Visualizing trypanosomes in a vertebrate host reveals novel swimming behaviours, adaptations and attachment mechanisms
Figures

Majority of trypanosomes in freshly drawn blood are tumblers.
Blood was freshly drawn from carp and T. carassii swimming behaviour analysed immediately using high-resolution microscopy at 240 frames per second (fps). (A) Relative percentage of tumblers, and intermediate or persistent swimmers (defined in the text) was calculated over a total number of 944 T. carassii, isolated from six different carp infections and imaged over 60 independent acquisitions. (B) Representative tracks of a tumbler (green), intermediate (yellow) and persistent (red) swimmer. The diameter of the circles (23 µm) indicates the average cell-body size of a trypanosome as also shown in (C). The inset table summarizes the straight-line distance covered by the trypanosome (between the first and last track point); the total track length, that is the path covered by the trypanosome in approximately 20 s of acquisition time, indicated in matching colours; and the average speed (μm/s) was calculated on a selection of the acquisitions used in (A). For tumblers, the displacement of the posterior end was used as tracking point. (C) Detailed image of two trypanosomes indicating the total body length including the flagellum (left) and the total cell-body length excluding the flagellum (right). Measurements were acquired on high-resolution images of at least 10 freshly isolated trypanosomes obtained from four independent infections, using more than 20 frames within the same acquisition. Quantification of trypanosome length, swimming speed and directionality was performed with ImageJ-Fijii using the MTrack plug-in. Video 1 displays high-speed videos of the swimming behaviour of tumblers, intermediate and persistent swimmers in carp blood, or of trypanosomes in serum or culture medium.
-
Figure 1—source data 1
Percentage of tumblers, intermediate and persistent swimmer.
- https://doi.org/10.7554/eLife.48388.004
-
Figure 1—source data 2
T. carassii speed in vitro.
- https://doi.org/10.7554/eLife.48388.005
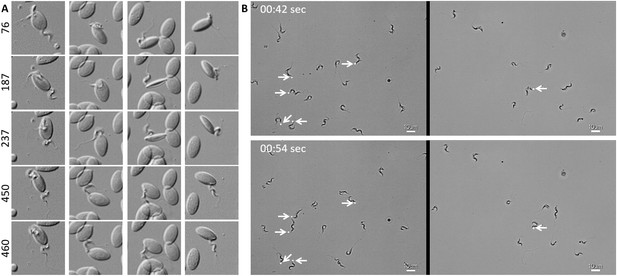
T. carassii attaches to cells or surfaces through its posterior end leaving the flagellum free to move.
(A) Blood was freshly drawn from carp and trypanosomes' swimming behaviour immediately imaged using high-resolution microscopy at 240 frames fps. Images are frames (indicated by the numbers) of four different locations within the same field of view, selected from the corresponding Video 2. Note how the posterior end of the parasites is attached to the red blood cell and the flagellum is free to move. (B) Selected frames from Video 2, at the indicated time points, show how T. carassii can also adhere to glass surfaces through the posterior end (white arrow) leaving the body and flagellum free to move.
-
Figure 2—source data 1
Susceptibility of zebrafish larvae to T. carassii infection and kinetics of parasitaemia.
- https://doi.org/10.7554/eLife.48388.008
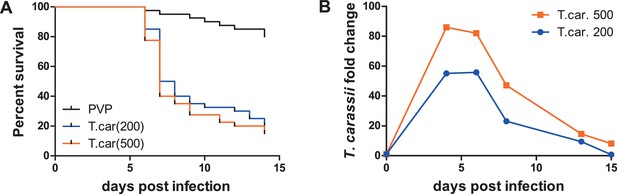
Zebrafish larvae are susceptible to T. carassii infection.
