R-propranolol is a small molecule inhibitor of the SOX18 transcription factor in a rare vascular syndrome and hemangioma
Figures
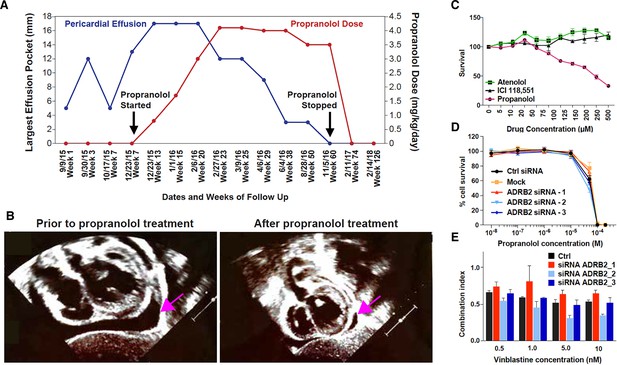
Propranolol treatment alleviates pericardial effusion severity in HLTRS patient and mediates β-adrenergic independent effects.
(A) A 17 months old HLTRS patient was treated with propranolol, starting at 0.8 mg/kg/day in three divided doses and increasing gradually every 2–3 weeks to maximum of 4.1 mg/kg/day (red curve). In parallel the volume of ventricular peri-cardial effusion was measured at the end of the diastole (blue curve). (B) Echocardiography revealed that pericardiocentesis was not required anymore after propranolol treatment due to significant reduction in pericardial effusion (pink arrow) which did not recur as of June 2018 (time of the study). (C) Fetal endothelial colony forming cells (ECFC) were isolated from term placenta from healthy donors, expanded for three passages, and subjected to propranolol treatment followed by analysis of survival (percentage) as compared to vehicle control (DMSO). Propranolol affected the survival of ECFC at equivalent doses whereas Atenolol (specific β1 blocker) and ICI118,551 (specific β2) did not. (D) Cell survival assay performed on ISO-HAS angiosarcoma cells after transfection with three different siRNA sequences targeting ADRB2 and following 72 hr incubation with propranolol (racemic mixture). Alamar Blue assay ; Points, mean of at least four independent experiments ; Error bars, standard error. (E) Combination indexes of propranolol and vinblastine in ISO-HAS angiosarcoma cells after transfection with three different siRNA sequences targeting ADRB2 and following 72 hr drug incubation (50uM). Alamar Blue assay ; Bars, mean of at least four independent experiments ; Error bars, standard error. Statistical analysis for C was performed using Mann-Whitney non parametric t-test and for D-E using an unpaired two-tailed t test.
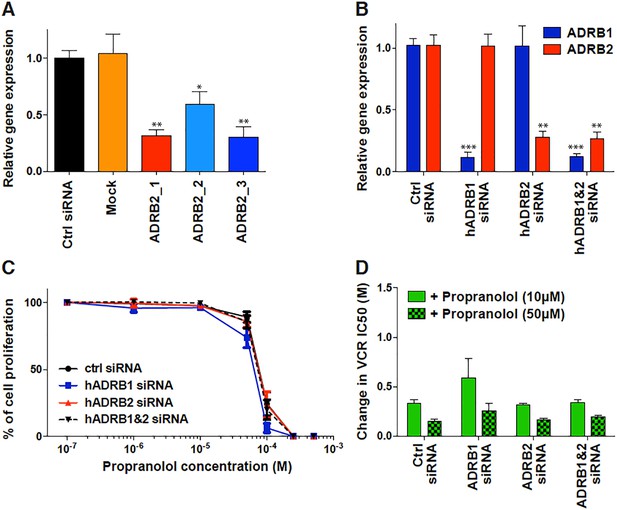
β-adrenergic independent effects of propranolol.
