In vivo identification of GTPase interactors by mitochondrial relocalization and proximity biotinylation
Figures
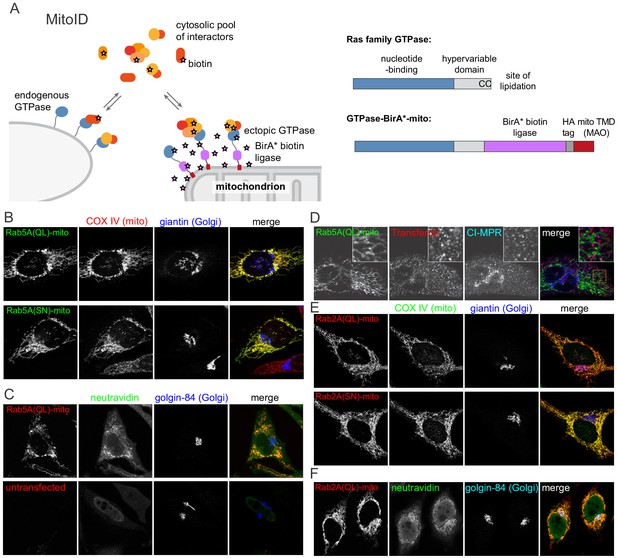
MitoID: Expression of BirA*-tagged GTPases on the surface of mitochondria.
(A) Schematic of the MitoID approach in which a GTPase is expressed as a chimera with BirA* and a mitochondrial targeting sequence. Also shown is the structure of a typical Ras superfamily GTPase and the of chimeric version used for MitoID. (B) Confocal images of HeLa cells expressing mitochondrially targeted forms of Rab5A-BirA* as in (A), and stained for the HA tag in the chimera as well as markers for the mitochondria and Golgi. The Rab5A chimeras have either the Q79L or the S34N versions that lock them in the GTP- or GDP-bound states respectively. (C) Confocal images of HeLa cells expressing mitochondrial Rab5A(Q79L)-BirA*, or untransfected, and labeled for the chimera and with biotin-binding neutravidin and a Golgi marker. (D) As (C) except that the cells were incubated with fluorescent transferrin for 45 min at 37°C prior to fixation to label endosomes, and then also stained for cation-independent mannos-6-phosphate receptor (CI-MPR) that recycles through endosomes. Neither endosomal marker is relocated to the Rab5A(Q69L)-covered mitochondria. (E, F). Confocal micrographs of cells expressing mitochondrial forms of Rab2A-BirA* in either the GTP-form (Q65L) or the GDP-form (S20N) and stained for mitochondria, the Golgi or with neutravidin as indicated. The Rab2A chimeras accumulate on mitochondria along with biotin, but not the Golgi markers.
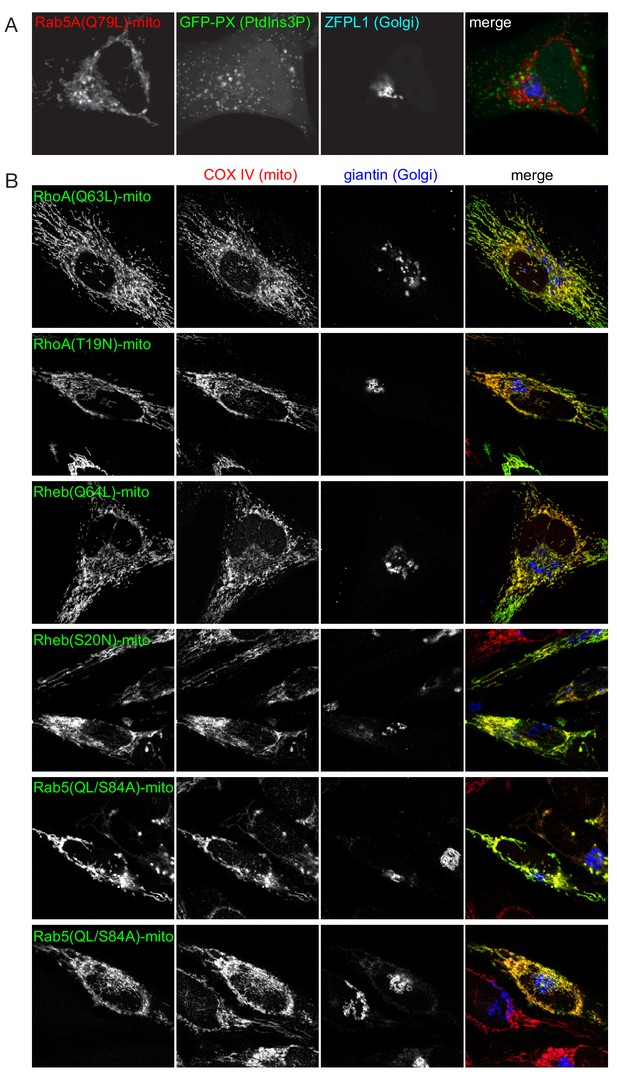
Expression of representative BirA*-tagged GTPases on the surface on mitochondria.
(A) Confocal images of HeLa cells expressing the mitochondrially targeted form Rab5A(QL)-BirA* and a GFP-fusion to a PX domain that binds PtdIns(3)P on endosomes. The cells were stained for the HA tag in the Rab chimera and a Golgi marker. The endosomes are not relocated to the Rab5A(Q69L)-covered mitochondria. (B) Confocal images of HeLa cells expressing mitochondrially targeted forms of the indicated GTPases, and stained for the HA tag in the chimera as well as markers for the mitochondria and Golgi.
