Dopamine neurons drive fear extinction learning by signaling the omission of expected aversive outcomes
Figures
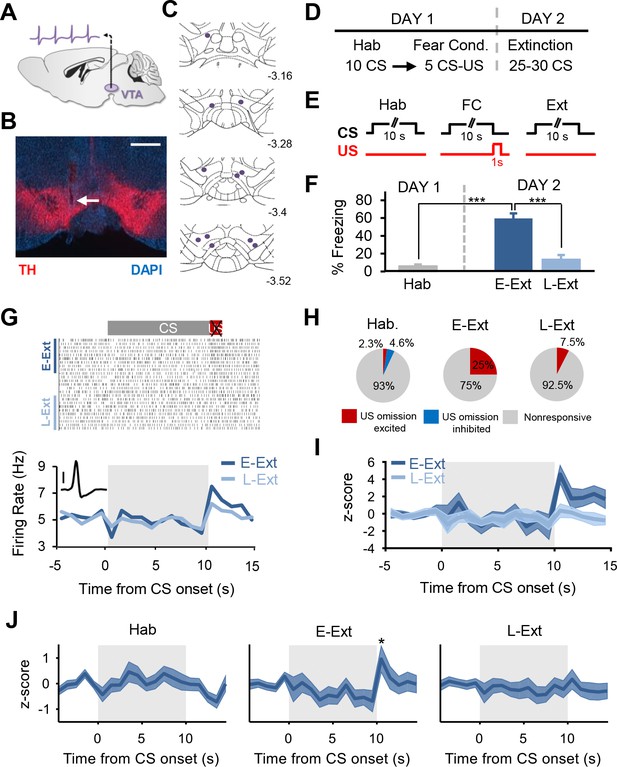
Putative dopamine neurons signal the unexpected omission of the aversive US during fear extinction learning.
(A) Schematic of single-unit recordings in the VTA. (B) Histological example showing an electrode placement in the VTA (white arrow). Red: immunostaining against tyrosine hydroxylase (TH), blue: DAPI staining. Scale bar: 0.5 mm. (C) Schematic coronal sections showing the location of the recording sites in the VTA. Numbers represent distance posterior to the bregma. (D) Schematic of the behavioral protocol. Hab: tone habituation, Fear Cond.: fear conditioning. (E) Schematic of CS and US presentations during tone habituation (Hab), fear conditioning (FC) and extinction (Ext). (F) Behavioral data. During tone habituation, mice (n = 11) showed low freezing levels in response to the CS. Twenty-four hours after fear conditioning, mice exhibited significant increase in freezing to the CS during E-Ext trials (first 10 CSs, paired t-test comparing E-Ext to Hab, t(10) = 8.30, p<0.0001). The CS evoked low freezing levels during L-Ext trials (last 10 CSs) indicating successful extinction learning (paired t-test: comparing L-Ext to Hab, t(10) = 1.32, p = 0.21; comparing L-Ext to E-Ext, t(10) = 11.9, p<0.0001). (***p<0.0001). (G) Raster plot (top) and peristimulus time histogram (1 s bins; bottom) of an example putative DA neuron (see inset; scale bar: 50 μV) responding to US omission during the extinction session. (H) The proportion of putative DA neurons that were significantly US omission (or CS offset for Hab) excited, inhibited or nonresponsive during the Hab, E-Ext and L-Ext trials. Proportion of US omission excited neurons significantly increased from Hab to E-Ext (Fisher’s exact test, p = 0.0028) and decreased back to Hab levels during L-Ext (Fisher’s exact test, p = 0.34). Note that there were no US omission inhibited neurons during E-Ext and L-Ext. (I) Peristimulus time histogram showing the z-scored population activity of all putative DA neurons that were significantly US omission excited during E-Ext (n = 10 of 40 putative DA neurons). (J) Peristimulus time histogram showing the z-scored population activity of all putative DA neurons during Hab (left), E-Ext (middle) and L-Ext (right). Note the significant increase in population activity at the time of the US omission during E-Ext (*p<0.05). Shaded regions represent mean ± s.e.m. across neurons.
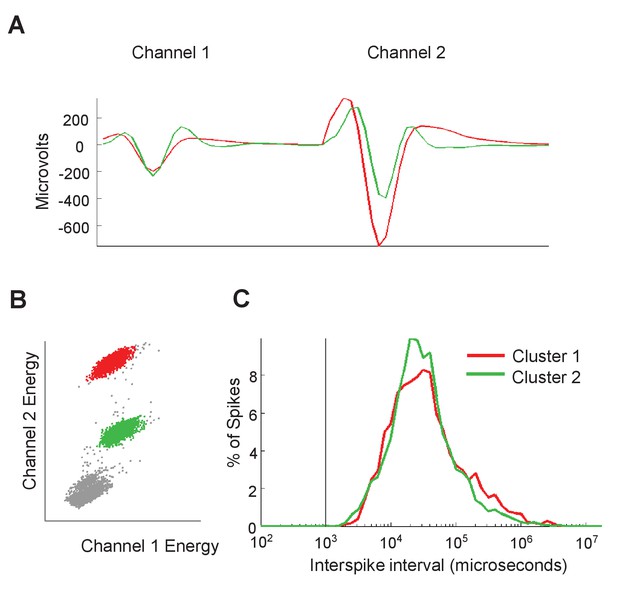
Isolation of single-units using steretrodes.
Recordings were performed in the VTA using microdrives containing bundles of 5–8 stereotrodes. (A) Examples of waveforms from two single-units recorded simulltaneously on the same stereotrode. (B,C) Scatterplot of energy on channel one against energy on channel 2 (B) and interspike interval distribution (C) for the single units in A.
