A multilayer circuit architecture for the generation of distinct locomotor behaviors in Drosophila
Figures
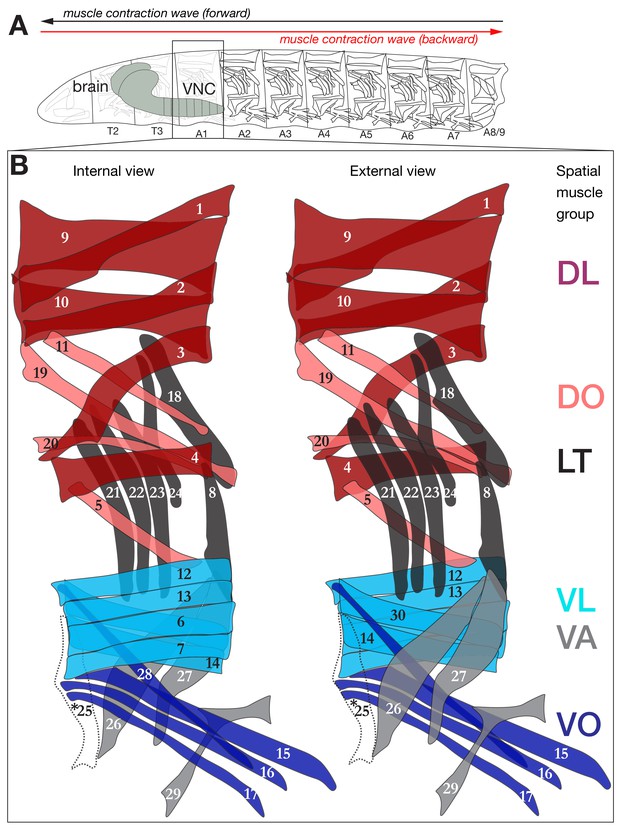
Schematic depiction of the larval neuromuscular system.
(A) Drosophila larva contain three thoracic and nine abdominal segments, the muscles of which are innervated by MNs located in the corresponding thoracic and abdominal segments of the CNS. (B) Schematic of the 29 muscles of abdominal segments (A1) from internal and external view. Segments A2-A6 are similar to A1, with the exception that they have a muscle 25 (dashed line, asterisk) here overlaid on the A1 muscle pattern.
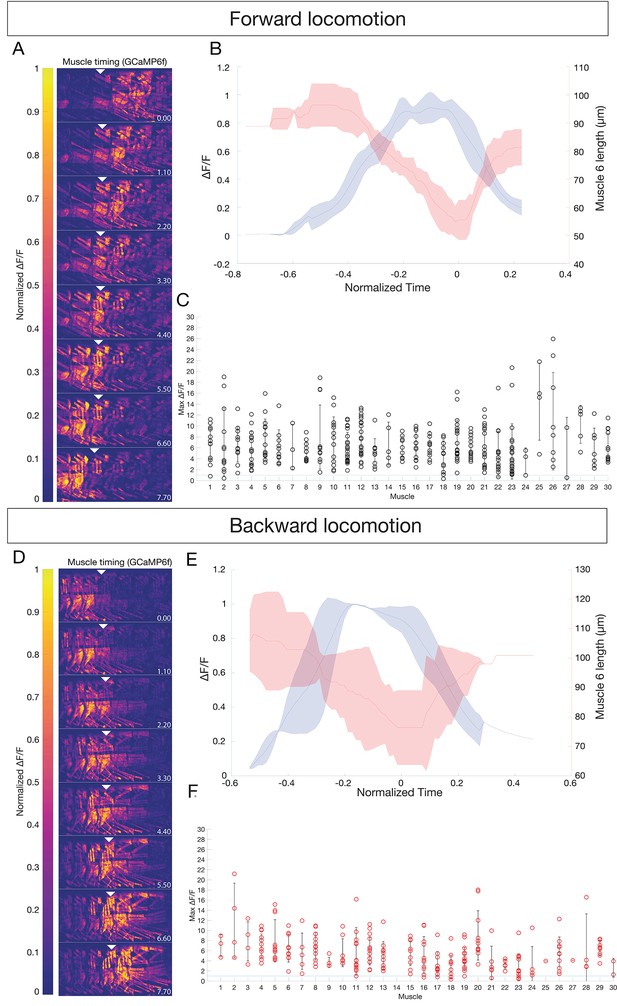
All body wall muscles are utilized during forward and backward locomotion.
