Positional information specifies the site of organ regeneration and not tissue maintenance in planarians
Figures
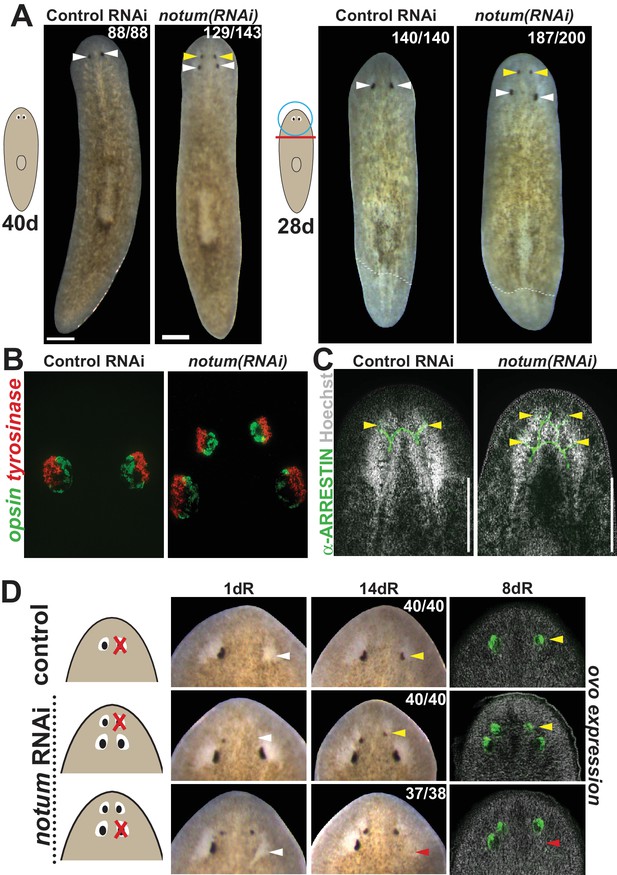
notum RNAi shifts the site of eye regeneration anteriorly.
(A) Animals were treated with notum or control dsRNA every 2–3 days for (top) 40 days in the absence of injury or (bottom) for four times over 9 days followed by decapitation and 28 days of head regeneration as indicated. notum(RNAi) animals produced an anterior set of eyes (129/143 notum(RNAi) homeostasis animals and 187/200 notum(RNAi) regenerating head fragments, yellow arrowheads) and retained a pre-existing set of eyes (white arrowheads). (B) FISH to detect expression of opsin and tyrosinase. (C) anti-ARRESTIN immunostaining to detect photoreceptor neuron axons. (D) Surgical removal of eyes in control and notum(RNAi) animals generated by homeostatic RNAi treatment as in (A), showing individuals at 1 day after surgery to confirm successful removal (white arrowheads) and 14 days to assess regeneration. In notum(RNAi) animals, 40/40 anterior supernumerary eyes regenerated after removal (yellow arrowheads) and 37/38 posterior pre-existing eyes failed to regenerate (red arrowheads). Right, FISH of ovo confirms lack of eye cells produced in the region of the resected notum(RNAi) posterior eyes. Scale bars, 300 microns.
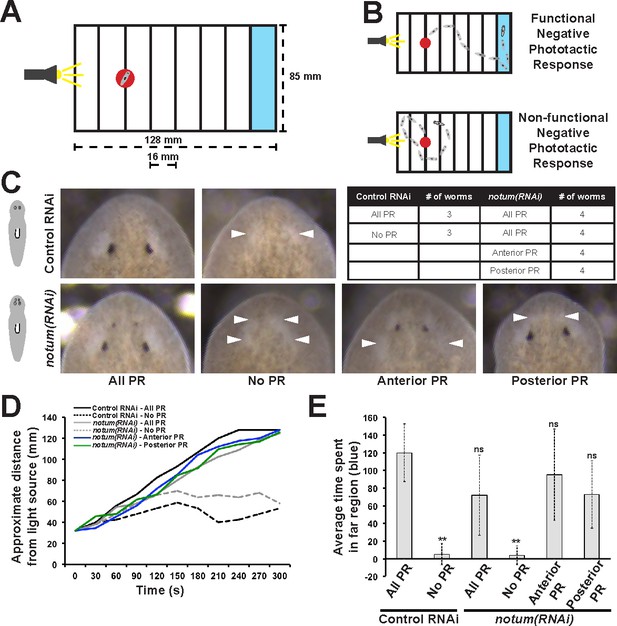
Regenerative and non-regenerative eyes both mediate negative phototaxis.
(A) Phototaxis behavior was measured by measuring the time of transit across an arena illuminated from one side. (B) Illustration of outcomes in the assay. (C) Control and notum(RNAi) animals were examined in phototaxis assays after no treatment, removal of all eyes or removal of only either the supernumerary or pre-existing eyes. (D) Time of transit data for animals after surgeries. Only removal of all eyes in either control or notum(RNAi) animals resulted in lack of negative phototaxis. (E) Quantification of data in D showing average time from the timeseries spent in the blue quadrant (greater than 100 mm from the illuminated side). **p<0.01 by 2-tailed t-test; n.s. denotes p>0.05 from the same test.