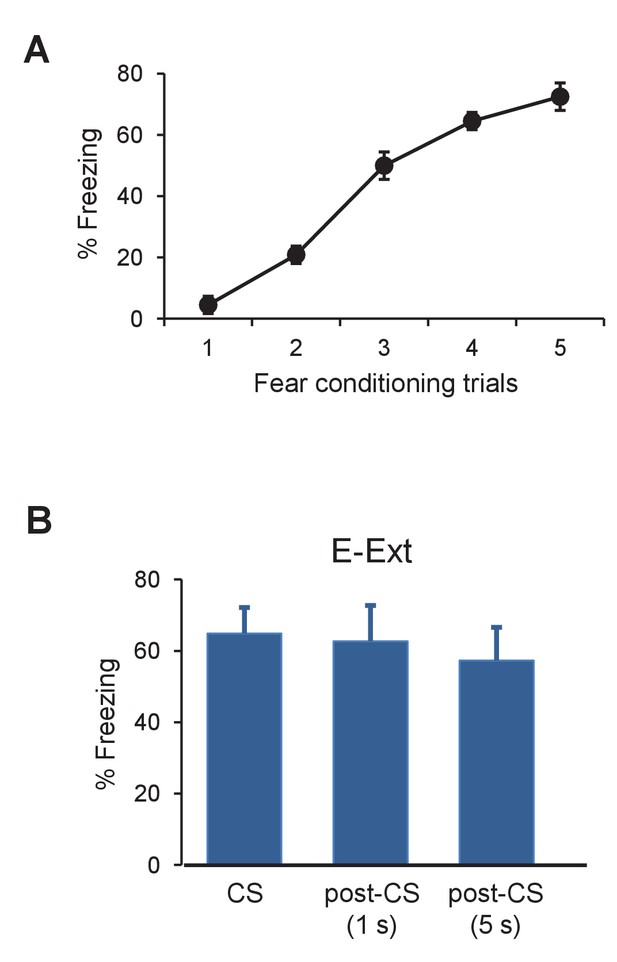
Freezing levels during fear conditioning and the post-CS periods during E-Ext.
(A) Behavioral freezing to the CS during fear conditioning for animals (n = 11) in the electrophysiology experiment. The CS-evoked freezing levels increased gradually during fear conditioning indicating successful fear acquisition (paired t-test comparing freezing during first CS to last CS, t(10) = 17.54, p<0.0001). (B) Bar plot showing behavioral freezing levels during the CS, 1 s post-CS and 5 s post-CS periods in E-Ext. Freezing levels during the post-CS periods were comparable to freezing during the CS (paired t-test comparing CS to 1 s post-CS, t(10) = 0.53, p = 0.6 and paired t-test comparing CS to 5 s post-CS, t(10) = 0.42, p = 0.67) ruling out the possibility that an increase in movement when the CS stops might have resulted in the observed increase in DA neuron firing at the CS offset (the time of the US omission) during E-Ext. E-Ext: early extinction. Data are presented as means and error bars represent s.e.m. across animals.
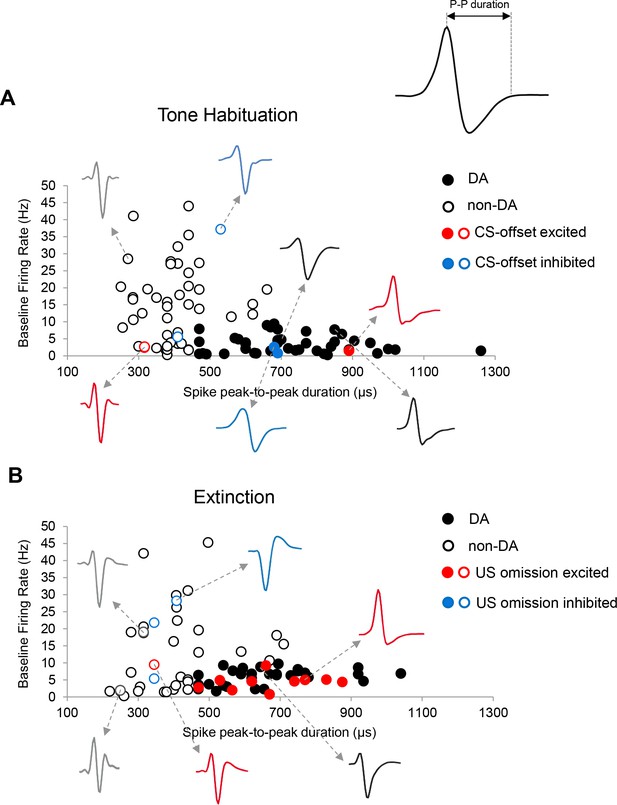
Classification of VTA neurons.
Plot of baseline firing rate (Hz) and spike peak-to-peak (P-P) duration (µs; see inset) of VTA neurons recorded during tone habituation (A) and extinction (B). Filled and unfilled circles indicate putative DA and non-DA neurons, respectively. Red dots (filled: DA, unfilled: non-DA) indicate neurons that showed significant CS-offset excited (A) and US omission excited (B) firing during tone habituation and extinction, respectively. Blue dots (filled: DA, unfilled: non-DA) indicate neurons that showed significant CS-offset inhibited (A) and US omission inhibited (B) firing during tone habituation and extinction, respectively. Black dots indicate non-responsive neurons.
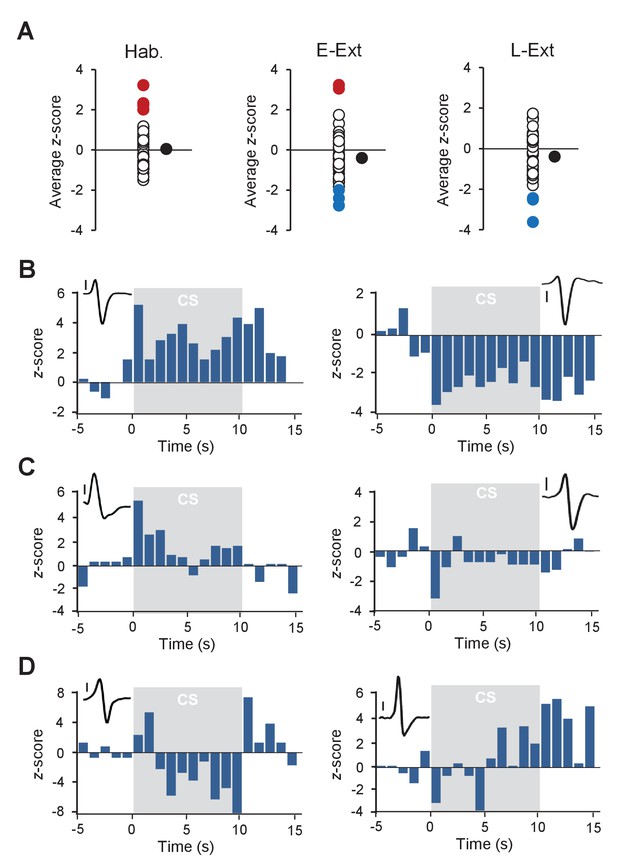
CS-evoked responses of putative DA neurons.
