Gi/o protein-coupled receptors in dopamine neurons inhibit the sodium leak channel NALCN
Figures
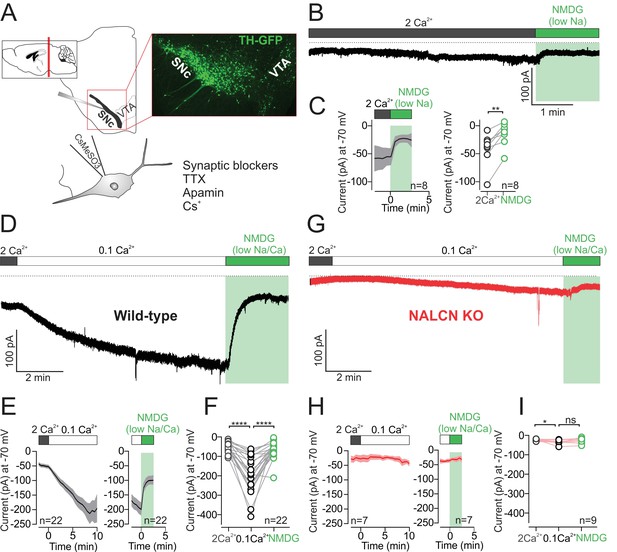
Low external calcium potentiates NALCN current in SNc dopamine neurons from adult mice.
(A) Top, schematic of a coronal section (left) and confocal image of coronal section from TH-GFP mouse. Bottom, schematic of recording solutions, with cesium based internal and external sodium leak isolation cocktail. (B) Example trace of isolated sodium current recorded at −70 mV from wild-type mouse SNc dopamine neuron before and after Na+ replacement with NMDG. (C) Left, Average time course of isolated sodium current before and after replacement of external Na+ by NMDG. Right, summary plot of isolated sodium current (pA) recorded at −70 mV in control (gray) and in NMDG solution (green). (D) Example trace of isolated sodium leak current showing potentiation of the sodium leak in 0.1 mM Ca2+ solution and the block induced by Na+ substitution by NMDG. (E) Time course of the averaged 0.1 mM Ca2+ mediated current (left) and the block of this current after replacement of Na+ by NMDG (right). (F) Summary plot of isolated sodium current amplitude at −70 mV in control (gray), 0.1 mM Ca2+ (black) and after Na+ substitution by NMDG (green). (G) Example trace of isolated sodium leak current in NALCN cKO mouse. Note, low external Ca2+ and Na+ substitution with NMDG has relatively little effect on holding current. (H) Same as in E but in NALCN KO mice. (I) Summary plot of isolated sodium current in control (open symbols, gray), 0.1 mM Ca2+ (black) and after sodium substitution (green). *p < 0.05; **p < 0.01****p < 0.0001.
-
Figure 1—source data 1
Data plotted in Figure 1.
- https://doi.org/10.7554/eLife.40984.003
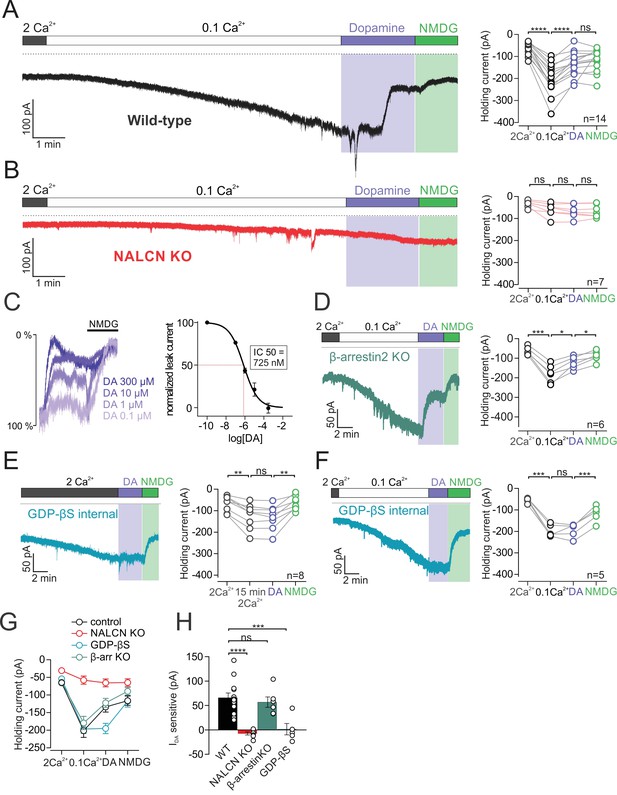
D2 receptor activation inhibits NALCN through a G-protein dependent mechanism.