(A and B) β-adrenergic receptor gene expression following 72 hr transfection with siRNA in ISO-HAS angiosarcoma cells (A) and SHEP neuroblastoma cells (B), as determined by qRT-PCR using specific primers for ADRB1 and ADRB2, and compared with YWHAZ as housekeeping gene. (C) Cell viability assay performed by Alamar Blue on SHEP neuroblastoma cells following siRNA transfection and 72 hr drug incubation; Data points, mean of at least four independent experiments; Error bars, standard error. (D) Change in vincristine EC50 (i.e. concentration causing a 50% reduction in cell viability after 72 hr drug incubation) in the presence of 10 or 50 µM propranolol as compared with vincristine alone following transfection of SHEP neuroblastoma cells with either ADRB1, ADRB2 or both siRNA. Bars, mean of at least four independent experiments ; Error bars, standard error. Classic unpaired Student's t test with the following legend: *p<0.05; **p<0,01; ***p<0001.
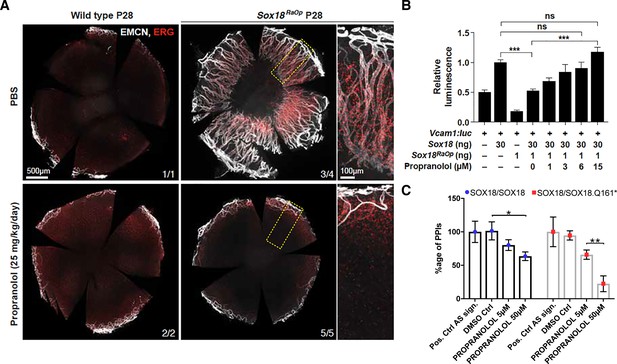
Propranolol rescues corneal neo-vascularization phenotype in a mouse pre-clinical model of HLTRS and SOX18 dominant-negative transcriptional repression via protein-protein interaction disruption.
(A) Fluorescent images of corneal flat mounts, showing blood vessel penetration into the cornea at P28 stage using endothelial cell markers ERG and endomucin (EMCN). Sox18 WT and RaOp mice were treated from P8 to P28 with either vehicle PBS or propranolol. Propranolol has no obvious effect on WT cornea, but prevents CNV in RaOp pups. Number of predominant phenotype shown in bottom right. Scale bar left 500 µm, right 100 µm. (B) COS-7 cells were transfected with SOX18 responsive Vcam1:luciferase construct and a combination of Sox18 wild type plasmid DNA and RaOp plasmid DNA. RaOp behaves in a dominant negative fashion and is capable to inhibit SOX18 WT function even at low 30:1 (w/w DNA) allelic ratios. Addition of propranolol to the media rescues SOX18 dependent activity of the Vcam1 promoter in presence of RaOp. Effect is concentration dependent and normal SOX18 activity on this construct is achieved at 15 µM propranolol. Sox18. *** p-value≤0.001, Kruskal-Wallis multiple comparison test. Data shown is mean ± SD of n ≥ 8. (C) The bar graph shows ALPHAScreen signal as a measure of the level of protein-protein interaction between SOX18 and its mutant counterpart RaOp (red square) and SOX18 homodimer formation (blue dot) in absence or presence of propranolol treatment. Propranolol is a small compound with the ability to disrupt SOX18 self-recruitment. Statistical analysis in 2B one-way ANOVA with Bonferroni post-hoc test and in 2C ANOVA Sidak’s multiple comparison test. Analysis of the protein pair by ALPHAScreen assay was performed in three different biological experiment with three technical replicates.
