Selective targeting of unipolar brush cell subtypes by cerebellar mossy fibers
Figures
Glt25d2 mouse line expresses Cre in VG neurons, their peripheral afferents and their primary projections to vestibular nucleus and vestibular cerebellum.
(A) Schematic of the vestibular cerebellar circuit. Primary afferent neuron somata in the VG have peripheral afferent dendrites that end in calyces, boutons, or both (dimorphic) recieving input from type I and/or type II hair cells in one of five vestibular end organs. Their axons project into the vestibular nuclei inclduing medial vestibular nucleus (MV) and into the vestibular lobes of cerebellum. MV neurons integrate input from multiple primary afferents carrying information from multiple end organs, in addition to inputs from other parts of the brain. MV neurons provide secondary mossy fiber input to the vestibular cerebellum. (B) Left- Sagittal section of Glt25d2::tdTomato brain showing mossy fibers projecting into lobe X and ventral lobe IX. Note that the medial vestibular nucleus (MV) is innervated by primary fibers but that the local neurons do not express tdTomato (no labeled somata), indicating that the labeled mossy fibers are not secondary vestibular afferents from this nucleus. Right- Magnification of lobe X showing tdTomato+ primary afferents. (C) Left- Coronal section showing tdTomato+ fibers (black). Note auditory nerve fibers in dorsal cochlear nucleus (DCN) and trigeminal nerve fibers in spinal trigeminal nucleus (SpV). Nucleus prepositus hypoglossi (NpH), which is known to project to lobe X, has no labeled somata. Right- Magnification of MV and NpH. (D) Whole-mount of VG showing tdTomato expression and colabeling for calretinin. (E) Two example areas of VG with a subset of tdTomato-expressing cells colabeled for calretinin. (F) In all five end organs, tdTomato+ peripheral afferents were found. Myo7A (cyan) was used to label hair cells. (G) Example of calyces of the utricle (view from the top) that express tdTomato and in some cases colabeled for calretinin, indicating their pure-calyx type.
Cre+ primary vestibular afferents carry angular acceleration signals from the semicircular canals.
(A) Cre-dependent retro-AAV was injected into lobe X of Glt25d2 mice to label the primary afferents with GFP to identify their dendritic endings in the vestibular organs. In this figure all of the images are from the same animal. (B) At the injection site labeled axons are apparent. GFP- black. (C) Somata in the VG were infected and expressed GFP. None colocalized with calretinin, a marker for vestibular primary afferents with pure-calyx endings. (D) Retrolabeled dimorphic calyx endings in the crista of the vertical canal. GFP- black. Below- Boxed region above expanded- Examples of retrolabeled (left- black, right- green) dimorphic calyces, a single retrolabeled bouton-only arbor (arrowhead) and calretinin+ pure calyces (magenta). Note that some hair cells also express calretinin in mice. (E) Retrolabeled dimorphic calyx endings in the crista of the anterior canal. Below- Boxed region above expanded- note the clearly dimorphic calyces having a flask shape with narrow top. Calretinin-expressing pure-calyx afferents (magenta) have a wider top and no bouton endings. The central region of the cristae only rarely had retrolabeled afferents and they were never pure-calyx. (F) Retrolabeled dimorphic calyx endings in the crista of the horizontal canal. Below- Boxed region above expanded. (G) The peripheral area of the utricle had few labeled afferents. Calretinin (magenta) was used to determine the extent of the utricular macula and to identify the striola (dashed outline). (H) Retrolabeled dimorphic calyces were found in the sacculus having many calyces and boutons per afferent fiber. See also Figure 2—figure supplements 1–2.
Injection that infected lobe IX in addition to lobe X labeled more fibers, especially in the medial utricle.
(A) Cre-dependent retrograde AAV (AAV2-retro-CAG-Flex-GFP) was injected in lobe X and IX of Glt25d2 mouse to infect the primary afferents. In this figure all of the images are from the same animal. (B) Sagittal section of cerebellum showing labeled primary afferents in both lobe X and IX. GFP is pseudocolored black. (C) Somata in the vestibular ganglion were infected with the virus and expressed GFP. None colocalized with calretinin, a marker for vestibular primary afferents with pure-calyx endings. (D) Retrolabeled dimorphic calyx endings in the anterior and horizontal canals and utricle. GFP is pseudocolored black. Right- Same image showing calretinin labeling (magenta) in addition to GFP (green). (E) Many more peripheral afferents were labeled in the posterior vertical canal compared to experiments in which lobe X was infected and lobe IX was not. (F) Many peripheral afferents in the sacculus were labeled.
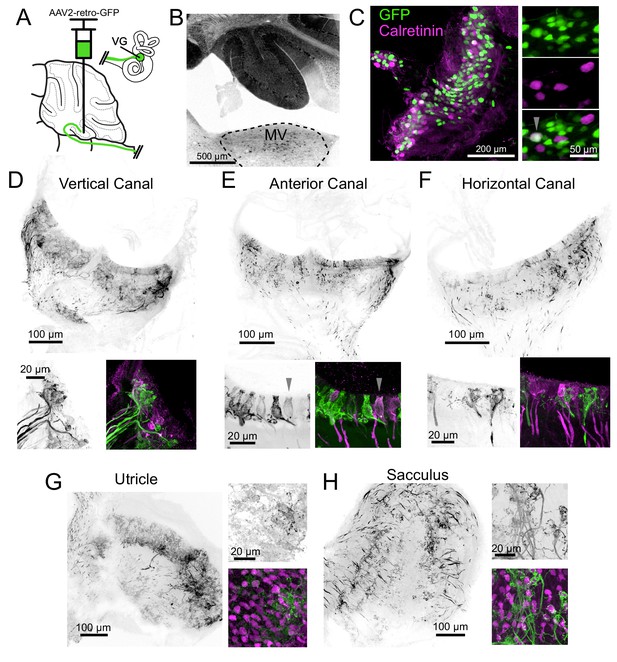
Non-Cre dependent retro-AAV reveals source of primary vestibular projections to cerebellum.