(A)Left, Example trace of isolated sodium leak current in wildtype dopamine neuron recorded at −70 mV in 2 mM Ca2+ and 0.1 mM Ca2+, followed by bath application of 100 µM dopamine and subsequent substitution of Na+ with NMDG. Right, Summary plot of data in different conditions. (B) Example trace of isolated sodium leak current in NALCN cKO mice. Note, dopamine inhibition of sodium leak is abolished. (C) Left, Example traces showing inhibition of sodium leak current induced by 100 nM (n = 5), 1 μM (n = 3), 10 μM (n = 3), and 300 μM (n = 4) dopamine followed by block of leak current in NMDG solution. Traces have been normalized to control current amplitude. Right, Concentration-response curve for inhibition of leak current by dopamine. (D) Example trace of leak current in β-arrestin2 KO mice. Right, Summary plot. (E) Left, Example trace of the isolated sodium leak current recorded with GDP-βS added to internal solutions. Dopamine applied to cell in 2 mM Ca2+ external solutions. Note the slow increase in leak current with GDP-βS internal. Right, Summary plot. (F) Example trace of sodium leak current with GDP-βS internal solution. Dopamine applied to cell in 0.1 mM Ca2+ external solutions. Right, Summary plot of data. (G) Summary plot of data shown in A, B, D and E. (H) Bar plot of the dopamine-sensitive current shown in A, B, D and E. *p < 0.05; **p < 0.01; ***p < 0.001; ****p < 0.001.
-
Figure 2—source data 1
Data plotted in Figure 2.
- https://doi.org/10.7554/eLife.40984.005
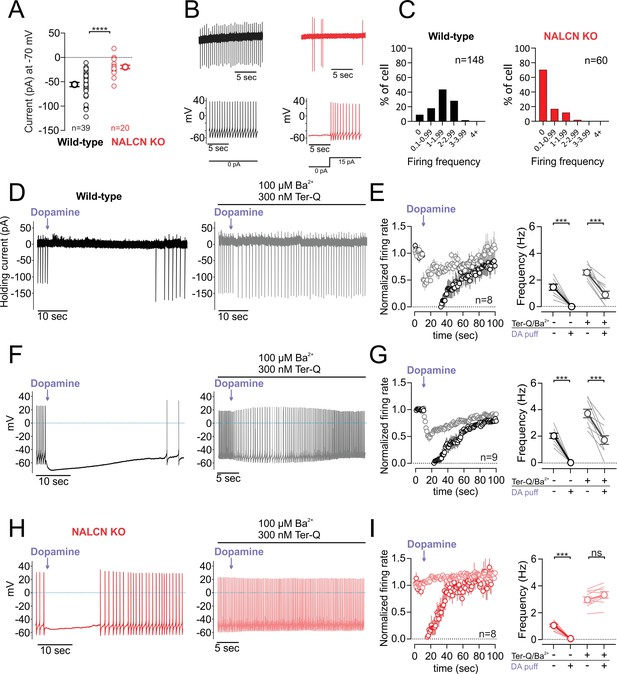
D2 receptor inhibition of NALCN slows spontaneous firing.
