Straightjacket/α2δ3 deregulation is associated with cardiac conduction defects in myotonic dystrophy type 1
Figures
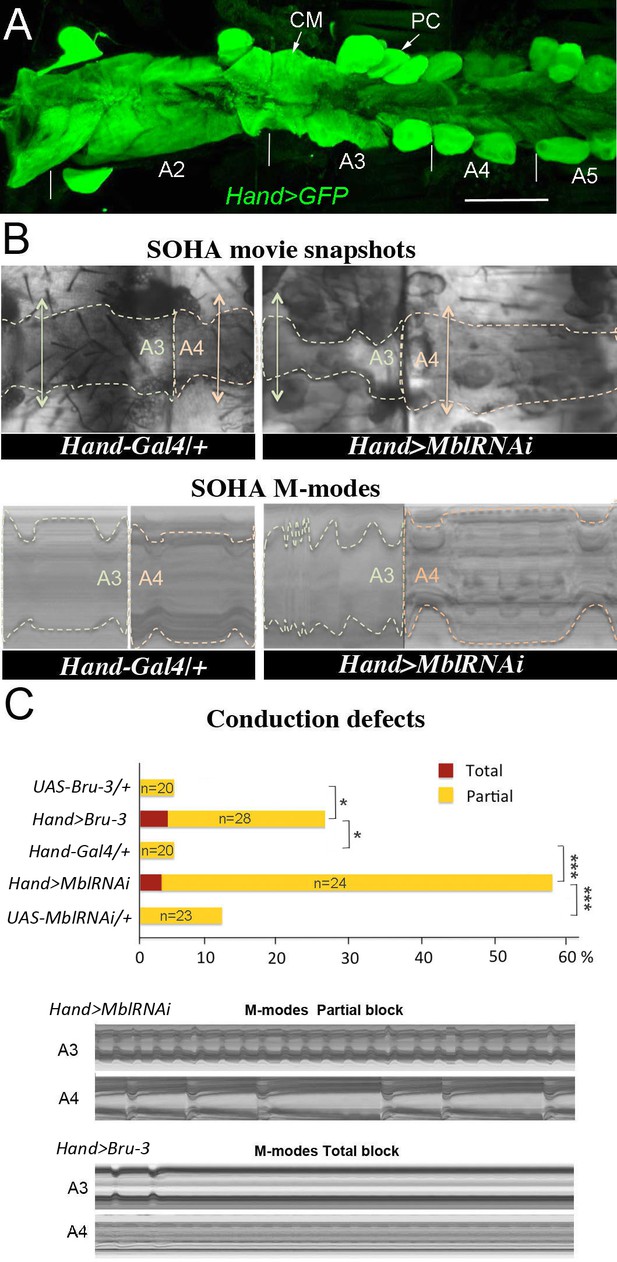
Cardiac-specific knockdown of MBNL1 ortholog and overexpression of CELF1 counterpart in Drosophila lead to asynchronous heartbeats.
(A) The adult Drosophila heart expressing Hand-Gal4-driven GFP (Hand>GFP). Note that this Gal4 line drives expression exclusively in the heart. Arrows indicate cardiomyocytes (CM) and pericardial cells (PC) and A2–A5 denote abdominal segments. Scale bar, 150 μm. (B) Movie and M-modes views illustrating asynchronous heartbeats in Hand>MblRNAi flies registered in two adjacent heart segments (A3 and A4). Two-sided arrows indicate heart diameter in diastolic state. (C) Barplot graph showing percentage of flies with conduction defects in the different genetic contexts tested. Note the higher impact of attenuation of Mbl compared to overexpression of Bru-3. Number of fly hearts tested (n) is indicated and statistical significance (Fisher’s exact test) denoted by * (p<0.05) and *** (p<0.001). Below are examples of M-modes illustrating ‘partial’ and ‘total’ heart blocks observed in Hand>MblRNAi and Hand>Bru-3 flies developing conduction defects.
