Large and fast human pyramidal neurons associate with intelligence
Figures
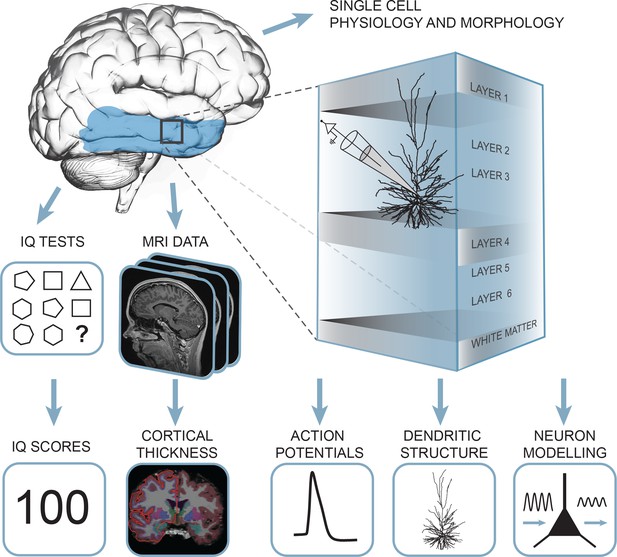
Summary of the approach: multidimensional data set from human subjects contained single cell physiology, neuronal morphology, MRI and IQ test scores (WAIS FSIQ).
The area of the brain highlighted in blue indicates the location of cortical thickness measurements, black square indicates the typical origin of resected cortical tissue.
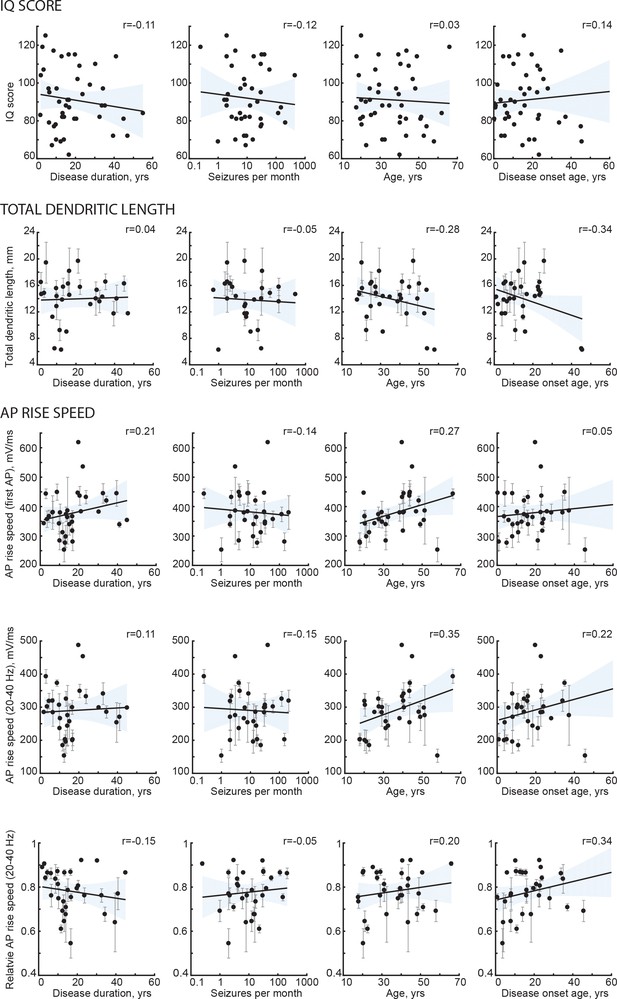
Subject disease history and age do not correlate with IQ and neuronal morphology.
Black lines are the regression lines, shading represents 95% confidence intervals, data are mean per subject ±standard deviation.
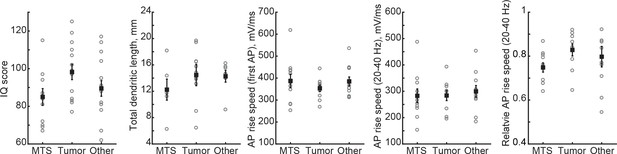
Neuronal morphology, IQ or AP rise speed are not different across patient groups.
Open circles represent mean per subject, filled circles are mean per group ±SEM.