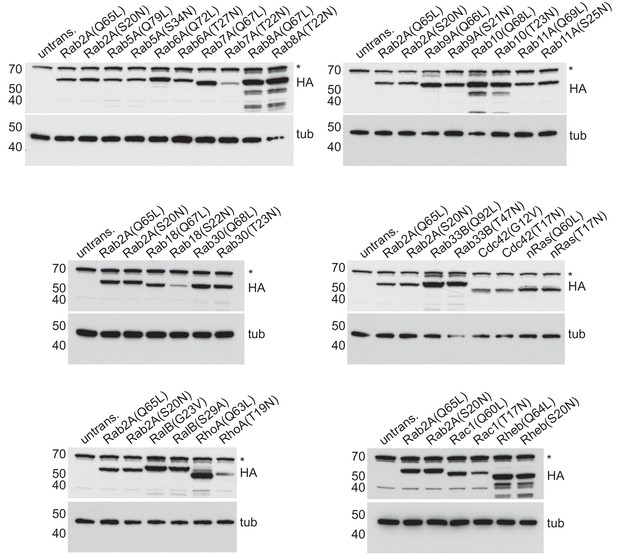
Expression levels of BirA*-tagged GTPases on mitochondria.
Immunoblots of cell lysates from HEK293 cells expressing mitochondrially targeted forms of the indicated GTPases, and stained for the HA tag in the chimera and α-tubulin (tub). The anti-HA blot has non-specific background band that is marked with an asterisk and also present in the lysate from untransfected cells (untrans.).
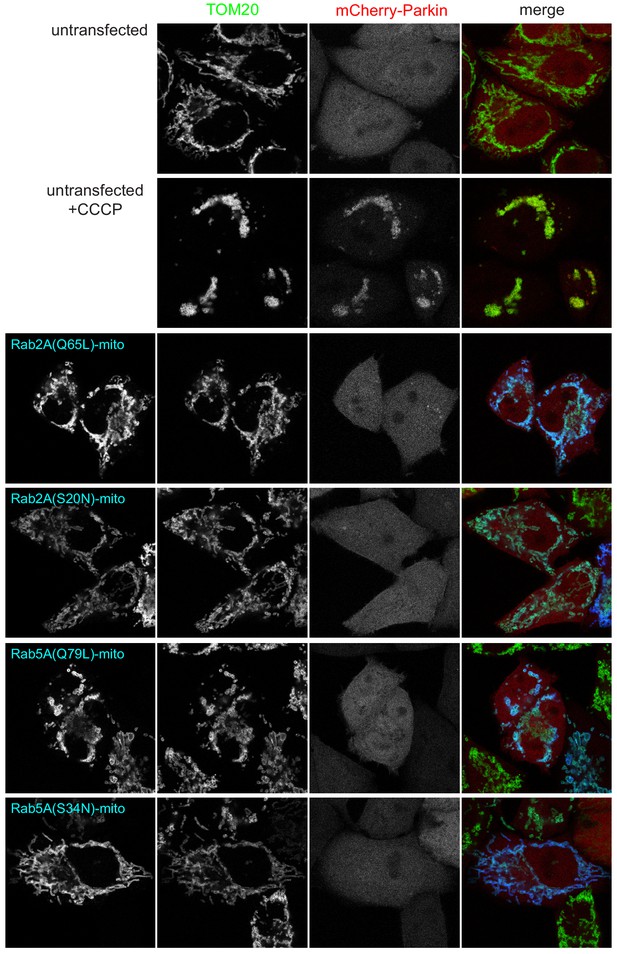
Expression of BirA*-tagged GTPases on mitochondria does not induce mitochondrial stress.
Confocal images of HeLa cells stably expressing mCherry-parkin and either untransfected or transfected with plasmids expressing the indicated GTPase chimeras and fixed after 48 hr. Untransfected cells were also treated with 10 μM carbonyl cyanide m-chlorophenyl hydrazine (CCCP) for three hours prior to fixation. The mitochondrial uncoupling agent CCCP induces translocation of the parkin stress sensor but this is not seen with the GTPase chimeras.
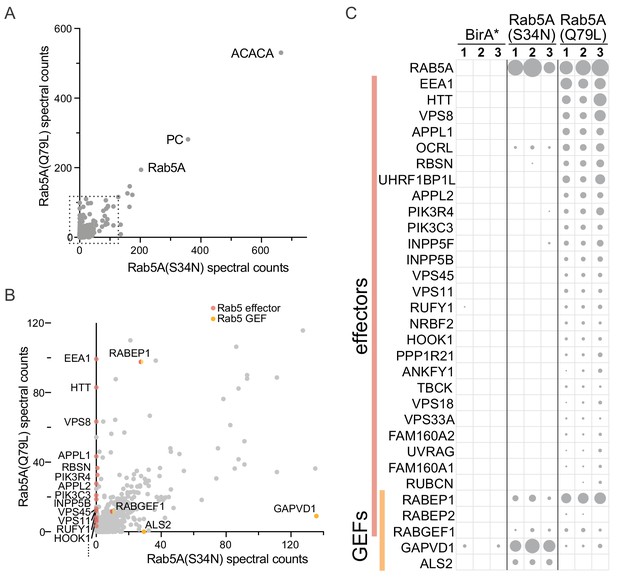
Expression of mitochondrial Rab5A-BirA* chimeras leads to biotinylation of known Rab5A effectors and regulators.