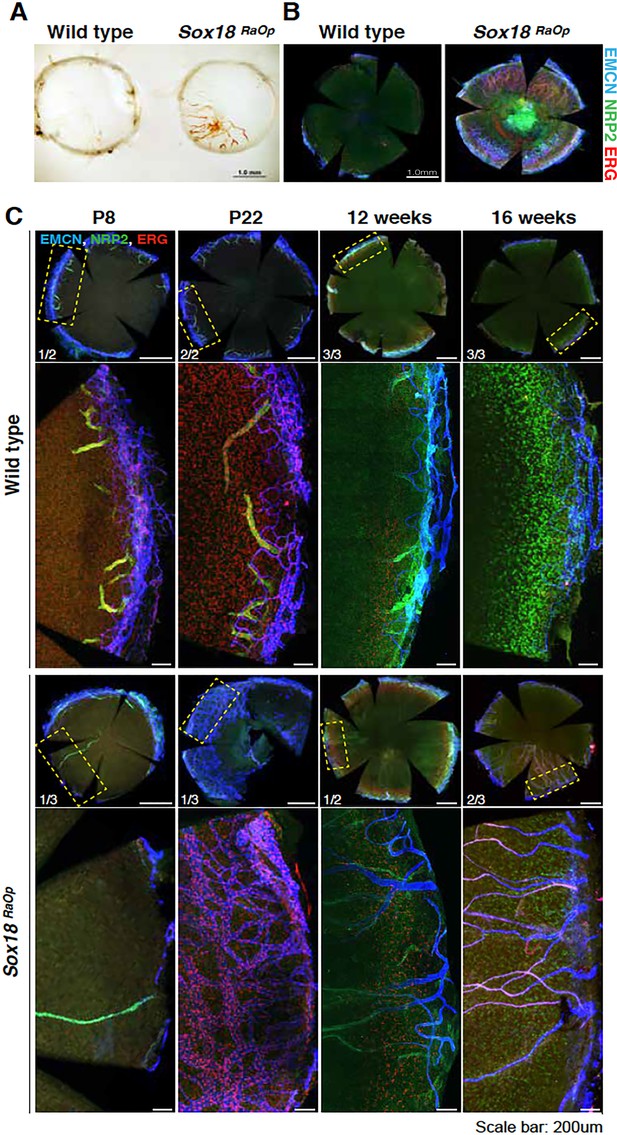
Time course of the corneal phenotype.
(A) Corneal flat mount from adults (8 weeks old mice) wild type control and RaOp ±animals showing blood vessel invasion. (B) Immunofluorescence further confirms blood (Endomucin, EMCN, blue, ERG, red) and lymphatic (Neuropillin-2, NRP2, green) vessels outgrowth in corneal tissues (8 weeks old mice). (C) Corneal tissues were harvested from mice at different stages, ranging from postnatal day 8 to 16 weeks of age. Penetrance of corneal vessels was analysed by immunofluorescence for vascular endothelial cells markers EMCN, NRP2 and ERG. At early stages (P8–P22), wild type pups have NRP2-positive, EMCN-negative vessels projecting from the sclera into the cornea. This is not observed at later stages. Onset of corneal neo-vessel formation in RaOp mice appears between P22 and 12 weeks. Scale bar whole corneas: 0.5 mm, detail: 0.2 mm.
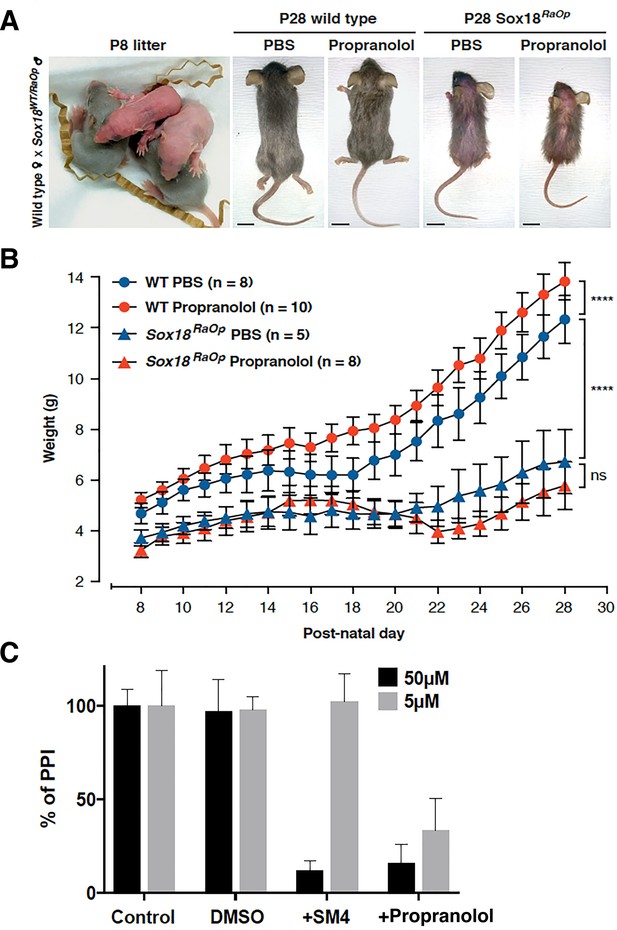
Effects of propranolol on the overall morphology of treated RaOp mice and disruption of SOX18/RBPJ protein-protein interaction.