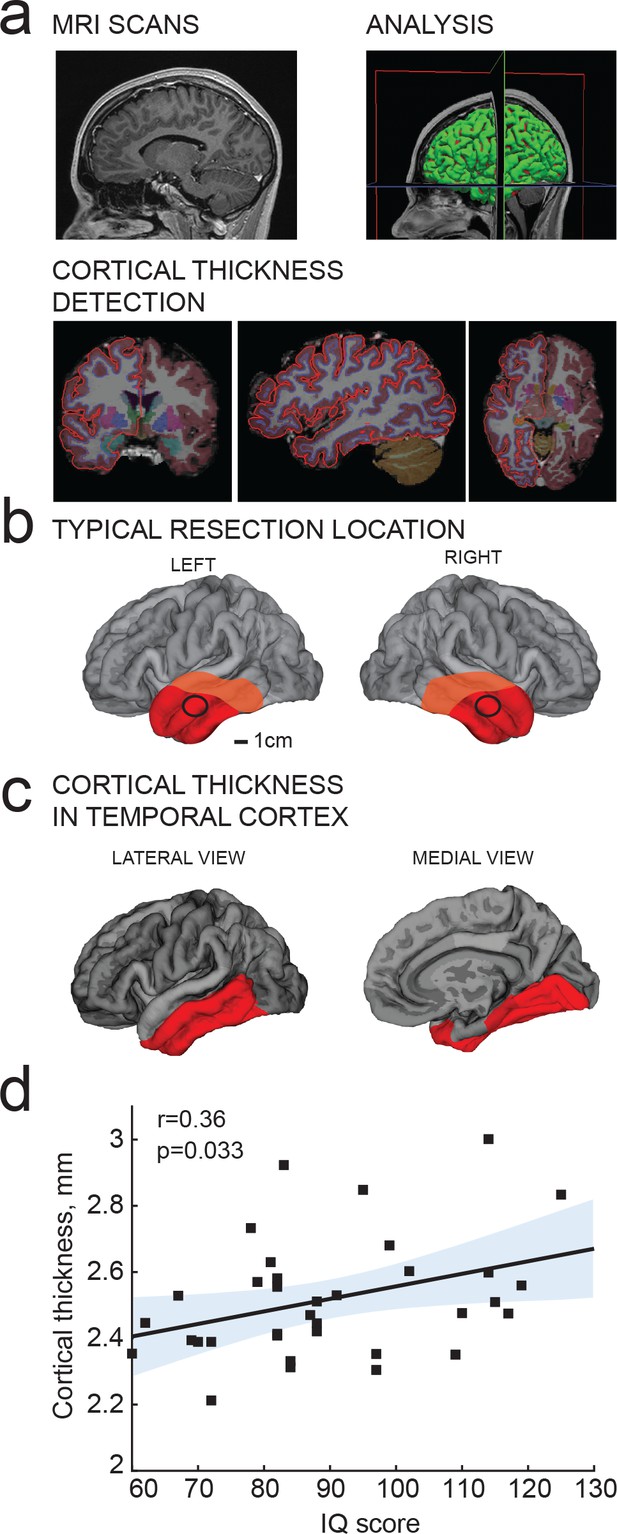
IQ scores positively correlate with cortical thickness of the temporal lobe.
(a) MRI analysis pipeline: (1) Presurgical MRI T1-weighted scans; (2) Morphometric analysis; (3) Detection of cortical thickness from pial and white-grey matter boundaries; (b) Typical resection location for tissue used in this study is marked by a black circle; average total resected area from the patient is shown in red and maximum resected area in orange; (c) selection of temporal cortical area for correlations with IQ in b (red). (d) Average cortical thickness in temporal lobe (from area highlighted in red in c) positively correlates with IQ scores from the same subjects (n subjects = 35). Here and in figures below, Pearson correlation coefficients and p-values are reported in graph insets, the solid line represents linear regression (R2 = 0.13), shaded area indicates 95% confidence bounds of the fit.
