Thalamic input to auditory cortex is locally heterogeneous but globally tonotopic
Figures
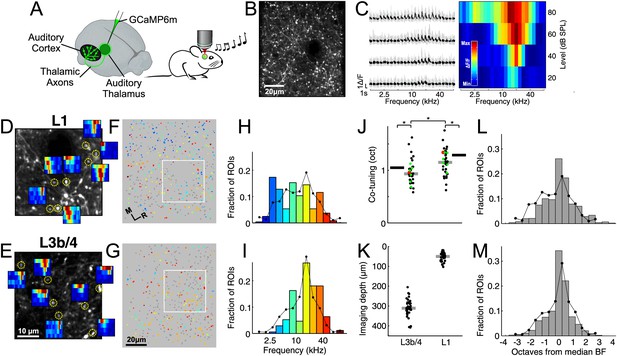
Local heterogeneity of thalamic input.
(A) Experimental schematic. Responses of thalamocortical axons in auditory cortex to pure tones were imaged using two-photon microscopy in anesthetized mice expressing the calcium indicator GCaMP6m in the auditory thalamus. (B) In vivo two-photon image of thalamic axons and axonal boutons in auditory cortex. (C) Left, example fluorescence traces of one bouton in response to randomized pure tones, shown here ordered according to sound frequency and level. Gray traces indicate responses to individual repetitions. Black traces indicate mean responses. Right, frequency response area corresponding to traces on left. Level-averaged tuning curves could generally be well approximated by a Gaussian (Figure 1—figure supplement 1). (D) In vivo two-photon image of thalamic axons and boutons in a small patch of L1 of the auditory cortex. FRAs are shown for several example boutons (locations indicated by yellow circles). (E) Same as D for L3b/4. (F) ROIs corresponding to putative L1 thalamocortical boutons from a single optical plane color-coded according to each bouton's BF. Non-responsive ROIs are shown in dark gray. White rectangle corresponds to area shown in (D). (G) Same as F for region imaged in L3b/4. (H) Distribution of BFs from the L1 region shown in F. Line shows overall BF distribution of all L1 boutons pooled from all imaged regions and animals. (I) Same as H for L3b/4. (J) Co-tuning (standard deviation of BF distribution) within individual regions imaged in L1 (n = 36) and L3b/4 (n = 36). Thick gray lines indicate medians. Thick black lines indicate co-tuning for overall BF distributions (black lines in H,I). Red dots indicate co-tuning of regions shown in F and G. Green dots indicate co-tuning of regions shown in Figure 2C,D. (K) Depth of all regions imaged in L1 and L3b/4. Thick gray lines indicate medians. There was no relationship between imaging depth within L3b/4 and co-tuning (Figure 1—figure supplement 2). (L) Bar graph shows the average of the normalized BF distributions for L1. In order to produce this average distribution the BF distributions of individual regions (such as the one in H) were normalized by setting the median BF to zero before averaging. Line re-plots the overall BF distribution of all pooled L1 boutons shown in H. (M) Same as L for L3b/4.
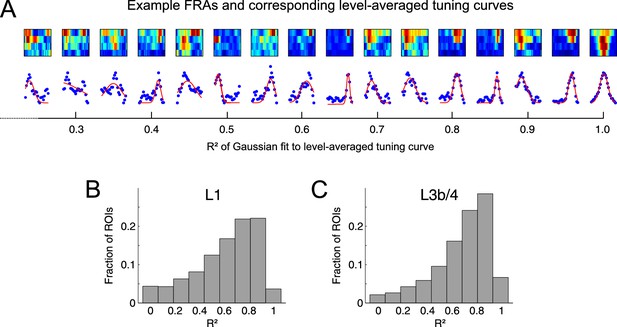
Frequency tuning of thalamocortical boutons.
(A) Top, Example frequency response areas (FRAs) of individual boutons. Bottom, Level-averaged frequency tuning curves (blue dots). In order to assess the quality of frequency tuning we fitted the level-averaged tuning curves with Gaussians (red). The R2 values given below indicate the quality of the fits. (B,C) The tuning curves could generally be well approximated by a Gaussian as shown by the R2 histograms. Median R2 for L1 = 0.67, for L3b/4 = 0.74.
