Proteolytic maturation of α2δ controls the probability of synaptic vesicular release
Figures
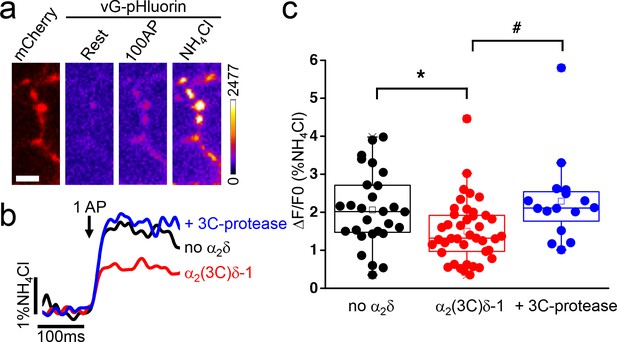
Effect of proteolytic cleavage of α2(3C)δ-1 on vesicular release in presynaptic terminals of hippocampal neurons.
(a) Fluorescence changes in presynaptic terminals of hippocampal neurons expressing vGlut-pHluorin (vG-pHluorin) in response to electrical stimulation. Left panel, mCherry-positive boutons. Three right panels, vG-pHluorin fluorescence: at rest (left), after 100 AP at 10 Hz (middle) and after a brief application of NH4Cl (right). Scale bar 5 μm. The pseudocolour scale is shown with the last panel. (b) Representative vG-pHluorin responses to a single AP (10–12 trial average, 25 to 50 boutons) from presynaptic terminals of neurons co-transfected with empty vector (black trace), α2(3C)δ-1 (red trace) or α2(3C)δ-1 + 3C-protease (blue trace). Arrow indicates stimulation with one AP. (c) vG-pHluorin fluorescence changes (expressed as % of NH4Cl response) in response to 1 AP from boutons co-transfected with empty vector (black), α2(3C)δ-1 (red) or α2(3C)δ-1 + 3C-protease (blue) (n = 28, 41 and 16 independent experiments, respectively). Box and whiskers plots; *p=0.044 and #p=0.014, one way ANOVA and Bonferroni post-hoc test.
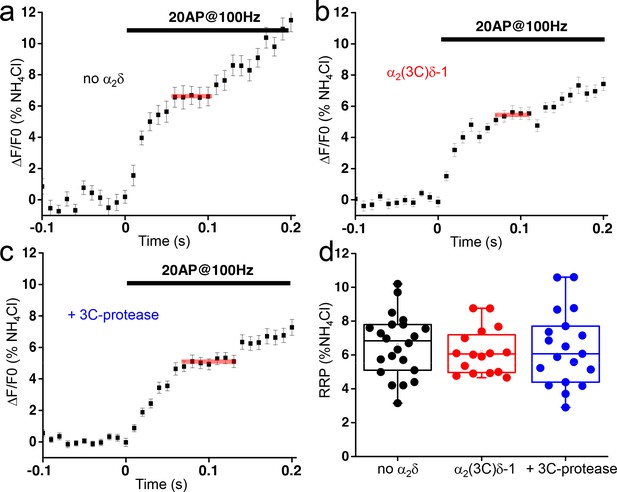
The proteolytic cleavage of α2(3C)δ-1 does not affect the readily releasable pool (RRP) in presynaptic terminals of hippocampal neurons.
(a–c) vG-pHluorin responses (mean ± SEM) to 20 AP at 100 Hz (5–6 trial average, 25 to 50 boutons) from presynaptic terminals of neurons co-transfected with empty vector (a), α2(3C)δ-1 (b) or α2(3C)δ-1 + 3C-protease (c). Horizontal red lines identify RRPs. (d) Average RRP (expressed as % of NH4Cl response) from boutons co-transfected with empty vector (black), α2(3C)δ-1 (red) or α2(3C)δ-1 + 3C-protease (blue) (n = 22, 16 and 19 independent experiments, respectively, p=0.78). Box and whiskers plots; one way ANOVA and Bonferroni post-hoc test.