(A) Retro-AAV (AAV2-retro-CAG-GFP) infected afferents in lobe X and IX. In this figure all of the images are from the same animal. (B) The virus infected many cells in the cerebellum. Retrolabeled somata were apparent in the vestibular nuclei. GFP is pseudocolored black. (C) Somata in the vestibular ganglion were infected with the virus and expressed GFP. Expanded view to the right with arrowhead indicating a retrolabeled VG neuron (green) colabeled for calretinin (magenta). In this experiment 4.6% of the retrolabeled somata were colabeled for calretinin. (D) Retrolabeled afferents in the vertical canal. GFP is pseudocolored black. Below- Left- Examples of retrolabeled afferents. Right- GFP (green) with calretinin labeling (magenta). (E) Retrolabeled afferents in the anterior canal. GFP is pseudocolored black. Below- Left- Examples of retrolabeled afferents. Right- GFP (green) with calretinin labeling (magenta). Arrowhead indicates a pure-calyx ending that was retrolabeled. (F) Retrolabeled afferents in horizontal canal. GFP is pseudocolored black. Below- Left- Examples of retrolabeled afferents. Right- GFP (green) with calretinin labeling (magenta). (G) Retrolabeled afferents in the utricle. GFP is pseudocolored black. Note the distribution of the majority of afferents is lateral to the striola. Below- Left- Examples of retrolabeled afferents. Right- GFP (green) with calretinin labeling (magenta). (H) Retrolabeled afferents in the posterior vertical canal. GFP is pseudocolored black. Below- Left- Examples of retrolabeled afferents. Right- GFP (green) with calretinin labeling (magenta).
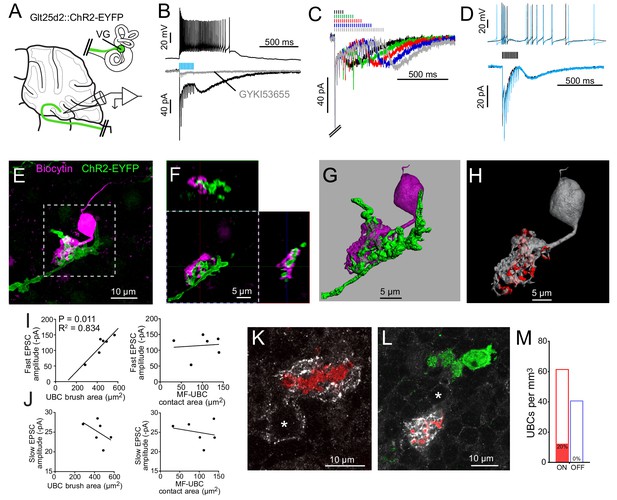
Primary vestibular afferents project to ON UBCs in lobe X.
(A) In the Glt25d2::ChR2-EYFP cross, primary afferents from the vestibular ganglion (VG) expressed ChR2 and were activated by blue light during whole-cell recordings of UBCs in an acute slice preparation. (B) Mossy fibers were activated by LED light pulses (50 Hz 0.25 ms) that evoked spiking responses in postsynaptic UBC that outlasted mossy fiber activation. In the same cell in voltage clamp (below) light pulses evoked fast EPSCs that depressed, followed by a slow inward current. 50 μM GYKI53655 blocked the majority of the inward current. This slow AMPAR-mediated current after the offset of stimulation is diagnostic of an ON UBC. This case was without 4-AP in the bath. All UBCs that had light evoked PSCs were ON UBCs (n = 13). (C) 50 Hz light stimulation of various train durations illustrated as lines above the traces. The slow AMPAR-mediated current begins at the offset of stimulation, consistent with re-activation of AMPARs as they recover from desensitization while glutamate gradually leaves the synapse. (D) Spiking response (top) and EPSCs (bottom) evoked by electrical stimulation (3.8 V, 50 Hz, 0.25 ms, black) were similar to those evoked by ChR2 stimulation (50 Hz 0.25 ms, blue) in the same cell. (E) UBCs were filled with biocytin and recovered in 6/13 cases. This UBC received input from ChR2-EYFP expressing primary vestibular afferent. Maximum intensity projection. (F) Orthogonal view of the boxed region in B, showing UBC brush wrapping around mossy fiber. (G) Surfaces were created on the fluorescence to characterize the structure of the mossy fiber-UBC synapse. (H) A one voxel thick contact layer between the UBC and mossy fiber surfaces was made to calculate the apposition area between the two surfaces (shown in red). The calculated apposition area of this mossy fiber to UBC contact was 137.66 μm2. (I) The postsynaptic EPSC correlated with the area of the UBC brush (left), but did not correlate with the contact area between the mossy fiber and brush (right). Currents are in the presence of 4-AP. (J) The slow EPSC did not correlate with the UBC brush area (left) or contact area between the mossy fiber and brush (right), suggesting that this current is due to the action of glutamate at distant receptors. Currents are in the presence of 4-AP. (K–L) In Glt25d2::tdTomato cross, tdTomato+ primary afferents were seen innervating the brushes of mGluR1+ UBCs (white), but not calretinin+ UBCs (green). Soma of mGluR1+ UBCs identified with *. Single image planes. (M) 20% of counted mGluR1+ UBCs were contacted by tdTomato+ primary afferents. No counted calretinin+ UBCs were contacted by these primary afferents. See also Figure 3—figure supplements 1–5.