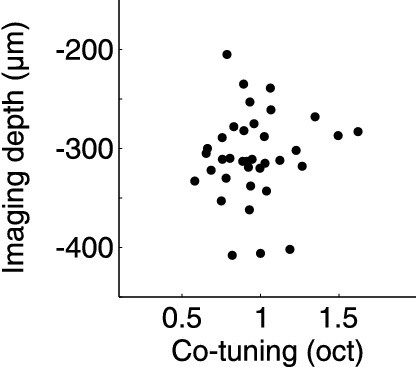
No relationship between imaging depth within L3b/4 and co-tuning.
https://doi.org/10.7554/eLife.25141.004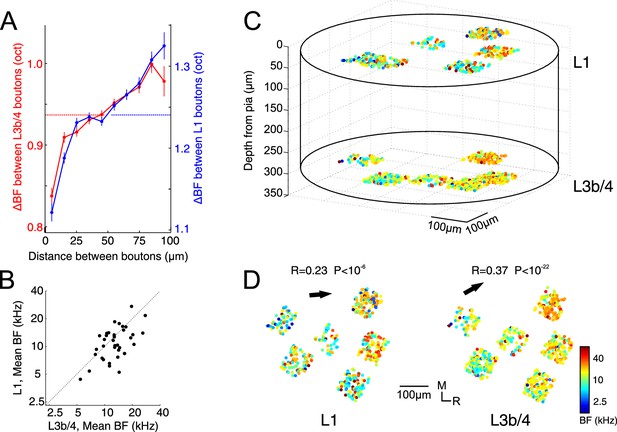
Tonotopic organization of thalamic input to auditory cortex.
(A) Pairwise difference in BF (ΔBF) between boutons as a function of topographic distance in L1 (blue) and L3b/4 (red) for all possible bouton pairs. Number of pairs per 10 µm wide bin is between 2330 and 36725. Horizontal lines indicate average ΔBF across all bouton pairs for L1 (blue) and L3b/4 (red). (B) Mean BF of individual imaged regions in L1 versus mean BF of regions in L3b/4. (C) Relative spatial locations of tone-responsive boutons from several regions in L1 and L3b/4 of the same animal reconstructed in 3D space and color-coded according to each bouton's BF. (D) Top view of the boutons shown in C separated into L1 (left) and L3b/4 (right). Arrows indicate direction of tonotopic axis in L1 and L3b/4.
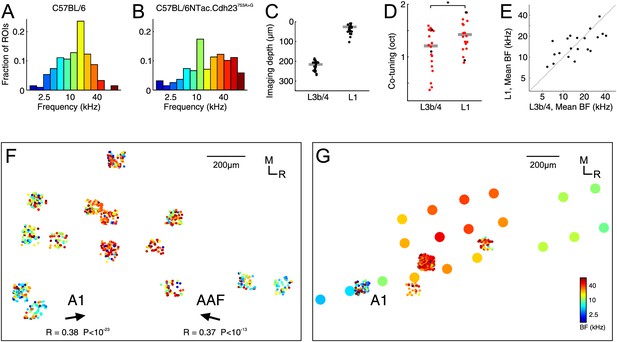
Characterization of thalamic input to the auditory cortex of awake C57BL/6NTac.
Cdh23753A>G mice. (A) Overall BF distribution of all boutons from C57BL/6 mice. (B) Overall BF distribution of all boutons from C57BL/6NTac.Cdh23753A>G mice. (C) Depth of all imaged regions in C57BL/6NTac.Cdh23753A>G mice. (D) Co-tuning (standard deviation of BF distribution) within individual regions imaged in L1 (n = 20) and L3b/4 (n = 20) in C57BL/6NTac.Cdh23753A>G mice. Red dots indicate co-tuning of regions identified to be in a primary auditory field. (E) Mean BF of individual imaged regions in L1 versus mean BF of regions in L3b/4 in C57BL/6NTac.Cdh23753A>G mice. (F) Locations of thalamic boutons from one animal color-coded by BF and collapsed onto the same horizontal plane. Arrows indicate direction of tonotopic axis of boutons deemed to be in A1 (left) and AAF (right). (G) Locations of thalamic boutons (small dots) and multi-unit recordings (large dots) from another animal color-coded by BF and collapsed onto the same horizontal plane. The color of the large dots indicates the mean BF of all tone-responsive multi-units recorded under anesthesia with multi-electrode arrays at the same site following completion of awake imaging.
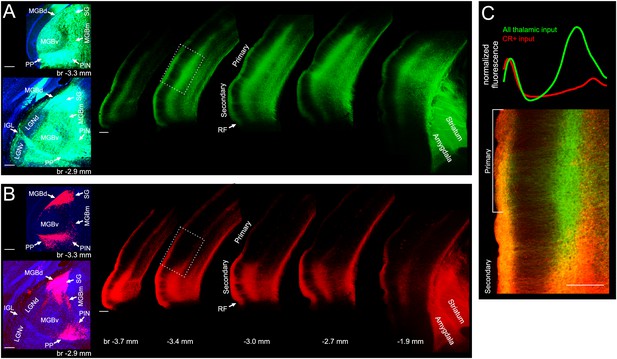
Input from non-lemniscal auditory thalamus targets almost exclusively non-primary auditory cortex.
