Palovarotene reduces heterotopic ossification in juvenile FOP mice but exhibits pronounced skeletal toxicity
Figures
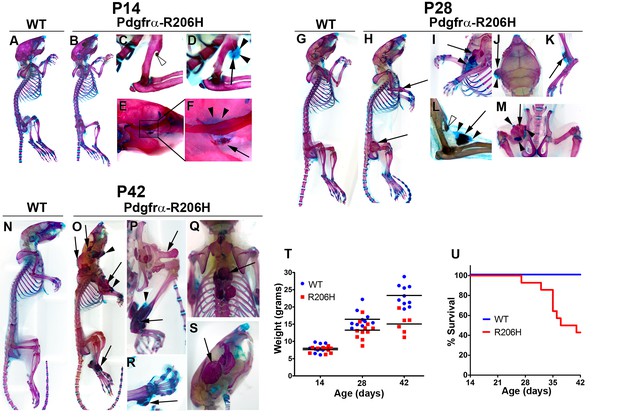
Natural history of juvenile Pdgfrα-R206H mice.
(A–S) Whole mount skeletal preparations of wild-type (WT) and Pdgfrα-R206H mice stained with Alcian Blue and Alizarin Red (ABAR) to detect cartilage and bone, respectively. (A, G, N) HO was never observed in WT mice. (B) Pdgfrα-R206H mice rarely exhibited HO at P14. (C) A stalk-like osteochondroma (open arrowhead) emanating from the deltoid tuberosity of the humerus. (D, E, F) When present, HO at P14 was primarily comprised of cartilage (arrowheads) with minimal bone (arrow). (H) All P28 Pdgfrα-R206H mice presented multiple sites of HO (arrows). (I–M) Higher magnification images of HO in P28 Pdgfrα-R206H mice, which contained both boney (arrows) and cartilaginous (arrowheads) regions. (L) An osteochondroma (open arrowhead) and HO (arrows/arrowheads) in the same forelimb limb. (O–S) P42 Pdgfrα-R206H mice exhibited widespread boney HO (arrows), which occasionally contained peripheral cartilage (arrowheads). (P–S) Higher magnification images of HO in P42 Pdgfrα-R206H. (T) Compared to WT mice, Pdgfrα-R206H mice exhibited a 16% and 35% reduction in mean body weight at P28 (n = 11; p<0.01) and P42 (n = 6; p<0.0001), respectively. Means are depicted (black bars), and statistical significance was assessed by one-way ANOVA. (U) Pdgfrα-R206H mice survived to a median age of 39 days and exhibited a significantly reduced survival rate of 43% at P42, as assessed by log-rank (n = 14; p<0.0001).
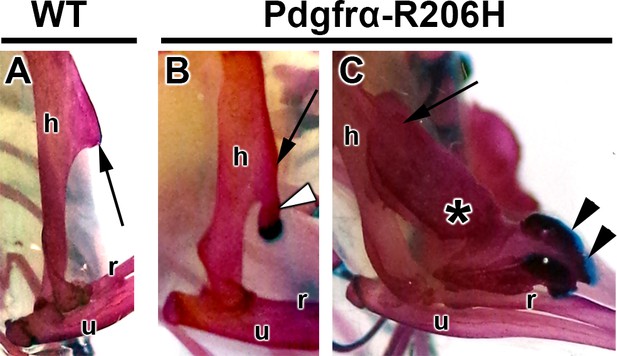
A comparison of osteochondroma and HO formation in the Pdgfrα-R206H mouse forelimb.
(A–C) Whole mount images of ABAR-stained forelimbs from (A) P42 WT and (B, C) Pdgfrα-R206H mice with the deltoid tuberosity denoted (arrows). (B) The deltoid tuberosity (arrow) was a common site of osteochondroma formation (open arrowhead), which presented as a stalk-like cartilage capped structure. (C) A higher magnification image of forelimb HO (asterisk) shown in Figure 1O, with the cartilaginous leading edge indicated (arrowheads). h, humerus; r, radius; u, ulna.
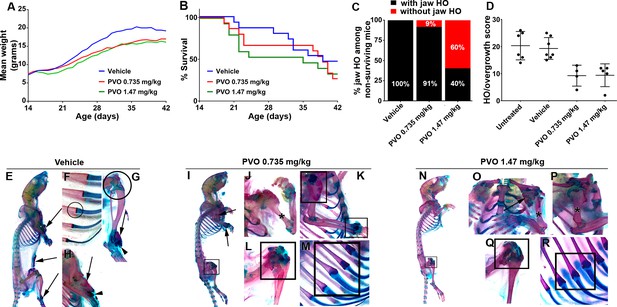
Palovarotene reduces the severity of HO in Pdgfrα-R206H mice.
