we also examined the scenario in which Homo naledi and other fossil specimens are included as a priori groups and the results are essentially identical.
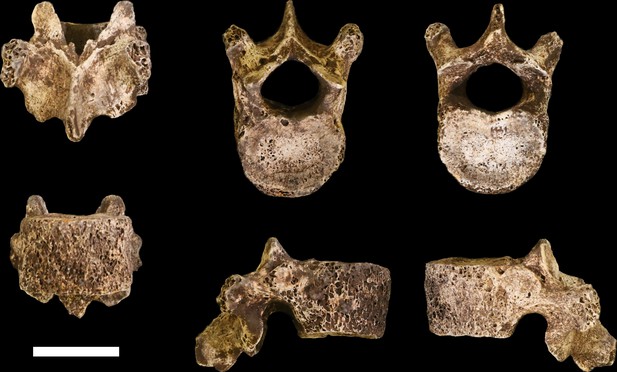
U.W. 102a-036 vertebra, T10.
Clockwise from top left: posterior, superior, inferior, left, right, and anterior views. Scale bar = 2 cm.
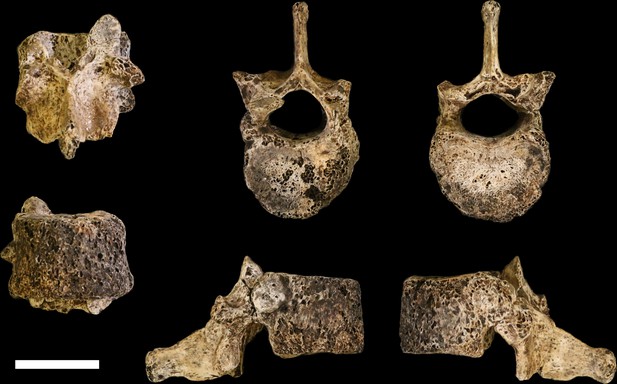
U.W. 102a-151 vertebra, T11.
Clockwise from top left: posterior, superior, inferior, left, right, and anterior views. Scale bar = 2 cm.
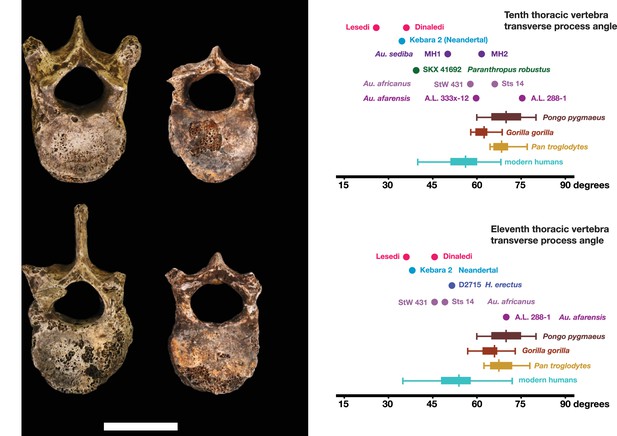
Vertebral transverse process orientation.
H. naledi is distinctive when compared to many other hominin species in having T10 and T11 vertebral transverse processes oriented with a relatively low angle. Left: U.W. 102a-036 compared to U.W. 101–855 from the Dinaledi Chamber (top), and U.W. 102a-151 compared to U.W. 101–1733 (bottom). All of these vertebrae have transverse processes oriented more posteriorly than those of most other hominins, U.W. 102a-036 is the most extreme. Right: charts showing the comparative orientation of transverse processes in humans, living great apes, and fossil hominins. For the T10 (top), the U.W. 102a-036 value (labeled ‘Lesedi’) is lower than that for any other hominins, while the Dinaledi T10 is similar to the Neandertal value and extremely low compared to that for modern humans. The T11 (bottom) shows a similar but less extreme pattern.
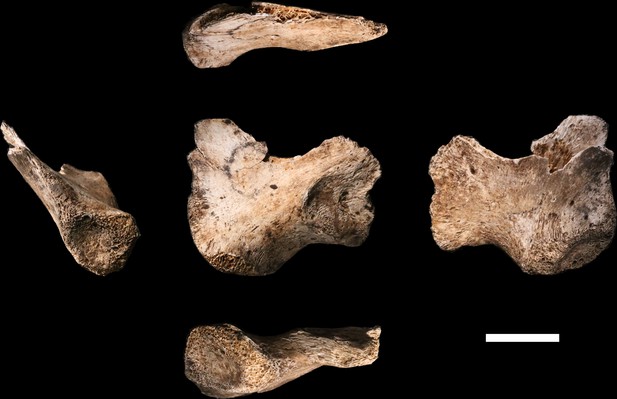
U.W. 102a-138 immature right os coxa fragment.
The medial view is at the center. Clockwise from top: superior, lateral, inferior and anterior views. The unfused triradiate suture is notable.

U.W. 102a-001 proximal femoral fragment.
From left: posterior, medial, anterior and lateral views. Right from top: proximal and distal views. Scale bar = 5 cm.