Zebrafish larvae (5 dpf) were injected with the indicated number of T. carassii per fish. PVP was used as injection control. (A) Kinetics of zebrafish larval survival. Fish (n = 40/group) were observed daily for signs of infection and survival. (B) Kinetics of parasitaemia. Trypanosome levels were quantified by real-time quantitative-PCR using T. carassii hsp70-specific primers. RNA of five fish was pooled at each time point. Expression was normalized relative to the host house-keeping gene elongation factor-1 alpha and expressed relative to fish injected with the corresponding trypanosome dose at time point zero.
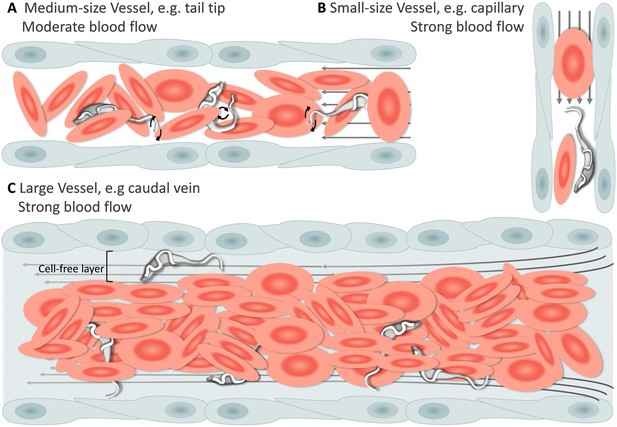
Schematic representation of T. carassii swimming behaviour in blood vessels of various sizes.
In general, in vessels with an intact blood flow (grey arrows) and a normal number of red blood cells (RBC), trypanosomes are dragged passively by the flow along with RBC. Under these conditions, trypanosomes were never seen swimming directionally against the flow or faster than RBC. (A) In medium-sized blood vessels with moderate flow, while being dragged passively by the flow, trypanosomes can either curl or stretch their body, as well as occasionally propel their flagellum in the same or opposite direction to the blood flow. (B) In small-sized blood vessels of one-cell diameter, such as intersegmental capillaries (ISC), trypanosomes are pushed forward by the blood flow and by colliding RBC. (C) In large-sized blood vessels, the blood flow is very strong and the density of RBC high, making it more difficult to detect trypanosomes without the aid of high-speed microscopy. Only the occasional trypanosome that would slow down by bouncing against the vessel wall would be visible in the cell-free layer. Video 3 contains high-speed videos showing details of the trypanosome movements schematically depicted above.
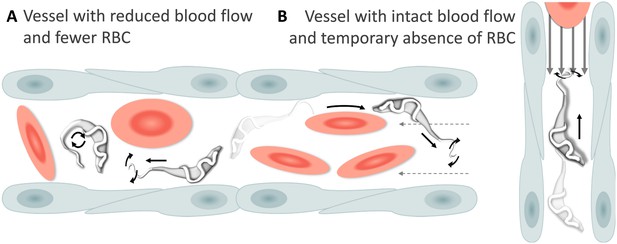
Schematic representation of T. carassii swimming behaviour in blood vessels with altered blood flow or red blood cell (RBC) number.
(A) Blood vessels with a reduced blood flow (dashed arrows) and fewer RBC. (B) Blood vessels with an intact blood flow (grey arrows) but with a temporary absence of RBC. In both cases, trypanosomes can swim directionally (black straight arrows) by propelling the flagellum (distorted circular arrows) in the same or opposite direction to the flow, or can tumble (circular arrows). Video 4 contains high-speed videos showing trypanosome movement in medium-sized blood vessels and in capillaries as schematically depicted.