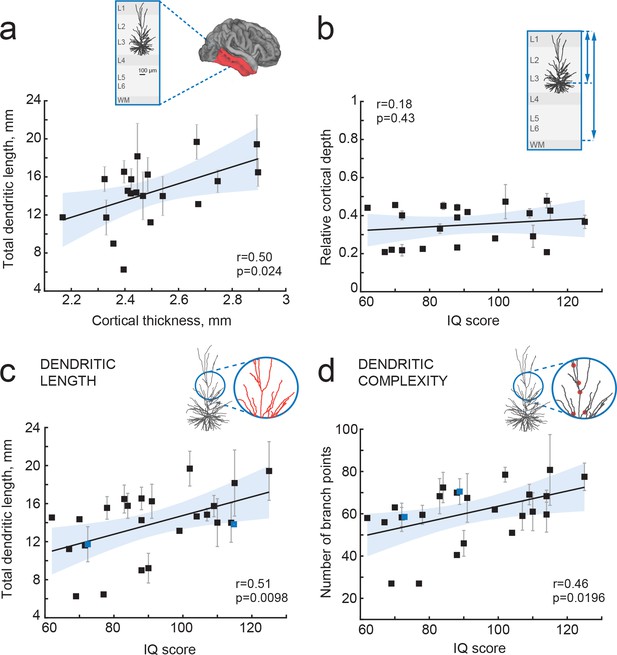
IQ scores positively correlate with dendritic structure of temporal cortical pyramidal cells.
(a) Average total dendritic length in pyramidal cells in superficial layers of temporal cortex positively correlates with cortical thickness in temporal lobe from the same hemisphere (area shaded in a, n subjects = 20; n neurons = 57, R2 = 0.25). Inset shows a scheme of cortical tissue with a digitally reconstructed neuron and the brain area for cortical thickness estimation (red) (b) Cortical depth of pyramidal neurons, relative to cortical thickness in temporal cortex from the same hemisphere, does not correlate with IQ score (n subjects = 21, R2 = 0.03). Inset represents the cortical tissue, blue lines indicate the depth of neuron and cortical thickness (c) Total dendritic length (TDL) and (d) number of dendritic branches positively correlate with IQ scores from the same individuals (n subjects = 25, n neurons = 72, TDL R2 = 0.26, Branch points R2 = 0.22). Symbols highlighted in blue were shifted along the x axis for display purposes. Data are mean per subject ±standard deviation.
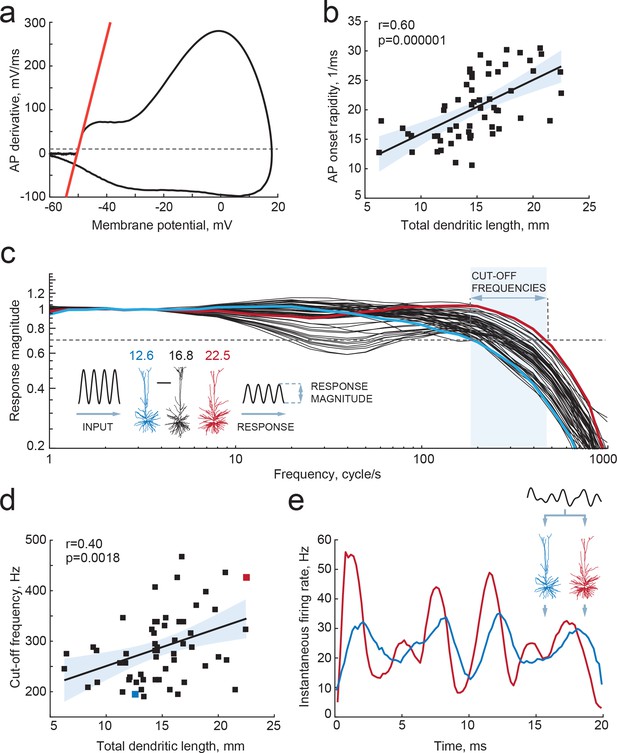
Larger dendrites lead to faster AP onset and improved encoding properties.
(a,b) Higher TDL results in faster onsets of model-generated APs: (a) example phase plot of an AP is shown with a red line representing onset rapidity - slope of AP derivative at 10 mV/ms (grey dashed line); (b) onset rapidity values of simulated APs positively correlate with TDL (R2 = 0.36). (c) Model neurons received simulated sinusoidal current-clamp inputs and generated spiking responses of different magnitudes and frequencies. Red and blue traces are response magnitudes of example neurons with low (blue) and large (red) TDLs; inset shows examples of morphological reconstructions with their TDLs in mm shown above. Cut-off frequencies are defined within the frequency range (shaded area) at which the model neuron can still track the inputs reliably (produce response of 0.7 response magnitude, dashed line). (d) Cut-off frequencies positively correlate with TDL (R2 = 0.16; example neurons from panel (c) are highlighted by the same colors). (e) Responses to the same input in two example neurons from panels (b) and (c): instantaneous firing frequency of the model neuron with large TDL (red) follows the input with higher temporal precision than the model neuron with smaller TDL (blue).