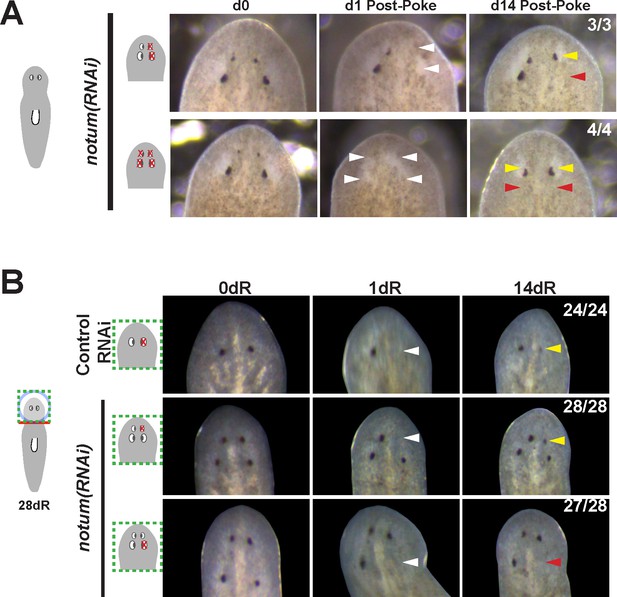
Additional controls for structure and regenerative ability of eyes from notum(RNAi) animals.
(A) Homeostasis notum(RNAi) animals were generated by dsRNA feeding for 40 days followed by surgeries as indicated by cartoons. Removal of both a supernumerary anterior eye and a posterior pre-existing eye resulted in regeneration only of an eye at the anterior eye position. Likewise, removal of all four eyes from such animals resulted in eye regeneration at the anterior position. (B) 4-eyed notum(RNAi) animals were generated by allowing decapitated head fragments to regenerate for 28 days, then tested for eye regeneration behavior. In such animals, the pre-existing eyes fail to regenerate whereas supernumerary eyes have regenerative ability.
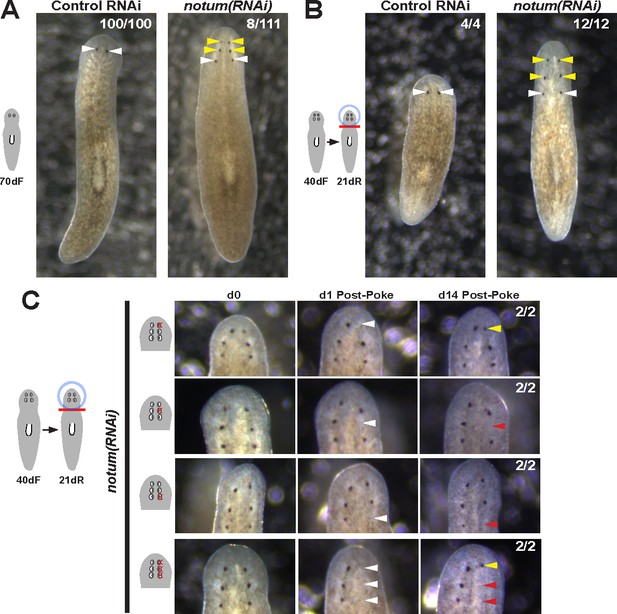
Prolonged notum RNAi and surgical strategies can create additional sets of ectopic eyes that track with regenerative ability.
(A) At a low frequency, notum(RNAi) homeostasis animals form an additional set of ectopic eyes (8/111) animals at 70d RNAi to generate 6-eyed animals. (B) Alternatively, 6-eyed animals can be produced at higher frequency by decapitating 4-eyed notum(RNAi) animals generated by homeostatic inhibition. (C) Experiments to test regenerative ability of the three sets of eyes from 6-eyed notum(RNAi) animals. Only the anterior-most set of eyes can regenerate (top panels vs. middle panels). Removal of a set of three eyes from the same side of these worms results in regeneration only of the anterior-most set of eyes.
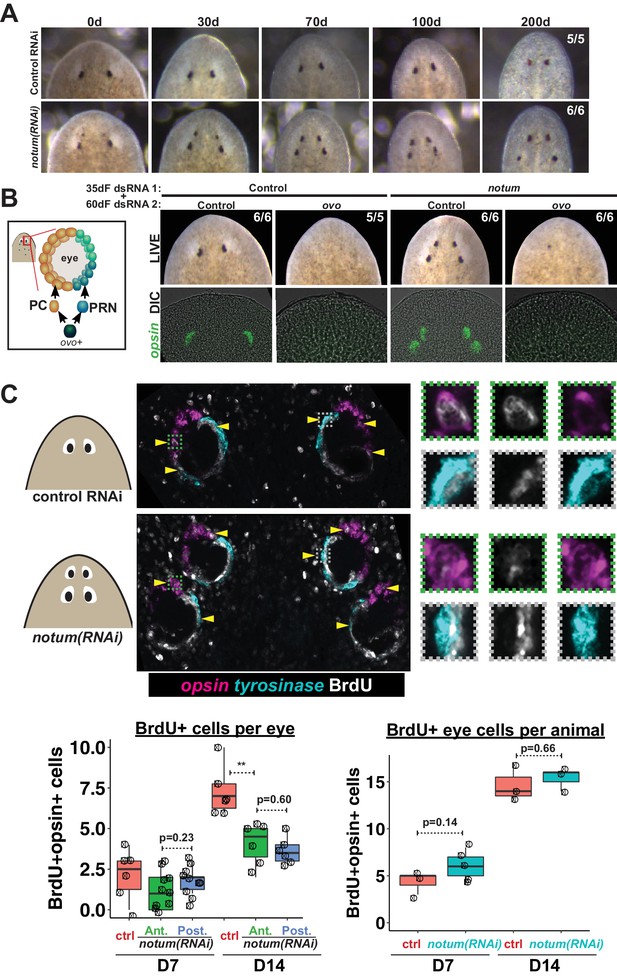
Both regenerative and non-regenerative eyes are homeostatically maintained.