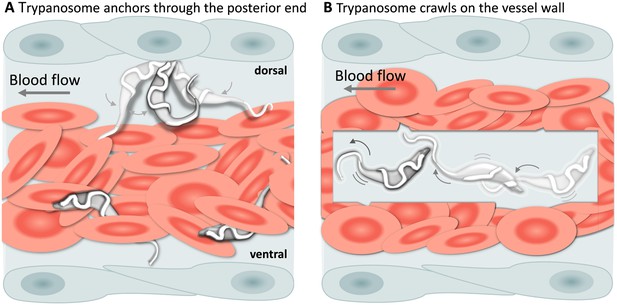
T. carassii attachment to the blood vessel wall.
(A) T. carassii anchor themselves by their posterior end, leaving the cell body and the flagellum free to move. Anchoring occurred only to the dorsal luminal side of the caudal vein and was not observed in other blood vessels. (B) Trypanosomes crawl along the vessel wall of the caudal vein; the transparent square allows visualization of trypanosomes through the pack of red blood cells. Crawling occurred everywhere in the vein and involved the entire cell body. Video 5 contains high-speed videos showing anchored and crawling T. carassii in the caudal vein as schematically depicted above.
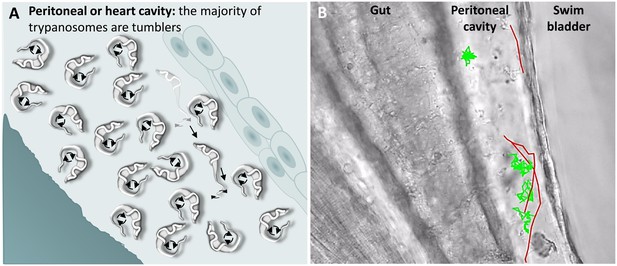
T. carassii swimming behaviour in tissue fluids outside blood.
(A) Schematic representation of T. carassii swimming in the peritoneal or heart cavity, both environments without hydrodynamic flow and red blood cells; here, most of the trypanosomes are tumblers, only occasionally was a persistent swimmer observed. (B) Selected frame from Video 6, capturing trypanosomes in the peritoneal cavity. More than 100 trypanosomes are present but are not all in focus in the selected frame; the majority are tumblers. The tracks of four representative tumblers (green) and of the only three persistent swimmers (red) are shown. Video 6 contains high-speed videos showing the location and swimming behaviours described above.

T. carassii swimming behaviour in tissues.
(A) Schematic representation of T. carassii swimming in compact tissues such as those in the fins. Most trypanosomes are directional swimmers and both forward and backward swimming were observed. (B) Selected frame from Video 7 showing the tracks of representative persistent swimmers identified in the fins. (C) In less compact tissues and in capillaries without blood flow, trypanosomes could invert their swimming direction in a ‘whip-like’ motion using the available three-dimensional space of the capillary or tissue. The ‘whip-like’ motion combines the swing of the flagellum along one plane (thin arc arrows), accompanied by a 180°C rotation of the flexible cell body along a third axis (rotational arrows). (D) In tissues where trypanosomes reach dead ends such as the interstitial space between vessels, persistent forward swimming translates into a drilling (auger) movement. Video 7 and Video 8 contain high-speed videos showing all locations and swimming behaviours schematically depicted.
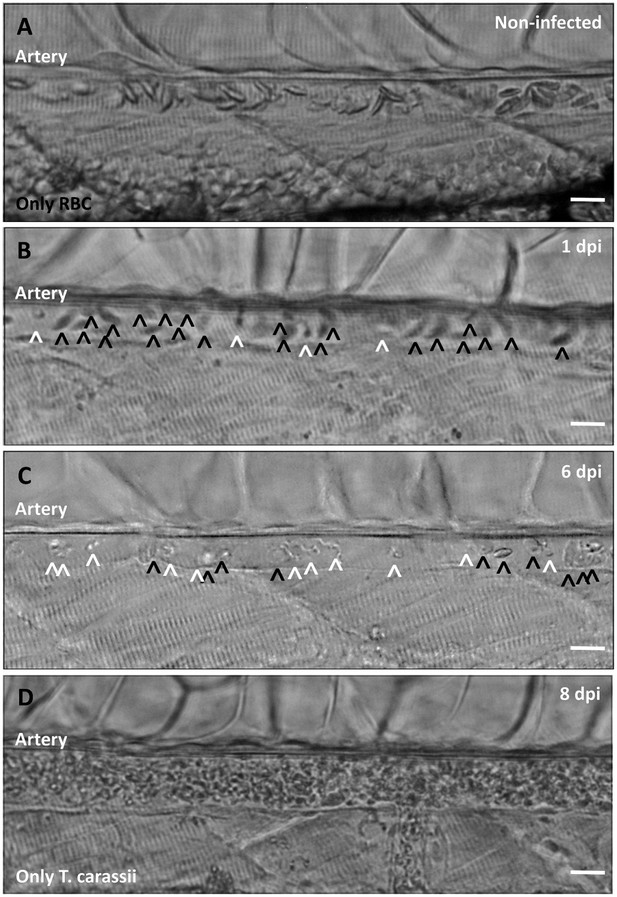
Onset of anaemia during T. carassii infection.