(A) Distribution of z-scores during the CS (averaged across the CS duration) for Hab (left), E-Ext (middle) and L-Ext (right). Each empty black circle represents a putative DA neuron. Filled black circles indicate the average z-score across neurons. Red and blue dots indicate neurons that were significantly excited and inhibited during the CS, respectively. Hab: tone habituation, E-Ext: early extinction, L-Ext: late extinction. (B) Example putative DA neurons (see insets, scale bar: 50 µV) that show sustained excitation (left) and inhibition (right) during the CS. (C) Example putative DA neurons (see insets, scale bar: 50 µV) that show transient increase (left) and decrease (right) in firing to the CS. (D) Example putative DA neurons (see insets, scale bar: 50 µV) that exhibit biphasic responses to the CS. These two neurons with biphasic responses are also US omission excited. Note the significant increase in firing at the time of the US omission. Gray shaded areas represent the CS.
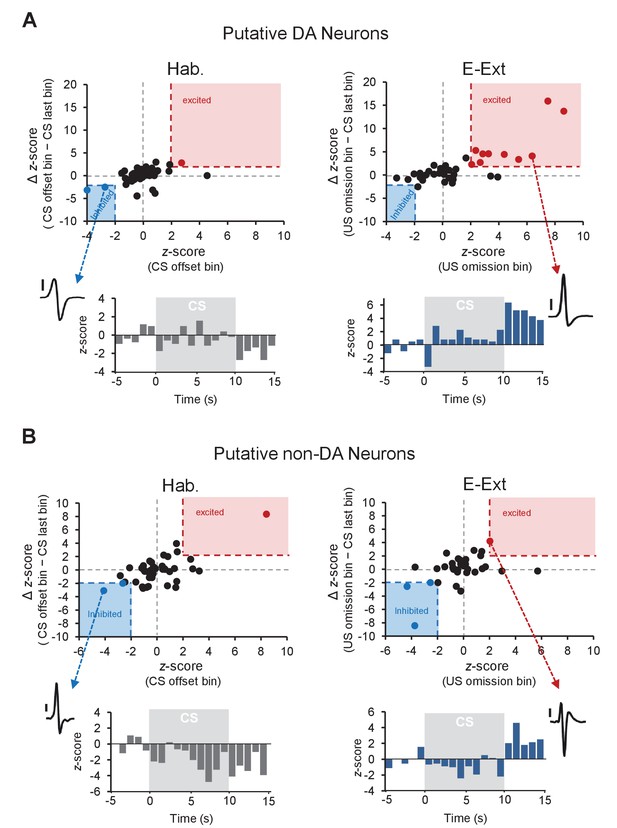
Distribution of z-scores during the US omission for putative DA and non-DA neurons.
(A) Top: plot of z-score (CS offset bin) against the ∆ z-score (CS offset bin − CS last bin) during tone habituation (Hab.; left) and z-score (US omission bin) against the ∆ z-score (US omission bin − CS last bin) during early extinction (E-Ext; right). Each dot represents a putative DA neuron. The red shaded areas (x-axis: 2, y-axis: 2) contain the significantly excited (red dots) and the blue shaded areas (x-axis: −2, y-axis: −2) contain significantly inhibited (blue dots) neurons. Black dots represent non-responsive neurons. Bottom: peristimulus time histogram (1 s bins) of an example neuron (see inset; scale bar: 50 μV) that showed significant inhibition to CS offset during Hab (left) and of an example neuron (see inset; scale bar: 50 μV) that showed significant excitation to the US omission during E-Ext (right). (B) The same as in (A), but for putative non-DA neurons.
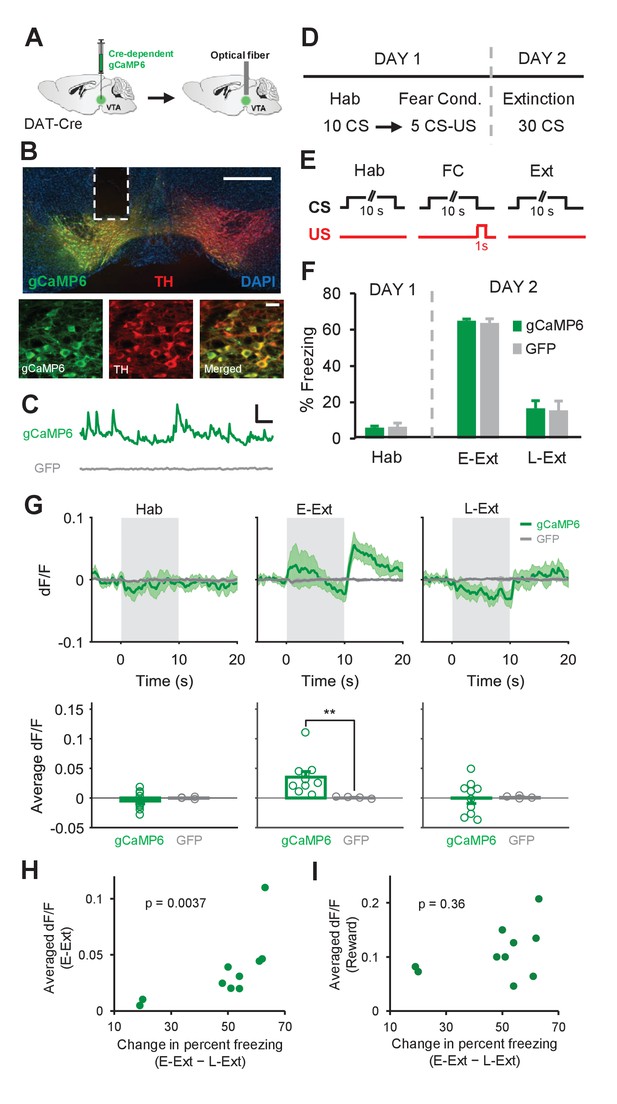
Calcium recordings in VTA dopamine neurons confirm signaling of the unexpected US omission during fear extinction learning.