U.W. 102a-003 left proximal femur fragment.
From left: posterior, medial, anterior and lateral views. Right from top: proximal and distal views. Scale bar = 5 cm.
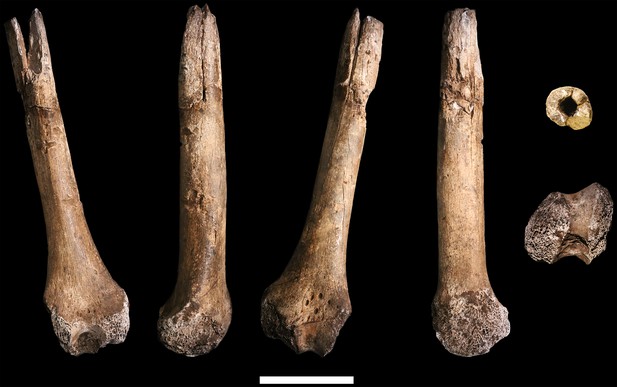
U.W. 102a-004 left distal femur fragment.
From left: posterior, medial, anterior and lateral views. Right from top: proximal and distal views. Scale bar = 5 cm.

Length estimation of femur based on U.W. 102a-003 and U.W. 102a-004.
Two specimens were used to estimate the missing proximal and distal ends of the femur. Top: U.W. 101–215 is a distal femur fragment that presents a similar morphology to the U.W. 102a-004 distal femur, while preserving the distal articular surface. Middle: U.W. 102a-004 and U.W. 102a-003 conjoined, in posterior view. Bottom: U.W. 102a-001 is comparable in size with U.W. 102a-003, and while the morphology of the muscle markings is different, the alignment of the lesser trochanters gives a good basis for estimating the proximal extent of the bone. The length estimate is 375 mm.
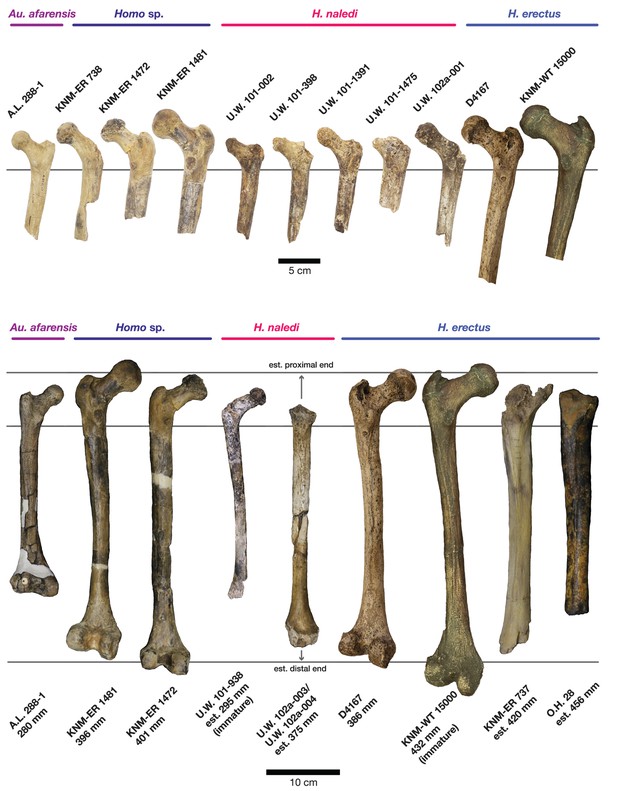
Comparison of H. naledi femora to those attributed to early Homo.
Top: roximal femora attributed to Au. afarensis, Homo sp., H. naledi, and H. erectus, all shown in posterior view. The femora have been aligned by matching the inferior point on the lesser trochanter to the horizontal line on the figure, and all are shown at approximately the same shaft angle. Many of the H. naledi femora have notably thin shafts, although the largest shown here, U.W. 101–1475, is greater in shaft thickness than the complete KNM-ER 1480, KNM-ER 1472, or D4167 femora. The specimens here attributed to ‘Homo sp.’ were surface finds without associated cranial material. Their anatomy has been considered consistent with Homo, although some have suggested that KNM-ER 738 may instead be Paranthropus. U.W. 102a-001 is a right femoral fragment, and left femora here have been mirrored for comparison, including KNM-ER 738, KNM-ER 1481, U.W. 101–398, U.W. 101–1475, KNM-WT 15000, and A.L. 288–1. Bottom: H. naledi femur length compared to those of other fossil femora. U.W. 102a-003 is shown conjoined with U.W. 102a-004. The top and bottom horizontal lines correspond to the proximal and distal limits of the maximum length estimate for this femur as illustrated in Figure 24. Femur maximum lengths and length estimates are as reported by McHenry (1991), D4167 length is from Lordkipanidze et al. (2007). As above, all femora are aligned by the lesser trochanter, which is preserved in all of these shaft fragments. The H. naledi U.W. 102a-003 femur has a shaft diameter comparable to that of A.L. 288–1, or even slightly shorter, but at an estimated 375 mm, it is nearly the same length as the D4167 femur of H. erectus at 387 mm. The U.W. 101–484 tibia specimen from the Dinaledi Chamber is also long and relatively narrow, and has an estimated length greater than that of the D3901 tibia from Dmanisi (Marchi et al., 2017). Several very thick, large femora have been attributed to H. erectus from the Early Pleistocene of Africa; these are very different from U.W. 102a-003 in their size, robustness, and more platymeric proximal shafts. U.W. 101–938 is an immature femur with no fusion of the head or distal epiphysis; it represents a younger developmental age than KNM-WT 15000, the immature H. erectus skeleton. Even at its young age, this H. naledi specimen is nonetheless longer than the A.L. 288–1 femur of Au. afarensis. A.L. 288–1 is shown here as reconstructed by P. Schmid. For comparison to the right U.W. 102a-003 femur, left femora are shown mirrored here, these include KNM-ER 1472, U.W. 101–938, and D4167. In this figure, KNM-ER 739, KNM-ER 1472, KNM-ER 1481, KNM-WT 15000, O.H. 28, and A.L. 288–1 are represented by casts.