(A) Body weight of Pdgfrα-R206H mice receiving palovarotene (PVO) trended lower but was not significantly different from those receiving vehicle, as assessed by two-way ANOVA. (B) Survival of Pdgfrα-R206H mice receiving PVO was not significantly different that those receiving vehicle, as assessed by log-rank. (C) Of Pdgfrα-R206H mice euthanized early or found dead prior to P42, only PVO-treated groups contained mice that lacked jaw HO. (D) Comparison of HO/overgrowth burden at P42. Data for untreated mice are from the natural history study. Both PVO-treated groups exhibited a ~ 50% reduction in HO severity. The HO/overgrowth score of each mouse is represented as a single point; group mean (black bar)±standard deviation (error bars) are shown. (E–R) Whole mount ABAR skeletal preparations of Pdgfrα-R206H mice at P42. (E–H) Vehicle treatment did not alter HO pathogenesis, which presented as bone (arrows) with limited cartilage (arrowheads). (F) Rib and (G) knee morphology was normal in vehicle-treated Pdgfrα-R206H mice (circles). (I–M) 0.735 mg/kg PVO-treated and (N–R) 1.47 mg/kg PVO-treated Pdgfrα-R206H mice exhibited reduced HO (arrows), and (J, O, P) osteochondromas were often absent (asterisks). (I, K–N, Q, R) PVO-treated Pdgfrα-R206H mice commonly presented abnormal shoulder, wrist, knee, and rib morphology at the costochondral junction (boxes).
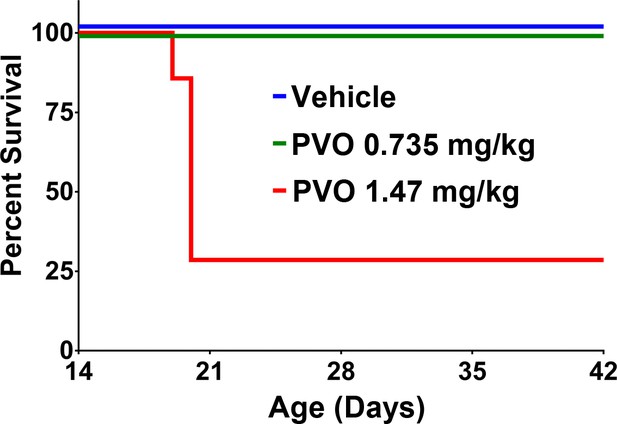
Palovarotene reduces survival in juvenile wild-type mice.
Although, wild-type mice receiving vehicle or 0.735 mg/kg palovarotene all survived to P42, treatment with 1.47 mg/kg palovarotene resulted in significantly reduced survival, as assessed by log-rank (n = 7; p<0.0001).

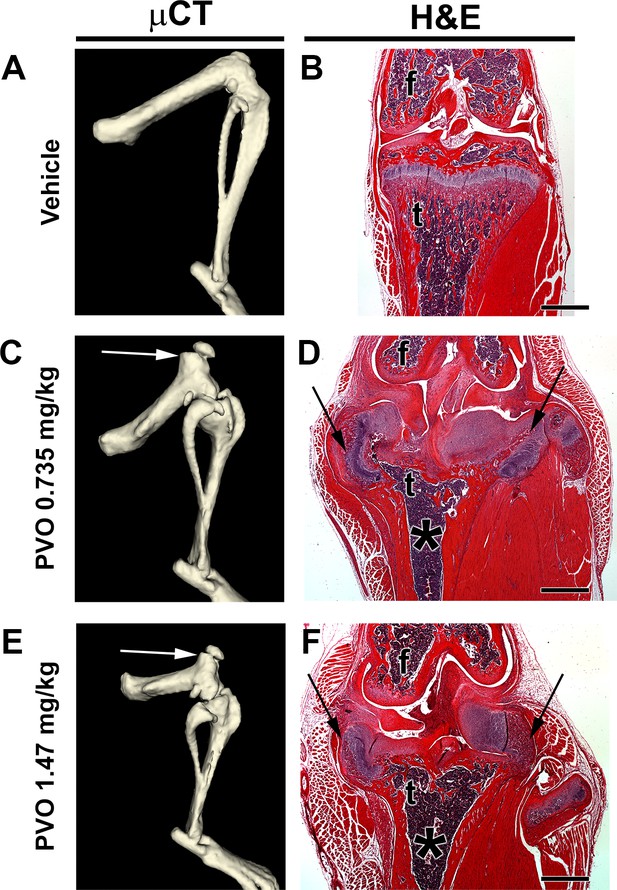
Palovarotene adversely affects synovial joints in juvenile Pdgfrα-R206H mice.