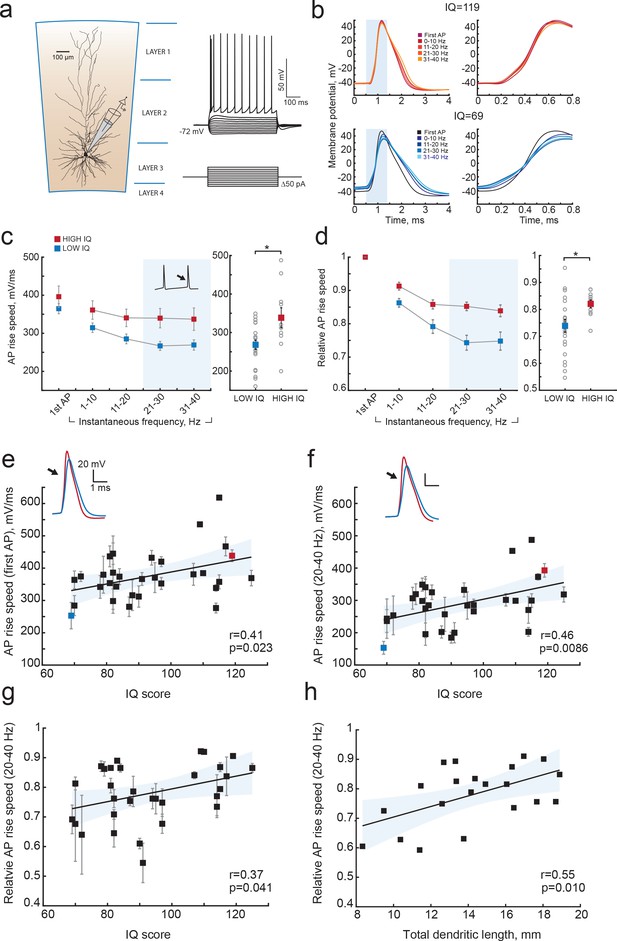
Higher IQ scores associate with faster AP initiation.
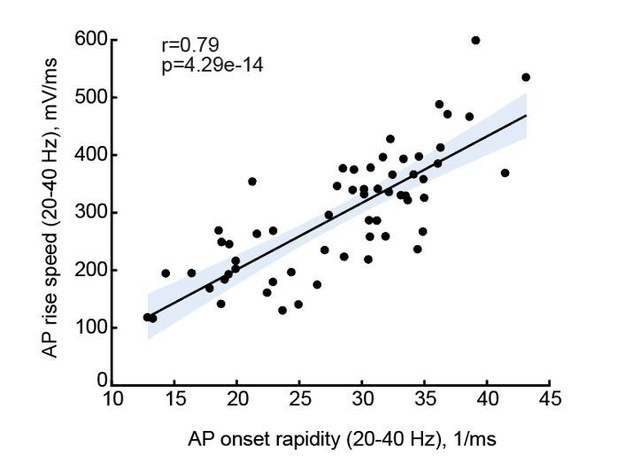
(a) Scheme of a whole-cell recording showing biocytin reconstruction of a pyramidal neuron from human temporal cortex. Right: typical voltage responses to depolarizing somatic current injections. (b) Examples of AP traces at increasing instantaneous firing frequencies (frequency is shown in color code in insets) recorded from a subject with IQ = 119 (above panel, red) and a subject with IQ = 69 (lower panel, blue). AP rising phase in shaded area is displayed to the right (c) APs from subjects with higher IQ are better able to maintain their rise speed at increasing frequencies. Average (per neuron and subject) AP rise speed and (d) relative to first AP rise speeds in neurons from subjects with IQ < 100 (red, n subjects = 21, n neurons = 91) and subjects with IQ > 100 (blue, n subjects = 10, n neurons = 38) are displayed against instantaneous firing frequency. Right: data points in shaded area are shown as averaged values for 20–40 Hz (filled squares are group means, open circles are mean rise speeds per subject), *p<0.05. (e) IQ scores positively correlate with the rise speeds of first AP in the train (n subjects 31, n neurons = 129; R2 = 0.17), (f) AP rise speed at 20–40 Hz (same data as right panel in (c), R2 = 0.21) and (g) relative AP rise speeds at 20–40 Hz (same data as right panel in (d), R2 = 0.14). (h) Larger neurons show less slowing of AP rise speed at higher frequencies: relative AP rise speeds at 20–40 Hz for individual neurons are plotted as a function of their TDL (n = 21 neurons, R2 = 0.30). In c,d data are mean per subject ±S.E.M; in e, f, g data are mean ±standard deviation.