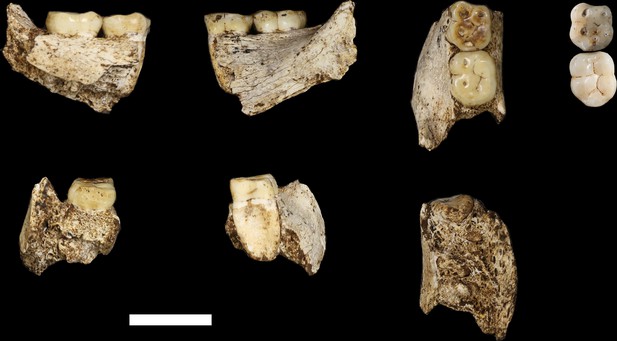
U.W. 102b-438 immature mandibular fragment.
From left: basal, lingual, occlusal, buccal and anterior views. The RP4 is within its crypt. Scale bar = 2 cm.

U.W. 102b-511 left mandibular canine crown from locality 102b.
Top row, from left: occlusal, lingual, distal, labial and mesial views. Bottom row: U.W. 102b-511 (left), compared to U.W. 101–1126 (middle) and U.W. 101–985 (right) relatively unworn left mandibular canines from the Dinaledi Chamber. All three teeth share a distinctive morphology, which is also present in the other Dinaledi mandibular canines, that includes an asymmetrical crown, higher mesial shoulder, and distal accessory cuspule. Scale bar = 1 cm.
U.W. 102 c-589 mandibular fragment.
Top row, from left: lingual view, buccal view and occlusal view. Top right: two H. naledi teeth from the Dinaledi Chamber that present comparable occlusal morphology and wear to the U.W. 102 c-589 teeth: U.W. 101–297 RM1 (top, reversed to represent left side) and U.W. 101–284 LM2 (bottom). Bottom row, from left: U.W. 102 c-589 posterior view, anterior view and basal view. Scale bar = 2 cm.
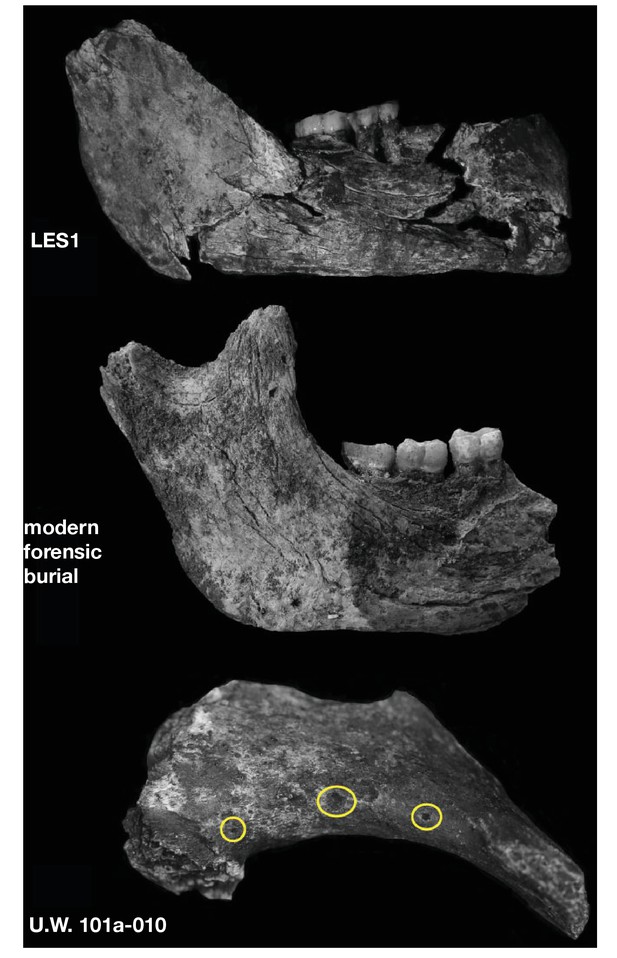
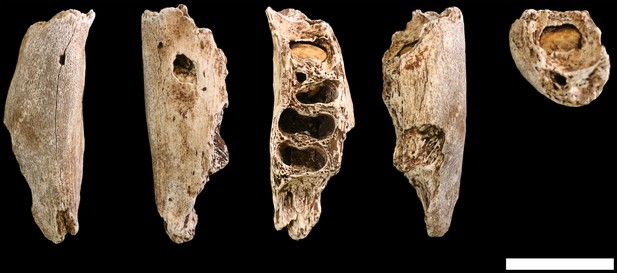
Surface cracking and pitting in Lesedi Chamber material.
