Synthetically modified guide RNA and donor DNA are a versatile platform for CRISPR-Cas9 engineering
Figures
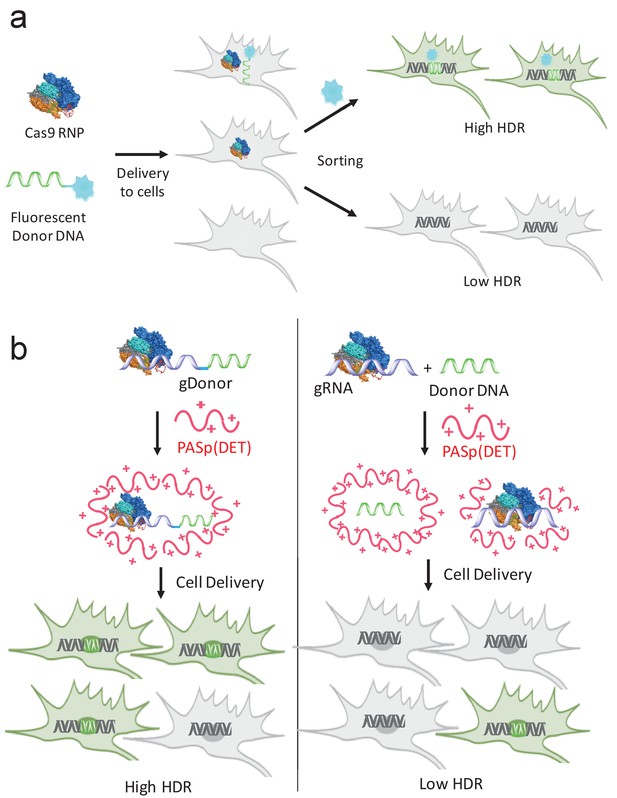
gRNA and donor DNA engineering enables the development of new strategies for enriching gene edited cells and for improving their delivery into cells.
(a) Fluorescently labeled donor DNA can be used as a marker for rapidly enriching HDR edited cells. Cells that internalize fluorescent donor DNA have a high probability of being gene edited via HDR, and can be isolated via FACS based on fluorescence intensity. (b) The gRNA and donor DNA can be conjugated together to generate a single molecule (gDonor). Cas9 complexed to gDonor is more efficient at inducing HDR in cells, after transfection with cationic polymers, than free gRNA complexed to Cas9 and donor DNA. The gDonor/Cas9 complex binds polycations, and the resulting nanoparticles have both gRNA and donor DNA in a single nanoparticle, leading to efficient HDR in cells (left). In contrast, Cas9 RNP + donor DNA forms heterogenous complexes with polycations, which are unable to encapsulate Cas9 RNP and donor within the same nanoparticle, leading to low efficiency HDR in cells (right).
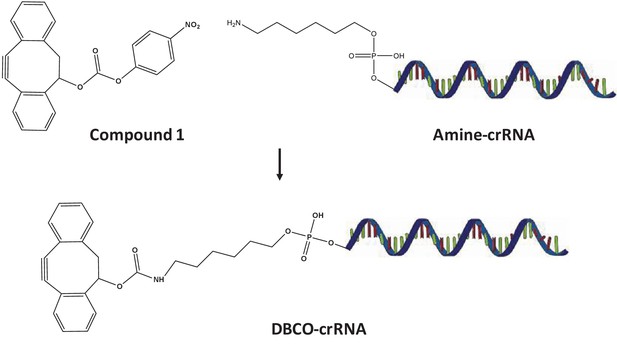
Synthesis of DBCO-crRNA.
https://doi.org/10.7554/eLife.25312.004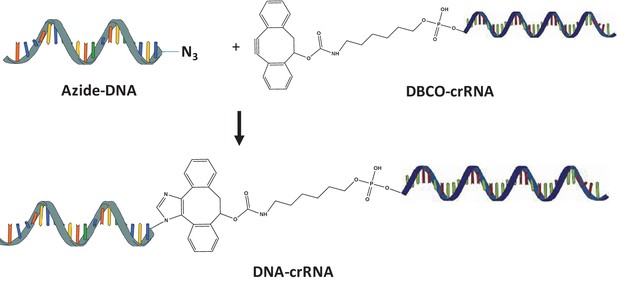
Synthesis of DNA-crRNA.