(A) DBA/2JArc wild type females were crossed with a heterozygous RaOp male (B6D2-RaOp/J) to generate Sox18 mutant pups heterozygous for the RaOp allele. RaOp mice have a distinct sparse coat, and are smaller than wild type littermates. Pups were treated daily through oral gavage with vehicle PBS or 25 mg/kg/day propranolol, from P8 until P28. No obvious gross morphological defects were observed in propranolol treated mice compared to vehicle PBS. Scale bar 1 cm. (B) Weight of the mice was recorded daily throughout the time course of the treatment. RaOp weighted less than wild type littermates. Propranolol increased the mean weight of the wild type mice, but not of RaOp mice. (C) The bar graph represents ALPHAScreen signals as a measure of the protein-protein interaction between SOX18 and RBPJ (NOTCH signaling effector) transcription factors. Control conditions (ctrl and DMSO ctrl) show the reference signal for SOX18/RBPJ interaction, results are shown as a fold response compared to control. Upon addition of SOX18 small molecule inhibitor Sm4, PPI is disrupted at 50 μM, whereas propranolol showed efficacy from 5 μM. Experiments were performed in three independent replicates. **** padj ≤1e-4, 2way ANOVA Tukey multiple comparisons. Data shown is mean ± SEM, n-number is indicated in graph.
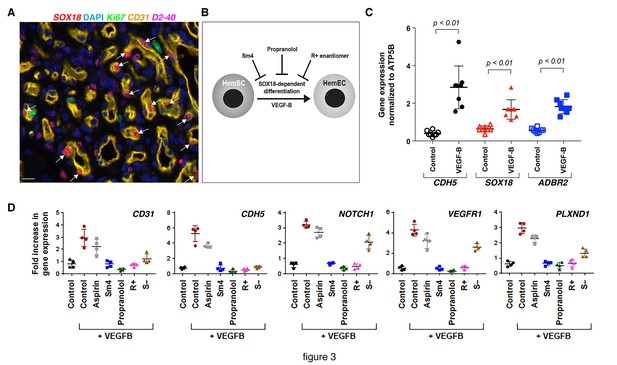
The R(+) enantiomer of propranolol and SOX18 small molecule inhibitor halt infantile hemangioma stem cell differentiation.
(A) Infantile) hemangioma tissue section stained for SOX18 (red), Ki67 (green), CD31 (orange), D2-40 (pink) and DAPI (blue) reveals the presence of SOX18 expression in a large subset of hemangioma endothelial cells (arrows). (B) Schematic representation of infantile hemangioma stem cell (HemSC) endothelial differentiation assay. VEGF-B stimulates HemSC to differentiate into hemangioma endothelial cells (HemEC). This differentiation process is inhibited by propranolol, the R(+) enantiomer of propranolol, and by SOX18 small molecule inhibitor Sm4 (all at 5 uM). (C) VEGF-B treatment of HemSC from four different infantile hemangiomas resulted in increased CDH5 (an endothelial cell marker), SOX18 and ADBR2 (β2 adrenergic receptor) mRNA. Means and standard deviations are shown. (D) The effects of SOX18 inhibitor Sm4, its scaffold aspirin as a negative control, propranolol and its purified R(+) and S(-) enantiomers on HemSC-to-HemEC differentiation from two infantile hemangioma patients. Endothelial differentiation markers, CD31 and CDH5 and hemangioma endothelial markers NOTCH1, PLXND1 and VEGFR1 under each treatment condition in four biological replicates, determined by qPCR, were standardized as described (Willems et al., 2008). Means and standard deviations are shown. Statistical analysis in 3C and 3D was done using one-way ANOVA, Fisher Tests, and two-tailed two independent sample T-Tests.
-
Figure 3—source data 1
Supplemental table for Figure 3D (p-values).