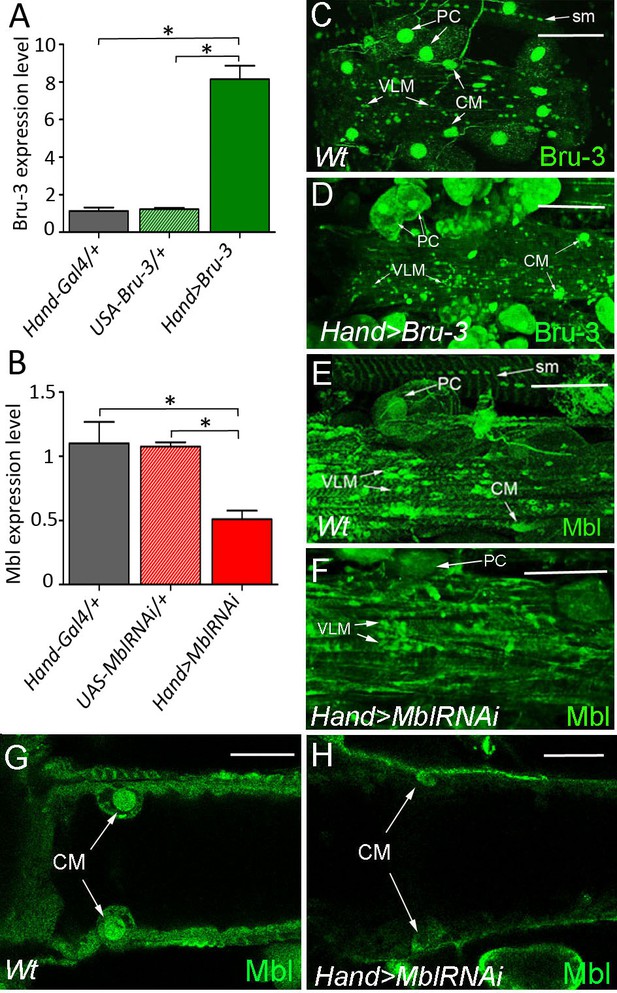
Bru-3 and Mbl are expressed in the adult fly heart and their transcript and protein levels are affected in Hand>Bru-3 and Hand>MblRNAi flies.
(A–B) RT-qPCR analyses of Bru-3 overexpression and Mbl attenuation in the adult hearts from Hand>Bru-3 (A) and Hand>MblRNAi (B) flies. Three biological replicates (n = 10–12 hearts per replicate) were analysed for each condition. Statistical significance (Mann Whitney U test) is denoted by * (p<0.05) (C–F) Optical projection views of A3 cardiac segment showing expression of Bru-3 (C–D) and Mbl (E–H) proteins in the hearts of wild-type (Wt) (C, E), Hand>Bru-3 (D) and Hand>MblRNAi (F) flies. (G, H) show confocal optical sections at the level of cardiomyocytes (CM) to document attenuation of Mbl in CMs nuclei. Bru-3 protein is detected in the nuclei of CMs, pericardial cells (PC) and ventral longitudinal muscle (VLM). A faint granular cytoplasmic expression could also be detected in PCs (C). Notice an increase of Bru-3 levels in Hand>Bru-3 context (D) with more cytoplasmic Bru-3 signal detected in PCs and CMs. Similarly, Mbl accumulation is observed in CMs, PCs and the VLM nuclei (E). Apparent sarcomeric localization of Mbl is also detected in circular fibers of CMs and in the VLM (E). Notice Bru-3 and Mbl expression in nuclei of somatic muscles (sm). Scale bars (C–F) 100 μm; (G,H) 50 μm.
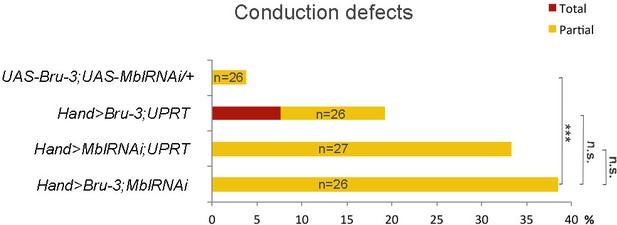
Effect of simultaneous cardiac attenuation of Mbl and overexpression of Bru-3 on conduction defects.
Barplot graph showing percentage of flies with conduction defects. Note a moderate additive effect of simultaneous attenuation of Mbl and overexpression of Bru-3. Number of fly hearts tested (n) is indicated and statistical significance (Fisher’s exact test) denoted by ns (p>0.05) and *** (p<0.001).
An example of asynchronous heartbeat phenotype observed in Hand>MblRNAi context.
High-speed 20 s time-lapse movie.
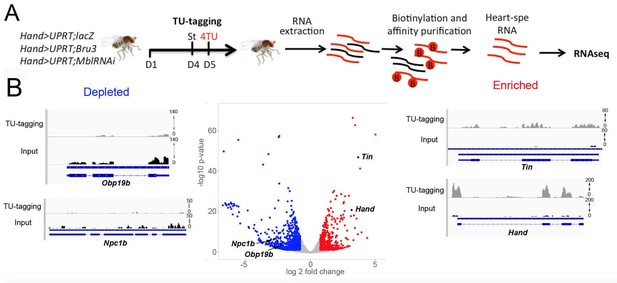
Cardiac-specific transcriptional profiling using TU-tagging method.