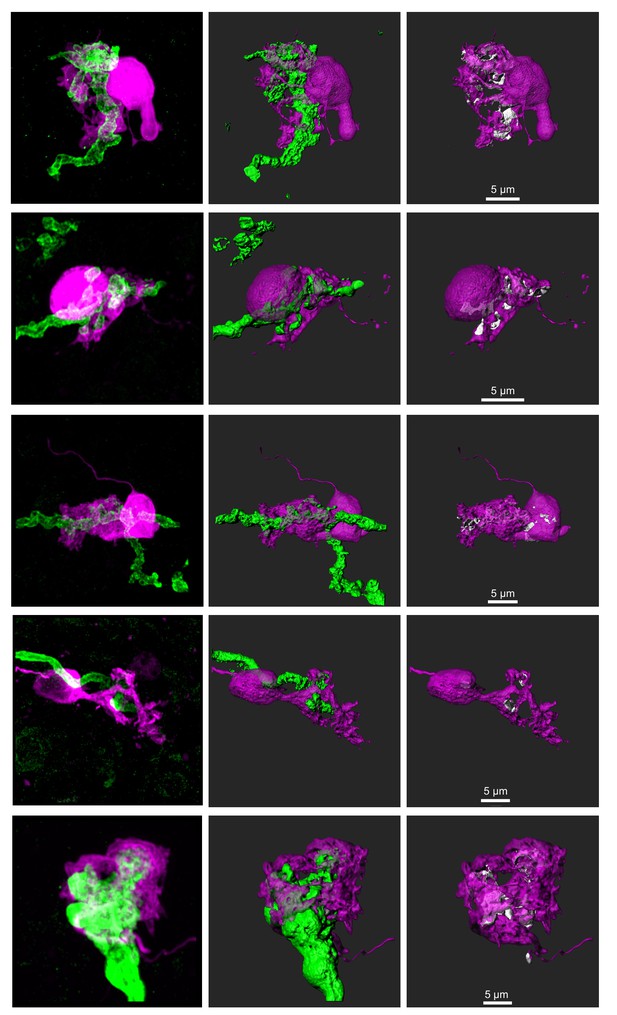
Additional UBCs that were filled with biocytin and recovered with an innervating primary afferent expressing ChR2-EYFP.
Left- Biocytin filled UBC- magenta, EYFP/ChR2 labeled primary afferent- green. Maximum intensity projection. Center- 3D rendering. Right- UBC showing contact with mossy fiber (white). UBC in middle row is also shown in Figure 7A and Figure 8E.
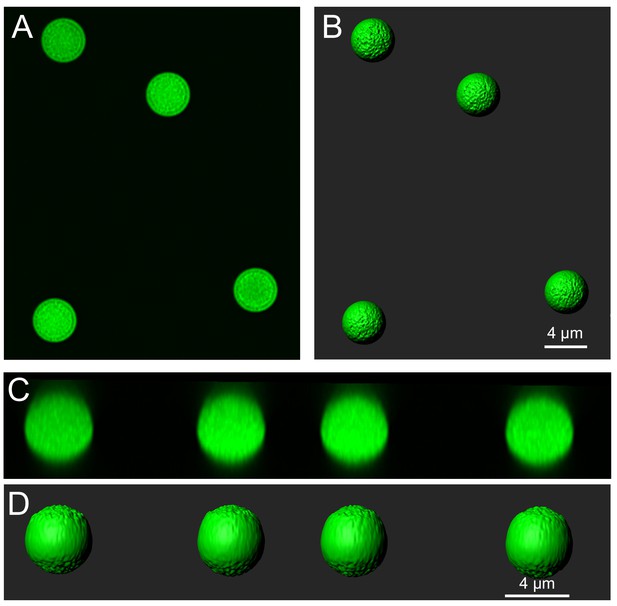
Imaging, surface construction and measurement of fluorescent spheres of known size.
(A) Fluorescent spheres were mounted on microscope slides and imaged following the procedures used for the biocytin filled cells (see Materials and methods). These spheres had a mean diameter of 4.23 μm according to the information provided by the supplier. (B) Surfaces were made following the procedures used for filled cells and mossy fibers. (C) A side view of the super-resolution image stack shows that the z-resolution was not elongated. (D) A side view of the surfaces indicates that they are less smooth on the top and bottom of the stack, but was well fit to the fluorescence and clearly represented the spherical shape. Surface areas were calculated following the procedures used for biocytin filled cells. The mean surface area was 56.61 ± 1.68 μm2, n = 18,~1% greater than the expected surface area (56.21 μm2) given the mean diameter of 4.23 μm provided by the supplier. The volume was calculated to be 37.22 ± 2.23 μm3,~6% less than the expected volume of 39.63 μm3. This indicates that the approach used to measure the surface area and volume of filled cells in super-resolution images is reasonably accurate.
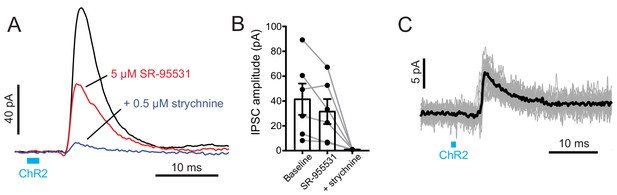
UBCs and granule cells receive disynaptic inhibitory input, likely via direct Golgi cell activation by primary vestibular afferents.
(A) UBC with ChR2 evoked IPSCs. 2 ms blue light (blue bar) activated primary afferents and evoked IPSCs that were delayed and mediated by both GABAA and glycine receptors. Amplitudes were 28.92 ± 27.73 pA (mean ± SD), n = 23. Note that these responses are in the presence of 50 μM 4-AP. (B) In 5/6 UBCs 5 μM SR-95531 blocked the IPSC 31.5 ± 16.7% (mean ± SD). The addition of 0.5 μM strychnine blocked 94.0 ± 2.0% (mean ± SD) of the remaining current. (C) Granule cell with ChR2 evoked IPSCs. Gray traces are individual trials and black trace is the average. Amplitudes were 11.32 ± 8.02 pA (mean ± SD), n = 10, in the presence of 50 μM 4-AP. Delays from onset of light to beginning of the IPSCs were 5.52 ± 0.40 ms (mean ± SD), n = 10.