Zebrafish larvae (5 dpf) were infected with 200 T. carassii or injected with PVP as non-infected control. Images are selected frames depicting the caudal artery, extracted from high-speed videos where trypanosomes (white open-arrow heads) and red blood cells (RBC, black open-arrow heads) were identified and tracked. (A) Artery of a control, non-infected, fish. Only RBC are present. (B) Artery of an infected fish, 1 dpi, showing a high ratio of RBC:trypanosomes. This frame corresponds to seconds 04:20-04:23 in Video 3, where the same trypanosomes were tracked. (C) Artery of an infected fish, 6 dpi, showing a reduced ratio of RBC:trypanosomes, indicating the onset of anaemia. (D) Artery of an infected fish suffering from severe anaemia, 8dpi, where only trypanosomes are present. The frame is extracted from the corresponding Video 9. Scale bars indicate 25 µm.
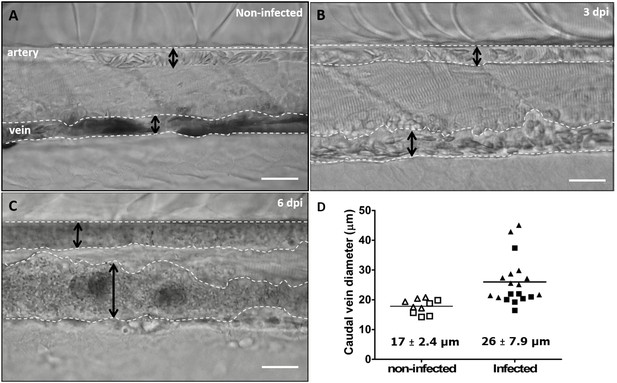
Advanced stages of T. carassii infection lead to vasodilation of the caudal vein.
Wild type zebrafish larvae (5 dpf) were infected with 200 T. carassii or injected with PVP as non-infected control. Images are selected frames from high-speed videos. (A-C) Representative images of caudal artery and caudal vein (dashed lines) region at various time points after infection. Scale bars indicate 50 µm. (D) Maximum diameter of the caudal vein in non-infected (open symbols) and infected individuals (closed symbols) at 2–3 dpi (squares) and 6–8 dpi (triangles). Each value is the average of at least three measurements taken at different locations within the caudal vein of the same individual. Numbers indicate average and standard deviation.
-
Figure 10—source data 1
Vasodilation in T. carassii infected larvae.
- https://doi.org/10.7554/eLife.48388.025
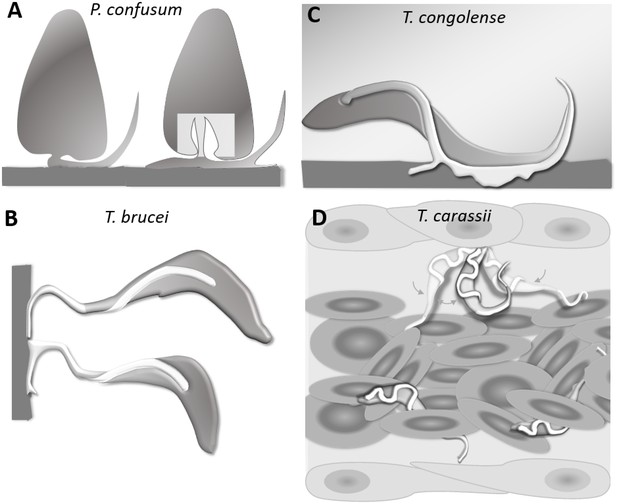
Schematic drawing depicting the attachment of various trypanosome species.