(A,D) Sequential images of muscle GCaMP6f ΔF/F signal during forward (A) or backward (D) locomotion. GCaMP6f levels were normalized to mCherry. Anterior to left, dorsal up; time in seconds. Genotype: GMR44H10-LexA lexAOP-GCaMP6f; -LexA lexAOP--mCherry. Arrowheads mark the same segment at each timepoint; A2 in (A) and A4 in (D). (B,E) Mean calcium transient (blue) vs mean muscle length (red) measurements for muscle six during forward (B) or backward (E) locomotion. N = 3 segments. T0 was set as the point of maximum contraction as determined by muscle length for each crawl. Shaded bars represent standard deviation. (C,F) All observed muscles show calcium transients greater than 100% ΔF/F during forward (C) or backward (F) locomotion. Each dot represents the maximum GCaMP ΔF/F signal in the indicated muscle during a single crawl, normalized to mCherry. Error bars represent standard deviation. Muscle names as in Figure 1.
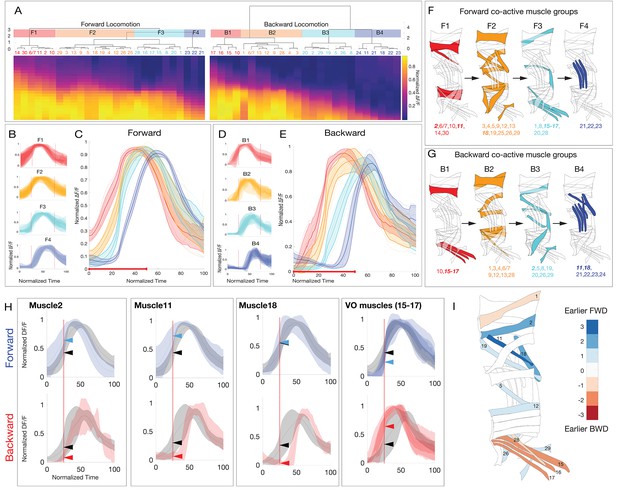
Larval body wall muscles form four co-activated muscle groups during forward and backward locomotion.
(A) Hierarchical clustering of mean activity for all observed muscles yields four co-activated muscle groups during forward locomotion (F1–F4) and a different group of four during backward locomotion (B1–B4). Heatmaps represent the mean range-normalized calcium activity of each muscle (n > 3 crawl bouts for each muscle, with a total of 337 individual muscles analyzed across 23 crawls for forward and 188 individual muscles analyzed across 14 crawls for backward locomotion). Muscles 6/7 are grouped because they are both innervated by the same MN. Clustering was performed only on the first half of the crawl cycle to determine the onset time for each co-activated muscle group. Cluster number was determined by visual inspection of the dendrogram as well as the gap-criterion optimal cluster number. (B,D) Plots of average muscle activity for muscles in each forward or backward co-activated muscle group. Error bars represent the standard deviation of individual muscles. (C,E) Plots of average forward or backward co-activated muscle group activity timing. Error bars represent the standard deviation of the average muscle activity of each muscle in a given co-activated muscle group. Dotted lines represent the average muscle activity for each muscle in a given co-activated muscle group. Red line along the x-axis represents the fraction of the crawl cycle that was used for clustering. (F,G) Schematic representation of the co-activated muscle group for forward or backward locomotion. (H) Plots of muscles that are differentially active during forward or backward crawling. For forward panels, the gray trace represents the mean calcium activity of all muscles during a forward crawl, while the blue trace represents the activity of the indicated muscle. For backward panels, the gray trace represents the mean calcium activity of all muscles during a backwards crawl, while the red trace represents the mean calcium activity of the indicated muscle. Error bars represent standard deviation. Dotted red line marks t = 25 (normalized time). Arrows represent the normalized ∆F/F of the two traces at t = 25. (I) Heatmap illustrating differential activity of muscles during forward versus backward crawling. Blue, or positive values indicate a given muscle is active earlier during forward crawling, while red or negative values indicate a given muscle was active earlier during backward crawling.