Top: right corpus and ramus of LES1 mandible prior to reconstruction, showing patterns of cracking consistent with the effects of sediment loading and wetting during the burial process and during skeletal decomposition. This pattern of taphonomic alteration is superficially similar to sub-aerial weathering processes, but is independent of surface exposure and occurs in both deep- and shallow-buried deposits. Middle: forensic known-history case for comparison. This specimen was recovered from a deep known-history inhumation. The fleshed body was deposited at a depth of 1.5 metres and recovered after a 30-year burial period. Note the similarities in surface texture, and superficial cracking, which follows the biomechanical stress lines (grain) of the bone. Bottom: U.W. 102a-010 acromial fragment showing surface modification with cortical pitting and punctate marks. Note that many of these marks (circled in yellow) penetrate pre-existing layers of manganese oxy-hydroxide deposited on the bone surface; the damage was therefore produced inside the Lesedi Chamber on bones that were already covered in coatings of manganese mineral.
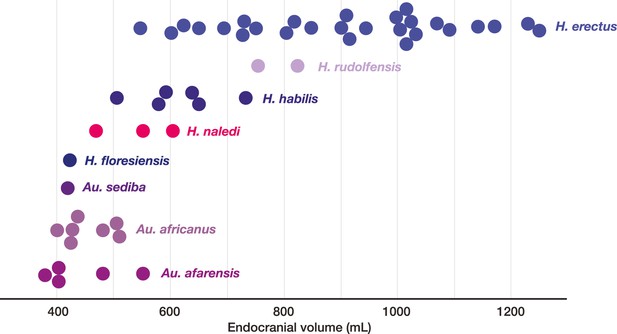
Endocranial volumes of hominin species.
With the addition of LES1 to the sample, the range of endocranial volume in H. naledi is extended slightly beyond the range represented in the Dinaledi Chamber. This range overlaps with two specimens of H. erectus, and LES1 is larger than the largest Au. africanus or Au. afarensis specimens. Data and sources are listed in Table 4.
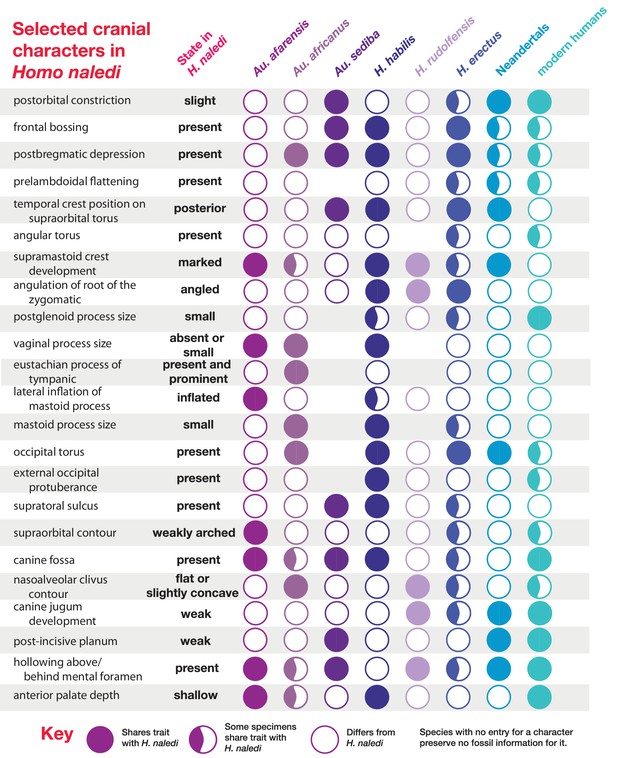
Selected cranial trait observations in H. naledi and other species.
A subset of observations of cranial traits reported in Supplementary file 1 that vary among species attributed to Homo. This list omits traits that are present in only one species of Homo or for which nearly all species exhibit more than one state. Here, the character state observed in H. naledi, including both Dinaledi Chamber and Lesedi Chamber material, is reported on the left. Character states for other species are reported in terms of whether they share the same state as H. naledi. Some traits that are shared with H. naledi may be interpreted as shared derived traits, based on their absence from Au. afarensis and Au. africanus, but some species of Homo also share primitive traits with H. naledi that may also be found in the australopiths. The pattern of shared morphological characters among these species and H. naledi is complex, and there is no consistent anatomical grouping of the characters shared with any given species. H. naledi shares more characters with H. erectus and H. habilis than with other species, and behind them, with Au. sediba and archaic and modern humans. H. erectus is variable for a large number of traits also found in H. naledi. The fact that variability is noted in H. erectus, Neandertals and modern humans for these traits is partially a function of the large samples available for these groups. It is probable that a larger sample of other species would likewise encompass greater variability. Any phylogenetic tree of these species would reveal a high degree of homoplasy for these cranial and mandibular traits.