(A) Control and notum(RNAi) animals were fed dsRNA food every three days for 35 days then starved and individually tracked for 200 days and imaged every 30–40 days to monitor stability of the duplicated eyes. (B) Left, cartoon of eye differentiation showing production of photoreceptor neurons (PRN) and pigment cup cells (PC) from ovo+ progenitors. Two-eyed control and four-eyed notum(RNAi) animals were generated by 35 days of dsRNA feeding were then treated with control or ovo dsRNA for 60 days by feeding. ovo inhibition caused loss of both the ectopic and pre-existing eyes of notum(RNAi) animals (12/12 sets of eyes). (C) Two-eyed control and four-eyed notum(RNAi) animals were injected with BrdU following 35 days of RNAi feeding, fixed 14 days later and stained by FISH for opsin (magenta), tyrosinase (cyan) and immunostained with anti-BrdU (gray). The head regions of BrdU-labeled notum(RNAi) animals had BrdU +cells in the anterior eyes (11/12 animals) and the posterior eyes (12/12 animals), a similar frequency as control animal eyes (14/14 animals). C (bottom), quantification of BrdU+ opsin+ cells after 7 or 14 days of BrdU pulsing measured per eye (left) or across all eyes (right) for each condition. p-values from 2-tailed t-tests, **p<0.01. Cartoons depict location of eyes imaged with insets showing single and multichannel enlarged images of BrdU +eye cells.
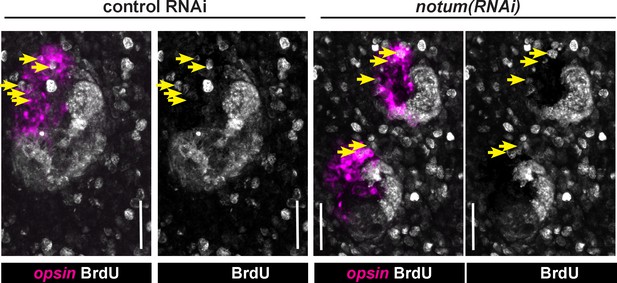
Quantification of BrdU-labeling in notum(RNAi) animals.
Maximum projections of eye cells labeled with opsin and fixed 14 days after BrdU pulsing and quantified in Figure 2C, with double and single channel images indicated along with BrdU +opsin+ cells (yellow arrows). Anterior, top. Bars, 25 microns.
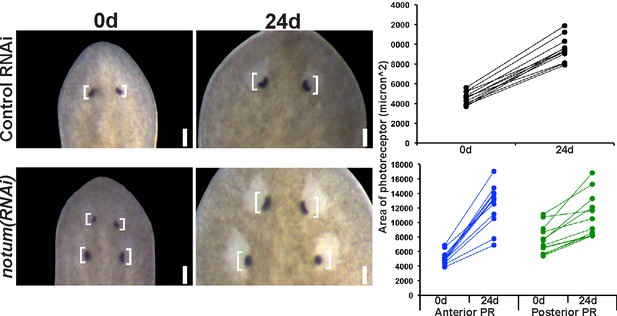
Regenerative and non-regenerative eye sizes respond to growth.
4-eyed notum(RNAi) animals were generated by dsRNA treatment followed by 28 days of head fragment regeneration, imaged (0d) fed dsRNA for 24 days, re-imaged, and the area size of the eye pigment cups measured in microns^2.
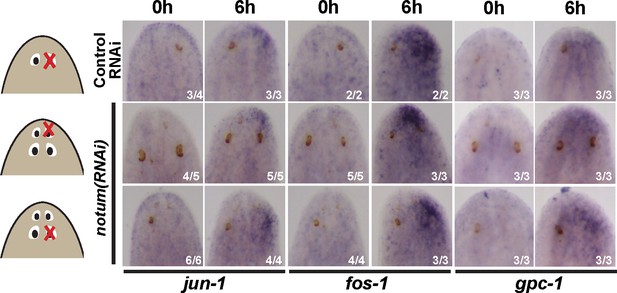
Injury-induced gene expression can occur near non-regenerative eyes.
Control RNAi and 4-eyed notum(RNAi) animals were prepared by homeostatic inhibition for 40 days, resected to remove either normal, anterior or posterior eyes as shown, then fixed at 0 hr or 6 hr and stained for expression of jun-1, fos-1, and gpc-1. Injury-induced gene expression was similar between control and notum(RNAi) animals and between removal of either anterior or posterior eyes.