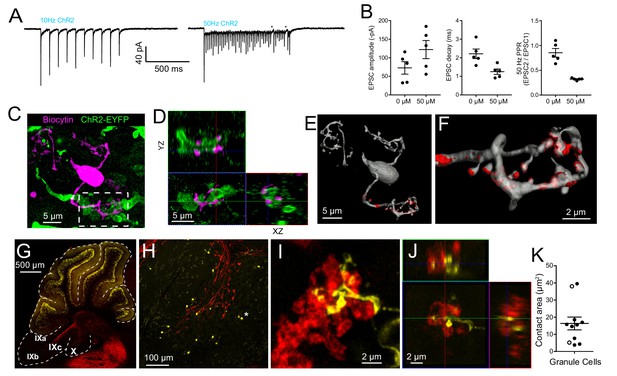
Granule cells receive primary vestibular synaptic input.
(A) Granule cell responds to 10 Hz and 50 Hz ChR2 evoked synaptic stimulation. The input to this granule cell can follow 50 Hz light flashes, but fails towards the end of a 50 pulse train (stars). This example is in 50 μM 4-AP. (B) Granule cell EPSCs evoked by ChR2 stimulation of primary afferents in 0 or 50 μM 4-AP. (C) A granule cell filled with biocytin and recovered post hoc. Maximum intensity projection. (D) Orthogonal view of the boxed region in C, showing granule cell claw wrapping around mossy fiber. (E) Surface reconstruction of granule cell showing mossy fiber contact area in red 37.98 μm2). (F) Granule cell claw showing contact with brush in red. (G) The Glt25d2::tdTomato mouse was crossed with the TCGO mouse line that expresses mCitrine (yellow) in a very small, presumably random, subset of granule cells in cerebellum. The expression is especially sparse in lobes IX and X. (H) Higher magnification of lobe X shows sparse labeling of granule cells and even sparser UBCs (a single UBC indicated by *). (I) Granule cell claw contacting primary afferent. Maximum intensity projection. (J) Orthogonal view of mossy fiber to granule cell claw contact. No granule cell was observed to contact multiple mossy fibers with different claws or the same mossy fiber with multiple claws. (K) Summary of contact areas measured between primary afferents and granule cell claws. White circles were measured from biocytin filled granule cells.
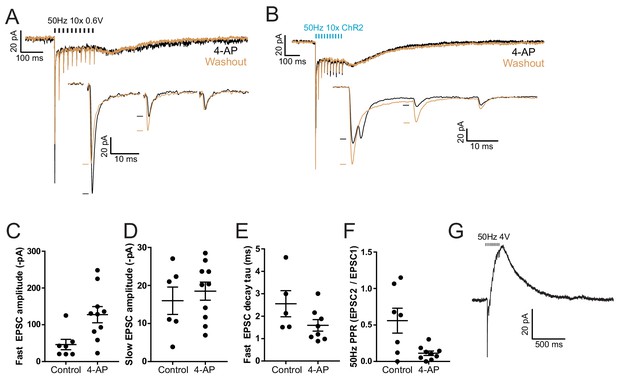
4-AP has effects on the ChR2-evoked EPSC, but does not change whether the UBC response is ON or OFF type.
(A) Example of response to electrical synaptic stimulation in 50 μM 4-AP (black) and after washout of 4-AP (brown). Inset shows the first 3 EPSCs in the train with a longer time base. 4-AP increases the amplitude of the EPSC, increases synaptic depression but has no effect on the recurrent slow EPSC that occurs at the offset of stimulation in ON UBCs. (B) In a different ON UBC, 4-AP washout had an effect on ChR2 evoked currents. In 4-AP, release was asynchronous. After washout the release became synchronous. In this cell and one other this asynchronous release was observed in 4-AP and changed after 4-AP washout. (C) The first ChR2 evoked fast EPSC amplitude was larger for cells that had 50 μM 4-AP in the bath. (D) Importantly, the slow EPSC at the offset of the stimulus train that is diagnostic of ON UBCs was inward whether or not 4-AP was present. (E) Decay of the first EPSC in the train with and without 4-AP. (F) Paired pulse ratio was lower in most cells in the presence of 50 μM 4-AP. (G) OFF UBC responses could be evoked by electrical stimulation in these Glt25d2::ChR2-EYFP slices with 4-AP present, indicating that the lack of OFF UBC responses evoked by primary fiber activation was not due to non-specific block of GIRK or mGluR2 or to the absence of OFF UBCs in this mouse line.
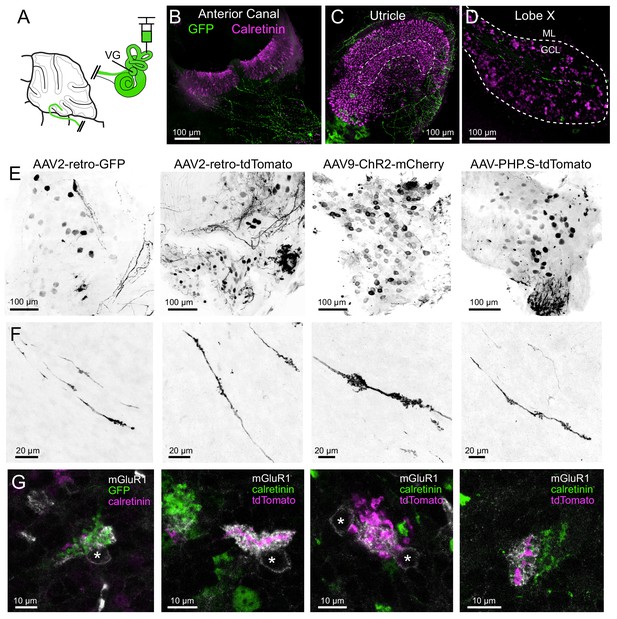
AAV infected vestibular primary afferents contact mGluR1-expressing, but not calretinin-expressing UBCs.