(A) Schematic of the surgical procedure showing the virus injection (left) and optical fiber implantation (right) in the VTA. (B) Top: example histological image showing Cre-dependent expression of gCaMP6 (green) along with immunostaining for tyrosine hydroxylase (TH, red) and DAPI (blue) staining in the VTA. White vertical track indicates the optical fiber placement in the VTA. Scale bar: 0.5 mm. Bottom: confocal images showing expression of gCaMP6 (left), TH (middle) and a merged image (right) showing co-expression. Scale bar: 20 μm. (C) Examples of changes in fluorescence (dF/F) over time in an animal expressing gCaMP6 (green) and an animal expressing the control fluorophore GFP (gray). Scale bar: 5 s, 0.2 dF/F. (D) Schematic of the behavioral protocol. Hab: tone habituation, Fear Cond.: fear conditioning. (E) Schematic of CS and US presentations during tone habituation (Hab), fear conditioning (FC) and extinction (Ext). (F) Behavioral freezing to the CS during tone habituation and extinction session for gCaMP6 (n = 10) and GFP (n = 4) groups. During Hab, all mice showed low freezing levels in response to the CS. Twenty-four hours after fear conditioning, both groups of mice increased freezing to the CS during E-Ext (first 10 CSs). The CS evoked low freezing levels during L-Ext (last 10 CSs) indicating successful extinction learning. (G) Top: Average change in fluorescence in animals expressing gCaMP6 (green, n = 10) or GFP (gray, n = 4) around the time of CS presentation (gray area) during Hab, E-Ext and L-Ext. Note the increase in fluorescence at the offset of the CS (the time of the unexpected US omission) during E-Ext. Bottom: Average change in fluorescence in the 5 s after CS offset during Hab, E-Ext and L-Ext. gCaMP6-expressing animals exhibited a significant increase in Ca+2 signal during E-Ext compared to the GFP control group (**p<0.01, rank-sum test). (H) Correlation between the average change in fluorescence in the 5 s after CS offset during E-Ext and the change in percent freezing from E-Ext to L-Ext (n = 10 mice; Spearman’s correlation = 0.83, p = 0.0037). (I) Correlation between the average change in fluorescence during the 0–3 s after reward delivery and change in percent freezing from E-Ext to L-Ext (n = 10 mice; Spearman’s correlation = 0.32, p = 0.36). Shaded regions and error bars represent mean ±s.e.m across animals.
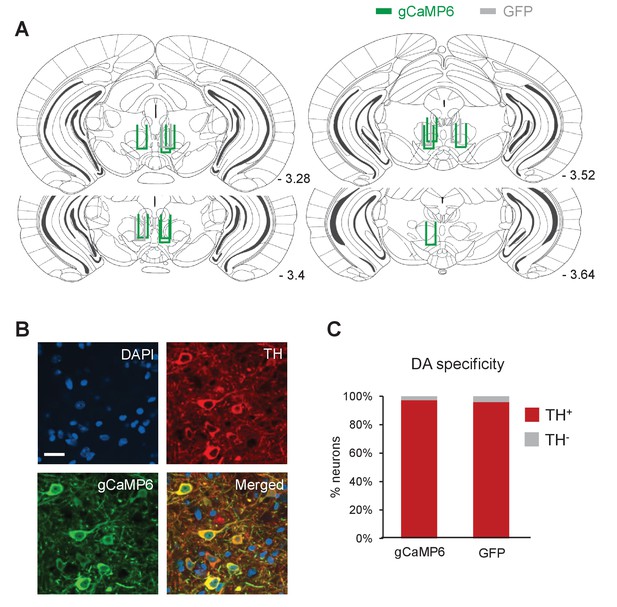
Placement of optical fibers and DA neuron-specific expression of GCaMP6.
(A) Schematic coronal sections showing the placement of the optical fibers for mice in gCaMP6 (green) and GFP (gray) groups. Numbers represent distance posterior to the bregma. (B) Representative confocal images showing DAPI staining (nuclear marker, blue), immunostaining for TH (dopamine neurons, red) and expression of gCaMP6 (green). Scale bar: 20 μm. (C) Percentage of neurons that are GFP+/TH+ (red) and GFP+/TH- (gray) in gCaMP6 (n = 1069 neurons in four mice) and GFP (n = 1163 neurons in four mice) groups. Both groups showed similarly high levels of DA specificity (gCaMP6: 97.1%; GFP: 95.8%).
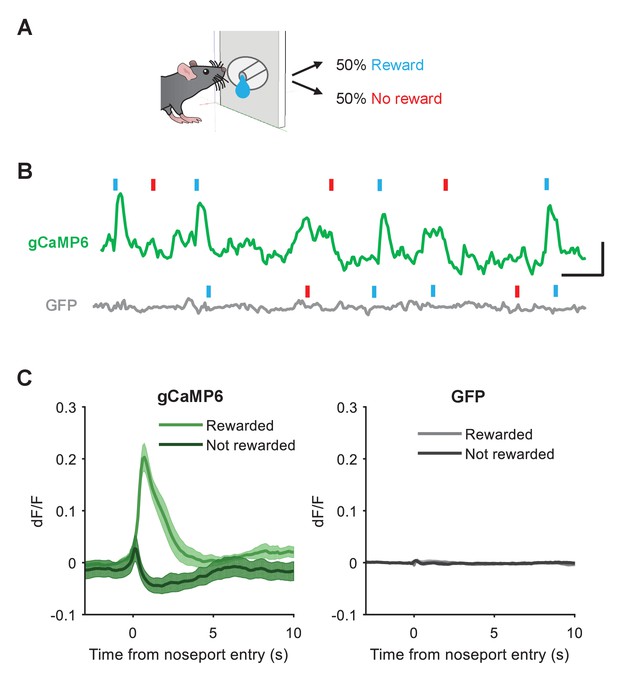
Responses of VTA DA neurons to reward.