- https://doi.org/10.7554/eLife.43026.012
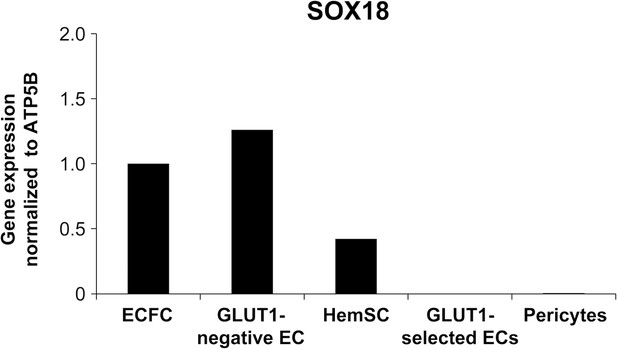
SOX18 expression in infantile hemangioma-derived cells.
Human ECFC served as a positive control for SOX18. Hemangioma-derived GLUT1-negative ECs, HemSC, GLUT1-positively selected ECs, and pericytes were analysed for SOX18 mRNA expression by qPCR analysis.
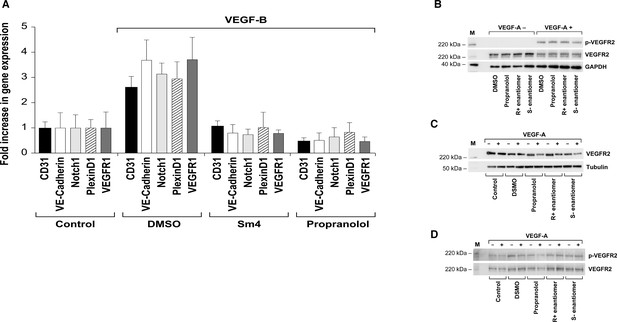
SOX18 inhibitor (Sm4) and propranolol block HemSC to EC differentiation.
(A) VEGF-B treatment of HemSC from four different infantile hemangiomas resulted in increased CD31 and CDH5 (endothelial cell markers), and hemangioma endothelial markers NOTCH1, PLXND1 and VEGFR1, determined by qPCR. Inclusion of SOX18 inhibitor (Sm4) or propranolol blocked the VEGF-B induced increases in these markers. Results from eight biological replicates were standardized as described (Willems et al., 2008). Means and standard deviations are shown. All P values were < 0.001 for Control versus DMSO (VEGF-B treated); DMSO versus Sm4 and DMSO versus propranolol. (B–D) VEGFR2 protein and phosphorylation were not affected by pre-treatment with propranolol, R + enantiomer or S(-) enantiomer (each tested at 5 uM). (B) Human endothelial colony forming cells (ECFC) were pre-treated for one hour with drug as indicated, stimulated with 25 ng/ml VEGF-A for 5 min, lysed and analysed by Western blotting. (C) HemSC were induced to differentiate for 5 days with VEGF-B; control indicates cells in differentiation media without VEGF-B. Differentiated cells were pre-treated for one hour with drugs as indicated, stimulated with 25 ng/ml VEGF-A for 5 min, and lysed for VEGFR2 Western blotting. (D) HemSCs were treated as in B, cell lysates were immunoprecipitated with anti-VEGFR2, followed by Western blotting for phosphorylated VEGFR2 and total VEGFR2. B-D, M indicates lane with molecular weight standards.