(A) Plot of spectral counts obtained for individual proteins following mass-spectrometric analysis of streptavidin precipitations from cells expressing mitochondrial forms of Rab5A-BirA*. These forms were either GTP-bound (Q79L) or GDP-bound (S34N), and the counts represent means of triplicate biological repeats. The most abundant proteins found with both are Rab5A and the endogenous biotin-containing proteins Acetyl-CoA carboxylase (ACACA) and pyruvate carboxylase (PC). (B) As (A) except only the region in the dashed box in (A) is shown. Known Rab5 effectors and exchange factors are labeled as indicated by their gene names. The effectors are specific for the GTP form (Q79L), whilst the exchange factors are found with the GDP form (S34N). The proteins encoded by RABEP1 and RABGEF1 are found with both forms, but they are known to form a heterodimer that has Rab5 GEF activity but is also a Rab5 effector. (C) Spectral counts for known Rab5 interactors obtained with the two mitochondrial forms of Rab5A shown in (A) and BirA* alone as a control. Values from three biological replicates are shown, with the area of the circle proportional to the number of counts. For full list of values for all panels see Supplementary file 1.
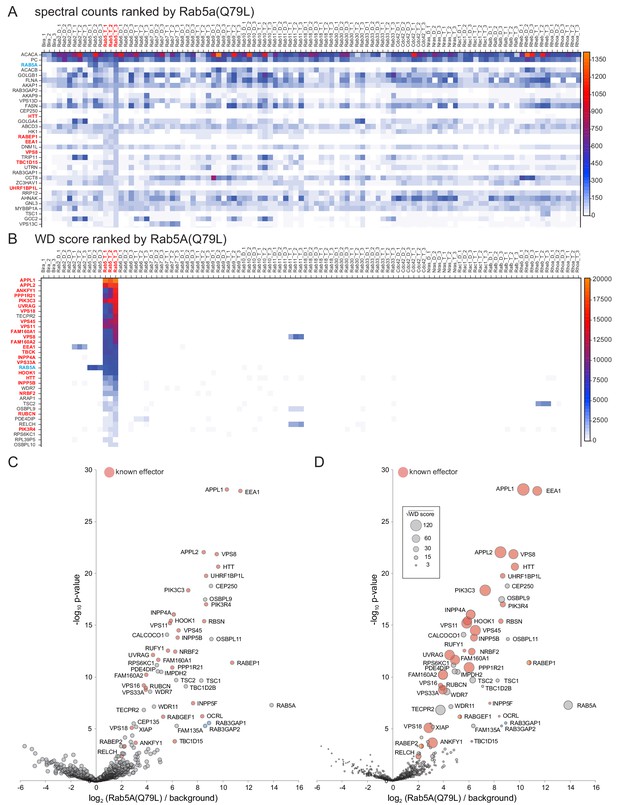
Comparison of Rab5A MitoID data to that obtained from sixteen other GTPases.
(A) Spectral counts for the indicated proteins obtained from triple replicates of MitoID with the indicated GTPases. All GTPases are in either the GDP (D) or GTP (T) form and the proteins are ranked by their highest spectral counts with the GTP form of Rab5A (Rab5A(Q79L)). Only the top 34 proteins are shown with the full list in Supplementary file 1. Known Rab5 effectors are shown red text and Rab5A with blue. (B) As (A) except that the spectral counts have been converted to WD scores and the proteins ranked by their highest WD score with Rab5A(Q79L). Known Rab5 effectors now dominate the highest positions in the list. For the full list of values see Supplementary file 1. (C) Volcano plot comparing the spectral intensities from MitoID with Rab5A(Q79L) to MitoID with the GDP-bound forms of Rab5A and sixteen other GTPases (background). Known Rab5 effectors are marked red. Values are in Supplementary file 3. (D) Bubble-volcano plot as in (C), but with the area of each point proportional to the root of the WD score. The root was used to ensure that the full range of bubble sizes was visible on the plot.
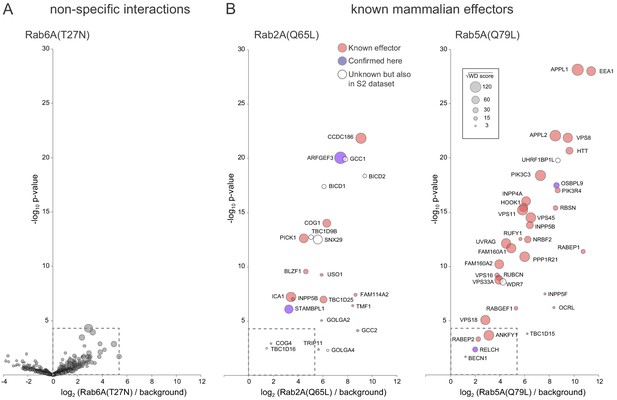
Relative distribution of specific and non-specific interactors on bubble-volcano plots.
(A,B) Comparison of bubble-volcano plots of the MitoID data for known effectors for mammalian Rab2A and Rab5A versus the non-specific interactions obtained with Rab6-GDP, a representative Rab form for which no known or plausible valid interaction was obtained (shown also in Figure 6). Also plotted are proteins that have not previously been reported to be effectors for the relevant Rab, but which were found as specific hits in an affinity-chromatography based screen in Drosophila and so seem highly likely to be valid (Gillingham et al., 2014). All values are in Supplementary file 5.
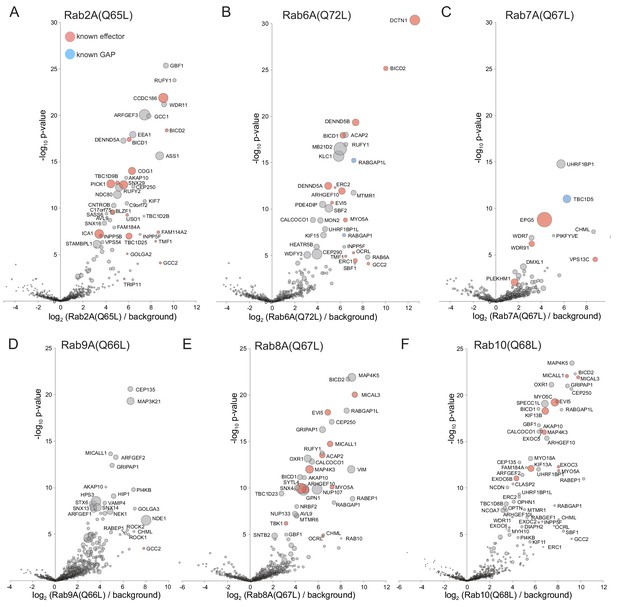
Application of MitoID to GTP-locked forms of Rab2A, Rab6A, Rab7A, Rab9A, Rab8A and Rab10.