(A) Pipeline of heart-targeted transcriptional profiling using TU-tagging. Note that Gal4- inducible UPRT transgene (UAS-UPRT) has been combined with UAS-MblRNAi and with UAS-Bru-3 for the purpose of TU-tagging. Hand>UPRT;lacZ is the control line used to identify pathogenic gene deregulations in Hand>UPRT;MblRNAi and Hand>UPRT;Bru-3 contexts. Flies were starved at day 4 for 6 hr before being transferred to 4TU containing food for 12 hr. (B) Volcano plot and IGV tracks from control Hand>UPRT;lacZ flies show examples of enrichment of heart-specific genes (e.g. Hand, Tin) (red, right side) and depletion of non-heart-expressed genes (blue, left side), thus validating the specificity of heart targeting.
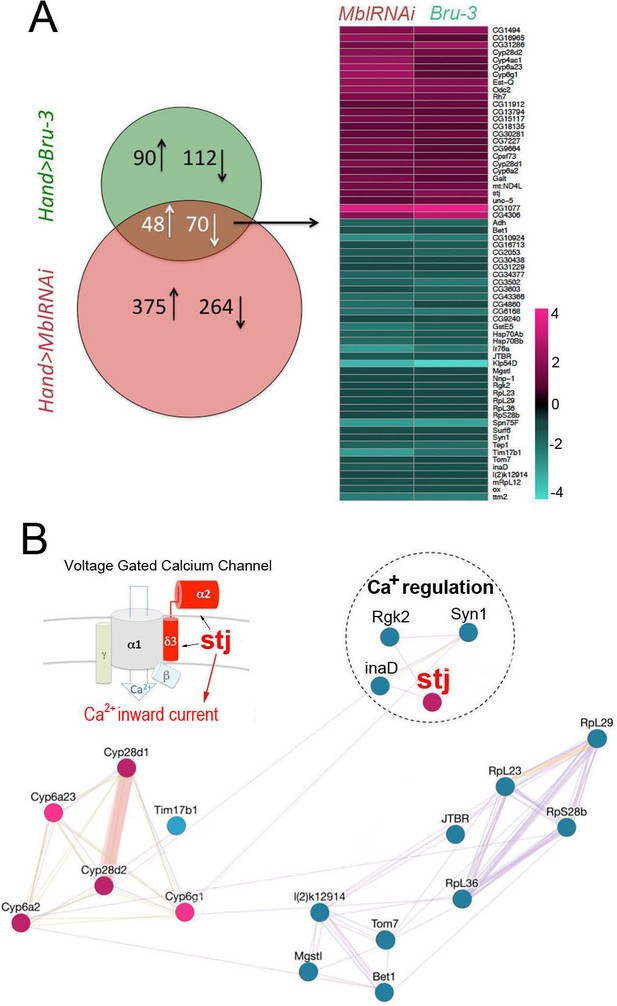
Heart-specific transcriptional profiling of DM1 flies identifies deregulation of genes controlling cellular calcium level.
(A) Venn diagrams of genes deregulated in Hand>UPRT;MblRNAi and in Hand>UPRT;Bru-3 contexts (FC > 1.7) followed by heatmap of commonly deregulated genes. (B) Genemania interaction network of conserved candidates including stj, Rgk2, Syn1 and inaD known to be involved in Ca2 regulation. A scheme presenting the structure of the voltage-gated calcium channel and its regulatory component Stj/α2δ3 is included. Color code in genemania network represents up and down regulation according to the heatmap. Cyan/Blue intensity code indicates fold change of genes expression in the heatmap.
-
Figure 3—source data 1
A list of Drosophila genes and their Human orthologs deregulated in both Hand>Bru-3 and Hand>MblRNAi contexts.
Orthology scores were obtained from DIOPT tool (DRSC Integrative Ortholog Prediction Tool) (Hu et al., 2011) which predicts orthologs, based on sequence homology, phylogenetic trees, and functional similarity (www.flyrnai.org/cgi-bin/DRSC_orthologs.pl).