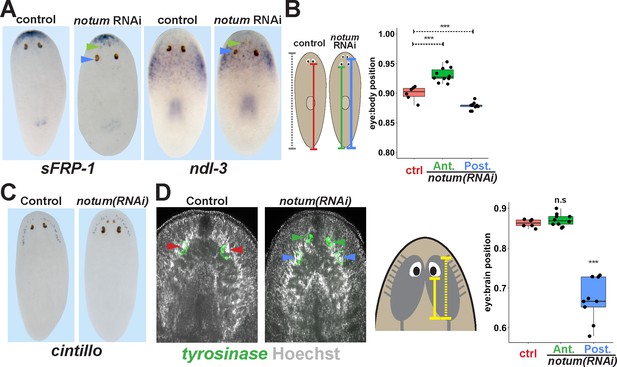
Non-regenerative eyes are mispositioned with respect to positional control genes and the brain.
(A) WISH to detect expression of sFRP-1 and ndl-3 in control and notum(RNAi) regenerating head fragments, marking regenerative eyes (green arrows) and non-regenerative eyes (blue arrows). Posterior eyes in notum(RNAi) animals were located more distantly from the sFRP-1 domain (3/3 animals) and within the ndl-3 expression domain (6/6 animals), whereas eyes from control animals were located outside of the ndl-3 domain (5/5 animals). (B) Measurement of control and notum(RNAi) eyes with respect to the body from fixed stained animals prepared as in (A). In notum(RNAi) animals, the supernumerary eyes are positioned more anterior and the pre-existing eyes are positioned more posterior than eyes from control animals. (C–D) Testing the position of eyes with respect to the brain. (C) Animals were prepared as in (A) and stained with a cintillo riboprobe labeling chemosensory neurons within a lateral territory of the head. The notum(RNAi) posterior eyes are located too far posterior with respect to the cintillo cell domain. (D) Measurement of the location of regenerative and non-regenerative eyes with respect to the brain, as visualized by FISH to detect tyrosinase and Hoechst staining that outlines the planarian cephalic ganglia. Right, quantifications of relative eye:brain position as determined by normalizing to the length of the brain as indicated with respect to the brain’s axis. Non-regenerative eyes from notum(RNAi) animals (blue) have a more posterior location than eyes from control animals (red) or regenerative eyes from notum(RNAi) animals (green). ***, p-value<0.001 by 2-tailed t-test. n.s., p>0.05 by 2-tailed t-test.
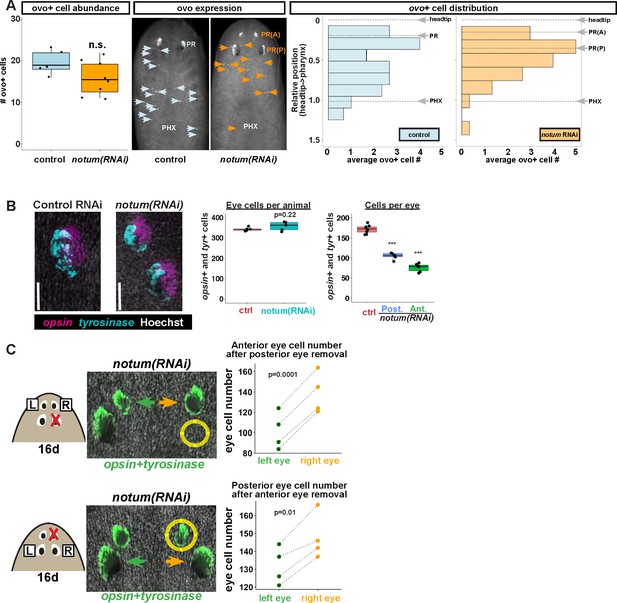
Non-regenerative eyes and regenerative eyes compete for progenitor acquisition.
(A) FISH to detect ovo+ progenitor cells located in the anterior animal region (middle panel, arrows) of control and notum(RNAi) animals. Left plots, ovo+ progenitor cell numbers were not significantly altered in 4-eyed notum(RNAi) animals. Right plots, histograms quantifying distribution of ovo+ eye cells showing regions anterior to the pharynx, with position normalized to the locations of the head tip and the pharynx. notum inhibition produced a slight anterior shift to the distribution of ovo+ cells, but they are present in a region that includes the posterior non-regenerative eyes. (B) FISH with opsin and tyrosinase riboprobes to detect numbers of eye cells from 4-eyed notum(RNAi) animals and 2-eyed control animals (bars, 25 microns). Hoechst counterstaining was used to count numbers of eye cells plotted below as total eye cell numbers per animal and cells per eye. notum RNAi did not significantly change total eye cell numbers, and reduced the number of cells per eye. Significance determined by 2-tailed t-test, ***p<0.001. (C) Four-eyed notum(RNAi) animals were generated by dsRNA feeding over 40 days prior to removal of either a posterior (top) or anterior (bottom) eye on one side of the animal (R,right), leaving both eyes on the left side (L) unaffected. After 16 days of recovery, animals were fixed and stained with a combination of riboprobes for opsin and tyrosinase (green), and eye cells were quantified by counting Hoechst-positive nuclei from opsin/tyrosinase +cells throughout the D/V eye axis. Right, quantifications of left and right eyes from several individuals are shown and connected by dotted lines. Top, removal of a posterior eye caused the ipsilateral anterior eye (orange) to become enlarged compared to the contralateral anterior eye (green). Bottom, removal of an anterior eye caused the ipsilateral posterior eye (orange) to become enlarged compared to the contralateral posterior eye (green). Significance was measured by 2-tailed paired sample t-tests.