(A) Summary plot of isolated sodium leak currents measured at −70 mV in wildtype (black) versus NALCN cKO mice (red). (B) Cell-attached (top) and whole-cell (bottom) recordings of pacemaking from dopamine neurons recorded in wildtype (black) and NALCN cKO mice (red). (C) Histogram plots of firing rates obtained during cell-attached recordings in wildtype and NALCN cKO mice. (D) Effect of dopamine puff (300 µM, 1 s) on the firing rate of dopamine neuron recorded in cell-attached mode with 2 mM external Ca2+, before (black) and after block of GIRK channels by bath application of Ba2+ and tertiapin-Q (gray). (E) Left, Averaged normalized firing frequency plotted against time showing the effect of dopamine puff in control conditions (black) and after the block of GIRK channels (gray). Right, Averaged absolute firing rate before and after dopamine puff in control conditions and after the block of GIRK channels. (F,G) Same experiment as D and E but in whole-cell configuration. (H) and (I) Same as F and G in NALCN cKO mice. ***p < 0.001.
-
Figure 3—source data 1
Data plotted in Figure 3.
- https://doi.org/10.7554/eLife.40984.011
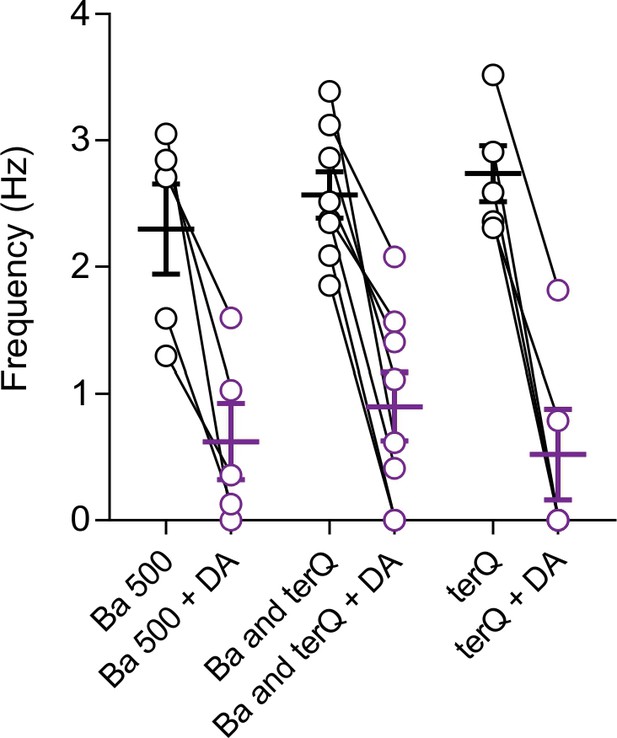
Effect of D2 receptor-mediated inhibition of NALCN on spontaneous firing recorded in the presence of different GIRK blocker combinations.
Summary plot showing dopamine inhibition of cell-attached firing. Data from individual neurons are shown as open symbols. Recordings were made in the presence of 500 μM barium chloride (left), 100 μM barium chloride plus 300 nM tertiapin-Q (middle), and 300 nM tertiapin-Q alone (right). For the barium (500 μM) only and barium (100 μM) plus terQ experiments, extracellular solutions contained CNQX, D-APV and picrotoxin to block AMPA, NMDA and GABA-A receptors. For terQ only experiments, CGP 55845, SCH 39166 and nomifensin were added to extracellular solutions to block GABA-B and D1 receptors as well as dopamine transporters. Addition of CGP 55845, SCH 39166 and nomifensin to the bath had no effect on the D2 receptor mediated inhibition of firing (dopamine inhibition of firing, Ba and terQ versus terQ condition, p = 0.54). This suggests that the inhibition of firing observed here is produced by direct activation of D2 receptors present on dopamine neurons and are unlikely to involve circuit effects.