Azide modified DNA was reacted with DBCO-crRNA via copper free click chemistry.
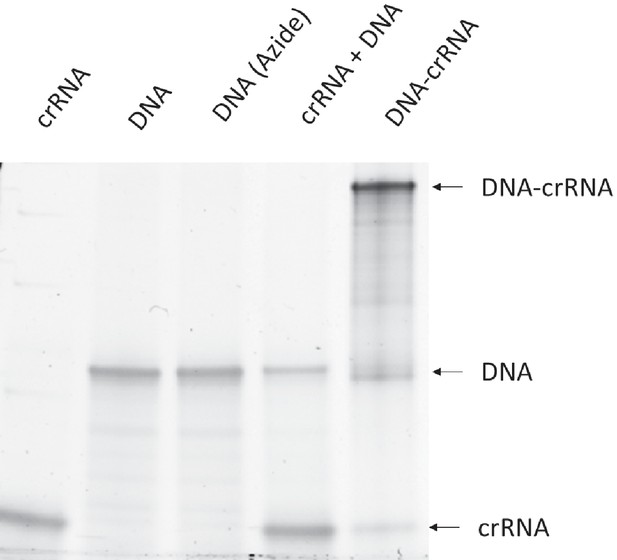
The synthesis of DNA-crRNA was confirmed with gel electrophoresis.
The low molecular weight band is crRNA. The middle band is Donor DNA. The high molecular weight band is DNA-crRNA.
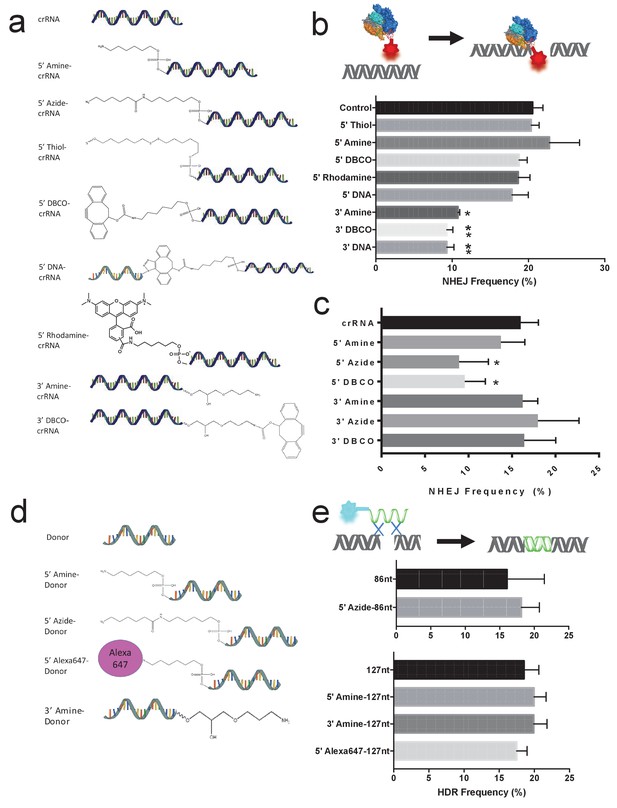
The gRNAs for Cas9 and Cpf1 and donor DNA tolerate large chemical modifications at their terminal ends.