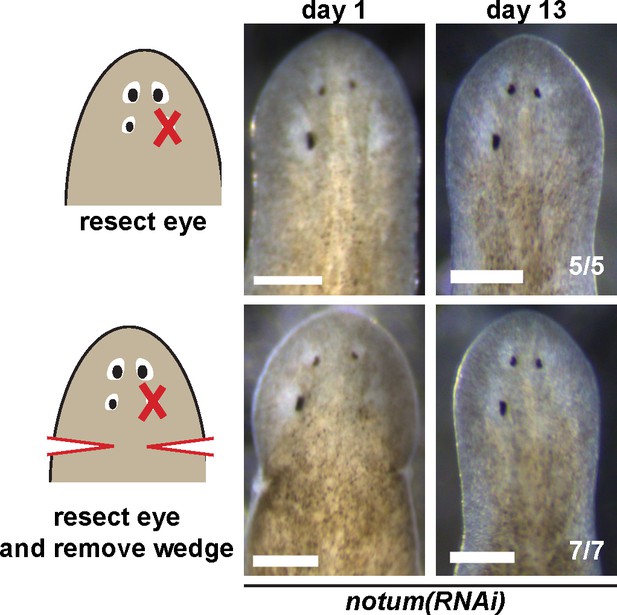
Effect of nearby tissue removal on posterior eye regeneration ability in notum(RNAi) animals.
Four-eyed notum(RNAi) regenerating head fragments obtained 28 days after decapitation were subjected to posterior eye resection with (bottom) or without (top) removal of a wedge of tissue posterior to the eyes. In all cases, animals did not regenerate the resected posterior eye by 14 days after surgery.
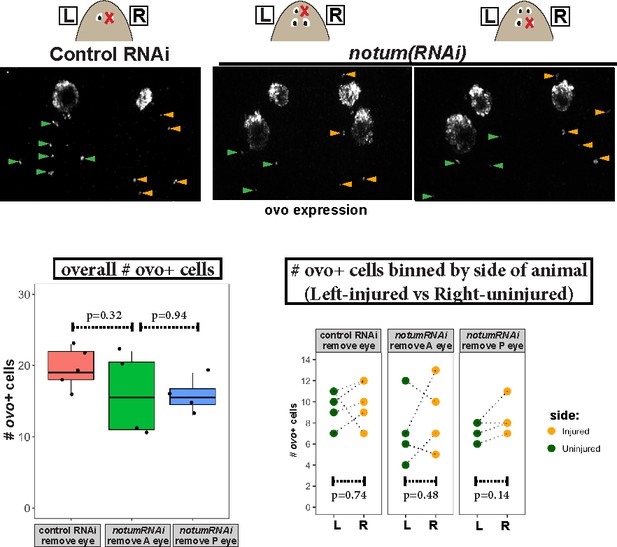
Measurement of ovo+ cell numbers after injury in control and notum(RNAi) animals.
Top, images of animals stained for ovo expression by FISH fixed 4 days after eye removal from control and notum(RNAi) animals as shown in cartoons. The anterior half of each animal was imaged and ovo+ cells manually scored from maximum projection images, scoring eye progenitors as ovo+ cells not residing within the mature eyes. Bottom left, quantification of overall numbers of ovo+ cells animals after each treatment. notum RNAi and either anterior or posterior eye removal did not substantially later numbers of ovo+ cells (p>0.05, 2-tailed t-tests). Bottom right, quantification of ovo+ cells based on localization on the uninjured (left) or injured (right) side of each animal. There was not a difference in number of ovo+ cells between uninjured and injured sides across all treatments (2-tailed paired t-tests).
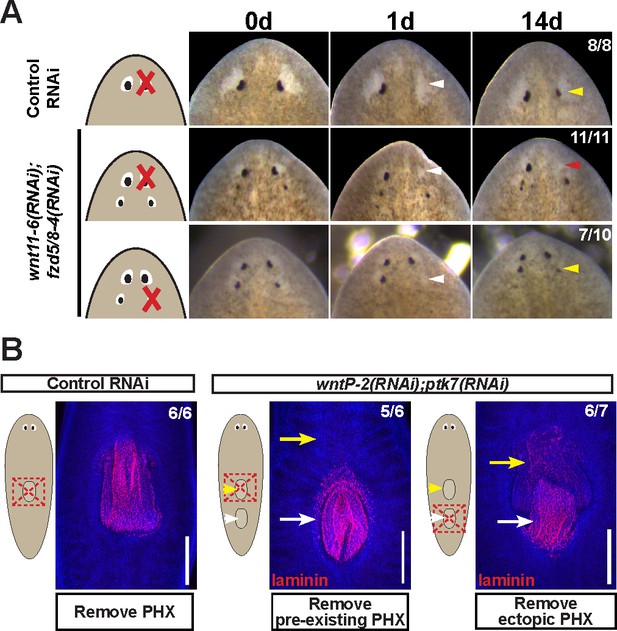
Modulation of other patterning factors alters the sites of eye or pharynx regeneration.