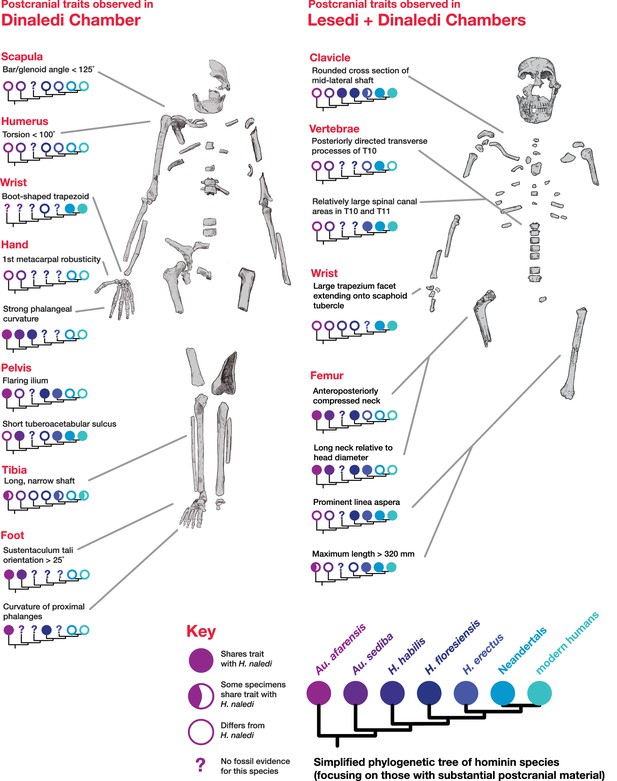
Postcranial traits in H. naledi compared to those in other hominin species.
Here, a subset of features of the postcranial skeleton that distinguish H. naledi from other species are summarized in comparison to these traits in other hominin species with substantial postcranial evidence. These features include some that H. naledi shares with Au. afarensis, some that H. naledi shares with modern humans or Neandertals, and some that are unique or shared with even more distantly related species such as Ardipithecus (not shown). These traits constitute a mosaic that distinguishes H. naledi clearly from other species of Homo. Many of the traits that are notable in the Dinaledi Chamber sample are also represented in the Lesedi Chamber material.

Lateral cranial comparison of H. naledi crania to crania of other hominin species.
H. naledi crania, DH1, LES1, and DH3 are in the center row. All crania are oriented as near as possible to the Frankfort plane, delineated by the light gray lines in the background of the figure. Compared to other hominin genera, including Australopithecus and Paranthropus, fossil Homo is often recognized by cranial and dental features such as a more vertical face profile, a reduced postcanine dentition, larger endocranial volume, a higher frontal, and a true supraorbital torus. Au. africanus (Sts 5, top left) represents the ancestral hominin condition lacking these traits. The other crania in the top two rows vary substantially in these features. LB1 has a vertical face, reduced dentition, and high, rounded frontal, but has comparatively small endocranial volume. KNM-ER 1470 has a large volume, a high frontal, and a more vertical face profile, but also is inferred to have a large postcanine dentition and has no true supraorbital torus. MH1 (Au. sediba) has a small volume, but shares features with Homo that include the less sloping face profile, a supraorbital torus, and reduced postcanine dentition. O.H. 24 has a low, sloping frontal, and a concave facial profile, but a true supraorbital torus and reduced postcanine teeth. This variability among species that are interpreted as ‘primitive’ Homo, such as H. habilis and H. floresiensis, and Homo-like australopiths makes it difficult to delineate the genus Homo (Wood and Collard, 1999; Dembo et al., 2016). H. erectus is also highly variable. It includes several crania with endocranial volumes below 700 ml, including KNM-ER 42700 and D2282, but also many larger crania, here represented by Sangiran 17 and Zhoukoudian L2 (ZKD L2). Specimens attributed to H. erectus tend to share a series of traits first noted in Asian H. erectus samples, including a long, low cranial profile, thick cranial bone, sagittal keeling, prominent supraorbital, angular, and occipital tori, a sharply angled occiput, and a postbregmatic depression. The smallest H. erectus crania share most of these features, with a low cranial profile, angled occiput and postbregmatic depression visible here in D2282. But these features do vary substantially and are less evident in the immature KNM-ER 42700. The H. naledi crania are similar to KNM-ER 1470 in having a transversely flat clivus contour, but all are smaller, with a much smaller palate and with very different frontal morphology. Like O.H. 24 and KNM-ER 1813, the H. naledi crania have relatively thin cranial bone and a thin and projecting supraorbital torus. But H. naledi manifests a different clivus shape, a projecting nasal spine, a greater cranial height, sagittal keeling and an angular torus. The H. naledi crania bear little resemblance to LB1, differing in face profile, size, and their larger postcanine dentitions. Known African Homo specimens from the later Middle Pleistocene other than H. naledi, such as the Kabwe skull (pictured), contrast strongly with H. naledi in cranial size and morphology. The Omo 2 skull, one of the earliest known modern human crania at approximately 196,000 years (McDougall et al., 2005), is vastly larger and very different from any H. naledi specimen, despite being near the same geological age. In this figure, O.H. 24, KNM-ER 1470, LB1, KNM-ER 42700, ZKD L2, and Omo 2 are represented by casts. Images have been adjusted to a common scale by maximum cranial length, or by glabella-bregma length where maximum length is not available. Photos of Sangiran 17 and D2282 are courtesy of Milford Wolpoff.