- https://cdn.elifesciences.org/articles/51114/elife-51114-fig3-data1-v1.xlsx
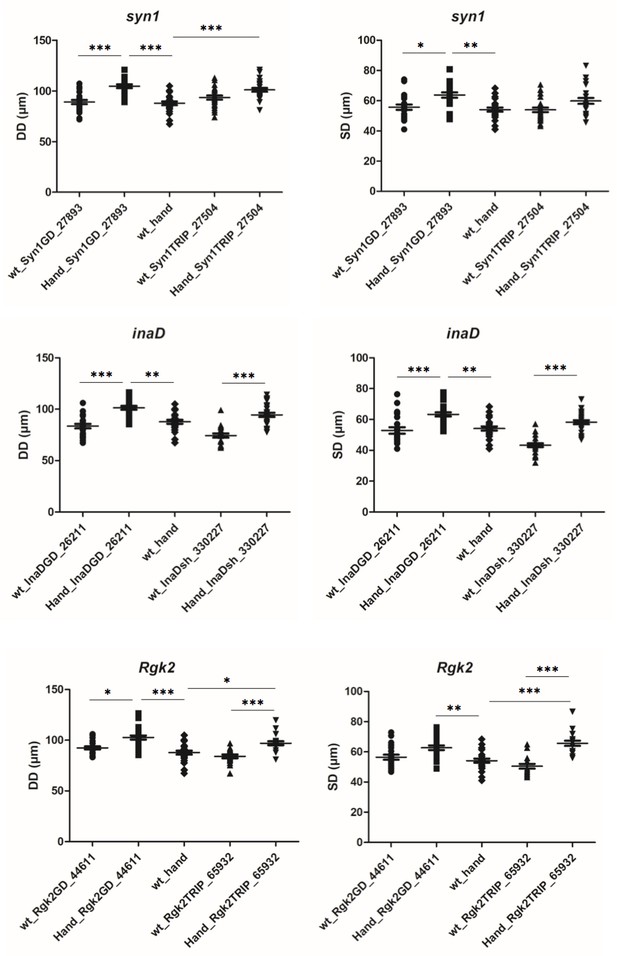
Attenuation of inaD, Syn1 and Rgk2 all lead to an increase of diastolic (DD) and systolic (SD) heart diameters.
For each candidate gene effects of attenuation were tested using two different UAS-RNAi lines crossed with Hand-Gal4 driver. More than 20 flies for each genotype were analysed by SOHA. Statistical significance of DD and SD changes was assessed using one-way ANOVA, Kruskal-Wallis Dunn’s multiple comparison post-test (* - p<0.05; ** - p<0.01; *** - p<0.001).
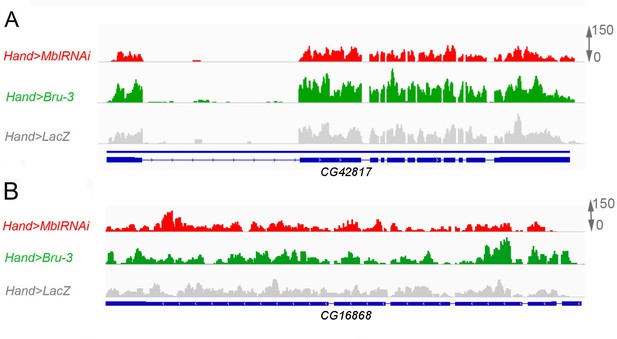
The expression levels of CG42617 and CG16868, two additional α2δ protein-coding genes are not affected in the heart of DM1 fly models.
Normalized RNAseq IGV tracks in control (Hand>lacZ) and in pathogenic DM1 contexts (Hand>MblRNAi and Hand>Bru-3) are shown aligned with genomic exon/intron organization of CG42617 (A) and CG16868 (B).
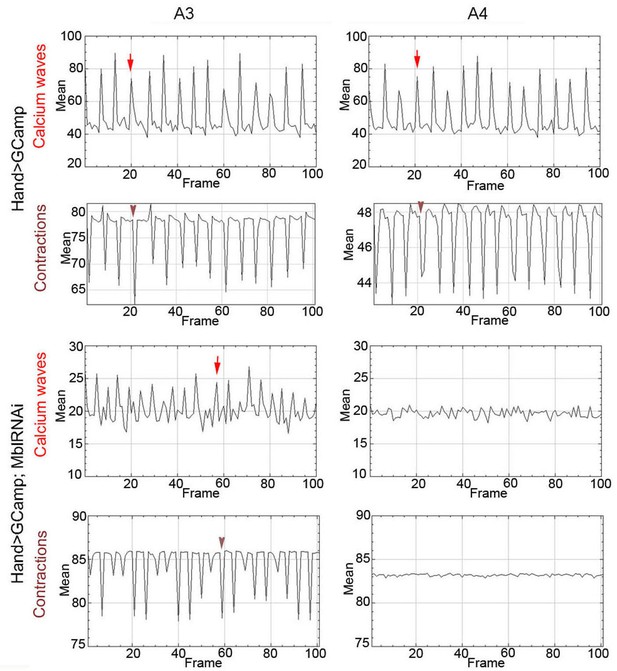
Calcium waves in asynchronous Drosophila heart.