(A) AAVs of various serotypes were injected into the posterior vertical semicircular canal of adult mice. (B–C) Example showing experiment using AAV2-retro-CAG-GFP. The anterior canal and utricle had numerous peripheral afferent fibers labeled (green). Calretinin (magenta) labels pure-calyx afferents and Type I hair cells. (D) Each 50 μm thick section of lobe X of cerebellum had a few virally-labeled afferents (green) shown among many calretinin-expressing UBCs (magenta). (E) Vestibular ganglia showing viral expression of fluorescent proteins (black) using four different viruses in separate experiments indicated above. Many neurons of various sizes were labeled. Images in F-G correspond to ganglia and viruses indicated in E. (F) Virally-labeled primary afferents were apparent in lobe X. Note the thickness of the afferents and the lack of branching. (G) Immunostaining for calretinin and mGluR1 was used to investigate to which type of UBC this population of fibers projected. Each terminal swelling along virally-labeled primary afferents was imaged, along with calretinin and mGluR1 labeling. 33% of afferent endings intercalated with the brush of an mGluR1+ UBCs, whereas no calretinin+ UBCs were contacted. Somata of mGluR1+ UBCs indicated with *. Single image planes.
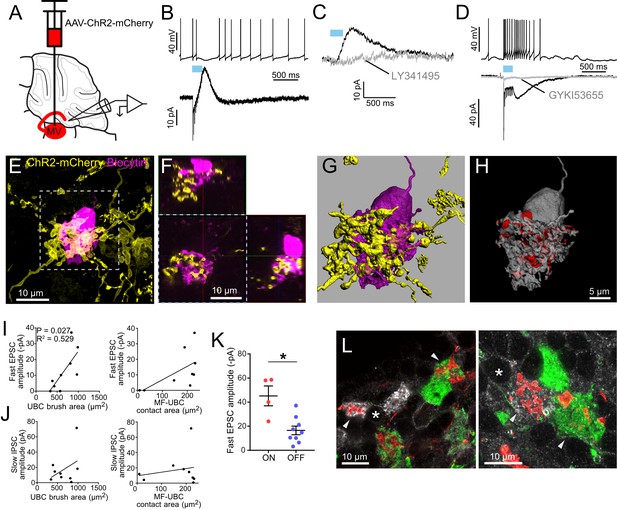
Secondary vestibular afferents project to OFF UBCs and ON UBCs.
(A) Medial vestibular nucleus (MV) was infected with a ChR2 expressing virus. (B) Example OFF UBC. 50 Hz 10x ChR2 activation of secondary vestibular mossy fibers caused a pause in spiking (above) and evoked a slow IPSC diagnostic of OFF UBCs (below). (C) The AMPAR-mediated fast EPSC varied across cells. In this example it was very small. In OFF UBCs, the IPSC was always blocked by mGluR2 antagonist LY341495 (1μM). (D) Example ON UBC. 50Hz10x ChR2 activation of secondary vestibular mossy fibers caused an increase in spiking (above) and evoked a fast and slow EPSC that were blocked by GYKI53655 (50μM) diagnostic of an ON UBCs (below). (E) The UBC shown in B was filled with biocytin (magenta) and recovered. Yellow: Immunohistochemical amplification of mCherry which is fused with the ChR2 protein. Maximum intensity projection. (F) Orthogonal view of boxed region in E, showing UBC brush wrapping around mossy fiber. (G–H) The mossy fiber and UBC shown in E-F was 3D rendered and the apposition area between the two volumizations was calculated. The calculated apposition area of this mossy fiber to UBC contact was 336 μm2 (red). (I) The postsynaptic EPSC of the OFF UBCs correlated with the area of the UBC brush. R2 = 0.529, p=0.027 (left), but did not correlate with the contact area between the mossy fiber and brush (right). (J) The slow IPSC of the OFF UBCs did not correlate with the UBC brush area (left) or contact area between the mossy fiber and brush (right). (K) OFF UBCs that received input from secondary afferents had smaller EPSCs than those of ON UBCs. t-test, p=0.003. (L) mCherry-expressing secondary mossy fibers (red) were seen innervating the brushes of mGluR1+ UBCs (white) and also calretinin+ UBCs (green). Soma of mGluR1+ UBCs identified with *. Arrowheads indicate contact between mossy fiber and brush. Single image planes. See also Figure 5—figure supplement 1.
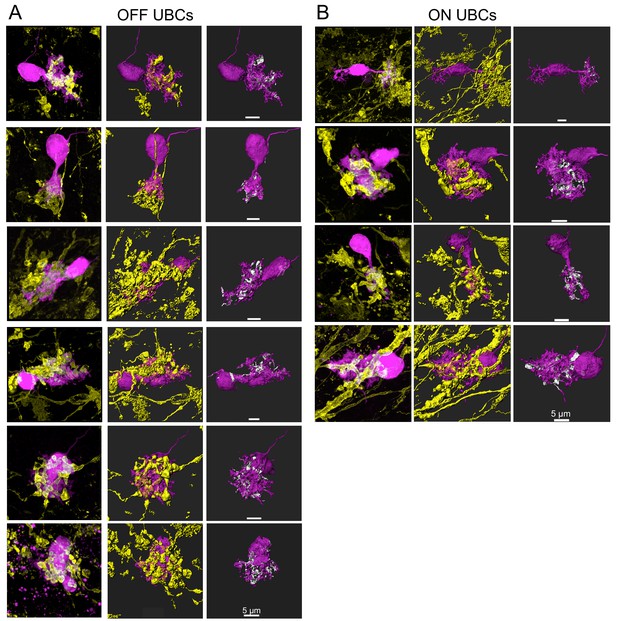
Additional UBCs that responded to secondary vestibular afferent stimulation.