(a) Chemical structure of modified gRNAs. gRNAs with 5’ or 3’ modifications were purchased or synthesized. (b) Cas9 crRNAs with 5’ or 3’ modifications and Cas9 were electroporated into BFP-HEK cells, and their activity was quantified by determining the amount of NHEJ they generated in cells (% BFP negative cells). crRNAs tolerate 5’ modifications but are sensitive to modifications at the 3’ end. DNA-crRNAs are crRNAs conjugated to an 87nt scrambled DNA oligonucleotide. One way ANOVA, post-hoc Tukey test, significant difference from control, *p<0.05, **p<0.01. (c) Cpf1 crRNAs with 5’ or 3’ modifications and Cpf1 were electroporated into BFP-HEK cells, and their ability to generate NHEJ was investigated. One way ANOVA, post-hoc Tukey test, significant difference from control crRNA, *p<0.05. (d) Chemical structures of modified donor DNA. Donor DNA with 5’ or 3’ modifications were purchased or synthesized. (e) Donor DNA with 5’ or 3’ modifications and Cas9 RNP were electroporated into BFP-HEK cells and their ability to induce HDR was investigated. The donor DNA tolerates both 5’ and 3’ modifications.
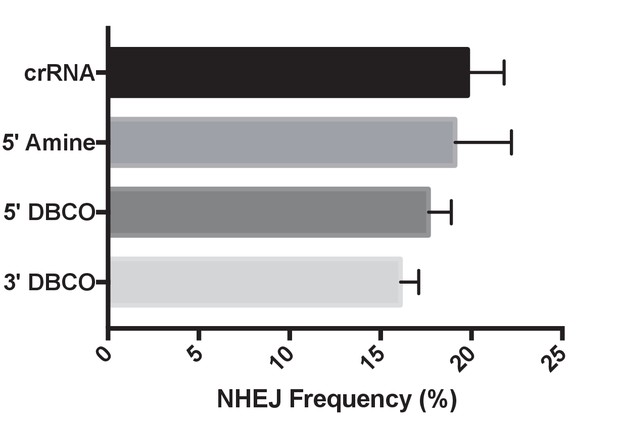
The NHEJ frequency of BFP-K562 cells transfected with chemically modified crRNAs.
The NHEJ frequency was quantified by performing flow cytometry on BFP-K562 cells, 7 days after nucleofection with Cas9 and chemically modified crRNAs/TracrRNA.
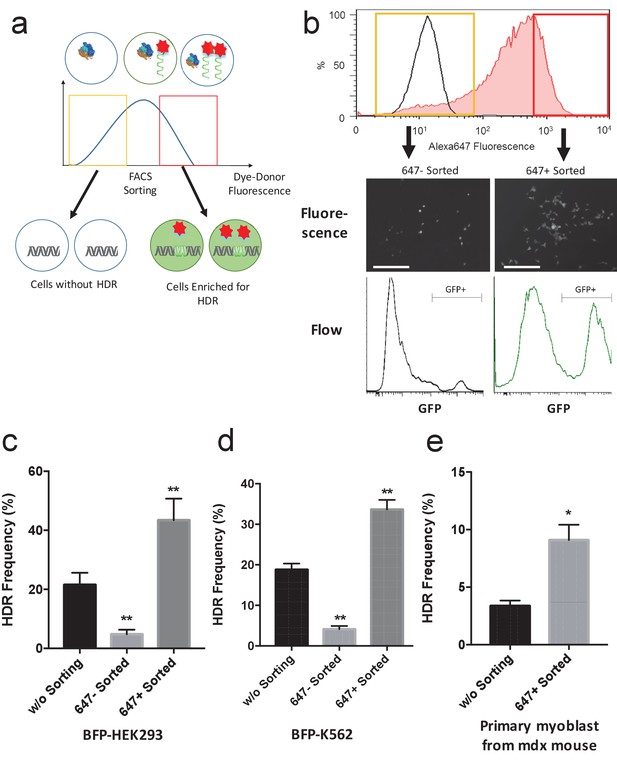
Fluorescently labeled donor DNA can be used to enrich for cells that have been edited via HDR.