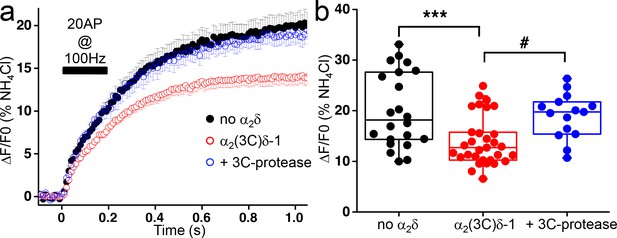
Effect of the proteolytic cleavage of α2(3C)δ-1 on delayed vesicular release in presynaptic terminals of hippocampal neurons.
(a) Average vG-pHluorin responses (mean ± SEM) to 20 APs at 100 Hz (5–6 trial average, 25 to 50 boutons) from presynaptic terminals of neurons co-transfected with empty vector (black), α2(3C)δ-1 (red) or α2(3C)δ-1 + 3C-protease (blue). The black bar indicates the stimulation period (20 AP at 100 Hz). (b) Average delayed vesicular release (expressed as % of NH4Cl response) measured 1 s after the beginning of the stimulation from boutons co-transfected with empty vector (black), α2(3C)δ-1 (red) or α2(3C)δ-1 + 3C-protease (blue) (n = 22, 31 and 15 independent experiments, respectively). Box and whiskers plots with superimposed individual experiments; ***p<0.001 and # p=0.021, one way ANOVA and Bonferroni post-hoc test.
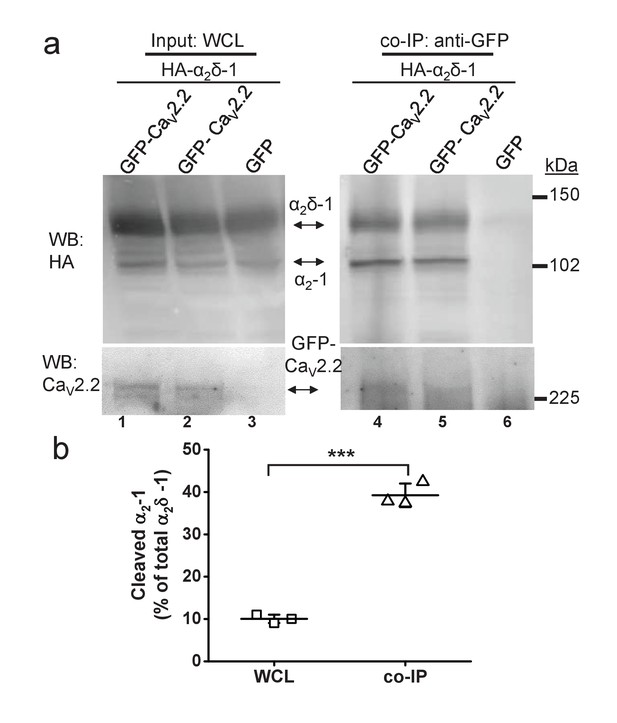
Quantified co-immunoprecipitation of CaV2.2 with cleaved and uncleaved fractions of wild-type α2δ-1 from WCL of tsA-201 cells.
(a) Left panels show WCL input from tsA-201 cells transfected with GFP-CaV2.2 (lanes 1 and 2) or GFP (lane 3), plus β1b and HA-tagged α2δ-1: upper panel, HA-α2δ-1 input; lower panel, CaV2.2-GFP input. Right panels show immunoprecipitation (IP) of GFP-CaV2.2 with anti-GFP Ab; immunoblots with CaV2.2 II-III loop Ab (lower panels, lanes 4 and 5) produced co-immunoprecipitation (co-IP) of HA-α2δ-1 (corresponding upper panels lanes 4 and 5), revealed by anti-HA mAb. All samples deglycosylated. (b) Proteolytic cleavage of α2δ-1 expressed as percentage of cleaved α2-1 moiety to total α2δ-1 calculated for input WCL fractions (squares) and for fractions co-immunoprecipitated with GFP- CaV2.2 (triangles). The cleaved α2-1 moiety in the co-IP fractions is increased by 29.2 ± 1.7% compared with the WCL fractions (average of 3 independent experiments). ***p=0.0032, paired t-test.
Additional files
-
Supplementary file 1
- https://doi.org/10.7554/eLife.37507.006
-
Transparent reporting form
- https://doi.org/10.7554/eLife.37507.007