Left- Biocytin filled UBC- magenta, mCherry/ChR2 labeled secondary afferent- yellow. Maximum intensity projection. Center- 3D rendering. Right- UBC showing contact with mossy fiber (white). (A) OFF UBCs. (B) ON UBCs.
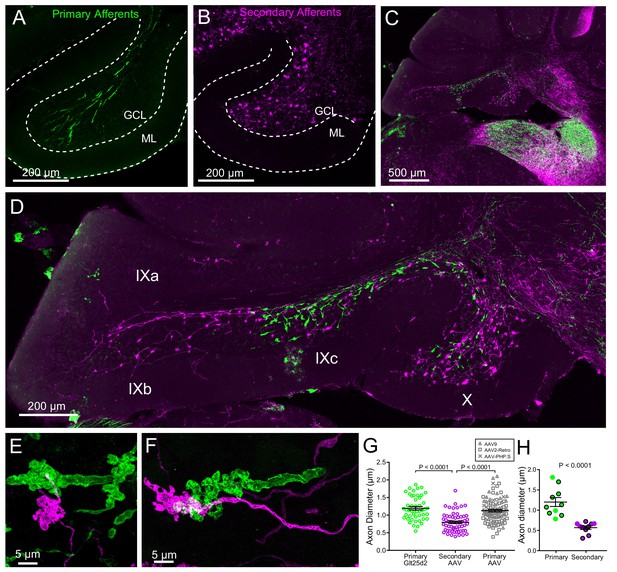
Primary and secondary vestibular mossy fiber morphology.
(A) Expression of a fluorescent protein in primary vestibular afferents (green). Note central location in the granule cell layer (GCL). ML- molecular layer. (B) Expression of a fluorescent protein in secondary vestibular mossy fibers (magenta). Note distribution of mossy fibers throughout the width of the GCL. (C–D) In a separate set of experiments Glt25d2::ChR2-EYFP mice had AAV1-CAG-ChR2-mCherry injected into MV, which allowed labeling of both primary (green) and secondary (magenta) mossy fiber projections in the same brain sections. Note the intermingling of primary and secondary fibers in the brainstem vestibular nuclei. Primary afferents only projected to rostral lobe IX (IXc), whereas secondary fibers innervated the entire ventral leaflet (IXb and IXc). Careful observation revealed few colabeled mossy fibers, suggesting possible but unlikely confound of virus infected primary fibers. (E–F) Higher magnification of primary and secondary vestibular afferents in the same section. Note the larger diameter axon of the primary afferent (green). The apparent overlap of the mossy fibers in these maximum intensity projections is due to the position of one above the other in the z dimension. (G) The mean diameter of primary afferents was significantly thicker than that of secondary afferents. Data points are individual mossy fiber axons. ANOVA, p=0.0001, post hoc t-tests, p<0.0001, n = 206 axons. (H) The diameter of primary and secondary afferents that contacted UBCs that were recorded from and recovered. Data points with black outline are UBCs that had ChR2 expressing mossy fiber making major contact with brush. Data points without outline had ChR2 expressing mossy fiber making small contact with brush and the ChR2-evoked response was small. t-test, p<0.0001, n = 20 axons.
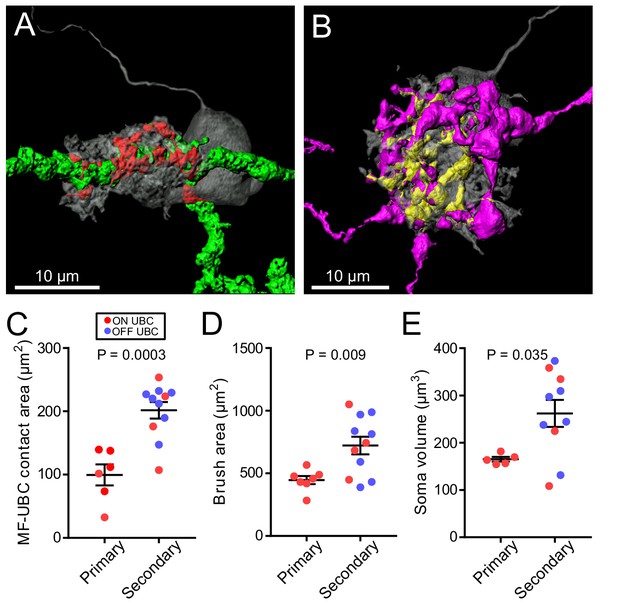
UBC dendritic brush size relates to source of input and not ON or OFF UBC subtype.
(A) 3D rendering of primary afferent showing characteristic thick axon (green). Biocytin fill- gray, contact between mossy fiber and UBC- red. (B) 3D rendering of secondary mossy fiber showing thin axons and rosette-like ending. Biocytin fill- gray, contact between mossy and UBC- yellow. (C) The contact areas between recovered primary afferents and UBC brushes was smaller than those of secondary mossy fibers (t-test, p=0.0003, n = 17). (D) The surface area of the UBC brushes that received primary afferents were smaller than those of secondary mossy fiber-receiving UBCs (t-test, p=0.009, n = 18). The volume of the UBC brushes that received primary afferents were also larger than those of secondary mossy fiber-receiving UBCs (t-test, p=0.014, n = 18), not shown. (E) The volume of the somas of UBCs that received primary afferents were smaller than those of secondary mossy fiber-receiving UBCs (t-test, p=0.035, n = 15).