-
Figure 3—figure supplement 1—source data 1
Data plotted in Figure 3—figure supplement 1.
- https://doi.org/10.7554/eLife.40984.008
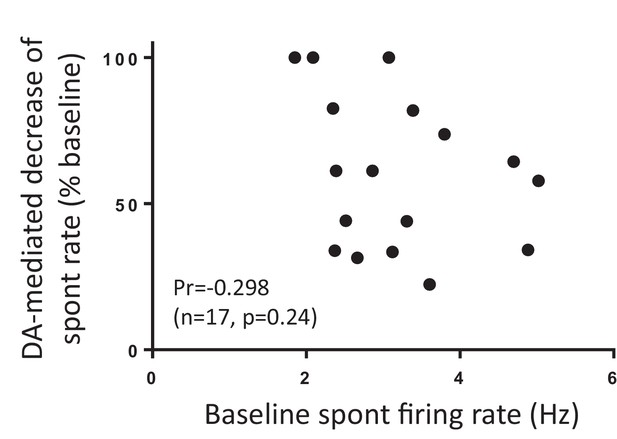
Lack of correlation between dopamine inhibition of firing and the rate of spontaneous firing.
Plot of the percent reduction of the firing rate following puff application of dopamine versus pacemaker firing rate immediately preceding the puff. Cells were recorded in the presence of 100 μM Ba2 +and 300 nM terQ. Analysis of points shows little to no correlation (Pr = −0.298, n = 17, p = 0.24).
-
Figure 3—figure supplement 2—source data 1
Data plotted in Figure 3—figure supplement 2.
- https://doi.org/10.7554/eLife.40984.010
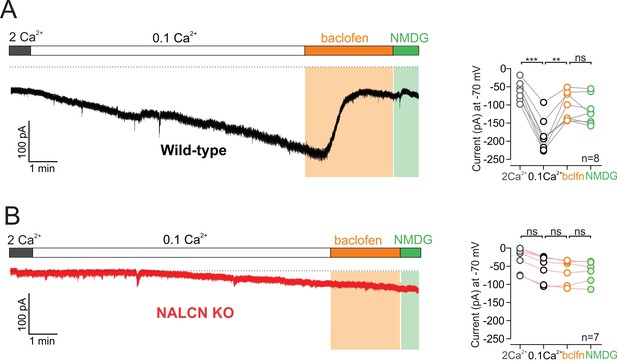
GABA-B receptor activation inhibits NALCN.
(A) Left, Example trace of showing effect of 10 µM baclofen on isolated sodium leak current recorded in wildtype mice. Note, Na+ substitution with NMDG produces no further effect following baclofen. Right, Summary plot. (B) Example trace of showing effect of baclofen on sodium leak current recorded in NALCN cKO mice.
-
Figure 4—source data 1
Data plotted in Figure 4.
- https://doi.org/10.7554/eLife.40984.013
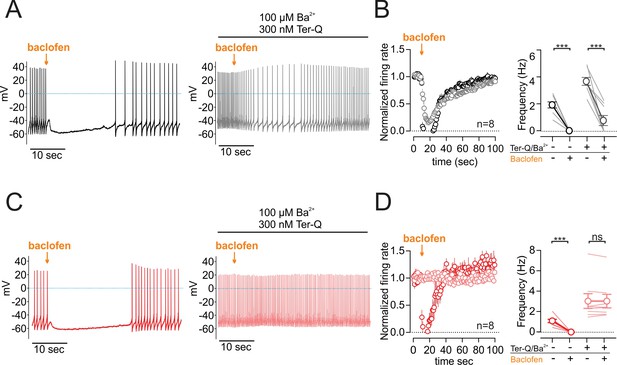
GABA-B receptor inhibition of NALCN slows spontaneous firing.