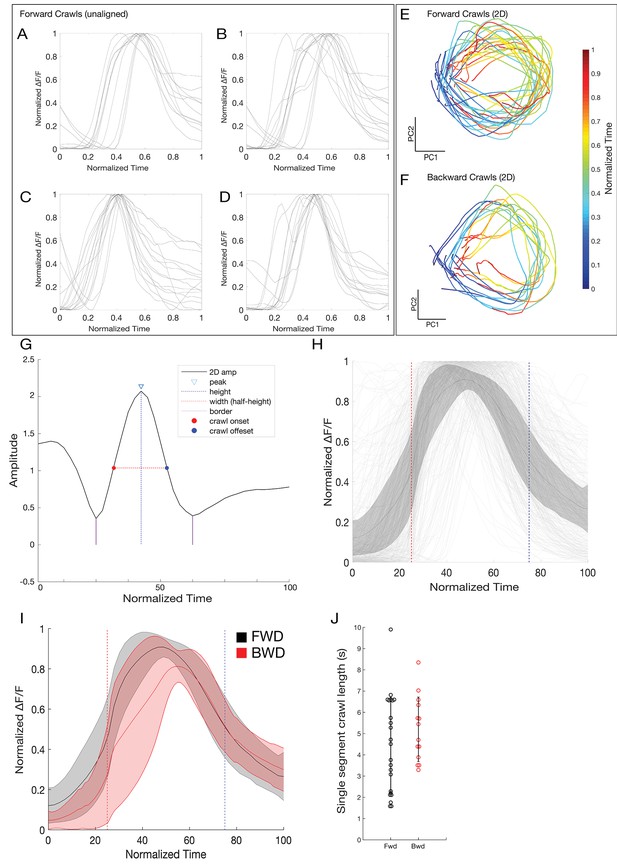
PCA-based alignment of crawl cycles.
(A–D) Plots of four representative forward crawls show a high degree of variability in the crawl structure. (E–F) 2D projection of forward and backward crawl cycles. Crawl cycles are represented as rotations away from and back towards the origin. Color changes (from blue to red) represent time. Directionality is not uniform given that the group of analyzed muscles in each crawl cycle is different in each case (all crawls used had at least 40% of the muscles analyzed in the segment). (G) Amplitude of a representative forward crawl in the same 2D space. The peak of this activity was defined as the center of a crawl cycle, and the peak width at half the height of the peak was used to find crawl-start and crawl-end times. (H) All analyzed forward muscles aligned. Gray lines represent individual muscles, black line represents the average activity of all muscles with error bars representing standard deviation. Red dotted line represents the crawl-start alignment point, and the blue line represents the crawl-end alignment point. (I) Average activity of forward (black) and backward (red) crawls across all experiments. (J) Single segment crawl length determined for each crawl (n = 24 forward/14 backward). Crawl length is determined by calculating the width of the 2D representation of the crawl cycle.
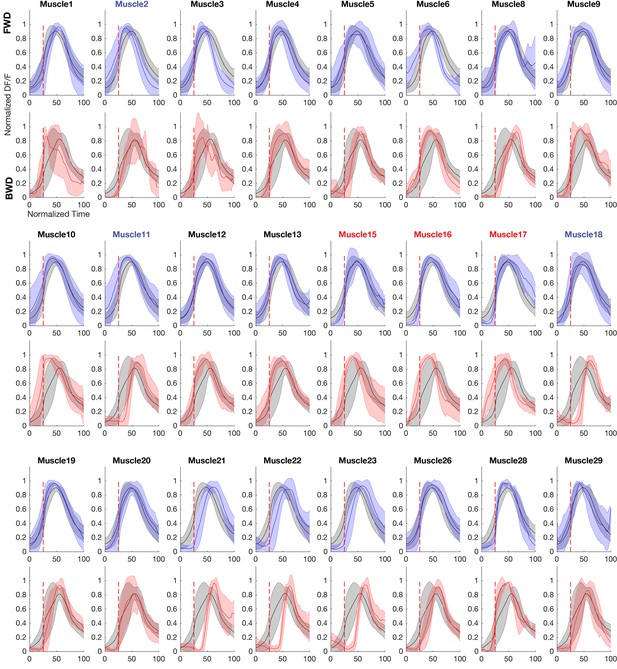
Muscles recruited at similar and different phases of the forward and backward crawl cycle.