(A–F). Bubble-volcano plots of MitoID with the indicated Rabs, each with a mutation predicted to lock it in the GTP-bound form. In each case the Rab is compared to a background comprising the GDP-locked/empty forms of all seventeen GTPases, with the size of the bubbles proportional to the WD scores. Indicated are known effectors (red) and GAPs (blue). For each plot the Rab is omitted and all values are in Supplementary file 3.
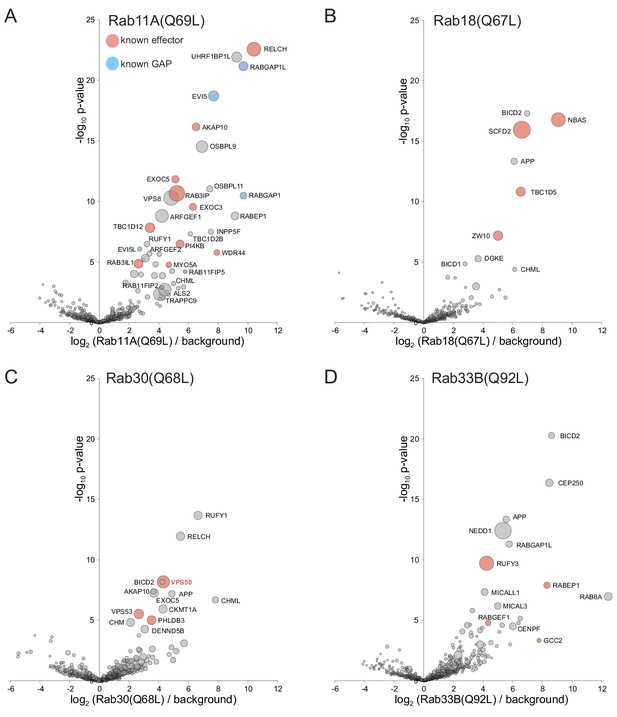
Application of MitoID to GTP-locked forms of Rab11A, Rab18, Rab30, and Rab33B.
(A–D). Bubble-volcano plots of MitoID with the GTP-locked forms of the indicated Rabs. In each case the Rab is compared to a background comprising the GDP-locked forms of all seventeen GTPases, with the size of the bubbles proportional to the WD scores. Indicated are known effectors (red) and GAPs (blue). For each plot the Rab is omitted and all values are in Supplementary file 3.
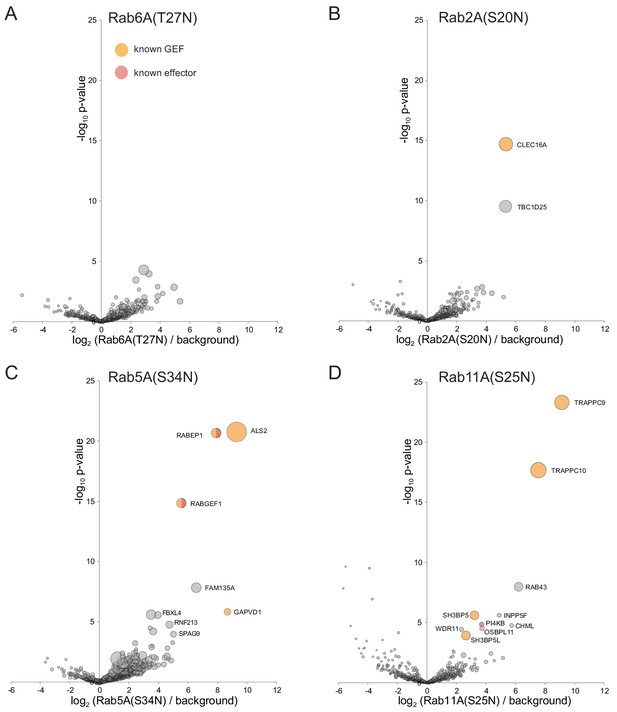
Application of MitoID to GDP-locked/empty forms of Rab6A, Rab2A, Rab5A, and Rab11A.
(A–D). Bubble-volcano plots of MitoID with the indicated Rabs, each with a mutation predicted to lock it in the GDP-bound form. In each case the Rab is compared to a background comprising the GDP-locked forms of seventeen GTPases, with the size of the bubbles proportional to the WD scores. Indicated are known effectors (red) and GEFs (yellow). Note that some of the lower scoring hits were also found with the GTP-bound form of the respective Rabs (eg TBC1D25 for Rab2A, FBXL4 and SPAG9 for Rab5A). In one case (PI4KB with Rab11A) the interaction has been reported to be nucleotide-independent, and so such hits seem more likely to be similar nucleotide-independent effectors rather than GEFs (Burke et al., 2014). For each plot the Rab is omitted and all values are in Supplementary file 3.