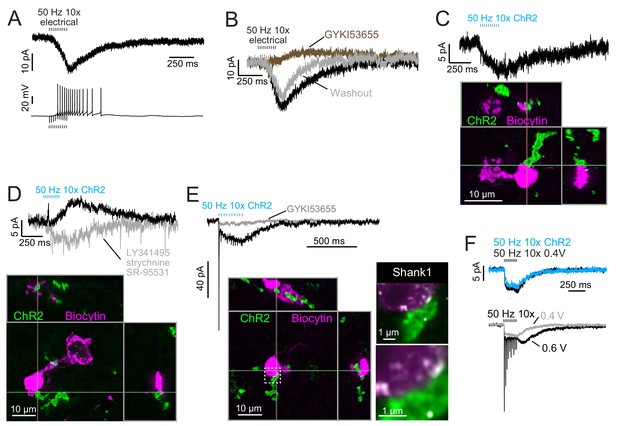
Build-up AMPAR-mediated EPSCs can be evoked by ChR2 or electrical evoked transmitter release and may be due to somatic synapses.
(A) Slow build-up EPSC evoked by presynaptic electrical stimulation in UBC. The build-up of inward current is sufficient to cause a burst of spikes beginning at the 5th stimulus. No 4-AP in bath. These currents are distinct from the usual slow EPSC seen in ON UBCs that occurs at the stimulus offset. (B) Another UBC with a similar slow EPSC evoked by 50 Hz train of presynaptic electrical stimulation. The current was blocked by AMPAR antagonist GYKI53655 (50 μM), revealing a small IPSC presumably mediated by mGluR2 known to be present in UBCs. (C) Slow EPSC evoked by ChR2 stimulation of primary vestibular afferent. This UBC was filled with biocytin and recovered post hoc. Super-resolution imaging revealed the mossy fiber made contact with the soma of the UBC and not the brush. (D) IPSCs evoked by ChR2 stimulation of primary vestibular afferent. The fast transient IPSC was blocked by strychnine (0.5 μM) and SR-95531 (5 μM) and the delayed slower IPSC was blocked by LY341495 (1 μM). Biocytin cell fill revealed that this UBC received input to the soma and to the shaft of its dendritic brush. This mGluR2 mediated IPSC was seen in 4 UBCs that received primary afferent input. The slow IPSC amplitude for primary afferent receiving UBCs was 5.43 ± 2.89 pA (mean ± SD), n = 4. (E) This UBC had a response that appeared to be a combination of dendritic and somatic synapses due to the fast EPSC at stimulus onset and buildup of inward current during the train, respectively. Below- Recovery of the cell and the input mossy fiber revealed that this was indeed possible, because the primary vestibular afferent made contact with both the soma and the dendritic brush. This is the cell shown in Figure 7A. Right- Boxed region expanded- Shank1 antibody staining (white) was seen on the periphery of the soma directly opposed to the primary afferent. (F) Slow EPSC evoked by 50 Hz train of either low intensity electrical synaptic stimulation (0.4 V, black) or ChR2 stimulation (blue). The similarity between responses suggests that the same input is being stimulated and the transmitter release is comparable between stimulation methods. Below- the same UBC with two levels of electrical synaptic stimulation. The observation that a stronger electrical stimulation evokes a typical ON UBC response suggests that this cell may have received multiple inputs.
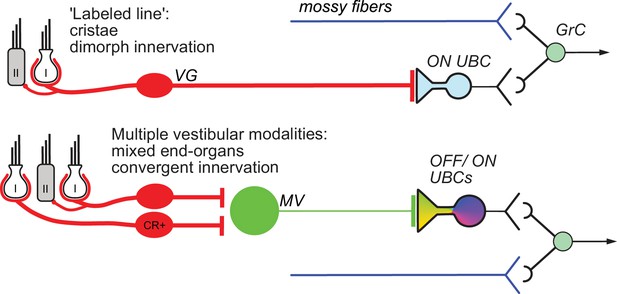
Summary of results.
Vestibular ganglion (VG) neurons having dimorphic peripheral endings that receive input from Type I and II hair cells project their primary afferents directly to ON UBCs in lobe X of cerebellum, and do not target OFF UBCs. These primary afferents also project to granule cells as well as inhibitory interneurons (Golgi cells) that provide feed-forward inhibition to ON UBCs (not shown). This direct projection to cerebellum may coordinate ensembles of granule cell activities in a ‘labeled line’ representing acceleration or velocity of the head in a single direction. Secondary mossy fibers arising from neurons in the medial vestibular nucleus (MV) project to both OFF and ON UBCs. MV neurons receive input from calretinin-expressing (CR+) VG neurons that make pure-calyx dendritic endings around Type I hair cells, in addition to dimorphic primary afferents. The signals carried by secondary afferents are integrated across vestibular end organs and processed by the local MV circuit. Thus, distinct anatomical pathways that vary in the convergence of inputs, level of processing and axon morphology target distinct UBC classes to transform specific sensory signals.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (M. musculus) | C57BL/6J | Jackson Laboratory | RRID: IMSR_JAX:000664 | |
| Genetic reagent (M. musculus) | B6.Tg(Colgalt2-cre)NF107Gsat/Mmucd | Dr. Chip Gerfen (NIH) PMID: 20023653 | RRID: MGI:2138232 | Referred to as Glt25d2 |
| Genetic reagent (M. musculus) | B6.TgN(grm2-IL2RA/GFP)1kyo | Dr. Robert Duvoisin (OHSU) PMID: 9778244 | RBRC: RBRC01194 | |