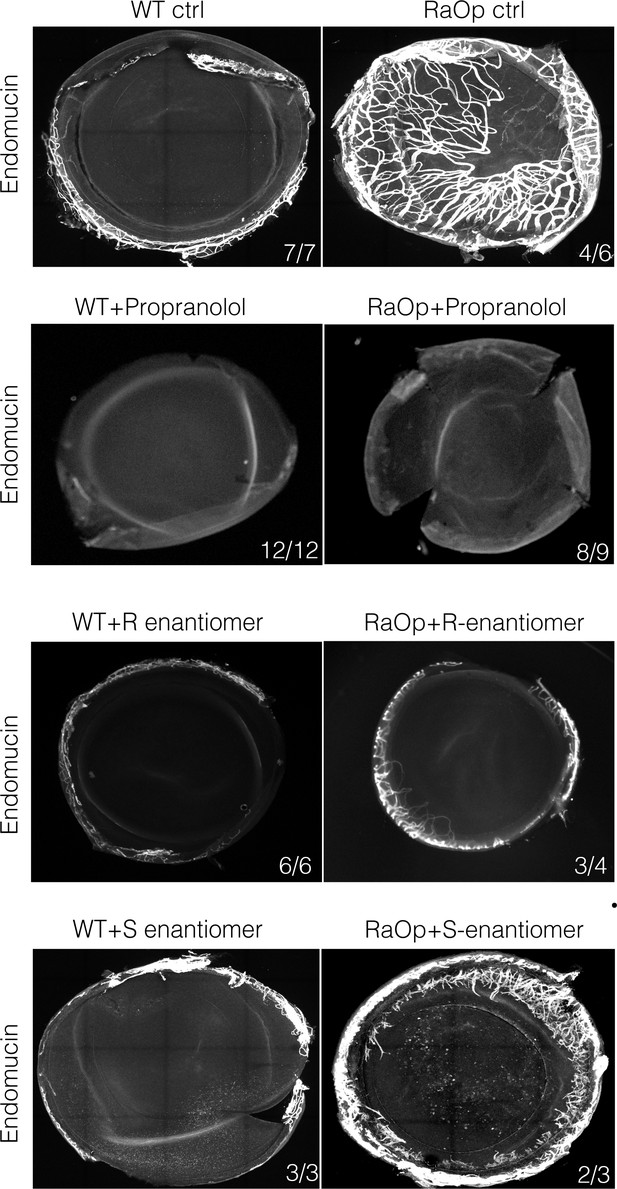
The racemic mixture Propranolol and its enantiomer (R(+) and S(-)) rescues corneal neo-vascularization phenotype in a mouse pre-clinical model of HLTRS.
Fluorescent images of corneal flat mounts, showing blood vessel penetration into the cornea at P28 stage using endothelial cell markers endomucin (EMCN). Sox18 WT and RaOp mice were treated from P8 to P28 with either vehicle PBS or propranolol or R-enantiomer or the S-enantiomer at 25 mg/Kg/day. Propranolol has no obvious effect on WT cornea, but prevents CNV in RaOp pups. Number of predominant phenotype shown in bottom right.
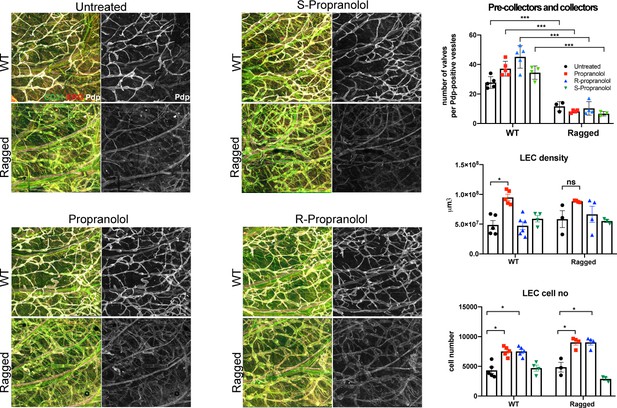
Propranolol and its R(+) and S(-) enantiomers do not rescue the dermal lymphatic vascular phenotype of the Ragged Opossum mutant mouse model.
Ear whole mount immunofluorescence for Podoplanin (white), ERG (red) and CD31 (green) was performed after wild type and RaOp mutant animals were treated by propranolol, S(-) and R (+)enantiomer (P1-P28 at 25 mg/kg/day). Quantification of the number of valves in pre-collector and collector vessels, LEC vessel density and LEC cell number showed that none of the drug treatment was efficient to mitigate the RaOp phenotype. n = 3–6 animal per genotypes and drug treatment.
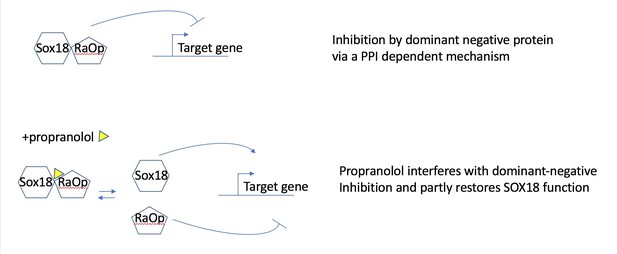
Propranolol rescues the RaOp protein-mediated inhibition by disrupting the interaction SOX18/SOX18RaOp.