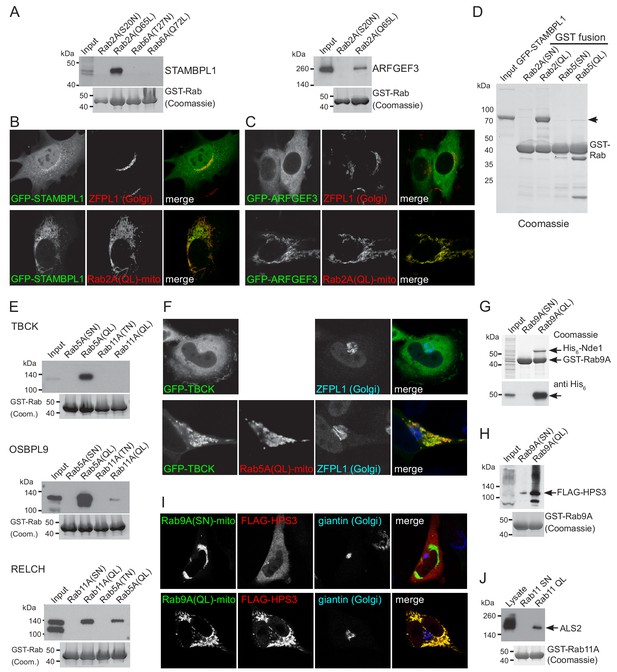
Validation of putative effectors for Rab2A, Rab5A and Rab9A.
(A) Affinity chromatography of cell lysates (input) using GST-fusions to the indicated GTP-locked (QL) or GDP-locked Rabs. The bound proteins were eluted and probed with antibodies to the indicated proteins. Lysates were from HEK293 cells and probed for STAMBPL1, or from rat brain and probed for ARFGEF3. (B) Confocal micrographs of MEFs expressing either GFP-STAMPBL1 alone and probed for a Golgi marker, or co-expressing the mitochondrial Rab2A(Q65L)-BirA* as indicated. (C) As (B) except that the cells were expressing GFP-ARFGEF3. (D) Coomassie blue stained gel showing affinity chromatography of purified recombinant GFP-STAMBPL1 using GST fusions to the indicate GTP-locked (QL) or GDP-locked (SN) Rabs. The recombinant GFP-STAMBPL1 binds specifically to the GTP form of Rab2A. (E) Affinity chromatography of HeLa cell lysates (input) using GST-fusions to the indicated GTP-locked (QL) or GDP-locked Rabs. The bound proteins were eluted and probed with antibodies to the indicated proteins. TBCK, OSBPL9 and RELCH show specific binding to the GTP form of Rab5, with RELCH also binding to Rab11-GTP as previously reported (Sobajima et al., 2018). (F) Confocal micrographs of cells expressing GFP-TBCK alone, or with co-expression of mitochondrial Rab5A(Q79L)-BirA* as indicated. (G) Coomassie blue stained gel showing affinity chromatography of lysate from E. coli expressing His6-Nde1 (input) using GST fusions to GTP-locked (Q66L) or GDP-locked (S21N) Rab9A. The samples were also immunoblotted for the His6 tag. The recombinant His6-Nde1 binds specifically to the GTP form of Rab9A. (H) Affinity chromatography of lysate from HEK293 cells expressing FLAG-HPS3 (Input) using GST-fusions to GTP-locked (QL) or GDP-locked (SN) Rab9A. The bound proteins were eluted and probed for the FLAG tag. (I) Confocal micrographs of HeLa cells expressing FLAG-HPS3 and mitochondrial Rab9A(Q66L)-BirA* (GTP-locked) or Rab9A(S21N)-BirA* (GDP-locked). The GTP-form specifically recruits HPS3 to mitochondria. (J) Affinity chromatography of HEK293 cell lysate (Input) using GTP-locked (QL) or GDP-locked (SN) GST-Rab11A. Bound proteins were eluted and probed for ALS2/alsin.
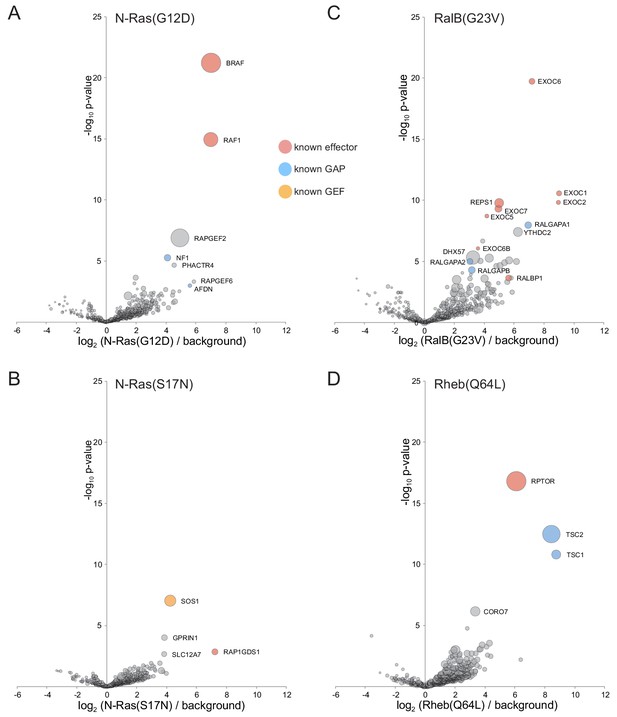
Application of MitoID to N-Ras, RalB and Rheb.
(A–F). Bubble-volcano plots of MitoID with the indicated Ras family GTPases, each with mutations predicted to lock it into the GTP-bound form, with N-Ras also shown with the GDP-bound/empty form. In each case the GTPase is compared to a background comprising the GDP-locked/empty forms of seventeen GTPases, with the size of the bubbles proportional to the WD scores. Indicated are known effectors (red), GAPs (blue) and GEFS (yellow). All values are in Supplementary file 4.