(a) Overview of the cell enrichment process. Cells are transfected with Alexa647-donor DNA (tDonor) and Cas9 RNP, and sorted based on their intracellular levels of Alexa647-donor DNA via FACS. Cells with high levels of Alexa647-donor DNA are enriched for HDR. (b) BFP-HEK cells were electroporated with Cas9 RNP and Alexa647-Donor, and were sorted via FACS based on Alexa647 fluorescence. The histogram shows the FACS analysis of Alexa647 fluorescence, control untreated cells are in black, and cells electroporated are in red. The yellow box shows the gating used to identify the Alexa 647 negative cells (bottom 20% gating), and the red box shows the gating used to identify the Alexa 647 positive cells (top 20% gating). Fluorescent images and histograms from flow cytometry of the sorted cells demonstrates that cells with high amounts of Alexa647-Donor had higher levels of HDR (bar: 100 μm). (c) The HDR rate in BFP-HEK cells was determined by quantifying GFP expression. The bulk population of transfected cells (without sorting), cells with low levels of Alexa647-Donor, and cells with high levels of Alexa647-Donor were analyzed by flow cytometry. Alexa647 based sorting enriches for cells that have a high probability of being edited via HDR. One way ANOVA, post-hoc Tukey test, significant difference from control, *p<0.05, **p<0.01. (d) The HDR rate in BFP-K562 cells was determined by quantifying GFP expression. The bulk population of transfected cells (without sorting), cells with low levels of Alexa647-Donor, and cells with high levels of Alexa647-Donor were analyzed by flow cytometry. Alexa647 based sorting enriches for cells that have a high probability of being edited via HDR. One way ANOVA, post-hoc Tukey test, significant difference from control, *p<0.05, **p<0.01. (e) Primary myoblasts from mdx mice were transfected with Cas9 RNP and Alexa647-Donor using lipofectamine, and were sorted via flow cytometry based on Alexa647 fluorescence. The correction of the dystrophin mutation in these cells via HDR was quantified by restriction enzyme analysis of the dystrophin gene. Flow sorted cells that internalized high amounts of the Alexa647 donor had more than a 2-fold increase in HDR frequency than unsorted cells. One way ANOVA, post-hoc Tukey test, significant difference from control, *p<0.05, **p<0.01.

BFP-K562 cells with high levels of fluorescently labeled donor DNA are enriched for HDR edited cells.
BFP-K562 cells were electroporated with Cas9 RNP and Alexa647-Donor, and were sorted via flow cytometry based on the Alexa 647 fluorescence. Cells that internalized high amounts of Alexa 647 donor were enriched for HDR (GFP expressing).
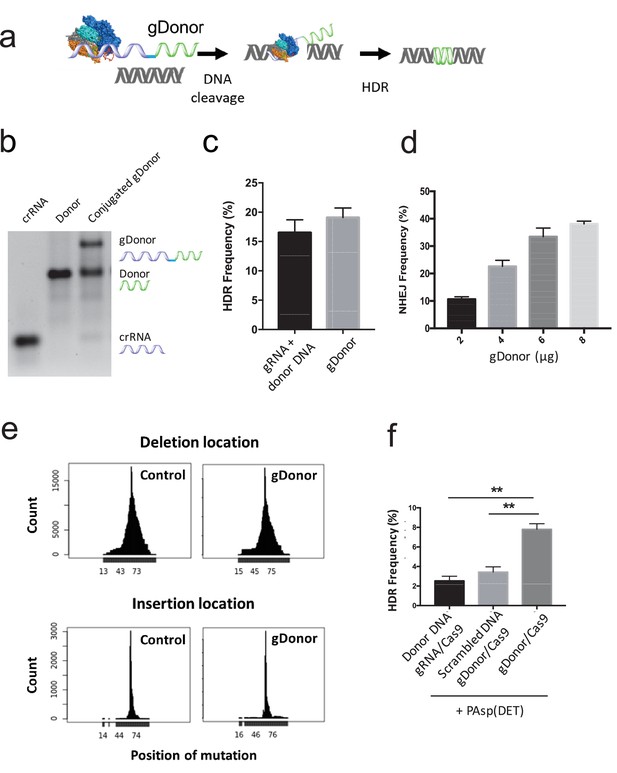
A gRNA-donor DNA conjugate (gDonor) transfects cells with higher efficiency than free gRNA and donor DNA.