In wild type hearts, calcium waves underlie cardiac contractions so that calcium peaks (arrows) align with the onset of contractions (arrowheads) in both A3 and A4 segments. In contrast, in asynchronously beating Hand>MblRNAi heart, in the anterior A3 segment, calcium peaks (arrows) are not always in perfect alignment with contractions (arrowheads) and could not be detected in A4 segment that does not beat. Notice that GFP signal in Hand>MblRNAi;GCaMP context is lower than in the control (Hand>GCaMP) most probably because of a lower Hand-Gal4 induction in two UAS transgene context.
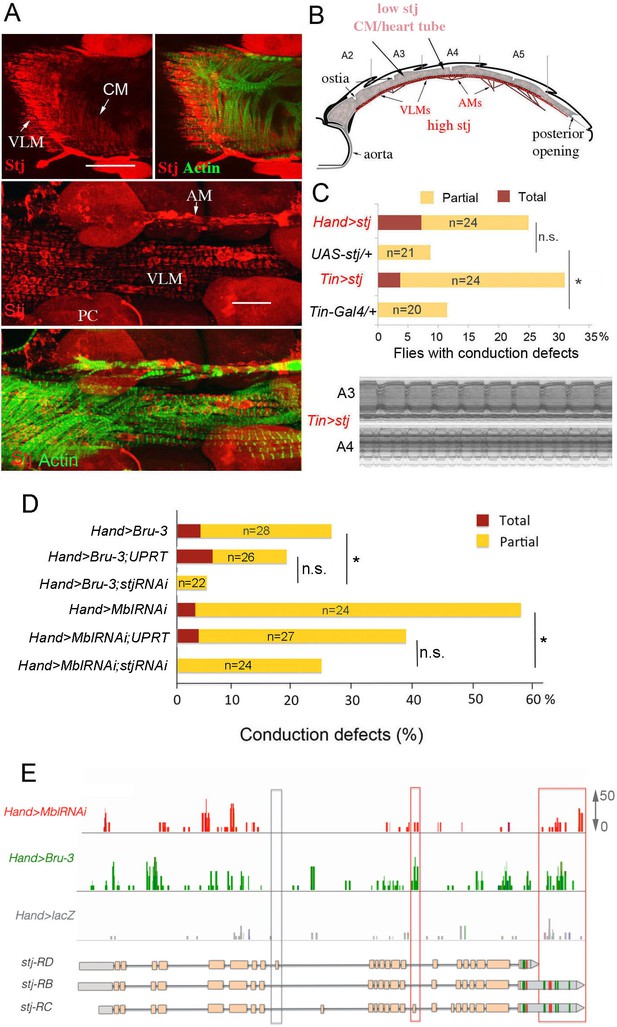
Increased cardiac expression of Stj contributes to the conduction defects observed in DM1 fly models.
(A) Stj protein is detected at a low level in cardiomyocytes (CM) but at a higher level in the ventral longitudinal muscles (VLMs) that underlie adult heart tube. Stj in VLMs and in heart tube-attaching alary muscles (AMs) marks the network of T-tubules. PC denotes pericardial cells. Scale bars, 50 μm. (B) Scheme of the adult Drosophila heart with representation of Stj expression. (C) Both Hand-Gal4 (whole heart) and Tin-Gal4 (cardiomyocytes only) driven overexpression of Stj lead to asynchronous heartbeats similar to those observed in Hand>MblRNAi and Hand>Bru-3 contexts. (D) Barplot representing percentage of Hand>MblRNAi and Hand>Bru-3 flies displaying asynchronous heartbeats after reducing cardiac Stj expression via RNAi (in stj rescue conditions). Notice that lowering Stj expression efficiently reduces the risk of asynchronous heartbeats in Hand>Bru-3 context. Number of fly hearts tested (n) is indicated and statistical significance (Fisher’s exact test) denoted by ns (p>0.05) and * (p<0.05). (E) IGV tracks showing RNAseq peaks over the stj locus in healthy control and pathological contexts. Red boxes highlight stj-RC – specific exon 15 tracks enriched in Hand>MblRNAi and Hand>Bru-3 contexts and 3’UTR-specific tracks.
-
Figure 5—source data 1
Corrected total cell fluorescence (CTCF) of Stj signal in circular fibers of cardiomyocytes.