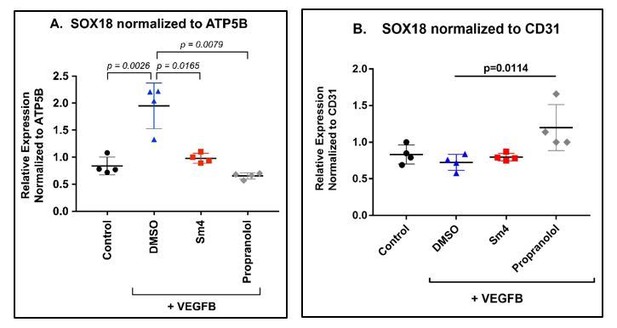
HemSC were treated without (control) or with VEGF-B for 5 days to induce endothelial differentiation.
Sm4 or propranolol were included in the differentiation medium at 5μM. DMSO, the vehicle, was added at the same dilution to serve as control. SOX18 was measured by qPCR and normalized to the house keeping transcript ATP5B (A) or to the pan-endothelial marker CD31 (B). Data points are from 4 different experiments carried out with HemSC from three different infantile hemangiomas.
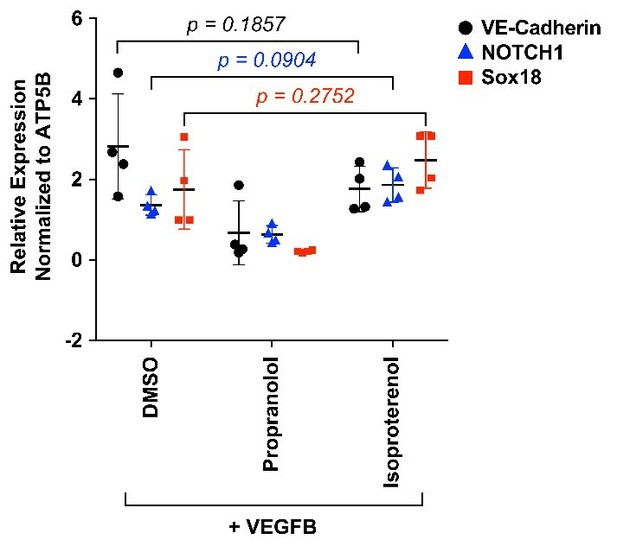
HemSC were treated with VEGF-B for 5 days to induce endothelial differentiation.
Propranolol or isoproterenol were included in the differentiation medium at 5μM, whereas DMSO, the vehicle, was added at the same dilution to serve as control. VE-cadherin, NOTCH1 and SOX18 were measured by qPCR. Data points are from 4 different experiments carried out with HemSC from three different infantile hemangiomas.
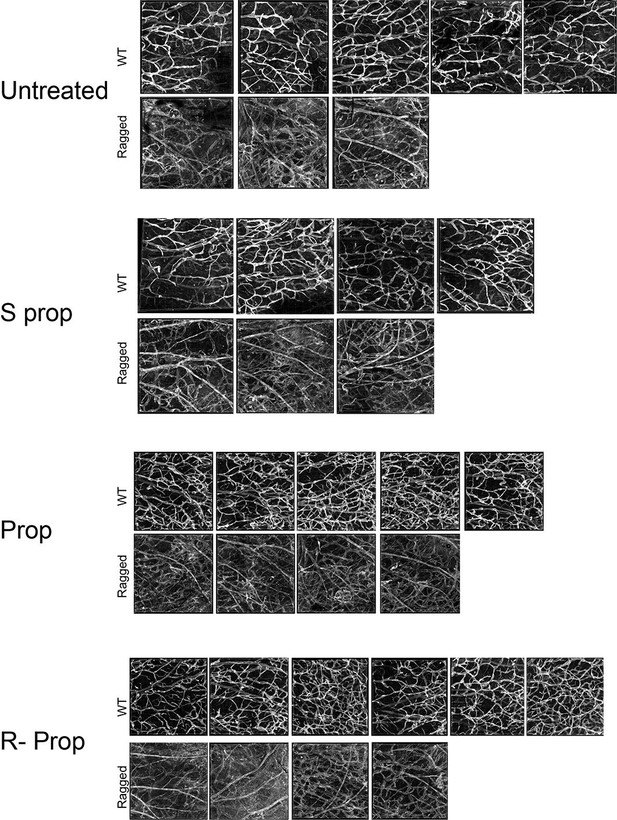
Images of whole mount skin immuno-stained for podoplanin.