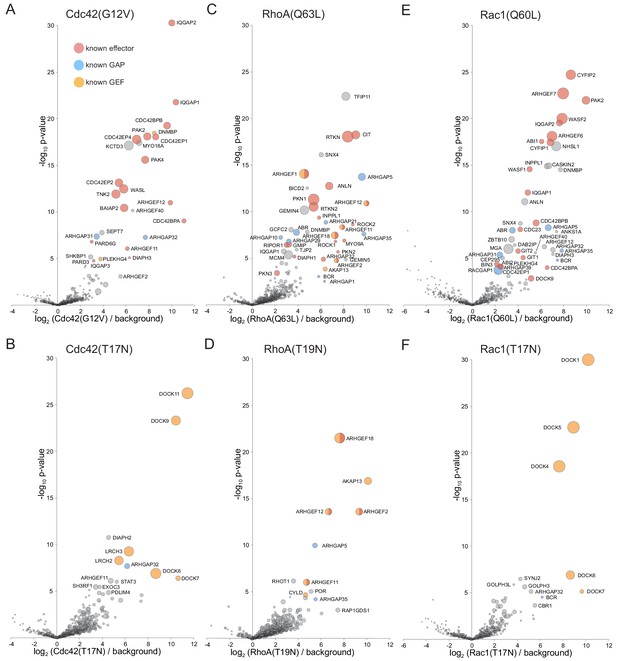
Application of MitoID to CDC42, RhoA and Rac1.
(A–F). Bubble-volcano plots of MitoID with the indicated Rho family GTPase, each with mutations predicted to lock it in to either the GTP-bound (A,C,E), or GDP-bound/empty form (B,D,G). In each case the GTPase is compared to a background comprising the GDP-locked/empty forms of seventeen GTPases, with the size of the bubbles proportional to the WD scores. Indicated are known effectors (red), GAPs (blue) and GEFS (yellow). All values are in Supplementary file 4.
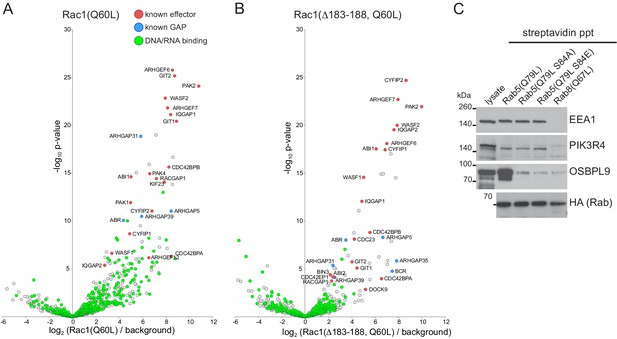
MitoID with sequence variants of GTPases.
(A,B) Bubble-volcano plots of MitoID with the GTP-locked form of Rac1 (Q60L) or with the same GTPase from which a C-terminal stretch of six basic residues has been deleted (Δ183–188). In each case the GTPase is compared to a background comprising the GDP-locked/empty forms of seventeen GTPases. Known effectors and GAPs are indicated along with proteins whose GO annotations include the term DNA-binding or RNA-binding. The removal of the basic stretch reduces contamination with the latter proteins. (C) Immunoblot of strepatividin precipitates from cells in which MitoID had been performed with the indicated versions of Rab5A(Q79L). A representative lysate, and the precipitates were blotted for the indicated Rab5 effectors. Mutation of Ser84 reduces biotinylation of OSBPL9, but has no effect on that of two other effectors, EEA1 and PIK3R4.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (E. coli) | BL21 GOLD (DE3) | Agilent Technologies | 230132 | Used for expression |
| Strain, strain background (E. coli) | Alpha-select gold | Bio-line | BIO-85027 | Used for cloning |
| Cell line (H. sapiens) | mCherry-Parkin, HeLa | (Lazarou et al., 2015) | ||
| Cell line (H. sapiens) | HEK293 | ATCC | ATCC CRL-11268 RRID:CVCL_1926 | |
| Cell line (H. sapiens) | HeLa | ATCC | ATCC CCL-2 RRID:CVCL_0030 | |
| Recombinant DNA reagent | pcDNA3.1 | Clontech | V79020 | |
| Recombinant DNA reagent | pGEX6p2 | GE Healthcare Life Sciences | GE28-9546-50 | |
| Detection reagent | Streptavidin-HRP | Cell Signaling Technology | 3999S RRID:AB_10830897 | WB: 1:300 |
| Antibody | Rat monoclonal anti-HA (3F10) | Roche | 867 423 001 RRID:AB_2314622 | IHC: 1:300 |
| Antibody | Goat polyclonal anti-giantin | Santa Cruz | sc-46993 RRID:AB_2279271 | IHC: 1:200 |
| Antibody | Rabbit polyclonal human COXIV (3E11) | New England Biolabs | 4850S RRID:AB_2085424 | IHC: 1:200 |
| Antibody | Rabbit polyclonal anti- ZFPL1 | Sigma | HPA014909 RRID:AB_1859055 | IHC: 1:300 |
| Antibody | Mouse monoclonal anti-TOM20 | BD Transduction Labs | 612278 RRID:AB_399595 | IHC: 1:200 |
| Antibody | Sheep polyclonal anti-TGN46 | ABD serotec | AHP500G RRID:AB_323104 | IHC: 1:300 |
| Antibody | Rabbit polyclonal anti-golgin-84 | Sigma | HPA000992 RRID:AB_1079009 | IHC: 1:300 |
| Antibody | Mouse monoclonal anti-HIS6 tag | Abcam | Ab18184 RRID:AB_444306 | WB: 1:1000 |
| Antibody | Rabbit polyclonal Anti-STAMBPL1 | Sigma | SAB4200146 RRID:AB_10622601 | WB: 1:1000 |
| Antibody | Rabbit polyclonal anti-NDE1 | Life Technologies | 711424 RRID:AB_2692258 | WB: 1:500 |
| Antibody | Rabbit polyclonal anti-TBCK | Cambridge Bioscience | HPA039951 RRID:AB_10795300 | WB: 1:500 |
| Antibody | Rabbit polyclonal anti-RELCH/KIAA1468 | Cambridge Bioscience | HPA040038 RRID:AB_10793860 | WB: 1:1000 |
| Antibody | Rabbit monoclonal anti-OSBPL9 | Abcam | ab151691 | WB: 1:1000 |
| Antibody | Mouse monoclonal anti-FLAG (M2) | Sigma | F1804 RRID:AB_262044 | WB: 1:5000 |
| Antibody | Rabbit polyclonal anti-ARFGEF3 | ThermoFisher Scientific | PA5-57623 RRID:AB_2638119 | WB: 1:1000 |
| Antibody | Alexa 488 Donkey polyclonal anti-Rat IgG | ThermoFisher Scientific | A21208 RRID:AB_2535794 | IHC: 1:400 |
| Antibody | Alexa 647 Donkey polyclonal anti-Goat IgG | ThermoFisher Scientific | A32849 RRID:AB_2762840 | IHC: 1:400 |
| Antibody | Alexa 555 Donkey polyclonal anti-Rabbit IgG | ThermoFisher Scientific | A32794 RRID:AB_2762834 | IHC: 1:400 |
| Antibody | Alexa 647 Donkey polyclonal anti-Rabbit IgG | ThermoFisher Scientific | A32795 RRID:AB_2762835 | IHC: 1:400 |