- https://cdn.elifesciences.org/articles/51114/elife-51114-fig5-data1-v1.xlsx
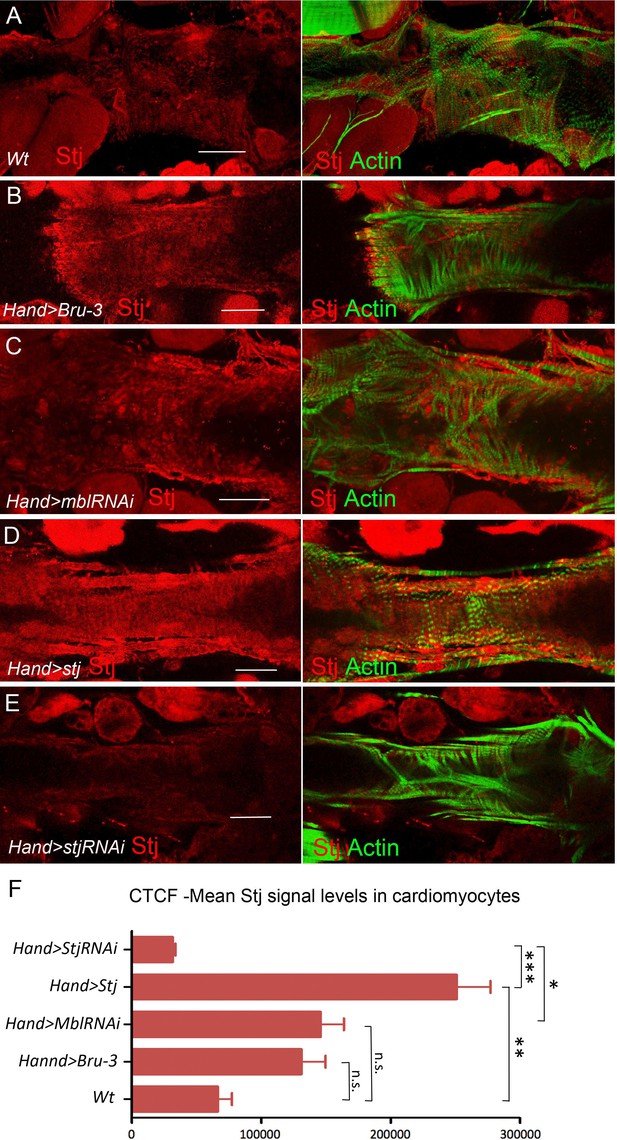
Stj protein levels in different genetic contexts visualized by immunostaining.
Stj protein expression (red) in circular fibers of cardiomyocytes in wild-type (wt) (A) and in different genetic contexts (B-E) tested. Circular fibers are visualized with phalloidin staining (green). Notice that Stj protein levels appear higher in Hand>Bru-3 and in Hand>MblRNAi compared to wt. High signal level in Hand>stj and loss of signal in Hand>stjRNAi demonstrate also the specificity of stj antibody. Scale bars, 50 μm. (F) Corrected total cell fluorescence (CTCF) of Stj signal in circular fibers of cardiomyocytes (Figure 5—source data 1) in different genetic contexts measured using Image J according to Burgess et al. (2010). Statistical significance (Kruskal-Wallis test) denoted by ns (p>0.05) * (p<0.05), ** (0.01) and *** (p<0.001).
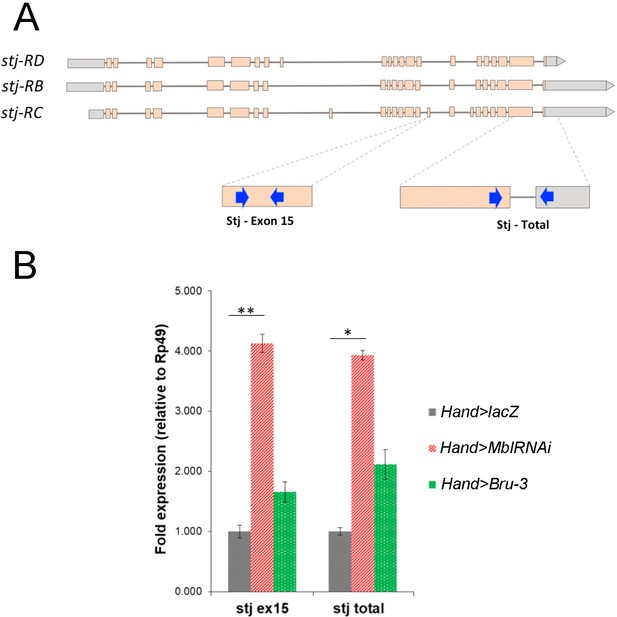
stj-RC transcript isoform carrying long 3’UTR and alternatively spliced exon 15 is up regulated in Hand>MblRNAi and in Hand>Bru-3 hearts.
(A) RT-qPCR probes for stj-RC specific exon 15 and for total stj transcripts are indicated on the scheme. (B) RT-qPCR analysis of stj-RC and total stj transcript levels in control (Hand>lacZ) fly hearts and in two DM1 contexts (Hand>MblRNAi and Hand>Bru-3). One-way ANOVA, Kruskall-wallis Dunn’s multiple comparison post-test was applied for assessing statistical significance (* - p<0.05; ** - p<0.001).