(A) Simultaneous inhibition of wnt11-6 and fzd5/8-4 resulted in the formation of ectopic eyes posterior to the original eyes. Removal of the supernumerary, posterior eyes resulted in regeneration (7/10 animals) whereas removal of the original, anterior eyes did not result in regeneration (11/11 animals), p=0.001 by Fisher’s exact test. (B) wntP-2(RNAi);ptk7(RNAi) animals form a supernumerary posterior pharynx while retaining a pre-existing central pharynx. Cartoons denote amputations used to test regenerative ability of pre-existing or supernumerary pharynges from control or wntP-2(RNAi);ptk7(RNAi) animals. wntP-2(RNAi);ptk7(RNAi) animals were prepared by dsRNA feeding for 3 weeks, then amputated using repeated punctures centrally in a box shape around the target pharynx. Regeneration of the wntP-2(RNAi);ptk7(RNAi) supernumerary posterior pharynx occurred at frequencies close to those of control animal pharynges, but regeneration ability of the wntP-2(RNAi);ptk7(RNAi) pre-existing anterior pharynx was markedly reduced (p=0.03 by Fisher’s exact test).
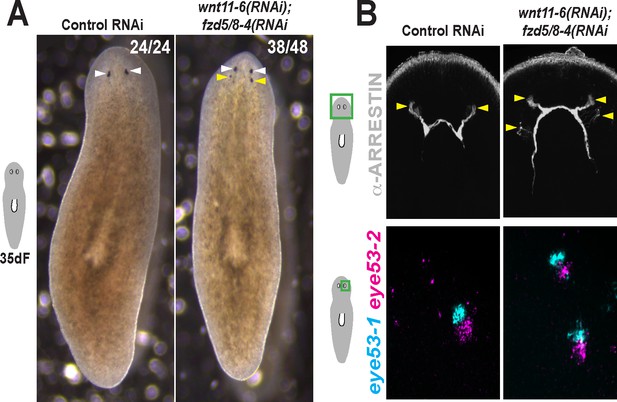
Additional staining and verification of the ectopic posterior eye phenotype of wnt11-6(RNAi);fzd5/8-RNAi(RNAi) animals.
(A) Live images of wnt11-6(RNAi);fzd5/8-RNAi(RNAi) animals after 35 days of RNAi feeding. (B) Images of control and wnt11-6(RNAi);fzd5/8-RNAi(RNAi) animals staining for ARRESTIN protein and eye53-1 and eye53-2 probes.
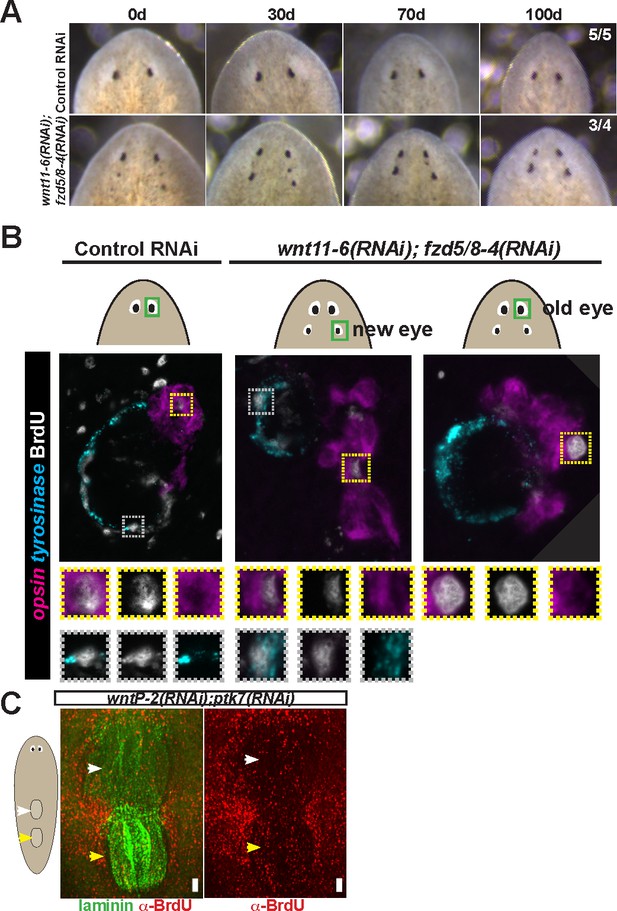
Tests to determine the homeostatic potential of supernumerary eyes and pharynges formed by RNAi of Wnt pathway components.