(a) The proposed mechanism of gene editing with gDonor/Cas9 complexes in cells. (b) Synthesis of gDonor. gDonor was synthesized via click chemistry and gel analysis confirms the synthesis of gDonor. (c) gDonor efficiently generates NHEJ in BFP-HEK cells after electroporation. The NHEJ frequency depends on the amount of gDonor. (d) gDonor with Cas9 can efficiently induce DNA cleavage and repair via HDR. BFP-HEK cells electroporated with gDonor/Cas9 had a similar HDR frequency as BFP-HEK cells electroporated with Cas9 RNP and donor DNA. (e) gDonor has a similar DNA cleavage pattern in cells as free gRNA and donor DNA (control). Deep sequencing analysis of BFP-HEK cells edited with gDonor/Cas9 and comparison to cells edited with Cas9 RNP and donor DNA (control). Cas9 with gDonor has an almost identical DNA cleavage profile as the unmodified control. The targeted Cas9 cleavage site for these experiments was at 64 locus (position of mutation), which is where most of the mutations were observed. (f) The gDonor/Cas9 complex was delivered into cells with cationic polymers, and the delivery efficiency was compared against cationic polymers complexed to unconjugated gRNA and donor DNA. gDonor/Cas9 complexed to PAsp(DET) was three times more efficient at generating HDR in BFP-HEK cells than PAsp(DET) complexed to Cas9 RNP and donor DNA. An additional control composed of a scrambled DNA conjugated to the gRNA did not increase the transfection efficiency of PAsp(DET). Student-t-test, significant difference from gDonor/Cas9, **p<0.01.
-
Figure 4—source data 1
Raw data from deep sequencing analysis.
Genome DNA of BFP-HEK cells edited with gDonor/Cas9 and compared to cells edited with Cas9 RNP and donor DNA (control) was analyzed.
- https://doi.org/10.7554/eLife.25312.012
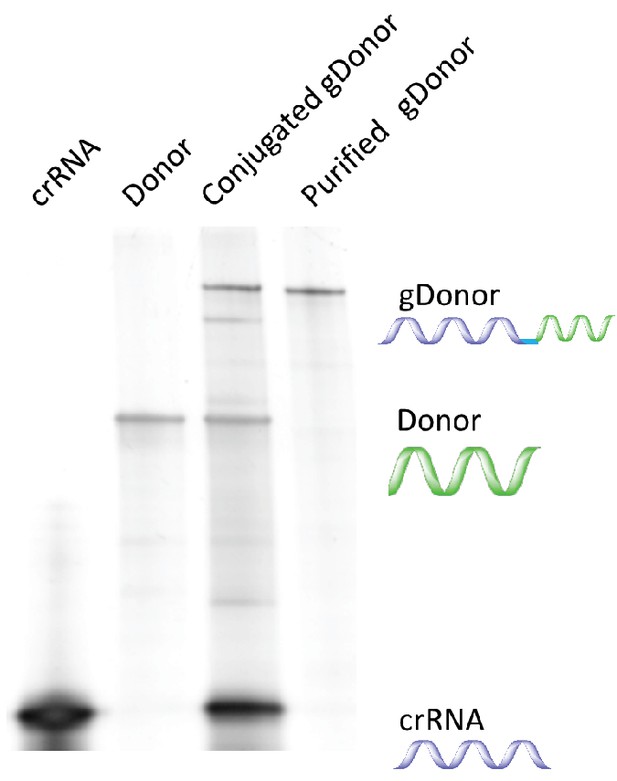
Synthesis and purification of gDonor.