| Antibody | Alexa 555Donkey polyclonal anti-mouse IgG | ThermoFisher Scientific | A32773 RRID:AB_2762848 | IHC: 1:400 |
| Recombinant DNA reagent | STAMBPL1 | Addgene | #22559 | |
| Recombinant DNA reagent | TBCK | Insight Biotechnology | RC203605 | |
| Recombinant DNA reagent | HPS3 | Insight Biotechnology | RC204569 | |
| Recombinant DNA reagent | NDE1 | Stratech Scientific | HG15586 | |
| Recombinant DNA reagent | Rab2AQ65L-BirA*-HA-MAO | This study | pN40 | Deposited at Addgene |
| Recombinant DNA reagent | Rab2AS20N-BirA*-HA-MAO | This study | JB2 | Deposited at Addgene |
| Recombinant DNA reagent | Rab5AQ79L-BirA*-HA-MAO | This study | JB28 | Deposited at Addgene |
| Recombinant DNA reagent | Rab5AS34N-BirA*-HA-MAO | This study | JB40 | Deposited at Addgene |
| Recombinant DNA reagent | Rab6AQ72L-BirA*-HA-MAO | This study | pO40 | Deposited at Addgene |
| Recombinant DNA reagent | Rab6AT27N-BirA*-HA-MAO | This study | JB4 | Deposited at Addgene |
| Recombinant DNA reagent | Rab7AQ67L-BirA*-HA-MAO | This study | JB93 | Deposited at Addgene |
| Recombinant DNA reagent | Rab7AT22N-BirA*-HA-MAO | This study | JB94 | Deposited at Addgene |
| Recombinant DNA reagent | Rab8AQ67L-BirA*-HA-MAO | This study | JB24 | Deposited at Addgene |
| Recombinant DNA reagent | Rab8AT22N-BirA*-HA-MAO | This study | JB23 | Deposited at Addgene |
| Recombinant DNA reagent | Rab9AQ66L-BirA*-HA-MAO | This study | JB84 | Deposited at Addgene |
| Recombinant DNA reagent | Rab9AS21N-BirA*-HA-MAO | This study | JB85 | Deposited at Addgene |
| Recombinant DNA reagent | Rab10Q68L-BirA*-HA-MAO | This study | JB81 | Deposited at Addgene |
| Recombinant DNA reagent | Rab10T23N-BirA*-HA-MAO | This study | JB82 | Deposited at Addgene |
| Recombinant DNA reagent | Rab11AQ69L-BirA*-HA-MAO | This study | JB20 | Deposited at Addgene |
| Recombinant DNA reagent | Rab11AS25N-BirA*-HA-MAO | This study | JB19 | Deposited at Addgene |
| Recombinant DNA reagent | Rab11BQ70L-BirA*-HA-MAO | This study | pX49 | Deposited at Addgene |
| Recombinant DNA reagent | Rab11BS25N-BirA*-HA-MAO | This study | pY49 | Deposited at Addgene |
| Recombinant DNA reagent | Rab18Q67L-BirA*-HA-MAO | This study | JB11 | Deposited at Addgene |
| Recombinant DNA reagent | Rab18S22N-BirA*-HA-MAO | This study | JB10 | Deposited at Addgene |
| Recombinant DNA reagent | Rab30Q68L-BirA*-HA-MAO | This study | JB15 | Deposited at Addgene |
| Recombinant DNA reagent | Rab30T23N-BirA*-HA-MAO | This study | JB3 | Deposited at Addgene |
| Recombinant DNA reagent | Rab33BQ92L-BirA*-HA-MAO | This study | JB26 | Deposited at Addgene |
| Recombinant DNA reagent | Rab33BT47N-BirA*-HA-MAO | This study | JB25 | Deposited at Addgene |
| Recombinant DNA reagent | nRasG12D-BirA*-HA-MAO | This study | JB95 | Deposited at Addgene |
| Recombinant DNA reagent | nRasS17N-BirA*-HA-MAO | This study | JB96 | Deposited at Addgene |
| Recombinant DNA reagent | Rac1Q60L-BirA*-HA-MAO | This study | JB42 | Deposited at Addgene |
| Recombinant DNA reagent | Rac1T17N-BirA*-HA-MAO | This study | JB54 | Deposited at Addgene |
| Recombinant DNA reagent | RalBG23V-BirA*-HA-MAO | This study | JB97 | Deposited at Addgene |
| Recombinant DNA reagent | RalBS29A-BirA*-HA-MAO | This study | JB98 | Deposited at Addgene |
| Recombinant DNA reagent | RhoAQ63L-BirA*-HA-MAO | This study | JB68 | Deposited at Addgene |
| Recombinant DNA reagent | RhoAT19N-BirA*-HA-MAO | This study | JB55 | Deposited at Addgene |
| Recombinant DNA reagent | Cdc42G12V-BirA*-HA-MAO | This study | JB48 | Deposited at Addgene |
| Recombinant DNA reagent | Cdc42T17N-BirA*-HA-MAO | This study | JB57 | Deposited at Addgene |
| Recombinant DNA reagent | RhebQ64L-BirA*-HA-MAO | This study | JB87 | Deposited at Addgene |
| Recombinant DNA reagent | RhebS20N-BirA*-HA-MAO | This study | JB88 | Deposited at Addgene |
| Peptide, recombinant protein | 3X FLAG peptide | Sigma | F4799 | |
| Commercial assay or kit | Amersham ECL Prime detection agent | GE Healthcare Lifesciences | RPN2232 | Western blots |