(A) wnt11-6(RNAi);fzd5/8-4(RNAi) animals with ectopic eyes were generated by dsRNA feeding for 40 days and animals were tracked for a subsequent 100 days after feeding. 3/4 animals maintained two sets of eyes during this time and 1/4 animals maintained three eyes during this time. (B) Four-eyed wnt11-6(RNAi);fzd5/8-4(RNAi) animals were generated as in (B), injected with BrdU then fixed and stained 7 days later with opsin and tyrosinase riboprobes and anti-BrdU antibody. notum(RNAi) animals labeled with BrdU had BrdU +cells in both the supernumerary posterior and pre-existing anterior eyes (5/5 animals), similar to control individuals (5/5 animals). (C) Tests using BrdU to determine homeostatic maintenance ability of new and pre-existing pharynx in wntP-2(RNAi);ptk7(RNAi) animals prepared as in Figure 5C then pulsed with BrdU prior to fixing and staining 7 days later with anti-BrdU antibody and laminin riboprobe that labels pharyngeal tissue. Both pharynges acquired BrdU +cells during the pulse (9/9 animals). Bars, 100 microns.
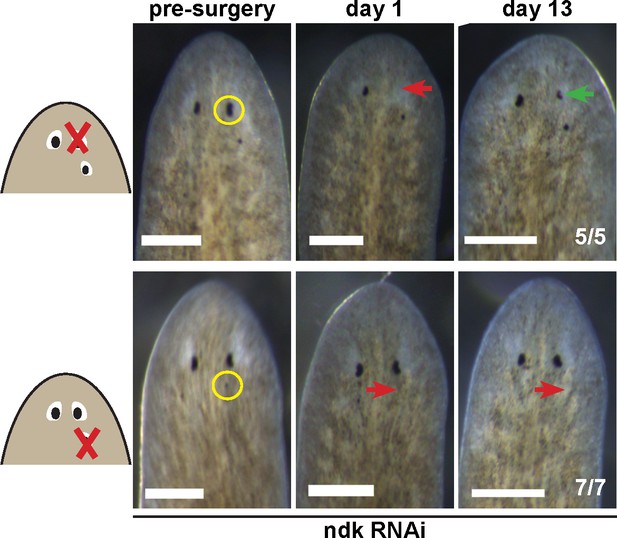
Tests to determine the regenerative potential of eyes in ndk(RNAi) animals.
Animals were fed ndk dsRNA 6 times over 2 weeks then decapitated and regenerating head fragments scored 21 days later for ectopic eyes (15/31 animals). Animals displaying this phenotype were selected for eye resection to remove either an original anterior eye or a supernumerary posterior eye. Removal of the anterior eye resulted in regeneration (5/5 animals), while regeneration was not observed after removal of posterior eyes (7/7) as scored 14 and 21 days later.
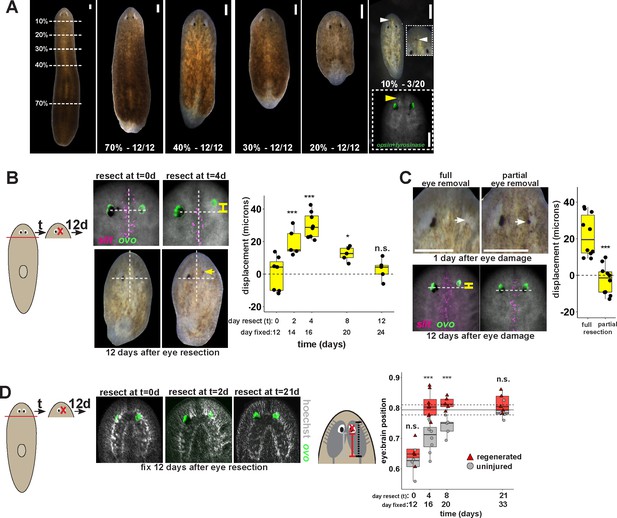
Tissue remodeling normally shifts the site of eye regeneration away from pre-existing eyes.
(A) Large animals were decapitated in a series of AP positions denoted by approximate percentage of anterior tissue remaining. Such fragments regenerate into small animals that ultimately regain proportionality, and the majority of fragments had a single set of eyes throughout this tissue remodeling process (65/68 animals). However, fragments resulting from far-anterior amputations occasionally formed an ectopic set of photoreceptors during regeneration (3/20 animals). (B) Large animals were decapitated to remove ~80% of the posterior and one of the eyes within the regenerating head fragments was resected in a timeseries. Animals were fixed 12 days after eye resection and stained with an ovo riboprobe to mark the site of eye regeneration, using midline expression of slit and the A/P position of the contralateral uninjured eye as a reference (dotted lines). Right, displacement from the reference position was modified by the time of eye resection as head fragments underwent remodeling. Maximal displacement from the location of the pre-existing eye occurred when resecting eyes from d4 regenerating head fragments. (C) Tests to determine whether eye damage or eye removal is necessary for revealing the altered location of regeneration. One eye from d4 regenerating head fragments was either fully removed (left) as in (B), or only damaged to partially resect it (right). Top panels show live animals 1 day after surgery indicating successful removal versus damage to the right eye. Bottom panels show animals fixed 12 days after eye removal or damage stained and quantified for eye displacement as in (C). Only complete eye removal caused eye regeneration at an anteriorly shifted site. (D). The position of eyes from animals treated as in (B) were measured with respect to the A/P brain axis as determined by Hoechst and ovo staining. Images are projections of optical sections taken from a mid-ventral position to highlight the cephalic ganglia and dorsal positions to highlight the location of the eye. The eye:brain ratio was calculated as in Figure 3D by measuring the eye’s distance to the posterior edge of the cephalic ganglia and normalizing to the length of the brain, with uninjured animals used to determine average eye:brain ratio at ideal proportions (solid line with dotted lines indicating standard error). Uninjured eyes successively regain proper position with respect to the brain axis as remodeling and regeneration proceed. Eye removal during this process results in eye regeneration at a more anteriorly displaced location that corresponds with the proper position with respect to the brain. Scale bars, 300 microns.