Plots of muscles that relative to the average activity of all muscles during forward or backward crawling. For forward panels, the gray trace represents the mean calcium activity of all muscles during a forward crawl, while the blue trace represents the activity of the indicated muscle. For backward panels, the gray trace represents the mean calcium activity of all muscles during a backwards crawl, while the red trace represents the mean calcium activity of the indicated muscle. Error bars represent standard deviation. Dotted red line marks t = 25 (normalized time). Muscles in red text are differentially recruited in forward and backward locomotion.
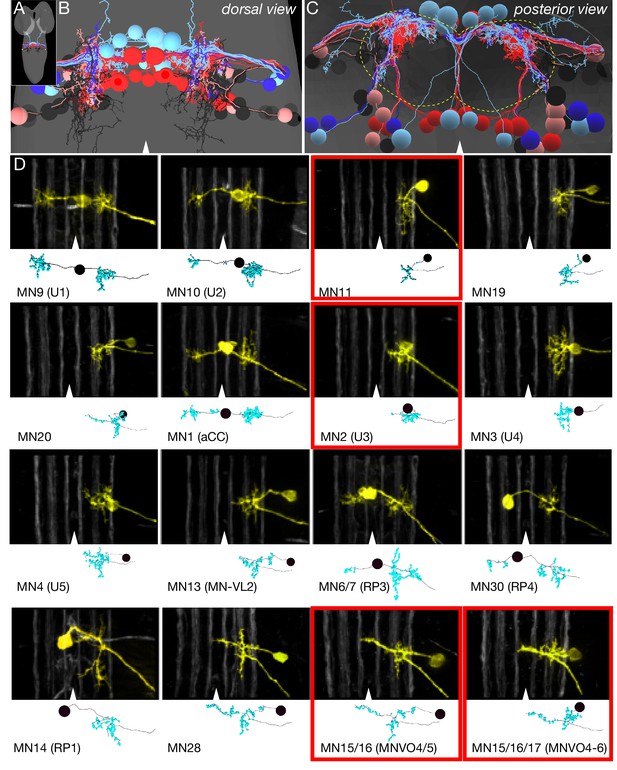
Identification of all motor neurons in segment A1 in the TEM volume.
(A) Dorsal view of the TEM reconstruction of the L1 CNS (gray shading) showing all bilateral MNs in A1 reconstructed at single synapse level. The one intersegmental dendrite is from RP3 in A1; it is not observed in other abdominal segments. (B) Dorsal view of centered on the A1 segment; midline, arrowhead. MNs are color-coded as in Figure 1A: DL MNs (red), DO MNs (light red), VL MNs (light blue), VO MNs (dark blue), LT MNs (black), VA MNs (gray). (C) Posterior (cross-section) view of the neuropil (outlined) and cortex in A1. Note the MN dendrites target the dorsal neuropil. Dorsal, up; midline, arrowhead; neuropil border, dashed outline. (D) Representative images showing the morphological similarity between MNs identified in vivo by backfills (Mauss et al., 2009) versus the most similar MN reconstruction from the TEM volume. The top section in each panel shows the morphology of the MN dendrites based on in vivo backfills; used with permission); six distinct Fas2 fascicles (three per hemisegment) are shown in white; midline, arrowhead. The bottom section shows MN dendrite morphology reconstructed from the TEM volume in A1. MNs highlighted in red boxes show differential recruitment timing during forward versus backward locomotion.