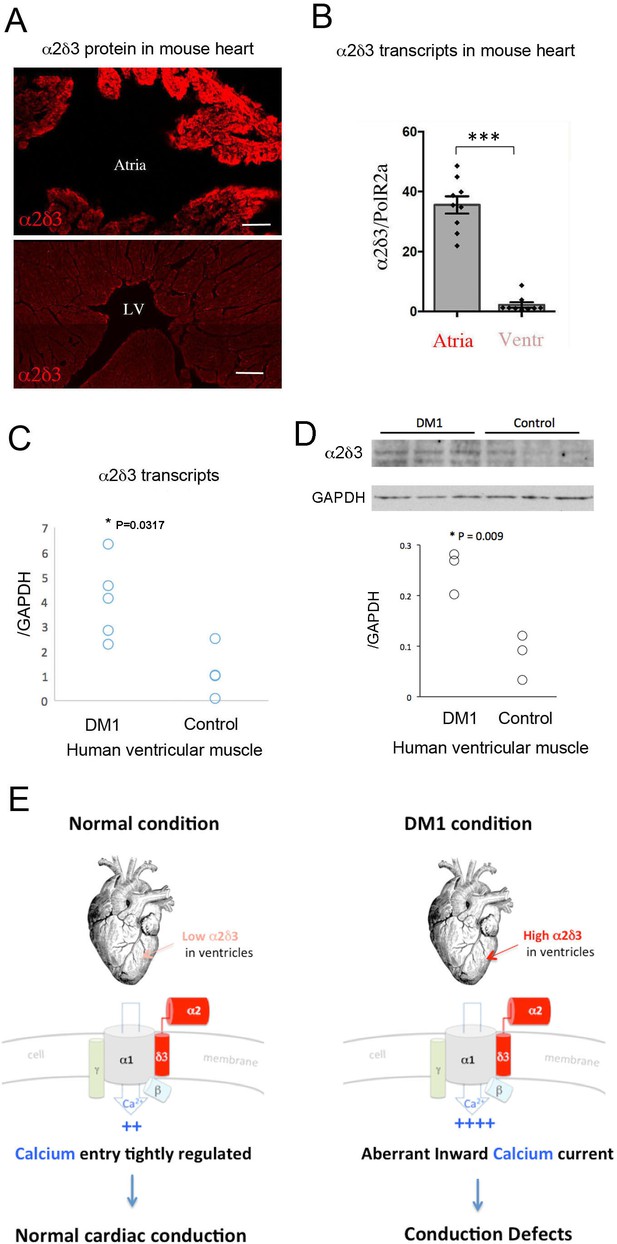
Increased cardiac expression of Straightjacket human ortholog α2δ3 is associated with conduction defects in DM1 patients.
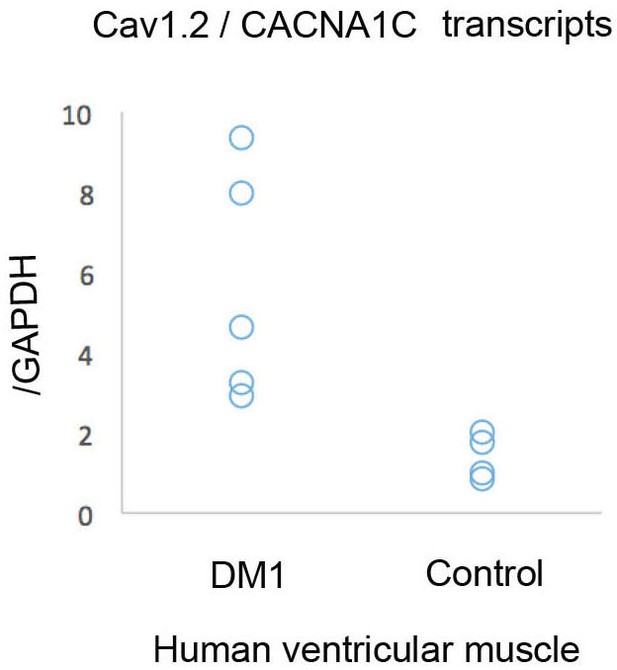
(A) In mouse, α2δ3 protein is detected at a high level in atrial cardiac cells and at a low level in ventricles. LV denotes left ventricle. Scale bars, 100 mm. (B) RT-qPCR analysis of mouse α2δ3 transcripts confirms low expression level in ventricles and high expression level in atria. Statistical significance was determined by Mann-Whitney U test (*** p<0.001) (C, D) α2δ3 (C) transcript and (D) protein levels analysed by RT-qPCR (C) and western blot (D) in human ventricular muscle from normal controls and DM1 patients with conduction defects. Note the statistically relevant increase in both α2δ3 transcript and α2δ3 protein levels in ventricles of DM1 patients (Mann-Whitney U test). (E) A scheme illustrating normal and DM1 conditions with low and increased α2δ3 levels in ventricular muscle, respectively. In pathological context, the aberrant inward calcium current in ventricular cardiomyocytes could lead to conduction defects and in particular to the IVCD.