(A) Effect of baclofen puff (10 µM, 1 s) on the firing rate of dopamine neuron recorded in whole-cell configuration with 2 mM external Ca2+, before (black) and after block of GIRK channels by Ba2+ and tertiapin-Q (gray). (B) Left, Averaged normalized firing frequency plotted against time showing the effect of 10 µM baclofen puff in control (black) and after the block of GIRK channels (gray). Right, Absolute firing rates before and after baclofen puff in control conditions and after the block of GIRK channels. (C) and (D). Same as C and D in NALCN cKO mice. **p < 0.01; ***p < 0.001.
-
Figure 5—source data 1
Data plotted in Figure 5.
- https://doi.org/10.7554/eLife.40984.015
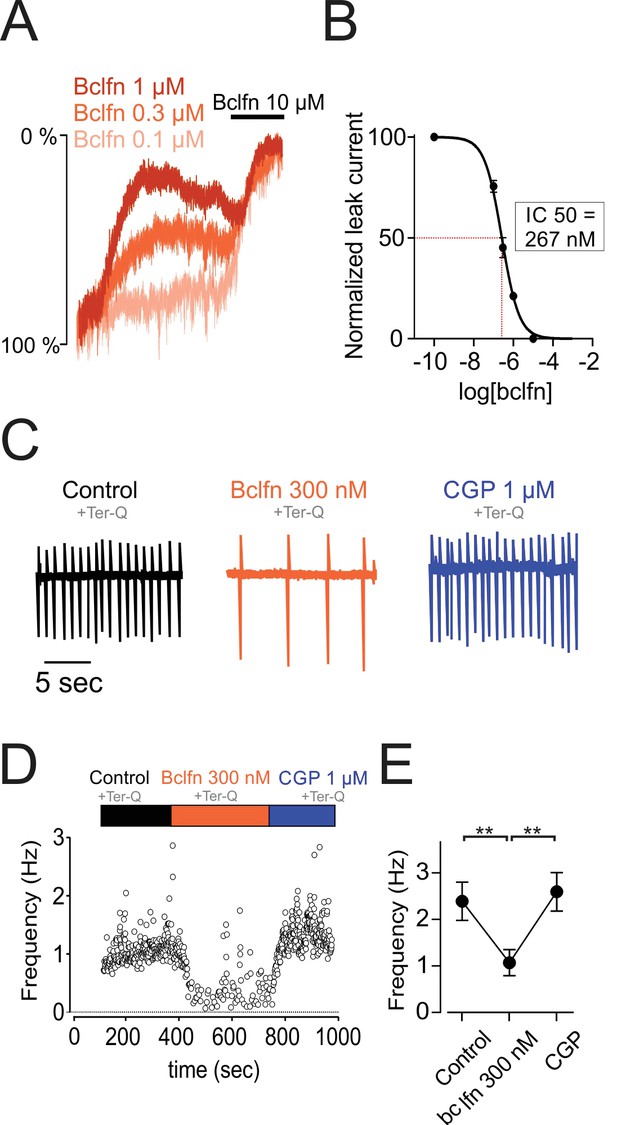
NALCN currents are highly sensitive to inhibition by GABA-B receptors.
(A) Example traces showing inhibition of sodium leak current by 100 nM (n = 4), 300 nM (n = 5), 1 μM (n = 5), and 10 μM (n = 5) baclofen. Traces have been normalized to control current amplitude. (B) Concentration-response curve of NALCN channel block by baclofen. (C) Cell-attached recording in control conditions (black), in the presence of 300 nM baclofen (orange) and after blocking GABA-B receptors with 1 µM CGP 55845 (blue). All recordings were made in the presence of tertiapin-Q (300 nM). (D) Time course of the instantaneous firing rate for the cell shown in C. (E) Summary plot of firing frequencies of 7 cells in experiments similar to those shown in C,D. Averaged values are shown as closed symbols. **p < 0.01.
-
Figure 6—source data 1
Data plotted in Figure 6.
- https://doi.org/10.7554/eLife.40984.017
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.40984.018





