Synapse-specific and compartmentalized expression of presynaptic homeostatic potentiation
Figures
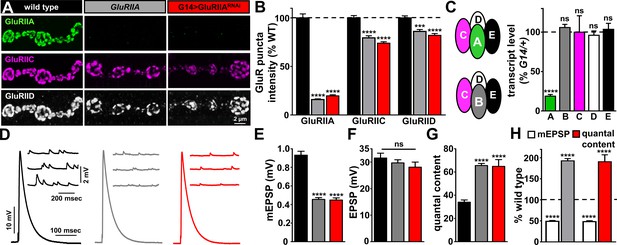
GluRIIA knock down phenocopies GluRIIA mutants.
(A) Representative images of NMJs on muscle six immunostained with antibodies that recognize GluRIIA, GluRIIC, and GluRIID receptor subunits in wild type (w1118), GluRIIA mutants (w1118;GluRIIASP16), and G14 > GluRIIARNAi (w1118;G14-Gal4/+;UAS-GluRIIARNAi/+). (B) Quantification of the mean fluorescence intensity of individual GluR puncta reveals GluRIIA subunits are virtually undetectable at NMJs of both GluRIIA mutants and G14 > GluRIIARNAi, while the essential subunits GluRIIC and GluRIID are moderately reduced, reflecting expression of the remaining GluRIIB-containing receptors. (C) Left: Schematic illustrating the composition of GluRIIA-containing and GluRIIB-containing postsynaptic receptor subtypes at the Drosophila NMJ. Right: Quantitative PCR analysis of GluR transcript levels for GluRIIA/B/C/D/E subunits in G14 >GluRIIARNAi normalized to G14/+. (D) Representative electrophysiological traces of EPSP and mEPSP recordings in the indicated genotypes. (E–G) Quantification of mEPSP amplitude (E), EPSP amplitude (F), and quantal content (G) in the indicated genotypes. Note that while mEPSP amplitudes are reduced to similar levels in GluRIIA mutants and G14 > GluRIIARNAi, EPSP amplitudes remain similar to wild type because of a homeostatic increase in presynaptic glutamate release (quantal content). (H) Quantification of mEPSP amplitude and quantal content values of the indicated genotypes normalized to wild-type values. Error bars indicate ± SEM. Asterisks indicate statistical significance using one-way analysis of variance (ANOVA), followed by Tukey’s multiple-comparison test: (****) p<0.0001, (ns) not significant. Detailed statistical information for represented data (mean values, SEM, n, p) is shown in Supplementary file 1.
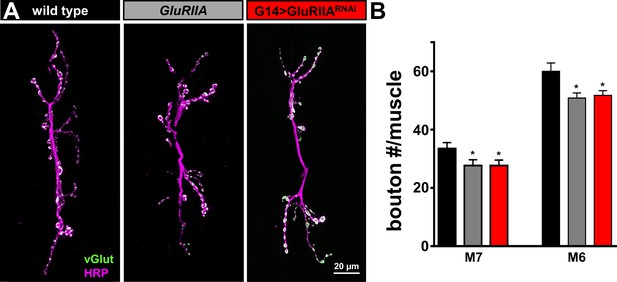
Synaptic growth is mildly reduced at NMJs of GluRIIA mutants and G14 > GluRIIARNAi.
(A) Representative images of muscle 6/7 NMJs of wild type, GluRIIA mutants, and G14 > GluRIIARNAi immunostained with antibodies against with the neuronal membrane (HRP) and the synaptic vesicle marker vGlut. (B) Quantification of bouton number per muscle segment in the indicated genotypes demonstrates that synaptic growth is reduced by ~18% in both GluRIIA mutants and G14 > GluRIIARNAi. Error bars indicate ± SEM. Asterisks indicate statistical significance using one-way analysis of variance (ANOVA), followed by Tukey’s multiple-comparison test: (*) p<0.05. Detailed statistical information for represented data (mean values, SEM, n, p) is shown in Supplementary file 1.