Reconstruction and identification of A1 MNs in the TEM volume.
MN names are shown in bottom left for each reconstruction. The morphology of each MN is shown in cross-sectional (left) and dorsal view (right). In the cross-sectional views, neuropil boundary is shown in gray. In the dorsal views, L, M, R stand for Left neuropil border, Midline, Right neuropil border, respectively. Cyan dots show MN postsynaptic sites.
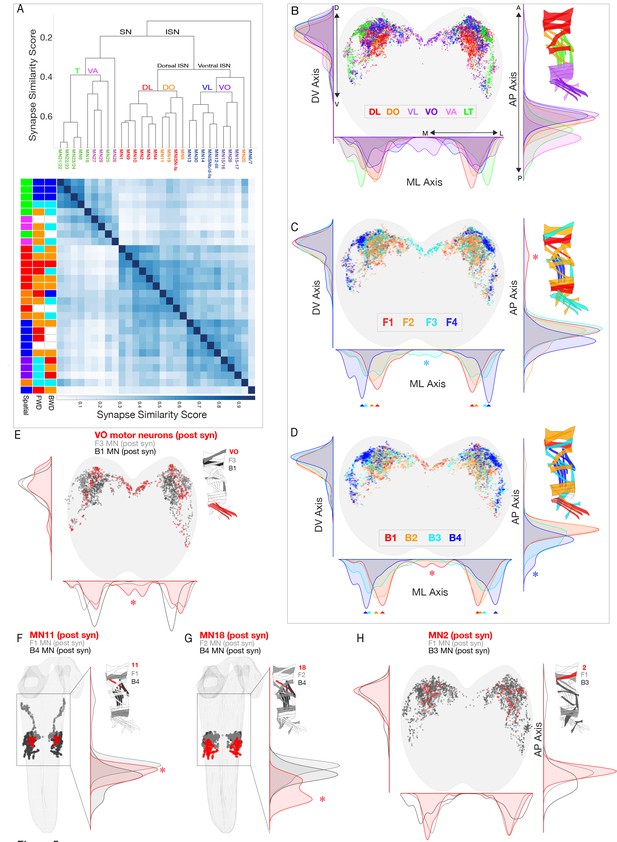
Motor neurons innervating spatial muscle groups or co-activated muscle groups have post-synapses in distinct regions of neuropil.
(A) Hierarchical clustering of MNs by their synapse similarity score reveals MN myotopic organization. To generate a similarity matrix, pairwise synapse similarity scores were generated separately for MNs exiting the left A1 nerve and right A1 nerve. The pairwise similarities for the left and right pools of MNs were highly correlated (r = .95); clustering was performed on the average of the left and right similarity matrices. (B–D) Spatial distributions of postsynaptic sites for the indicated spatial or co-active MNs. Plots are 1D kernel density estimates for the mediolateral (ML), dorsoventral (DV) and anteroposterior (AP) axes. Asterisks, postsynapses from a single group that are enriched in a specific region of the neuropil. (E–H) Spatial distribution of postsynapses for the differentially recruited MNs (red) compared to the MNs in their forward or backward co-active group (gray or black, respectively).
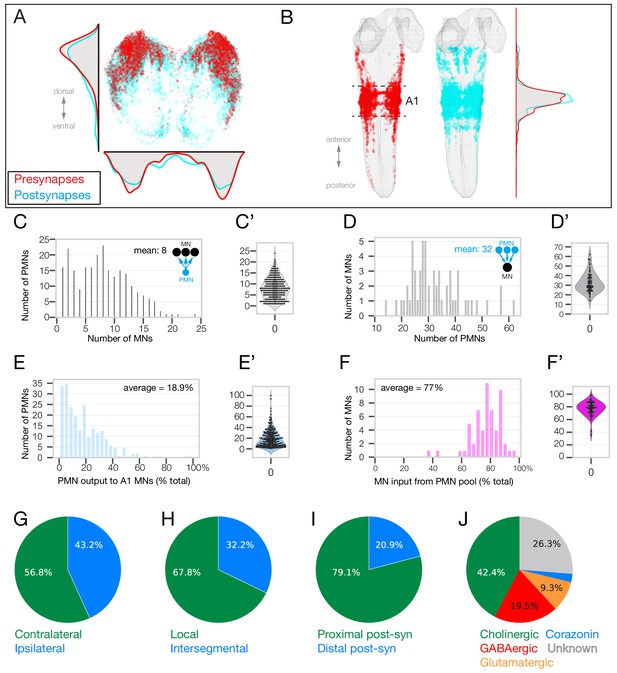
Identification of 118 premotor neurons at synapse-level in the TEM volume.