(A) Schematic of reward task. Animals received reward 50% of the time after entering the noseport. (B) Example traces showing changes in fluorescence in an animal expressing gCaMP6 (top, green) or GFP (bottom, gray) in the VTA during the reward task. Rewarded and unrewarded nosepokes are indicated by blue and red ticks, respectively. Note the increases in fluorescence during reward delivery in the gCaMP6 animal. Scale bar: 5 s, 0.1 dF/F. (C) Average change in fluorescence during rewarded and unrewarded nosepokes in gCaMP6 (left, n = 10) or GFP-expressing (right, n = 4) animals. Data are presented as means and error bars represent s.e.m across animals.
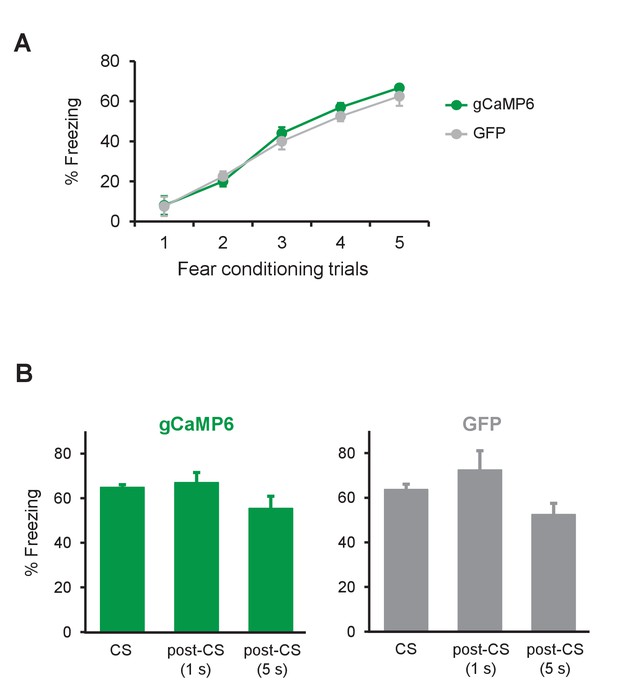
Freezing levels during fear conditioning and the post-CS periods during E-Ext.
(A) Percent freezing to the CS during fear conditioning for gCaMP6 (green, n = 10) and GFP (gray, n = 4) groups. The CS evoked freezing levels increased gradually during fear conditioning indicating successful fear acquisition. There was no difference between the groups in their fear acquisition (two-way repeated measures ANOVA, no main effect of group, F1,48 = 0.72, p = 0.41 or no group × trial interaction, F4,48 = 0.3, p = 0.87). (B) Bar plots showing freezing levels during the CS, 1 s post-CS and 5 s post-CS periods in E-Ext for gCaMP6 and GFP groups. For both groups, freezing levels during the post-CS periods were comparable to freezing during the CS (gCaMP6: paired t-test comparing CS to 1 s post-CS, p = 0.65 and paired t-test comparing CS to 5 s post-CS, p = 0.1; GFP: paired t-test comparing CS to 1 s post-CS, p = 0.4 and paired t-test comparing CS to 5 s post-CS, p = 0.15) ruling out the possibility that an increase in movement when the CS stops might have resulted in the observed increase in Ca+2 signal at the CS offset (the time of the US omission) during E-Ext. Data are presented as means and error bars represent s.e.m. across animals.
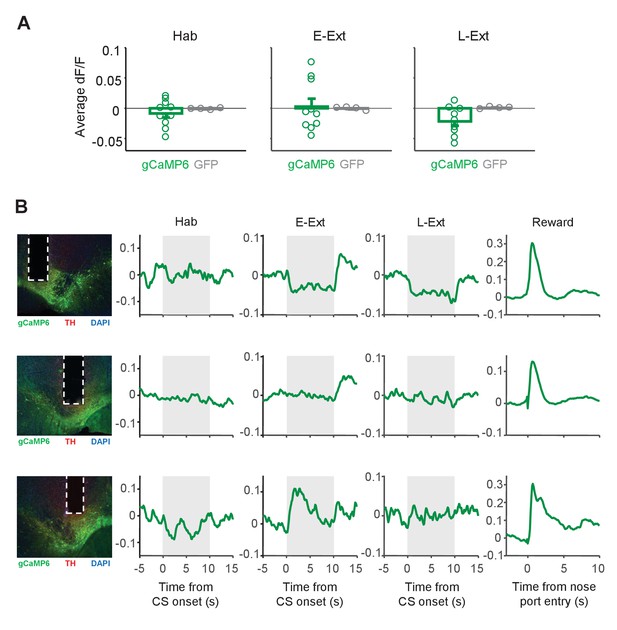
Changes in the calcium signal during CS presentations.
(A) Average change in fluorescence (dF/F) during the CS (10 s) during tone habituation (Hab), early extinction (E-Ext) and late extinction (L-Ext). There was no difference in responses to the CS between the gCaMP6-expressing (n = 10) and GFP-expressing (n = 4) animals (rank-sum test: Hab, p = 1.0; E-Ext, p = 0.37; L-Ext, p = 0.11). (B) Example CS responses in gCaMP6-expressing animals that showed decreased (top), unchanged (middle) and increased fluorescence during the CS (gray area). Left: histology image showing the location of the optical fiber in VTA. Green: cre-dependent gCaMP6 expression, red: immunostaining for tyrosine hydroxylase (TH), blue: DAPI staining. White vertical tracks indicate the optical fiber placement in the VTA. Right: Changes in fluorescence during Hab, E-Ext, L-Ext and Reward.