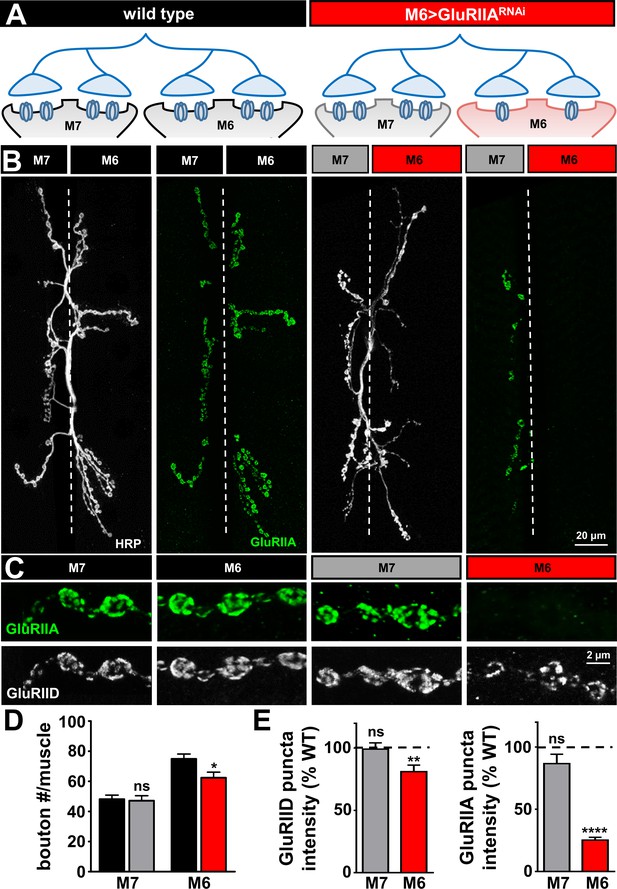
GluRIIA-containing receptors can be knocked down specifically on muscle six using M6 >Gal4.
(A) Schematic of the Type Ib motor neuron innervating both muscles 6 and 7 at the Drosophila NMJ. Gal4 is expressed specifically on muscle six in combination with UAS-GluRIIARNAi using M6 > Gal4 (M6 > GluRIIARNAi: w1118;Tub-FRT-STOP-FRT-Gal4, UAS-FLP, UAS-CD8-GFP/+; H94-Gal4, nSyb-Gal80/UAS-GluRIIARNAi). GluRIIA expression is reduced on muscle 6, while expression of GluRIIA on muscle 7 is unperturbed. (B) Representative images of muscle 6/7 NMJs immunostained with antibodies that recognize the neuronal membrane (HRP) and the postsynaptic GluR subunit GluRIIA in wild type and M6 > GluRIIARNAi. (C) Representative images of individual Ib synaptic boutons immunostained with anti-GluRIIA and anti-GluRIID on the indicated muscles in wild type and M6 > GluRIIARNAi. (D) Quantification of bouton numbers on muscles 6 and 7 in wild type and M6 > GluRIIARNAi. (E) Quantification of mean fluorescence intensity of GluRIIA and GluRIID puncta normalized to wild type on the indicated muscles. Asterisks indicate statistical significance using a Student’s t test: (*) p<0.05; (**) p<0.01; (****) p<0.0001, (ns) not significant. Error bars indicate ± SEM. Detailed statistical information for represented data (mean values, SEM, (n, p) is shown in Supplementary file 1.
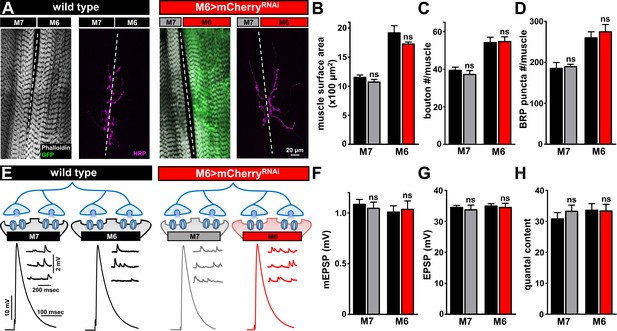
Biased Gal-4 expression on muscle six does not alter synaptic growth or function.
(A) Representative images of muscle 6/7 NMJs of wild type and M6 > mCherryRNAi (w1118;Tub-FRT-STOP-FRT-Gal4, UAS-FLP, UAS-CD8-GFP/+; H94-Gal4, nSyb-Gal80/UAS-mCherryRNAi) labeled with phalloidin (actin marker), GFP, and the neuronal membrane marker HRP. (B–D) Quantification of muscle surface area (B), bouton number (C), and BRP puncta number per NMJ (D). No significant differences in any of these parameters were observed. (E) Schematic and electrophysiological traces of recordings from wild type and M6 >mCherryRNAi NMJs. (F–H) Quantification of mEPSP amplitude (F), EPSP amplitude (G), and quantal content (H) from the indicated genotypes finds no significant difference in synaptic physiology. Error bars indicate ± SEM. Asterisks indicate statistical significance using a Student’s t test: (ns) not significant. Detailed statistical information for represented data (mean values, SEM, n, p) is shown in Supplementary file 1.