Each image corresponds to the same region of interest from the inner skin flap of a mouse ear. Untreated WT, n=4; untreated ragged n=3; S(-) propranolol WT n=4; S(-) propranolol ragged n=3; propranolol WT n=5, propranolol ragged n=4; R(+) propranolol WT n=6, R(+) propranolol ragged n=4.
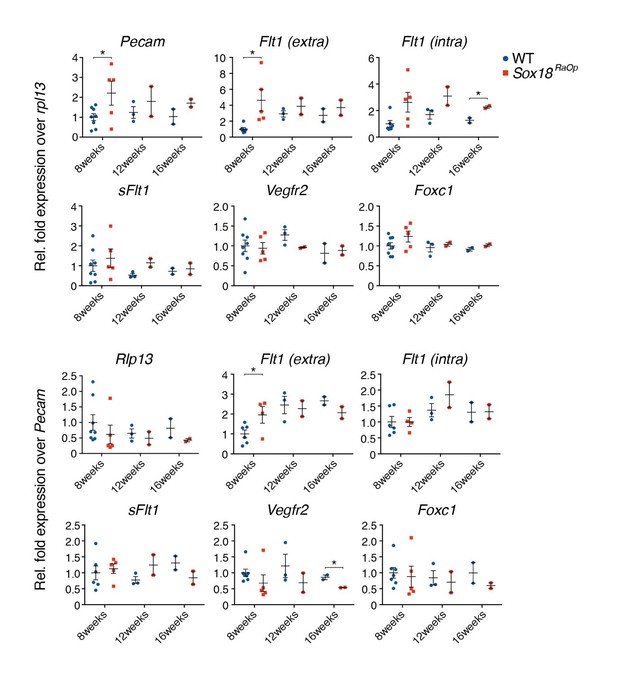
Whole corneas were harvested from 8, 12 and 16 week old mice and prepared for qRT-PCR analysis.
Gene transcripts for Pecam, Flt1, Vegfr2 and Foxc were normalized to either housekeeper gene Rpl13 (top row) or vascular gene Pecam (bottom row) to normalize for total amount of endothelial cells (EC). SOX18RaOp mice at 8 weeks of age had increased levels of Pecam, indicating an increase in ECs, and increased levels of the transcripts corresponding to the extracellular domain of the Flt1 receptor (Flt1-extra), the intracellular domain (Flt1-inra) and the exclusively soluble variant of Flt1 (sFlt1). * P-value ≤ 0.05, Holm-Sidak multiple comparison test. Individual biological replicates are shown, including mean ± s.e.m of n=2-8..
Tables
qPCR primer sequences.
https://doi.org/10.7554/eLife.43026.013| Gene | Forward primer | Reverse primer |
|---|---|---|
| CD31 | CACCTGGCCCAGGAGTTTC | AGTACACAGCCTTGTTGCCATGT |
| CDH5 | CCTTGGGTCCTGAAGTGACCT | AGGGCCTTGCCTTCTGCAA |
| PLXND1 | CAAGTTTGAGCAGGTGGTGGCTTT | ATTTCCCAGTCTGAGTCACAGGCA |
| NOTCH1 | CGGTGAGACCTGCCTGAATG | GCATTGTCCAGGGGTGTCAG |
| VEGFR1 | CTCAAGCAAACCACACTGGC | CGAGCTCCCTTCCTTCAGTC |
| SOX18_2 | GTGTGGGCAAAGGACGAG | AGCTCCTTCCACGCTTTG |
| ADBR2 | CACCAACTACTTCATCACTTCAC | GACACAATCCACACCATCAG |
| ATP5B | CCACTACCAAGAAGGGATCTATCA | GGGCAGGGTCAGTCAAGTC |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.43026.014