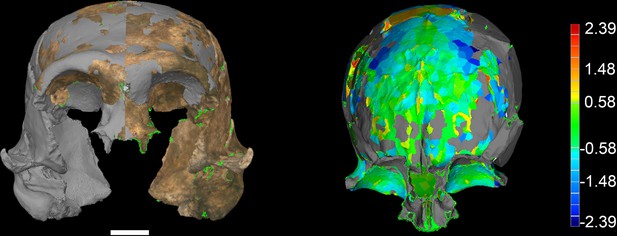
LES1 model congruency.
Congruency between the original LES1 3D scan and an aligned mirror-image. Scale bar = 2 cm. (A) Frontal view of a 3D scan of the original LES1 specimen (brown) aligned with the mirror-image (grey), illustrating congruency between overlapping regions. (B) Deviation map of the internal view of the frontal region. Deviation scale bar in mm.
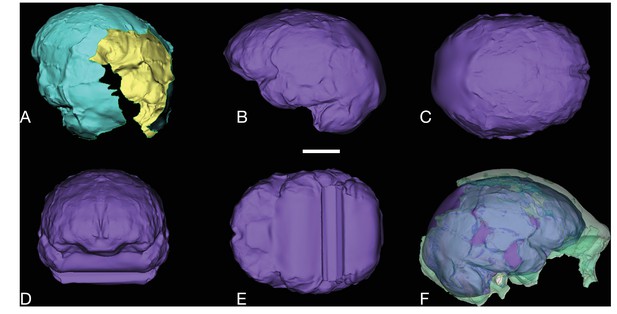
LES1 endocast reconstruction.
Virtual reconstruction of LES1 endocast for endocranial volume estimation. Scale bar = 3 cm. (A) Oblique posterior view illustrating missing portions in the endocranial reconstruction that were filled to close the model for volume estimation. (B-E) Left lateral (B), superior (C), anterior (D), and inferior (E) views of the completed endocranial model. (F) Right lateral view of the completed endocranial model within the surrounding cranium.
Tables
Hominin fossil material from the Lesedi Chamber. All diagnostic hominin specimens are listed, with attribution to element. Specimens that have been refitted are not listed separately. Most Locality 102a cranial fragments are presumed to be part of LES1 and are not listed separately.
| Specimen number | Element | Notes |
|---|---|---|
| LOCALITY 102a | ||
| LES1 | cranium | constituted of 57 specimens, not listed separately |
| U.W. 102a-001 | proximal right femur | |
| U.W. 102a-002 | proximal right humerus | |
| U.W. 102a-003 | proximal left femur | |
| U.W. 102a-004 | distal left femur | |
| U.W. 102a-010 | right scapula fragment | acromion |
| U.W. 102a-013 | humeral head fragments | |
| U.W. 102a-015 | right proximal ulna | |
| U.W. 102a-018 | long bone fragment | immature |
| U.W. 102a-019 | partial rib | |
| U.W. 102a-020 | right ulna fragment | |
| U.W. 102a-021 | right clavicle | |
| U.W. 102a-025 | right radius shaft fragment | |
| U.W. 102a-028 | right fourth metacarpal | |
| U.W. 102a-036 | T10 vertebra | |
| U.W. 102a-039 | rib fragments | |
| U.W. 102a-040 | long bone shaft fragment | |
| U.W. 102a-117 | right scaphoid | |
| U.W. 102a-138 | right ilium fragments | immature |
| U.W. 102a-139 | L5 vertebra fragments | |
| U.W. 102a-148 | sternum fragment | |
| U.W. 102a-151 | T11 vertebra | |
| U.W. 102a-152 | rib fragments | |
| U.W. 102a-154 | T12 and L1 vertebrae | found in articulation |
| U.W. 102a-155 | mid-thoracic vertebral body | |
| U.W. 102a-171 | atlas fragment | |
| U.W. 102a-172 | atlas fragment | |
| U.W. 102a-189 | rib fragment | |
| U.W. 102a-195 | rib fragment | |
| U.W. 102a-206 | left clavicle fragment | |
| U.W. 102a-207 | rib fragment | |
| U.W. 102a-210 | sacral element | immature, possibly S1 |
| U.W. 102a-231 | rib fragment | |
| U.W. 102a-232 | rib fragment | |
| U.W. 102a-236 | humerus head fragment | |
| U.W. 102a-239 | left clavicle fragment | |
| U.W. 102a-247 | right scapula fragment | coracoid process |