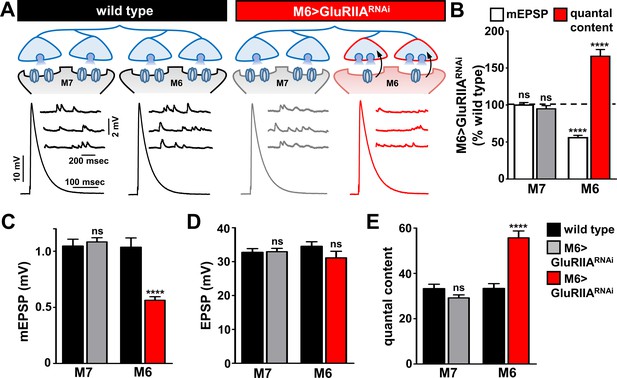
Presynaptic homeostatic potentiation can be induced and expressed exclusively at synapses innervating muscle 6.
(A) Schematic and electrophysiological traces of recordings from muscles 7 and 6 of wild type and M6 > GluRIIARNAi NMJs. Note that mEPSPs are reduced only on muscle 6 of M6 > GluRIIARNAi, while EPSP amplitudes are similar across all muscles. Thus, the expression of PHP is restricted to synapses innervating muscle 6 of M6 > GluRIIARNAi and does not impact neurotransmission at neighboring synapses within the same motor neuron. (B) Quantification of mEPSP amplitude and quantal content values of M6 > GluRIIARNAi normalized to wild-type values. (C–E) Quantification of mEPSP amplitude (C), EPSP amplitude (D), and quantal content (E) in the indicated muscles and genotypes. Asterisks indicate statistical significance using a Student’s t test: (****) p<0.0001, (ns) not significant. Error bars indicate ± SEM. Detailed statistical information for represented data (mean values, SEM, (n, p) is shown in Supplementary file 1.
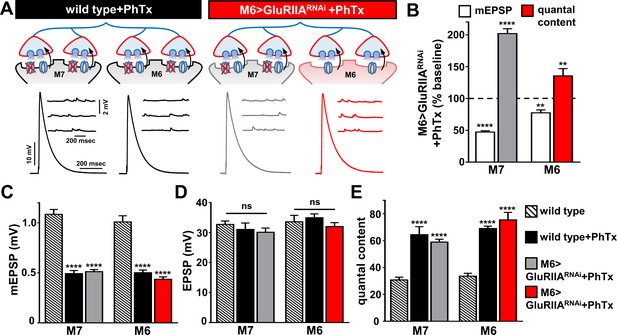
PHP can be induced and expressed acutely on muscle 7 following chronic GluRIIA knock down on M6.
(A) Schematic and representative traces illustrating the acute application of PhTx on wild type and M6 >GluRIIARNAi NMJs. The acute expression of PHP was observed on previously non-potentiated muscle 7 synapses in M6 >GluRIIARNAi NMJs. mEPSP amplitudes are diminished at wild type and M6 >GluRIIARNAi NMJs on both muscles following PhTx application, while EPSP amplitudes are maintained at baseline levels due to a homeostatic increase in presynaptic neurotransmitter release. (B) Quantification of mEPSP and quantal content values normalized to baseline values (-PhTx) at muscles 6 and 7 NMJs of M6 >GluRIIARNAi larvae. (C–E) Quantification of mEPSP amplitude (C), EPSP amplitude (D), and quantal content (E) values in the indicated genotypes. Asterisks indicate statistical significance using a Student’s t test and one-way analysis of variance (ANOVA), followed by Tukey’s multiple-comparison test: (**) p<0.01, (****) p<0.001, (ns) not significant. Error bars indicate ± SEM. Detailed statistical information for represented data (mean values, SEM, (n, p) is shown in Supplementary file 1.