(A) Left, coronal sections showing GCaMP6m labelled neurons throughout the auditory thalamus of a CR-IRES-cre mouse injected with AAV1.Syn.GCaMP6m.WPRE.SV40 and AAV1.CAG.Flex.tdTomato.WPRE.bGH. Right, coronal sections showing GCaMP6m labelled thalamic axons in auditory cortex, amygdala and striatum. (B) Left, same coronal sections as in left panels of A, showing tdtomato labelled neurons exclusively in non-lemniscal and paralaminar nuclei. Right, same coronal sections as in right panel of A, showing tdtomato labelled thalamic axons almost exclusively in secondary auditory cortex, amygdala and striatum. (C) Bottom, overlay of GCaMP6m (all thalamic input) and tdtomato (CR+ input) labelled axons in transition area from secondary to primary auditory cortex indicated by white rectangle in A and B. Top, cortical depth profile of labelling from non-lemniscal thalamic axons (red, CR+) versus all thalamic axons (green) within primary auditory cortex (average across area within white bracket in bottom panel). The red and green lines were normalized to have same peak height in layer I. MGBd, dorsal division of medial geniculate body; MGBm, medial division of medial geniculate body; MGBv, ventral division of medial geniculate body; SG, suprageniculate nucleus; PIN, posterior intralaminar nucleus; PP, peripeduncular nucleus; LGNd, dorsal division of lateral geniculate nucleus; LGNv, ventral division of lateral geniculate nucleus; IGL, intergeniculate leaf; br, bregma; CR+, calretinin-positive; RF, rhinal fissure. Scale bars, 200 µm. Locations of thalamic subdivisions adopted from Lu et al., 2009. CR+ boutons typically responded very poorly to acoustic stimulation. See Figure 4—figure supplement 1 for In vivo two-photon image of CR+ thalamic axons of CR-IRES-cre mouse injected with AAV1.Syn.Flex.GCaMP6m.WPRE.SV40 and example FRAs.
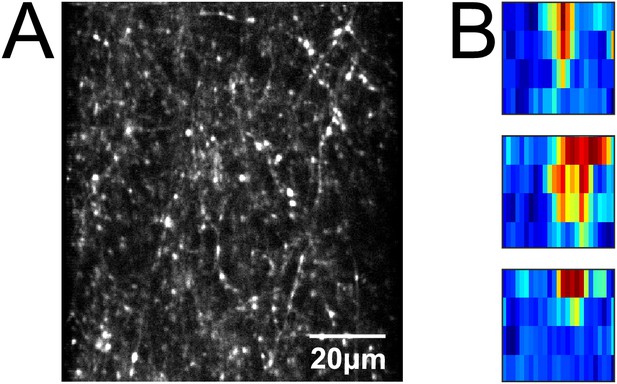
Calcium imaging of CR+ thalamic axons.
(A) In vivo two-photon image of CR+ thalamic axons in A2 of a CR-IRES-cre mouse injected with AAV1.Syn.Flex.GCaMP6m.WPRE.SV40. (B) Example FRAs.
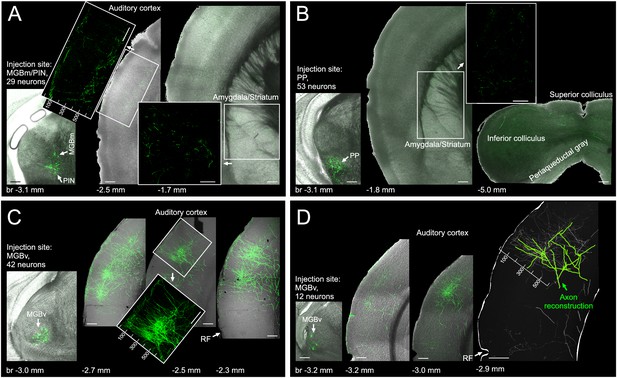
Of the different regions of auditory thalamus, only MGBv provides substantial input to primary auditory cortex.
(A) Left, eGFP labelled neurons (green) in MGBm and PIN after very small injection of highly diluted AAV1.hSyn.Cre.WPRE.hGH and AAV1.CAG.Flex.eGFP.WPRE.bGH. Middle, thalamic axons in auditory cortex. Numbers indicate distance from cortical surface in µm. Right, thalamic axons in amygdala and striatum. (B) Left, eGFP labelled neurons following an injection in PP. Middle/Right, thalamic axons in amygdala, striatum and midbrain. No labelling was found in auditory cortex. (C) Left, eGFP labelled neurons following an injection in MGBv. Right, thalamic axons in auditory cortex. Numbers indicate distance from cortical surface in µm. Total rostrocaudal spread of thalamic axons in cortex exceeded 1 mm. (D) Left, eGFP labelled neurons in MGBv. Middle, thalamic axons in auditory cortex. Total rostrocaudal spread of thalamic axons in cortex exceeded 1 mm. Right, partial reconstruction of a single MGBv axon within a 100 µm thick section of auditory cortex. Numbers indicate distance from cortical surface in µm. Scale bars, 200 µm.
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.25141.010