Tables
| 1 | Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|---|
| 2 | Gene (D. melanogaster) | stj | FLYB: FBgn0261041 | ||
| 3 | Gene (D. melanogaster) | mbl | FLYB: FBgn0265487 | ||
| 4 | Gene (D. melanogaster) | bru3 | FLYB: FBgn0264001 | ||
| 5 | Genetic reagent (D. melanogaster) | UAS-stj | FlyORF Zurich ORFeome Project | FlyORF ID: F001252 | |
| 6 | Genetic reagent (D. melanogaster) | stj TRIP | Bloomington Drosophila Stock Center | RRID:BDSC25807 | RNAi line |
| 7 | Genetic reagent (D. melanogaster) | Mbl RNAi | VDRC Vienna Drosophila Resource Center | VDRC: GD 28732 | RNAi line |
| 8 | Genetic reagent (D. melanogaster) | P{XP}bru3d09837 | Exelixis at Harvard Medical School | Harvard: d09837 | UAS carrying P element insertion line |
| 9 | Genetic reagent (D. melanogaster) | UAS-GFP | Bloomington Drosophila Stock Center | RRID:BDSC_32201 | |
| 10 | Genetic reagent (D. melanogaster) | UAS-HA-UPRT | Bloomington Drosophila Stock Center | RRID:BDSC_27604 | |
| 11 | Genetic reagent (D. melanogaster) | UAS-lacZ | Bloomington Drosophila Stock Center | RRID:BDSC_1776 | |
| 12 | Genetic reagent (D. melanogaster) | UAS-GCaMP3 | Bloomington Drosophila Stock Center | RRID:BDSC_32116 | |
| 13 | Genetic reagent (D. melanogaster) | Ca-alpha1D TRIP | Bloomington Drosophila Stock Center | RRID:BDSC_25830 | RNAi line |
| 14 | Genetic reagent (D. melanogaster) | Rgk2 TRIP | Bloomington Drosophila Stock Center | RRID:BDSC_65932 | RNAi line |
| 15 | Genetic reagent (D. melanogaster) | Rgk2 RNAi | VDRC Vienna Drosophila Resource Center | VDRC: GD44611 | RNAi line |
| 16 | Genetic reagent (D. melanogaster) | Syn1 RNAi | VDRC Vienna Drosophila Resource Center | VDRC: GD27893 | RNAi line |
| 17 | Genetic reagent (D. melanogaster) | Syn1 RNAi | Bloomington Drosophila Stock Center | RRID: BDSC_27504 | RNAi line |
| 18 | Genetic reagent (D. melanogaster) | inaD RNAi | VDRC Vienna Drosophila Resource Center | VDRC: GD26211 (unavailable) | RNAi line |
| 19 | Genetic reagent (D. melanogaster) | inaD RNAi | VDRC Vienna Drosophila Resource Center | VDRC: 330227 | RNAi line |
| 20 | Genetic reagent (D. melanogaster) | Hand-GAL4 | Laurent Perrin, TAGC, Marseille, France | GAL4 driver line | |
| 21 | Genetic reagent (D. melanogaster) | TinCΔ4-GAL4 | Rolf Bodmer, SBPMD Institute, San Diego, US | GAL4 driver line | |
| 22 | Antibody | anti-Stj | Hugo Bellen, Baylor College of Medicine, Huston, USA | guinea pig polyclonal | Use 1:500 (IHC) |
| 23 | Antibody | anti-α2δ3 | Greg Neely, University of Sidney, Australia | rabbit polyclonal | Use 1:200 (IHC) |
| 24 | Antibody | anti-α2δ3 | GeneTex | GTX16618 - rabbit polyclonal | Use 1:500 (WB) |
| 25 | Antibody | anti-Bru3 | Millegen, Toulouse, France | rabbit polyclonal | Use 1:1000 (IHC) |
| 26 | Antibody | anti-Mbl | Darren Monckton, Glasgow, UK | sheep polyclonal | Use 1:200 (IHC) |
| 27 | Antibody | anti-GFP | Abcam | ab5450 – goat polyclonal | Use 1:500 (IHC) |
| 28 | Antibody | Anti-GAPDH | Sigma Aldrich | SAB2108266 – rabbit polyclonal | Use 1:3000 (WB) |