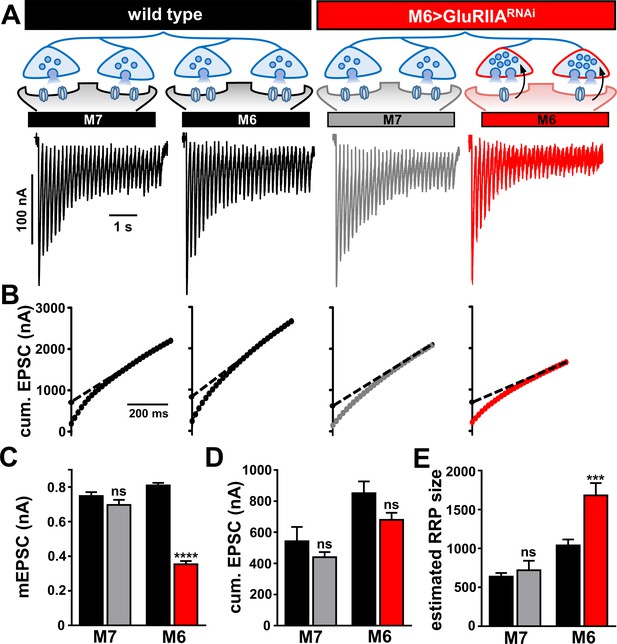
Homeostatic modulation of the readily releasable vesicle pool is restricted to synapses innervating muscle 6 at M6 >GluRIIARNAi NMJs.
(A) Schematic illustrating the size of the RRP is enhanced specifically at the terminals innervating muscle six in M6 > GluRIIARNAi. Representative traces of two-electrode voltage clamp recordings (30 stimuli at 60 Hz in 3 mM extracellular Ca2+) from muscles 7 or 6 in the indicated genotypes. (B) Averaged cumulative EPSC amplitude plotted as a function of time. A line fit to the 18-30th stimuli was back-extrapolated to time 0. (C–E) Quantification of mEPSC amplitude (C), average cumulative EPSC values (D), and estimated readily releasable pool (RRP) sizes (E) for the indicated muscles and genotypes. Note that RRP size is significantly increased at muscle 6 NMJs, but no change is observed at muscle 7. Error bars indicate ± SEM. Asterisks indicate statistical significance using a Student’s t test: (*) p<0.05; (***) p<0.001; (ns) not significant. Detailed statistical information for represented data (mean values, SEM, n, p) is shown in Supplementary file 1.
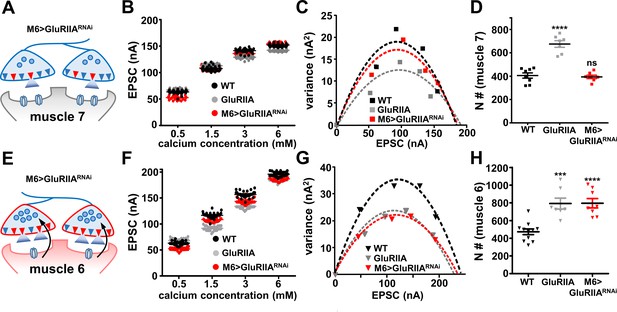
The number of functional release sites are specifically enhanced at synapses innervating muscle 6 at M6 >GluRIIARNAi NMJs.
(A) Schematic illustrating the number of functional release sites (marked as red triangles) at synapses innervating muscle 7 of M6 > GluRIIARNAi NMJs. (B) Scatter plot EPSC distribution of recordings derived from muscle 7 of wild type, GluRIIA mutants, and M6 > GluRIIARNAi in the indicated extracellular Ca2+ concentrations. (C) Variance-mean plots for the indicated genotypes. Variance was plotted against the mean amplitude of 30 EPSCs recorded at 0.2 Hz from the four Ca2+ concentrations detailed in (B). Dashed lines are the best fit parabolas to the data points. (D) Estimated number of functional release sites (N) obtained from the variance-mean plots in (C). Note that the number of functional release sites are enhanced only at muscle 7 NMJs of GluRIIA mutants. (E) Schematic illustrating the number of functional release sites (marked as red triangles) at synapses innervating muscle 6 of M6 > GluRIIARNAi NMJs. (F) Scatter plot EPSC distribution of recordings derived from muscle 6 of wild type, GluRIIA mutants, and M6 > GluRIIARNAi in the indicated extracellular Ca2+ concentrations. (G) Variance-mean plots for the indicated genotypes. (H) Estimated number of functional release sites (N) obtained from the variance-mean plots in (G). Both GluRIIA mutants and M6 > GluRIIARNAi showed a significant enhancement of functional release site number at muscle 6 NMJs compared to wild type. Error bars indicate ± SEM. Asterisks indicate statistical significance using one-way analysis of variance (ANOVA), followed by Tukey’s multiple-comparison test: (***) p<0.001; (****) p<0.0001; (ns) not significant. Detailed statistical information for represented data (mean values, SEM, (n, p) is shown in Supplementary file 1.