| U.W. 102a-250 | right first rib | |
| U.W. 102a-252 | rib fragment | |
| U.W. 102a-256 | left scapula fragment | portion of body, spine, and acromion |
| U.W. 102a-257 | left proximal humerus | |
| U.W. 102a-279 | left scapula fragment | partial glenoid fossa |
| U.W. 102a-280 | rib fragment | |
| U.W. 102a-300 | vertebral fragment | |
| U.W. 102a-306 | L4 vertebra body | |
| U.W. 102a-322 | L2 vertebra body | |
| U.W. 102a-337 | vertebral fragment | neural arch |
| U.W. 102a-348 | right pubic ramus fragment | |
| U.W. 102a-349 | vertebral fragment | neural arch |
| U.W. 102a-358 | rib fragments | |
| U.W. 102a-360 | vertebral fragment | |
| U.W. 102a-455 | ulna shaft fragment | |
| U.W. 102a-456 | ulna shaft fragment | |
| U.W. 102a-470 | rib fragments | |
| U.W. 102a-471 | right distal radius fragment | |
| U.W. 102a-474 | long bone fragment | immature |
| U.W. 102a-476 | right capitate | |
| U.W. 102a-477 | partial right lunate | |
| U.W. 102a-479 | rib fragment | |
| LOCALITY 102b | ||
| U.W. 102b-178 | LI2 | |
| U.W. 102b-437 | rdm2 | |
| U.W. 102b-438 | right mandibular corpus fragment | immature, RP4 in crypt |
| U.W. 102b-502 | cranial fragments | |
| U.W. 102b-503 | RP4 crown | |
| U.W. 102b-506 | cranial fragment | |
| U.W. 102b-507 | cranial fragment | |
| U.W. 102b-509 | cranial fragment | |
| U.W. 102b-511 | LC1 crown | |
| U.W. 102b-514 | cranial fragment | |
| U.W. 102b-515 | LI2 | |
| U.W. 102b-516 | cranial fragment | |
| LOCALITY 102c | ||
| U.W. 102 c-589 | left mandibular fragment | LM1 and LM2 in place |
Dental measurements for Lesedi Chamber specimens.
| Specimen | Mesiodistal diameter | Buccolingual (or labiolingual) diameter |
|---|---|---|
| U.W. 102b-437 ldm2 | 10.7 | 8.7 |
| U.W. 102b-503 RP4 | 8.4 | 10.9 |
| U.W. 102b-515 LI2 | 6.8 | 6.5† |
| U.W. 102b-178 LI2 | 5.6 | 5.9 |
| U.W. 102b-511 LC1 | 6.8 | 6.8† |
| U.W. 102 c-589 LM1 | 11.4 | 10.6 |
| U.W. 102 c-589 LM2 | 13.1 | 11.3 |
| LES1 maxillary | ||
| RI1 | 7.6* | 6.9 |
| RI2 | 6.8* | 7.0 |
| RC1 | 7.5 | 8.7 |
| RP3 | 8.1 | 10.8 |
| RP4 | 8.1 | 11.3 |
| RM1 | 10.6* | 11.8 |
| RM2 | 11.7 | 12.7 |
| RM3 | 11.4 | 12.7 |
| LI1 | 7.4* | 6.9 |
| LI2 | 6.1* | 6.8 |
| LC1 | 7.4 | 8.7 |
| LP3 | 8.0 | 10.9 |
| LP4 | 8.1 | 11.3 |
| LM1 | 10.7* | 11.9 |
| LM2 | 12.1 | 12.8 |
| LM3 | 11.4 | 13.6 |
| LES1 mandibular | ||
| RI1 | 5.8* | 6.3 |
| RI2 | 5.4* | 6.1 |
| RC1 | 7.1 | 7.7 |
| RP3 | 8.4 | 9.3 |
| RP4 | 8.2 | 9.1 |
| RM1 | 10.8* | 10.6 |
| RM2 | 12.3 | 11.5 |
| RM3 | 13.3 | 11.7 |
| LC1 | 7.8 | 7.5 † |
| LP3 | 8.4 | 9.3 |
| LP4 | 8.2 | 9.1 |
| LM1 | 11.2* | 10.6 |
| LM2 | 12.3 | 11.5 |
| LM3 | 13.3 | 11.7 |
-
*Denotes measurements where the tooth is extremely worn, and mesiodistal diameter reported here has not been corrected for the degree of wear.
-
†Denotes instances where we report a minimum value for labiolingual measurements because the crown is not complete or is broken.