| Genetic reagent (M. musculus) | Ai9(RCL-tdT) | Jackson Laboratory PMID: 22446880 | RRID: IMSR_JAX:007909 | |
| Genetic reagent (M. musculus) | Ai32(RCL-ChR2(H134R)/EYFP) | Jackson Laboratory PMID: 22446880 | RRID: IMSR_JAX:024109 | |
| Genetic reagent (M. musculus) | B6.Cg-Et(tTA/mCitrine)TCGOSbn | Dr. Adam Hantman (Janelia Farm) PMID: 26999799 | ||
| Antibody | Chicken polyclonal anti-GFP | Aves Labs | Cat # GFP-1020 RRID: AB_10000240 | IHC (1:2000) |
| Antibody | Rabbit polyclonal anti-DsRed | Clontech | Cat# 632496 RRID:AB_10013483 | IHC (1:2000) |
| Antibody | Goat polyclonal anti-mCherry | Sicgen | Cat# AB0040-200 RRID:AB_2333092 | IHC (1:2000) |
| Antibody | Mouse monoclonal anti-rat mGluR1a | BD Pharmingen | Cat# 556389 RRID:AB_396404 | IHC (1:800) |
| Antibody | Rabbit polyclonal anti-calretinin | Swant | Cat# 7697 RRID:AB_2619710 | IHC (1:500–2000) |
| Antibody | Goat polyclonal anti-calretinin | Swant | Cat# CG1 RRID:AB_10000342 | IHC (1:500–2000) |
| Antibody | Rabbit polyclonal anti-shank1 | Synaptic Systems | Cat# 162 013 RRID:AB_2619859 | IHC (1:1000) |
| Antibody | Mouse monoclonal anti-Myo7A | Dr. Peter Barr-Gillespie (OHSU) | IHC (1:500) | |
| Antibody | Donkey polyclonal anti-chicken Alexa Fluor 488 | Jackson ImmunoResearch Labs | Cat# 703-545-155 RRID:AB_2340375 | IHC (1:500) |
| Antibody | Donkey polyclonal anti-mouse Alexa Fluor 488 | Jackson ImmunoResearch Labs | Cat# 715-545-150 RRID:AB_2340846 | IHC (1:500) |
| Antibody | Donkey polyclonal anti-rabbit Cy3 | Jackson ImmunoResearch Labs | Cat# 711-165-152 RRID:AB_2307443 | IHC (1:500) |
| Antibody | Donkey polyclonal anti-goat Cy3 | Jackson ImmunoResearch Labs | Cat# 705-165-147 RRID:AB_2307351 | IHC (1:500) |
| Antibody | Donkey polyclonal anti-mouse Alexa Fluor 647 | Jackson ImmunoResearch Labs | Cat# 715-605-151 RRID:AB_2340863 | IHC (1:500) |
| Antibody | Donkey polyclonal anti-chicken Alexa Fluor 647 | Jackson ImmunoResearch Labs | Cat# 703-605-155 RRID:AB_2340379 | IHC (1:500) |
| Antibody | Streptavidin-Alexa Fluor 647 | ThermoFisher Scientific | Cat# S21374 RRID:AB_2336066 | IHC (1:2500) |
| Chemical compound, drug | Alexa Fluor 594 hydrazide sodium salt | ThermoFisher Scientific | Cat# A10438 | |
| Chemical compound, drug | GYKI-53655 | Tocris | Cat # 2555 | |
| Chemical compound, drug | JNJ-16259685 | Tocris | Cat # 2333 | |
| Chemical compound, drug | LY-341495 | Tocris | Cat # 1209 | |
| Chemical compound, drug | (+)-MK-801 hydrogen maleate | Sigma | Cat # M107 | |
| Chemical compound, drug | Strychnine hydrochloride | Sigma | Cat # S8753 | |
| Chemical compound, drug | SR-95531 hydrobromide | Tocris | Cat # 1262 | |
| Chemical compound, drug | 4-Aminopyradine | Tocris | Cat # 940 | |
| Recombinant DNA reagent | AAV1-CAG-ChR2(H134R)-mCherry (2.92E12 GC/ml) | University of Pennsylvania Vector Core | Cat # CS0949 | |
| Recombinant DNA reagent | AAV9-CAG-ChR2(H134R)-mCherry (2.96E12 GC/ml) | University of Pennsylvania Vector Core | Cat # CS0916 | |
| Recombinant DNA reagent | AAV2-retro-CAG-GFP (1.0E13 GC/ml) | Janeila Farm Vector Core PMID: 27720486 | Dr. Adam Hantman (Janelia Farm) | |
| Recombinant DNA reagent | AAV2-retro-CAG-tdTomato (7.0E12 GC/ml) | Addgene PMID: 27720486 | Cat # 59462-AAVrg | |
| Recombinant DNA reagent | AAV2-retro-CAG-Flex-GFP (9.86E12 GC/ml) | Janeila Farm Vector Core PMID: 27720486 | Dr. Adam Hantman (Janelia Farm) | |
| Recombinant DNA reagent | AAV-PHP.S-CAG-tdTomato (1.7E13 GC/ml) | Addgene PMID: 28671695 | Cat # 59462-PHP.S | |
| Software, algorithm | pClamp 10 | Molecular Devices | RRID:SCR_011323 | |
| Software, algorithm | Igor Pro 6 | WaveMetrics | RRID:SCR_000325 | |
| Software, algorithm | Prism 7 | GraphPad | RRID:SCR_002798 | |
| Software, algorithm | Excel | Microsoft | RRID:SCR_016137 | |
| Software, algorithm | Imaris | Bitplane | RRID:SCR_007370 | |
| Software, algorithm | Zen Black | Zeiss | RRID:SCR_013672 | |
| Software, algorithm | FIJI | https://fiji.sc | RRID:SCR_002285 | |
| Software, algorithm | ImageJ | https://imagej.nih.gov/ij/ | RRID:SCR_003070 | |
| Software, algorithm | Affinity Designer | Serif | RRID:SCR_016952 |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.44964.020