(A, C, E, G) µCT images of P42 mouse knees displaying lateral, frontal, and medial orientation. (B, D, F, H) Hematoxylin and eosin (H and E) staining of the same knee (p, patella; f, femur, t, tibia), oriented on the frontal plane. (A, B) WT knee morphology is comparable to that of (C, D) vehicle-treated Pdgfrα-R206H mice. (E–H) PVO-treated Pdgfrα-R206H mice exhibited overgrowth of knee articular cartilage, as detected by µCT (white arrows) and histology (black arrows). Scale bars = 1 mm for (B, D, F, H).
Palovarotene treatment results in skeletal toxicity and reduced survival in wild-type mice.
(A, C, E) µCT images of P42 mouse knees displaying lateral orientation. (B, D, F) H and E staining of the same knee (f, femur, t, tibia), oriented on the frontal plane. (A, B) Knee morphology is not altered by vehicle treatment. (C–F) PVO-treated wild-type mice lacked a growth plate (asterisk) and exhibited overgrowth of knee articular cartilage, as detected by µCT (white arrows) and histology (black arrows). Scale bars = 1 mm for (B, D, F).
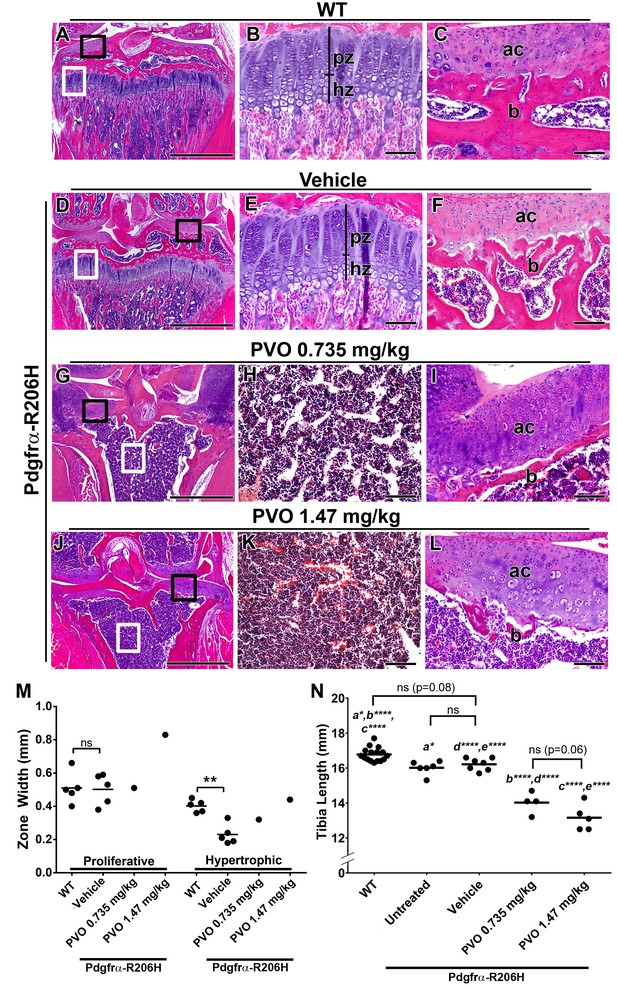
Palovarotene results in growth plate loss in juvenile Pdgfrα-R206H mice.
(A–L) H and E analysis of the proximal P42 tibia, oriented on the frontal plane. (A, D) Low magnification images with the growth plate (white boxes) and articular cartilage (black boxes) denoted. Corresponding high magnification images of the (B, E) growth plate and (C, F) articular cartilage revealed that WT mice and vehicle-treated Pdgfrα-R206H mice exhibit similar growth plate (pz = proliferative zone; hz = hypertrophic zone), subchondral bone (b), and articular cartilage (ac) morphology. (G, J) Low magnification images showing growth plate loss (white boxes) and thickened articular cartilage (black boxes) in PVO-treated Pdgfrα-R206H mice. Corresponding high magnification images revealed (H, K) an open marrow space lacking trabeculation, and (I, L) thinner subchondral bone (b) coupled with thickened articular cartilage (ac). (M) Growth plate proliferative and hypertrophic zone width at P42. (N) Tibial length of WT and Pdgfrα-R206H mice at P42. Each dot represents data from a single mouse, and group mean is represented by a horizontal bar. Statistical significance was determined by one-way ANOVA (ns, not significant; *=p ≤ 0.05; **=p ≤ 0.01; ***=p ≤ 0.001; ****=p ≤ 0.0001). Scale bars = 1 mm for A, D, G, J and 100 μm for B, C, E, F, H, I, K, L.