| Commercial assay or kit | Supersignal West Femto Maximum Sensitivity Substrate | Thermo Scientific | 34095 | Western blots |
| Chemical compound | Biotin | Sigma | B4501 | |
| Software, algorithm | Perseus | (Tyanova et al., 2016). | RRID:SCR_015753 | |
| Other | Dynabeads MyOne Streptavidin C1 | Invitrogen | 65002 | Isolation of biotinylated proteins |
| Other | Anti-FLAG M2 affinity resin | Sigma | A2220 RRID:AB_10063035 | Anti-FLAG immunoprecipitation |
| Other | Fugene 6 | Promega | E2691 | Transfection |
| Other | Neutravidin-FITC | Invitrogen | 31006 | |
| Other | Glutathione sepharose 4B | GE Healthcare Lifesciences | 17075601 | GST affinity chromatography |
| Other | Vectashield | Vector Laboratories Inc | H-1000 | Mounting media |
| Other | Novex 4–20% Tris-Glycine gels | Thermofisher | XP04202BOX | Pre-cast gels |
| Other | Gibco Opti-MEM | Fisher Scientific | 31985070 | Media |
Additional files
-
Supplementary file 1
Analysis of mass-spectral data from GTPase MitoID based on spectral counts.
Proteins identified by MitoID with at least one GTPase. Total spectral counts for biological triplicates of GTP-bound (T) and GDP-bound forms (D) are in Sheet S1A. Spectral counts were converted into D scores and WD scores based on the CompPASS platform. D scores in Sheet S1B, mean D scores in Sheet S1C, WD scores in Sheet S1D and mean WD scores in Sheet S1E (see tabs at bottom of sheet). Excel (.xlsx) file.
- https://doi.org/10.7554/eLife.45916.016
-
Supplementary file 2
Analysis of mass-spectral data from GTPase MitoID based on peak intensities.
Label free quantitation (LFQ) intensities from MaxQuant are shown for all proteins identified by MitoID with at least one GTPase. Excel (.xlsx) file.
- https://doi.org/10.7554/eLife.45916.017
-
Supplementary file 3
Volcano plot data from mass spectral peak intensities from MitoID with GTPases of the Rab family.
For each Rab form indicated in the tabs, the LFQ intensities of every protein found in at least two of the triplicates was compared to the values obtained with the GDP forms of all GTPases by using the Perseus platform. The difference, expressed as log2 of the ratio, and the P-value for the significance of the difference are shown, with these being used to generate the volcano plots shown in the figures. Also shown are the WD score for each interaction as obtained from analysis of spectral counts. Excel (.xlsx) file.
- https://doi.org/10.7554/eLife.45916.018
-
Supplementary file 4
Volcano plot data from mass spectral peak intensities from MitoID with GTPases of the Rho and Ras families.
For each GTPase form indicated in the tabs, the LFQ intensities of every protein found in at least two of the triplicates was compared to the values obtained with the GDP forms of all GTPases by using the Perseus platform. The difference, expressed as log2 of the ratio, and the P-value for the significance of the difference are shown, with these being used to generate the volcano plots shown in the figures. Also shown are the WD score for each interaction as obtained from analysis of spectral counts. Excel (.xlsx) file.
- https://doi.org/10.7554/eLife.45916.019
-
Supplementary file 5
Coverage of previously reported effectors of mammalian Rab2A and Rab5A by MitoID and S2 cell affinity chromatography.
Previously reported effectors for mammalian Rab2 and Rab5 are listed along with their coverage by MitoID and by a previous screen for Rab effectors based on affinity chromatography of Drosophila S2 cell lysates (Gillingham et al., 2014). Additional tables show for the MitoID interactions the comparisons of LFQ intensities and the WD scores as in Supplementary file 4. This illustrates typical such scores for bona fide effectors. The FAM71 family (also called the GARI family) have also been reported to bind human Rab2 (Fukuda et al., 2008). However, this interaction is specific for Rab2B and not the Rab2A used for MitoID, and the family is only present in vertebrates, and so it is not included in the comparison. Excel (.xlsx) file.
- https://doi.org/10.7554/eLife.45916.020
-
Transparent reporting form
- https://doi.org/10.7554/eLife.45916.021