(A) Haptomonads stages of Paratrypanosoma confusum: adhesion occurs through an attachment pad forming from the bulge at the base of the flagellum involving extensive remodelling of the flagellum membrane (based on Skalický et al., 2017). The square indicates the location of the flagellar pocket. (B) T. brucei epimastigotes attached through the flagellum to the brush border of the salivary gland epithelium (based on Beattie and Gull, 1997; Schuster et al., 2017; Vickerman and Tetley, 1990). (C) T. congolense adhesion to bovine aorta endothelial cell line via extensive membrane protrusions (filopodia) of the membrane-attached flagellum (based on Beattie and Gull, 1997; Hemphill and Ross, 1995). (D) T. carassii attached through the posterior end, leaving the cell body and flagellum free to move, as also shown in Figure 5.
Videos
Swimming behaviour of T. carassii in freshly drawn carp blood, or blood diluted with either carp serum or culture medium, showing representative tumblers, intermediate and persistent swimmers.
https://doi.org/10.7554/eLife.48388.006T. carassii attaches to cells or surfaces through its posterior end leaving the flagellum free to move.
Trypanosomes were also observed to swim backwards.
Swimming behaviour of T. carassii in small, medium and large-sized vessels with an intact blood flow.
Under these conditions, trypanosomes are dragged passively by the flow or pushed forward by colliding RBC.
Swimming behaviour of T. carassii in medium-sized blood vessels with interrupted blood flow, or in capillaries with intact blood flow and reduced number of red blood cells.
Under these conditions, trypanosomes can repeatedly invert direction, swim directionally or tumble.
T. carassii anchor to the dorsal luminal side of the caudal vein and crawl along the vessel wall of veins.
T. carassii movement in fluids outside the blood vessels and in tissues.
Swimming behaviour of T. carassii extravasated from blood vessels into tissue fluids that lack blood flow and red blood cells.
These include the peritoneal and heart cavities.
Swimming behaviour of T. carassii in the narrow spaces of the fins, capturing persistent forward and backward swimmers.
https://doi.org/10.7554/eLife.48388.020Swimming behaviour of T. carassii in various tissues.
https://doi.org/10.7554/eLife.48388.021Anaemia is a hallmark of T. carassii infection.
https://doi.org/10.7554/eLife.48388.023Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Danio rerio) | elongation factor-1α (ef1a) | NA | ZDB-GENE-990415–52 | template for primers for RQ-PCR analysis |
| Gene (Trypanosoma carassii) | heat-shock protein-70 (hsp70) | NA | GeneBank-FJ970030.1 | template for primers for RQ-PCR analysis |
| Strain, strain background (Cyprinus carpio) | Wild type common carp, R3xR8 strain | doi:10.1016/0044-8486(95)91961 T | ||
| Strain, strain background (Danio rerio) | Wild type zebrafish, AB strain | European Zebrafish Resource Center (EZRC) | https://www.ezrc.kit.edu/index.php | |
| Strain, strain background (Danio rerio) | casper strain | White et al. (2008) | optically transparent | |
| Strain, strain background (Danio rerio) | Tg(fli:egfp)y1 (casper) | doi:10.1038/nrg888 | optically transparent line, marking the vasculature with green fluorescent protein | |
| Strain, strain background (Trypanosoma carassii) | TsCc-NEM strain | Overath et al. (1998) |
Additional files
-
Transparent reporting form
- https://doi.org/10.7554/eLife.48388.027