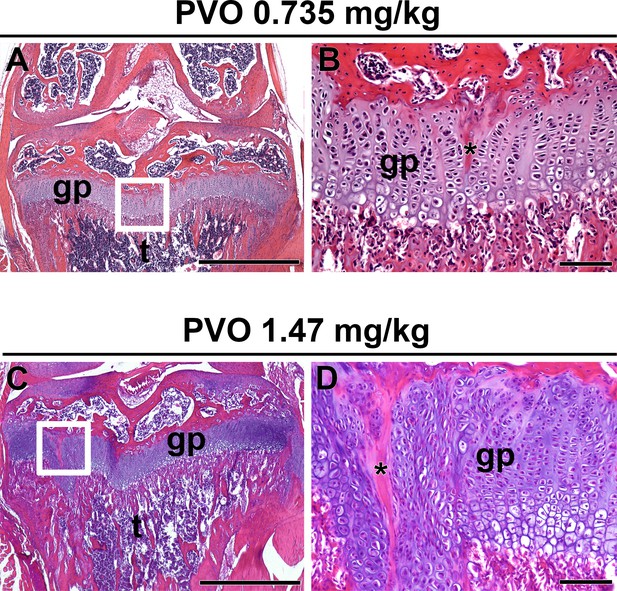
Growth plate trauma in palovarotene-treated Pdgfrα-R206H mice.
H and E stained proximal tibia of PVO-treated Pdgfrα-R206H mice that retained a tibial growth plate at P42. (A, C) Low magnification of the tibia (t) oriented on the frontal plane with the growth plate (gp) trauma region denoted (white boxes). (B, D) High magnification of the boxed region with trauma sites denoted (asterisk). Scale bars = 1 mm for A, C and 100 μm for B, D.
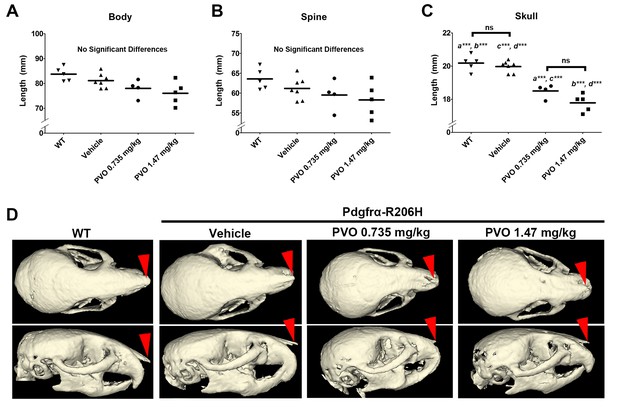
Palovarotene reduced skull length in Pdgfrα-R206H mice.
(A, B) At P42, differences in (A) total body length and (B) spine length trended lower in PVO-treated mice, but were not significantly different from WT or vehicle-treated Pdgfrα-R206H mice. (C) PVO-treated Pdgfrα-R206H mice exhibited reduced skull length as compared to WT and vehicle-treated Pdgfrα-R206H mice. Each point represents a single mouse and group mean is represented with a horizontal bar. Statistical significance was determined by one-way ANOVA (ns, not significant; *=p ≤ 0.05; **=p ≤ 0.01; ***=p ≤ 0.001). (D) µCT images of the skull showing superior (top row) and lateral (bottom row) views with nasal bone denoted (red arrowheads).
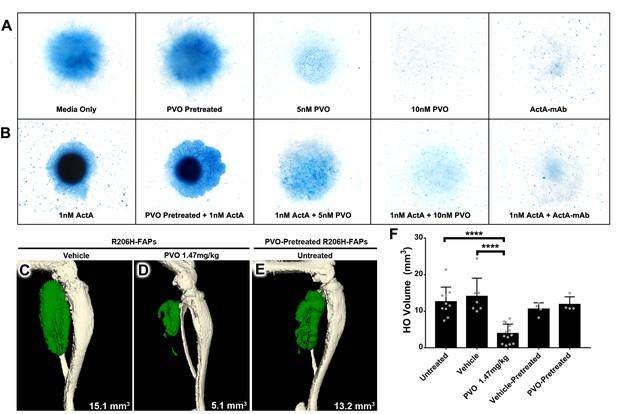
PVO treatment inhibits chondrogenic and osteogenic differentiation of R206H-FAPs.