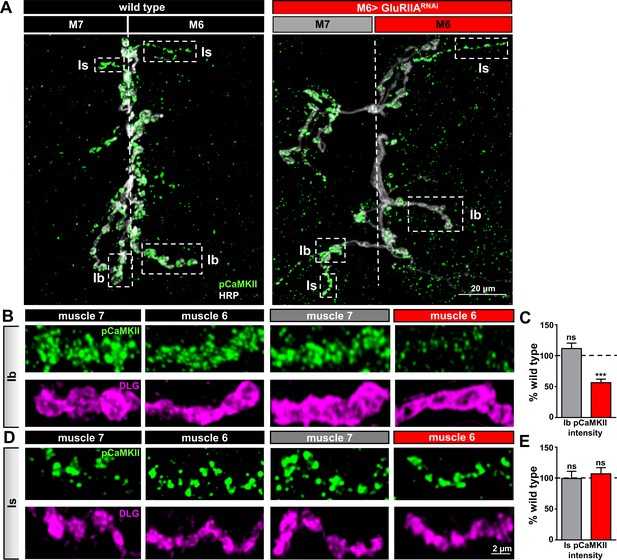
Compartmentalized changes in CaMKII activity at postsynaptic densities of M6 > GluRIIARNAi NMJs.
(A) Representative images of muscle 6/7 NMJs from wild type and M6 > GluRIIARNAi immunostained with antibodies against with the active (phosphorylated) form of CaMKII (pCaMKII) and the presynaptic neuronal membrane marker HRP. Note that pCaMKII intensity is specifically reduced at Ib postsynaptic densities on muscle 6 of M6 >GluRIIARNAi (dashed areas). (B,C) Images (B) and quantification (C) of pCaMKII fluorescence intensity at Ib boutons on muscles 7 and 6 normalized to wild-type values. A reduction in pCaMKII intensity is observed on muscle 6 of M6 > GluRIIARNAi, while no significant change is observed at synapses innervating the adjacent muscle 7. (D,E) Images (D) and quantification (E) of pCaMKII intensity at Is boutons shows no significant difference between wild type and M6 > GluRIIARNAi at NMJs of either muscle. Asterisks indicate statistical significance using a Student’s t test: (***) p<0.001, (ns) not significant. Error bars indicate ± SEM. Detailed statistical information for represented data (mean values, SEM, (n, p) is shown in Supplementary file 1.
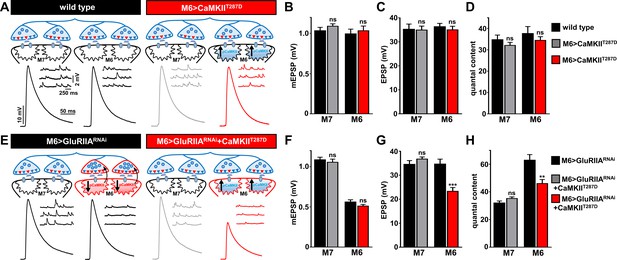
Overexpression of constitutively active CaMKII inhibits retrograde PHP signaling at M6 > GluRIIARNAi NMJs.
(A) Schematic and representative traces illustrating that overexpression of a constitutively active form of CaMKII only on muscle 6 (M6>CaMKIIT287D) has no significant impact on neurotransmission. (B–D) Quantification of mEPSP amplitude (B), EPSP amplitude (C), and quantal content (D) in the indicated muscles. (E) Schematic and representative traces illustrating that expression of constitutively active CaMKII with GluRIIA knock down (M6 > GluRIIARNAi + CaMKIIT287D) on muscle 6 inhibits the retrograde potentiation of presynaptic glutamate release. (F–H) Quantification of mEPSP amplitude (F), EPSP amplitude (G), and quantal content (H) in the indicated muscles. Asterisks indicate statistical significance using a Student’s t test: (**) p<0.01; (***) p<0.001; (ns) not significant. Error bars indicate ± SEM. Detailed statistical information for represented data (mean values, SEM, n, p) is shown in Supplementary file 1.
Additional files
-
Supplementary file 1
Absolute values for all data and additional statistics.
The figure and panel, genotype, and conditions used are noted (muscle segment, external calcium concentration, PhTx application). All electrophysiological recordings were performed in 0.4 mM external calcium unless specified otherwise. Average values for mEPSP, EPSP, quantal content, resting potential, input resistance, number of data samples (n), p values, and significance are shown. Average values for confocal imaging intensity levels are also indicated. Standard error values are noted in parentheses.
- https://doi.org/10.7554/eLife.34338.013
-
Transparent reporting form
- https://doi.org/10.7554/eLife.34338.014