Tables
Subject details.
https://doi.org/10.7554/eLife.41714.007| Patient number | IQ | Age | Diagnosis | Gender | Antiepileptic drugs |
|---|---|---|---|---|---|
| 1 | 88 | 41 | Tumor | M | CBZ |
| 2 | 78 | 21 | Other | F | LEV; VPA |
| 3 | 119 | 66 | Tumor | F | None |
| 4 | 88 | 31 | Tumor | F | CBZ; LEV |
| 5 | 81 | 51 | Other | F | CLB; LTG; OXC |
| 6 | 69 | 58 | MTS | F | CZP |
| 7 | 107 | 28 | Tumor | M | LTG; LEV |
| 8 | 115 | 29 | MTS | F | LTG; TPM |
| 9 | 125 | 20 | Tumor | M | CBZ; LEV |
| 10 | 84 | 27 | Tumor | F | CBZ, LTG |
| 11 | 110 | 41 | Tumor | M | CBZ; LTG |
| 12 | 87 | 18 | MTS | M | OXC |
| 13 | 67 | 23 | MTS | F | LEV; OXC |
| 14 | 72 | 53 | MTS | M | CBZ; CLB |
| 15 | 97 | 25 | Tumor | M | None |
| 16 | 104 | 19 | Other | M | CLB; OXC |
| 17 | 88 | 48 | Other | F | CBZ |
| 18 | 65 | 38 | MTS | F | CBZ; LEV |
| 19 | 62 | 40 | Other | F | None |
| 20 | 84.5 | 31 | Other | F | None |
| 21 | 88 | 35 | Other | F | CZP; LCS; LTG; LEV |
| 22 | 77 | 54 | Tumor | M | VPA |
| 23 | 91 | 25 | Other | M | CLB; LCS; LEV |
| 24 | 70 | 31 | MTS | F | CBZ; CLB |
| 25 | 114 | 49 | Other | M | CBZ; CLB; LEV |
| 26 | 83 | 25 | Tumor | M | None |
| 27 | 109 | 45 | Other | F | CBZ; CLB; LTG |
| 28 | 102 | 47 | Tumor | F | CBZ |
| 29 | 67 | 22 | Other | M | CLB; LTG; LEV |
| 30 | 97 | 38 | MTS | M | CBZ |
| 31 | 79 | 40 | MTS | F | CBZ, CLB, LTG, LEV |
| 32 | 117 | 44 | Other | M | LCS; VPA |
| 33 | 99 | 30 | Tumor | F | CLB; OXC |
| 34 | 72 | 44 | MTS | M | LTG; LEV |
| 35 | 82 | 41 | Other | F | CBZ, LEV, TPM |
| 36 | 95 | 29 | Other | M | CBZ; PB |
| 37 | 91 | 20 | Other | F | CBZ; LEV |
| 38 | 82 | 21 | Tumor | M | CBZ; LCS; LTG; LEV |
| 39 | 115 | 40 | MTS | M | CBZ; LEV |
| 40 | 97 | 48 | MTS | F | CBZ; ZNS |
| 41 | 94 | 40 | MTS | F | CLB; LTG; ZNS |
| 42 | 81 | 44 | MTS | M | CBZ; LTG |
| 43 | 70 | 33 | MTS | F | CBZ; CLB; LEV |
| 44 | 82 | 51 | Other | M | CBZ |
| 45 | 114 | 18 | Tumor | F | OXC |
| 46 | 90 | 23 | Other | M | OXC |
-
M = male; F = female;
Antiepileptic drugs specified: Carbamazepine (CBZ); Lamotrigine (LTG); Levetiracetam (LEV); Topiramate (TPM); Clobazam (CLB); Oxcarbazepine (OXC); Clonazepam (CZP); Phenobarbital (PB); Phenytoin (PHT); Lacosamide (LCS); Sodium valproate (VPA); Zonisamide (ZNS)
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.41714.011