(A, B) Chondrogenic differentiation of high-density R206H-FAPs cultures in (A) base media or (B) media with 25 ng/mL (~1 nM) activin A was assessed by Alcian Blue staining to detect cartilage matrix proteoglycans at day 10 (n = 3 experiments). ActA-mAb was used at ~7 fold molar excess to activin A ligand (inhibin βA dimers). (C–E) µCT of representative distal hindlimbs 14 days after transplantation of R206H-FAPs into SCID hosts. HO is pseudocolored green and volume given in mm3. (F) Quantification of HO volume at day 14 post-transplantation of R206H-FAPs. Each dot represents a single transplantation with group mean (black bar) and ± standard deviation (error bars) shown. Statistical significance was assessed by one-way ANOVA; ****=p ≤ 0.0001. PVO-pretreated FAPs received 1 µM PVO during the 7 day expansion period prior to in vitro or transplantation assays.
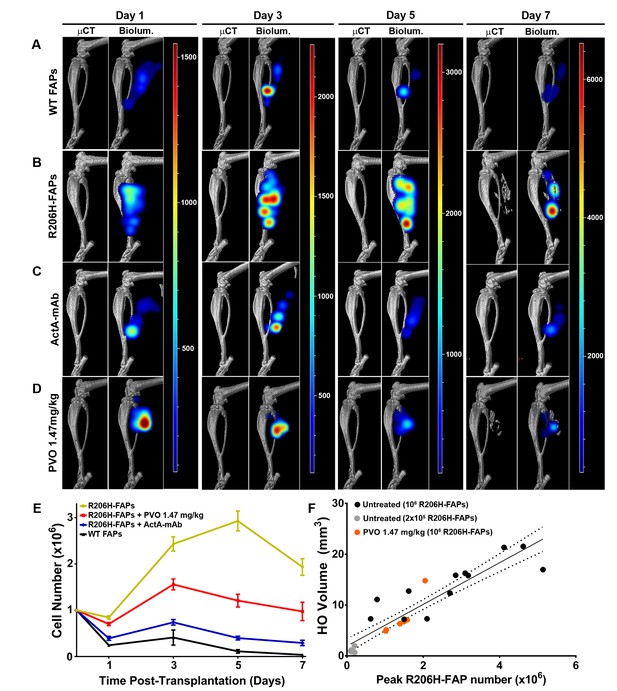
Activin sequestration restores wild-type population dynamics to bioluminescent R206H-FAPs.
(A–D) 3D tomographic bioluminescent source reconstruction following transplantation of 106 bioluminescent FAPs into SCID hosts. Paired images show µCT alone (left panel) and µCT combined with the corresponding 3D bioluminescent reconstruction (right panel). For each experimental group, the same mouse is shown from days 1–7. (E) Graphical representation of bioluminescent FAP population dynamics following transplantation into SCID hosts. Error bars represent ±standard error of the mean; see Figure 6—source data 1 for statistical analysis. (WT, n = 5; R206H-FAPs, n = 12; R206H-FAPs + PVO 1.47 mg/kg, n = 6; R206H-FAPs + ActA mAb, n = 4). (F) Linear regression analysis of the relationship between HO volume and peak FAP number. Untreated SCID hosts were transplanted with either 106 (black dots; n = 12) or 2 × 105 (grey dots; n = 6) R206H-FAPs, and PVO-treated SCID hosts were transplanted with 106 (red dots; n = 6) R206H-FAPs. Line of best fit (black line) and 95% confidence interval (dotted lines) are shown. R2 = 0.85 for untreated and 0.83 for PVO 1.47 mg/kg-treated groups.
-
Figure 6—source data 1
Two-way ANOVA analysis of bioluminescent FAP population dynamics eLife’s transparent reporting form.