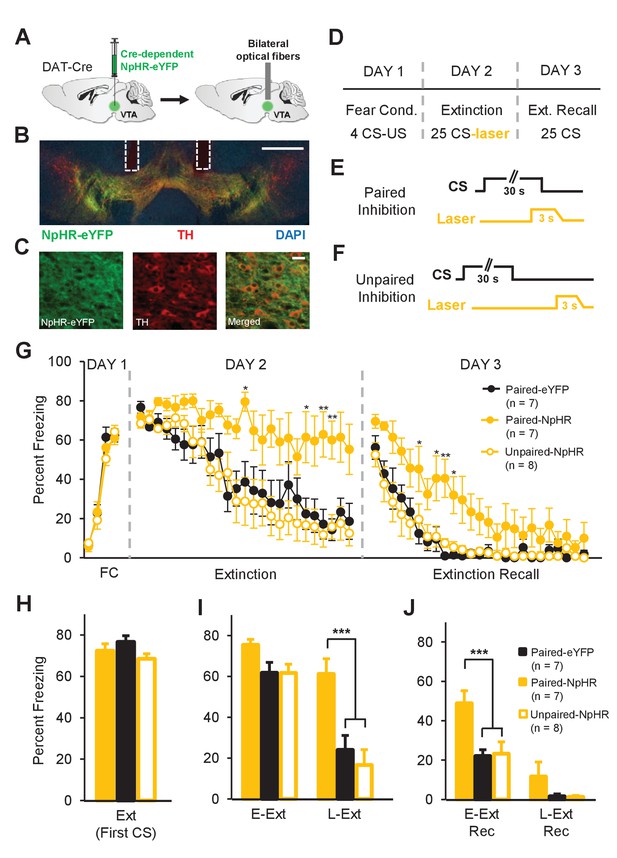
Inhibition of dopamine neuron firing at the time of the US omission impairs fear extinction learning.
(A) Schematic of the surgical procedure showing bilateral virus injection (left) and optical fiber implantation (right) in the VTA. (B) Example histological image showing Cre-dependent expression of NpHR-eYFP (green) along with immunostaining for tyrosine hydroxylase TH (red) and DAPI (blue) staining in the VTA. White vertical tracks indicate the bilateral optical fiber placements in the VTA. Scale bar: 0.5 mm. (C) Confocal images showing expression of NpHR-eYFP (left), TH (middle) and merged image (right) showing co-expression. Scale bar: 20 μm. (D) Schematic of the behavioral protocol. Fear Cond.: fear conditioning, Ext Recall: extinction recall. (E) Schematic of paired optogenetic inhibition of DA neurons at the time of the US omission. (F) Schematic of unpaired optogenetic inhibition during intertrial intervals. (G) Percent freezing to the CS during fear conditioning (FC), extinction and extinction recall sessions. The Paired-NpHR group showed impaired extinction learning and extinction recall. (**p<0.01, *p<0.05). (H) No difference in freezing to the CS between groups at the start of extinction (first CS). Ext: extinction. (I) Freezing levels during E-Ext (average of first 10 CSs) and L-Ext (average of last 10 CSs; two-way repeated measures ANOVA, main effect of group: F2,19 = 9.05, p = 0.0017; group × trial interaction: F2,19 = 7.38, p = 0.0043). The Paired-NpHR group (n = 7) showed significantly higher freezing to the CS compared to the Paired-eYFP (n = 7) and Unpaired-NpHR (n = 8) groups during L-Ext trials (***p<0.001). E-Ext: early extinction, L-Ext: late extinction. (J) Freezing levels during E-Ext Rec (average of first 10 CSs) and L-Ext Rec trials (average of last 10 CSs; two-way repeated measures ANOVA, main effect of group: F2,19 = 7.21, p = 0.0047). The Paired-NpHR group exhibited significantly higher freezing to the CS compared to the control groups during E-Ext Rec (***p<0.001). E-Ext Rec: early extinction recall, L-Ext Rec: late extinction recall. Error bars represent mean ± s.e.m. across animals.
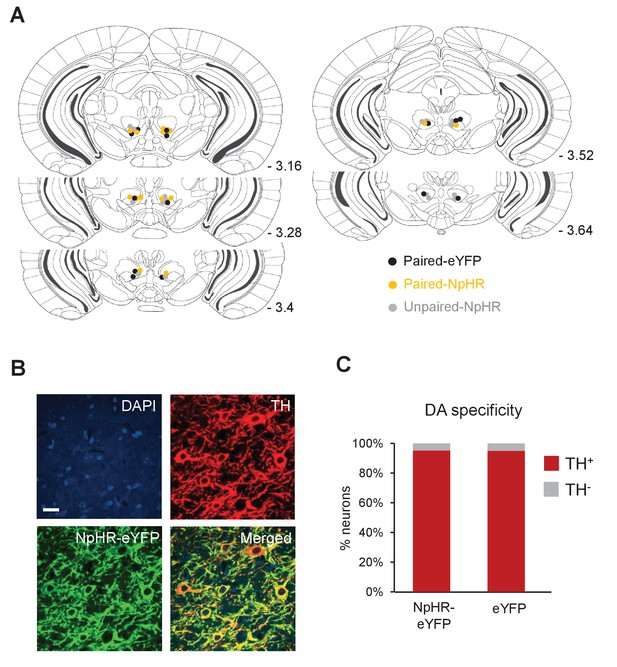
Placement of optical fibers and DA neuron-specific expression of NpHR-eYFP.
(A) Schematic coronal sections showing the bilateral placement of the tips of the optical fibers for mice in Paired-NpHR (yellow) Paired-eYFP (black) and Unpaired-NpHR (gray) groups. Numbers represent distance posterior to the bregma. (B) Representative confocal images showing DAPI staining (nuclear marker, blue), immunostaining for TH (dopamine neurons, red) and expression of NpHR-eYFP (green). Scale bar: 20 μm. (C) Percentage of neurons that are eYFP+/TH+ (red) and eYFP+/TH- (gray) in NpHR-eYFP (n = 1464 neurons in four mice) and eYFP (n = 1589 neurons in four mice) groups. Both groups showed similarly high levels of specificity for DA neurons (NpHR-eYFP: 95.3%; eYFP: 95%).