Mammal species recorded in the Lesedi Chamber. Several specimens from the classes Aves, Amphibia and Reptilia were also recovered, but individual counts and taxonomic identifications are pending further examination.
| Class | Order | Family | Subfamily | Genus/species | MNI | NISP |
|---|---|---|---|---|---|---|
| Mammalia | Lagomorpha | Leporidae | 1 | 1 | ||
| Soricomorpha | Soricidae | 3 | 3 | |||
| Crocidurinae | Crocidura | 1 | 1 | |||
| Rodentia | 1 | 1 | ||||
| Bathyergidae | Bathyerginae | 1 | 1 | |||
| Muridae | 1 | 1 | ||||
| Otomyinae | Otomys | 1 | 1 | |||
| Murinae | 5 | 7 | ||||
| Mus | 1 | 2 | ||||
| Nesomyidae | Mystromyinae | Mystromys | 1 | 1 | ||
| Dendromurinae | 3 | 4 | ||||
| Steatomys | 3 | 6 | ||||
| Carnivora | Felidae | Felis aff. F. sylvestris | 1 | 1 | ||
| Felis cf. sylvestris | 1 | 6 | ||||
| Felis sp. | 1 | 2 | ||||
| Herpestidae | cf. Mungos | 1 | 1 | |||
| cf. Herpestidae | 1 | 3 | ||||
| Canidae | Canis aff. C. familiaris | 1 | 14 | |||
| Canis cf. mesomelas | 1 | 3 | ||||
| Canis sp. | 1 | 21 | ||||
| Vulpes cf. chama | 1 | 3 | ||||
| cf. Vulpes | 1 | 6 | ||||
| Vulpes sp. | 1 | 5 |
Endocranial volume of LES1 compared to key specimens of other hominin species. Estimates from Asfaw et al. (1999), Kubo et al. (2013), Holloway et al., (2014), Lee and Wolpoff (2003), Kimbel (2004); Berger et al. (2010), (2015), Lordkipanidze et al. (2006) and Lordkipanidze et al. (2013).
| Specimen | Cranial capacity in cc | |
|---|---|---|
| Au. afarensis | Mean | 444 |
| AL 162–28 | 400 | |
| AL 288–1 | 387 | |
| AL 333–45 | 485 | |
| AL 333–105 | 400 | |
| AL 444–2 | 550 | |
| Au. africanus | Mean | 455 |
| MLD 1 | 510 | |
| MLD 37/38 | 425 | |
| Sts 5 | 485 | |
| Sts 19 | 436 | |
| Sts 60 | 400 | |
| Sts 71 | 428 | |
| StW 505 | 505 | |
| Au. sediba | MH1 | 420 |
| H. floresiensis | LB1 | 426 |
| H. naledi | Mean | 513 |
| DH1 | 560 | |
| DH3 | 465 | |
| LES1 | 610 | |
| H. habilis | Mean | 616 |
| KNM-ER 1805 | 582 | |
| KNM-ER 1813 | 509 | |
| O.H. 7 | 729 | |
| O.H. 13 | 650 | |
| O.H. 16 | 638 | |
| O.H. 24 | 590 | |
| H. rudolfensis | Mean | 789 |
| KNM-ER 1470 | 752 | |
| KNM-ER 1590 | 825 | |
| KNM-ER 3732 | 750 | |
| H. erectus | Mean | 917 |
| BOU-VP-2/66 | 995 | |
| D2280 | 730 | |
| D2282 | 650 | |
| D2700 | 601 | |
| D3444 | 625 | |
| D4500 | 546 | |
| KNM-ER 3733 | 848 | |
| KNM-ER 3883 | 804 | |
| KNM-ER 42700 | 691 | |
| KNM-WT 15000 | 900 | |
| O.H. 9 | 1,067 | |
| O.H. 12 | 727 | |
| Sangiran 2 | 813 | |
| Sangiran 4 | 908 | |
| Sangiran 17 | 1,004 | |
| Zhoukoudian DI | 915 | |
| Zhoukoudian LI | 1,025 | |
| Zhoukoudian LII | 1,015 | |
| Zhoukoudian LIII | 1,030 | |
| Ngandong 1 | 1,172 | |
| Ngandong 5 | 1,251 | |
| Ngandong 6 | 1,013 | |
| Ngandong 9 | 1,135 | |
| Ngandong 10 | 1,231 | |
| Ngandong 11 | 1,090 | |
| Trinil | 940 |
Additional files
-
Supplementary file 1
Traits of the LES1 cranium in comparison to H. naledi and other hominin species.
- https://doi.org/10.7554/eLife.24232.045
-
Supplementary file 2
Cranial and mandibular measurements.
- https://doi.org/10.7554/eLife.24232.046
-
Supplementary file 3
Postcranial measurements.
- https://doi.org/10.7554/eLife.24232.047
-
Supplementary file 4
Canonical variates analysis of carpal morphology.
- https://doi.org/10.7554/eLife.24232.048
-
Supplementary file 5
Taphonomic observations by specimen from the Lesedi Chamber.
- https://doi.org/10.7554/eLife.24232.049