- https://doi.org/10.7554/eLife.40814.017
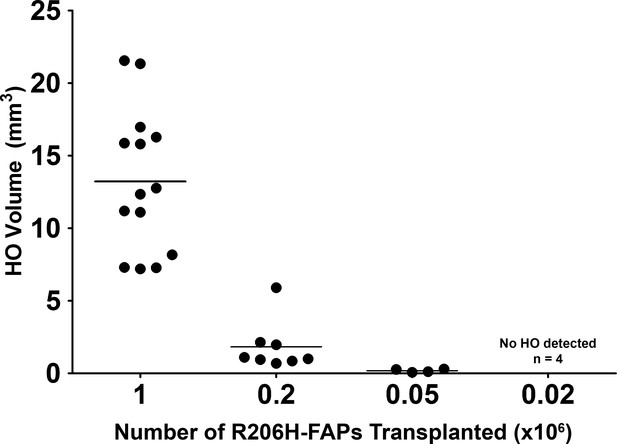
HO induction requires a threshold number of R206H-FAPs.
HO volume was correlated with the number of R206H-FAPs transplanted. HO was imaged by μCT and quantified with 3D Slicer software. Each dot represents a single transplantation and group mean is shown as a horizontal bar. HO was not detected following transplantation of 2 × 104 R206H-FAPs (n = 4).
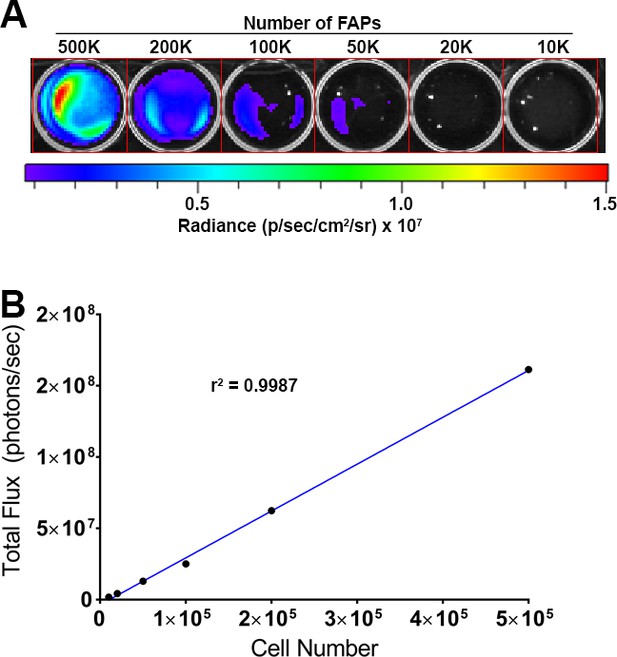
Quantification of FAP bioluminescence.
(A) Bioluminescent light emission from serially diluted luciferase-expressing FAPs. (B) Graphical representation of light emission from bioluminescent FAPs. Line of best fit (blue line) is shown. R2 = 0.9987.
Tables
Incidence of HO/overgrowth in untreated Pdgfrα-R206H mice
https://doi.org/10.7554/eLife.40814.004| Site | P14 (n = 10) | P28 (n = 11) | P42 (n = 6) |
|---|---|---|---|
| Ankle | 20% | 73% | 100% |
| Knee | 0% | 0% | 33% |
| Hip | 10% | 36% | 33% |
| Wrist | 0% | 45% | 66% |
| Abdomen | 0% | 0% | 0% |
| Forearm | 0% | 9% | 83% |
| Elbow/Upper Arm | 40% | 64% | 100% |
| Shoulder | 0% | 9% | 33% |
| Thoracic/Cervical Vertebrae | 0% | 18% | 50% |
| Lumbar/Sacral Vertebrae | 0% | 27% | 33% |
| Jaw | 10% | 18% | 100% |
| At least one site of HO/Overgrowth | 40% | 100% | 100% |
Incidence of HO/overgrowth in vehicle and PVO-treated P42 Pdgfrα-R206H mice
https://doi.org/10.7554/eLife.40814.007| Site | Vehicle (n = 7) | 0.735 mg/kg PVO (n = 4) | 1.47 mg/kg PVO (n = 5) |
|---|---|---|---|
| Ankle | 86% | 0% | 20% |
| Knee | 29% | 75% | 80% |
| Hip | 14% | 0% | 40% |
| Wrist | 0% | 50% | 0% |
| Abdomen | 57% | 25% | 40% |
| Forearm | 14% | 0% | 0% |
| Elbow/Upper Arm | 100% | 50% | 0% |
| Shoulder | 57% | 75% | 80% |
| Thoracic/Cervical Vertebrae | 14% | 25% | 80% |
| Lumbar/Sacral Vertebrae | 29% | 75% | 100% |
| Jaw | 43% | 0% | 40% |
| At least one site of HO/Overgrowth | 100% | 100% | 100% |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.40814.018