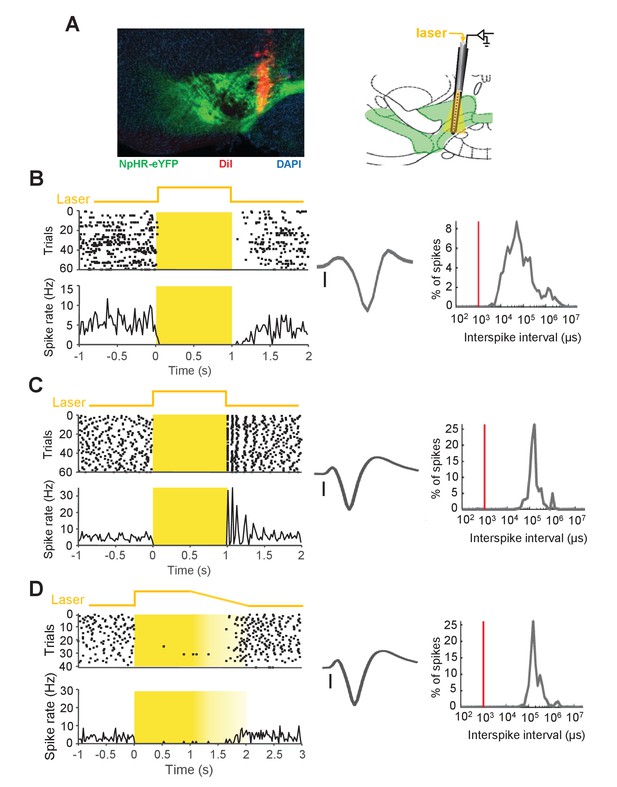
Optical activation of NpHR inhibits DA neuron firing in awake behaving mice.
(A) Left: histological example of a silicon probe track (red) in the VTA. Green: NpHR-eYFP expression, red: DiI, blue: DAPI staining. Right: Schematic coronal section showing the location of the opto-probe in the VTA. (B) A representative DA neuron showing inhibition of spiking in response to laser illumination. Left: Schematic of the laser illumination (top, laser was applied from 0 to 1 s); raster plot (middle) and spike rate plot (bottom). Middle: spike waveform (scale: 100 µV). Right: inter-spike interval distribution. (C) The same as (B), but for a representative DA neuron that shows rebound excitation at the laser offset. (D) The same DA neuron as in (C) in response to a laser pulse with a ramp-like reduction in light power (1 s ramp) similar to the ramp (1 s) used in optogenetic inhibition experiment in Figure 5. Note the reduced rebound excitation.
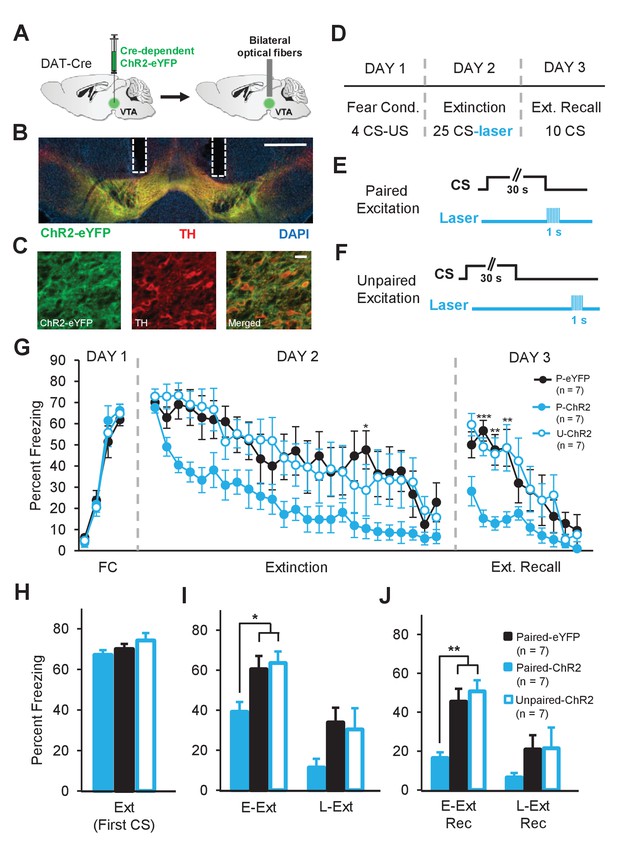
Enhancing dopamine neuron firing at the time of the US omission accelerates fear extinction learning.
(A) Schematic of the surgical procedure showing the bilateral virus injection (left) and optical fiber implantation (right) in the VTA. (B) Example histology showing Cre-dependent expression of ChR2-eYFP (green) along with immunostaining for tyrosine hydroxylase (TH, red) and DAPI (blue) staining in the VTA. White vertical tracks indicate the bilateral optical fiber placements in the VTA. Scale bar: 0.5 mm. (C) Confocal images showing expression of ChR2-eYFP (left), TH (middle) and merged image (right) showing co-expression. Scale bar: 20 μm. (D) Schematic of the behavioral protocol. Fear Cond.: fear conditioning, Ext Recall: extinction recall. (E) Schematic of the paired optogenetic excitation of DA neurons at the time of the US omission. (F) Schematic of the unpaired optogenetic excitation during intertrial intervals. (G) Percent freezing to the CS during fear conditioning (FC), extinction and extinction recall sessions. The Paired-ChR2 (P-ChR2) group showed accelerated extinction learning and better extinction recall. (***p<0.001, **p<0.01, *p<0.05). (H) No difference in freezing to the CS between groups at the start of extinction (first CS). Ext: extinction. (I) Freezing levels during E-Ext (average of first 10 CSs) and L-Ext (average of last 10 CSs; two-way repeated measures ANOVA, significant group effect: F2,18 = 3.88, p = 0.03). The P-ChR2 group (n = 7) exhibited significantly lower freezing compared to the Paired-eYFP (P-eYFP; n = 7) and Unpaired-ChR2 (U-ChR2; n = 7) control groups during E-Ext (*p<0.05). E-Ext: early extinction, L-Ext: late extinction. (J) Freezing levels during E-Ext Rec (average of first 5 CSs) and L-Ext Rec (average of last 5 CSs; two-way repeated measures ANOVA, main effect of group: F2,18 = 5.5, p = 0.01 and group × trial interaction: F2,18 = 4.81, p = 0.021). The P-ChR2 group exhibited significantly lower freezing to the CS compared to control groups during E-Ext Rec (**p<0.01). E-Ext Rec: early extinction recall, L-Ext Rec: late extinction recall. Data are presented as means and error bars represent s.e.m.