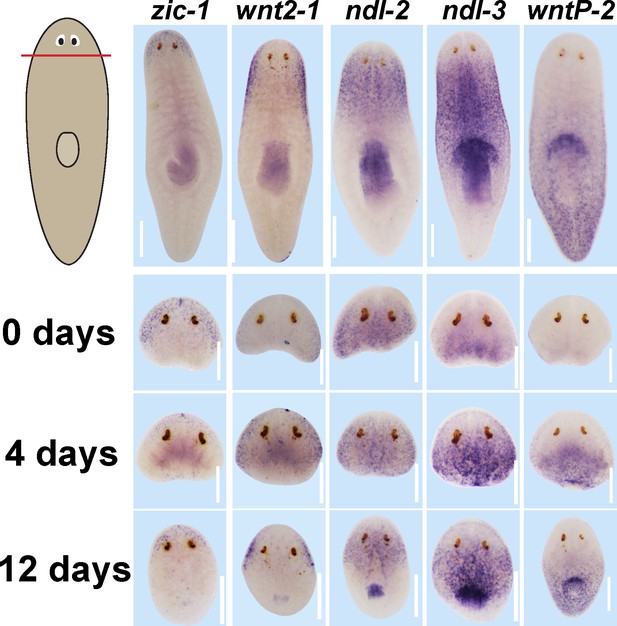
Expression of positional control genes is modified early during remodeling.
WISH to detect expression of five different positional control genes in a timeseries during the regeneration of head fragments (zic-1, wnt2-1, ndl-2, ndl-3 and wntP-2). At d4 of regeneration, positional control gene expression domains have altered but not yet acquired their final distributions. For example, zic-1 expression appears overly reduced compared to 12 days of regeneration, and wnt2-1, ndl-2, and ndl-3 expression occupies too much of the axis, and the wntP-2 expression axis has not yet resolved. These observations suggest that early in regeneration, positional control genes are mispositioned with respect to pre-existing tissues. All images representative of at least n = 4 animals per timepoint and condition. Bars, 300 microns.
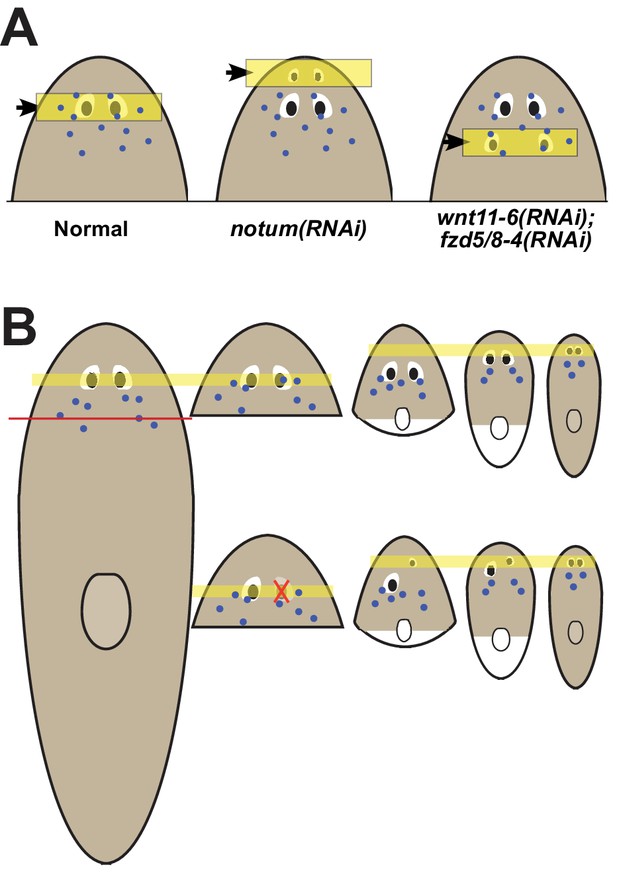
Pattern alteration uncouples the sites of regeneration and homeostasis.
(A) Model showing shifts to the anteroposterior target site of eye regeneration (yellow box) in animals undergoing notum RNAi or wnt11-6 and fzd5/8-4 RNAi. Eye progenitors (purple dots) are present in a broader anterior domain and can renew pre-existing eyes left behind by the pattern alteration. (B) Shifts to the location of eye regeneration during the remodeling of head fragments (top series). Eye removal during this process results in eye regeneration at the target location for proportion re-establishment (bottom series).
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.33680.021