https://doi.org/10.7554/eLife.25312.013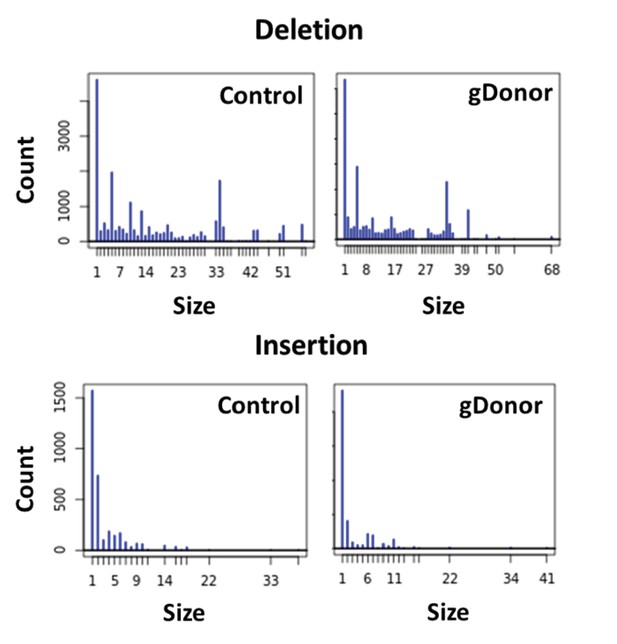
Mutation patterns made by gDonor and Cas9 are similar to that of free gRNA and Cas9.
Deep sequencing analysis was conducted for BFP-HEK cells edited with gDonor/Cas9 and compared to cells edited with Cas9 RNP and donor DNA (control). Top images show deletion size (bp) from gene editing and bottom images show insertion size (bp) from gene editing from both Control and gDonor.
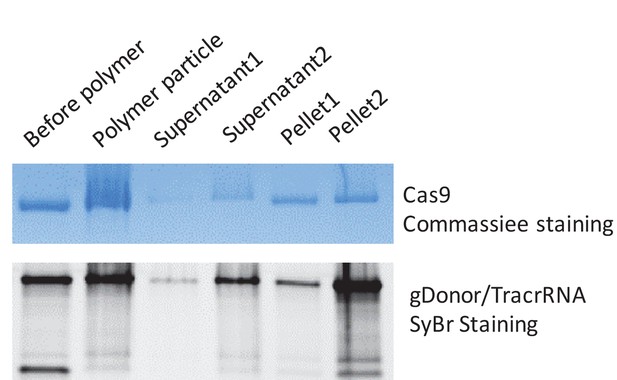
PAsp(DET) polymer nanoparticles complex both gDonor and Cas9.
The gDonor/Cas9 complex was mixed with PAsp(DET) and centrifuged at 17,000 rpm to spin-down polymer nanoparticles that were formed. Gel electrophoresis was performed on the isolated pellets and supernatants; the gels were then stained for either protein or nucleic acids, to determine if the gDonor-Cas9 complex was encapsulated within PAsp(DET) nanoparticles. Samples were prepared in duplicate, and duplicate samples of supernatants and pellets were run on the gel. The PAsp(DET) pellet contains both Cas9 and gDonor, demonstrating efficient complexation. Before polymer: sample without polymer addition, Polymer particle: complete polymer nanoparticle, Supernatant: supernatant collected from the centrifuged polymer nanoparticle, Pellet: pellet collected from the centrifuged polymer nanoparticle. SyBr safe stained gel was imaged first and the same gel was stained with Coomassie blue to visualize Cas9 protein.
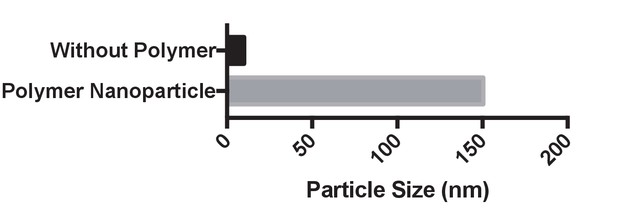
Dynamic light scattering analysis of PAsp(DET) complexes with Cas9 and gDonor.
The particle size was measured after 5 min of incubation and had a size of approximately 150 nm.