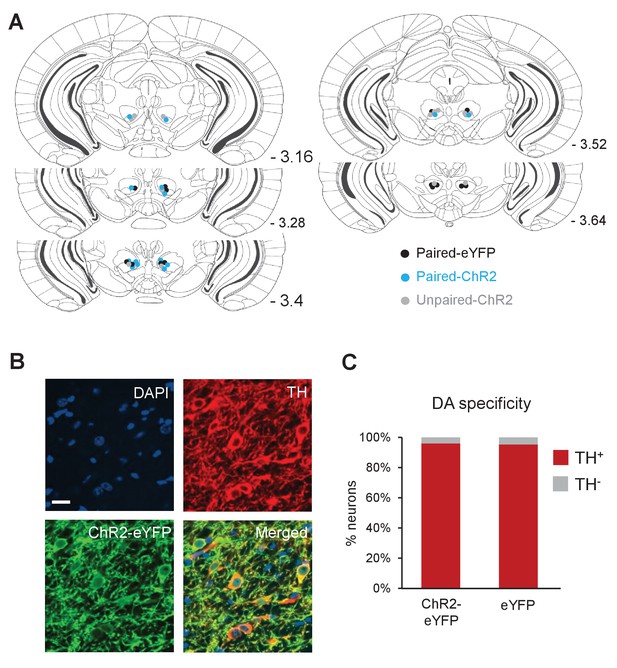
Placement of optical fibers and DA neuron specific expression of ChR2-eYFP.
(A) Schematic coronal sections showing the bilateral placement of the tips of the optical fibers for mice in Paired-ChR2 (blue) Paired-eYFP (black) and Unpaired-ChR2 (gray) groups. Numbers represent distance posterior to the bregma. (B) Representative confocal images showing DAPI staining (nuclear marker, blue), immunostaining for TH (dopamine neurons, red) and expression of ChR2-eYFP (green). Scale bar: 20 μm. (C) Percentage of neurons that are eYFP+/TH+ (red) and eYFP+/TH- (gray) in ChR2-eYFP (n = 1267 neurons in four mice) and eYFP (n = 1538 neurons in four mice) groups. Both groups showed similarly high levels of specificity for DA neurons (ChR2-eYFP: 95.9%; eYFP: 95.1%).
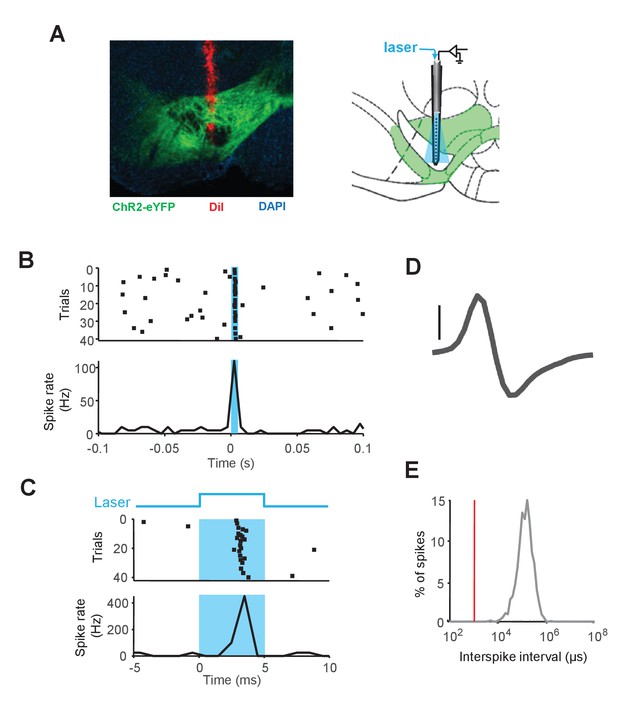
Optical stimulation of ChR2 induces firing of DA neurons in awake behaving mice.
(A) Left: histological example of a silicon probe track (red) in the VTA. Green: ChR2-eYFP expression, red: DiI, blue: DAPI staining. Right: Schematic coronal section showing the location of the opto-probe in the VTA. (B) A representative DA neuron showing short-latency excitation to laser illumination (5 ms laser pulse). Raster plot (top) and spike rate plot (bottom). (C) Response of the same neuron as in (B) shown with higher temporal resolution. Schematic of the laser illumination (top; laser was applied from 0 to 5 ms), raster plot (middle) and spike rate plot (bottom). Spike waveform (scale bar: 50 µV) (D) and Inter-spike interval distribution (E) of the neuron in (B) and (C).
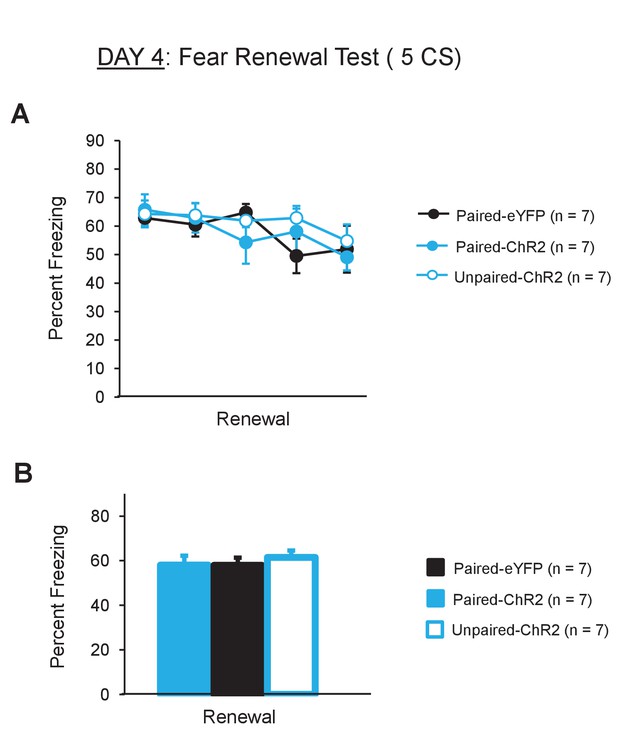
All animals showed comparable levels of fear renewal.
(A) Percent freezing to the CS during the renewal test (5 CS presentations in the conditioning context) on day 4. All groups showed comparable levels of freezing throughout the renewal test. (B) Average freezing levels (average of 5 CSs) during the renewal test were comparable between the groups (one-way ANOVA, F2,18 = 0.32, p = 0.72). Data are presented as means and error bars represent s.e.m.
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.38818.021