(A) Posterior (cross-section) view of the PMN pre-synapse location (red) and postsynapse location (cyan) within the A1 neuropil. Density plots shown for the dorsoventral axis (left) and mediolateral axis (bottom). Dorsal, up. (B) Dorsal view of entire larval neuropil to show anteroposterior distribution of presynapses (red) and postsynapses (cyan). Density plots shown for the anteroposterior axis. (C–F) Quantification of PMN-MN connectivity. All A1 MNs, A2 MN-25, and 118 pair of PMNs were used to generate these histograms. (C) PMNs innervate an average of 8 MNs. X-axis shows binned number of MNs receiving inputs from PMNs. Y-axis shows number of PMNs in each bin (C’) Swarm-violin plot representation of the same dataset used in C. (D) MNs receive inputs from an average of 32.5 PMNs from this population of PMNs. X-axis shows binned number of PMNs providing output to MNs. Y-axis shows number of MNs in each bin. (D’) Swarm-violin plot representation of the same dataset used in D. (E) Histogram showing binned fraction of PMN output to MNs. Y-axis shows number of PMNs in each bin. (E’) Swarm-violin plot representation of the same dataset used in E. (F) Histogram showing binned fraction of MN inputs from PMNs. Y-axis shows number of MNs in each bin. (F’) Swarm-violin plot representation of the same dataset used in F. (G–J) Quantification of PMN morphology and neurotransmitter expression. We did not assay Corazonin+ neurons for fast neurotransmitter expression, but a recent RNAseq study shows promiscuous expression of fast neurotransmitters in Corazonin+ neurons (Brunet Avalos et al., 2019).
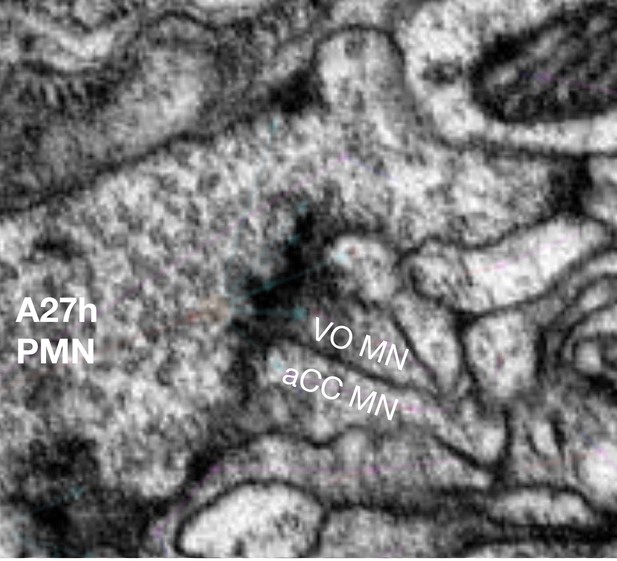
Pre-motor neuron/motor neuron synapse identification in the TEM volume.
The presynaptic neuron A27h is shown along with two of its postsynaptic partners (MN1 and MN15/16, also called aCC and VO, respectively). Note the accumulation of vesicles at the A27h presynapse and the electron density in the motor neuron postsynapses.

All premotor neurons traced in the TEM volume.
Dorsal view (top) and posterior view (bottom) of all 118 premotor neurons. In all panels, neuron names are at the top; red and cyan dots indicate pre-synaptic and postsynaptic sites respectively. Neurons with pre- and postsynaptic sites intermixed are boxed.
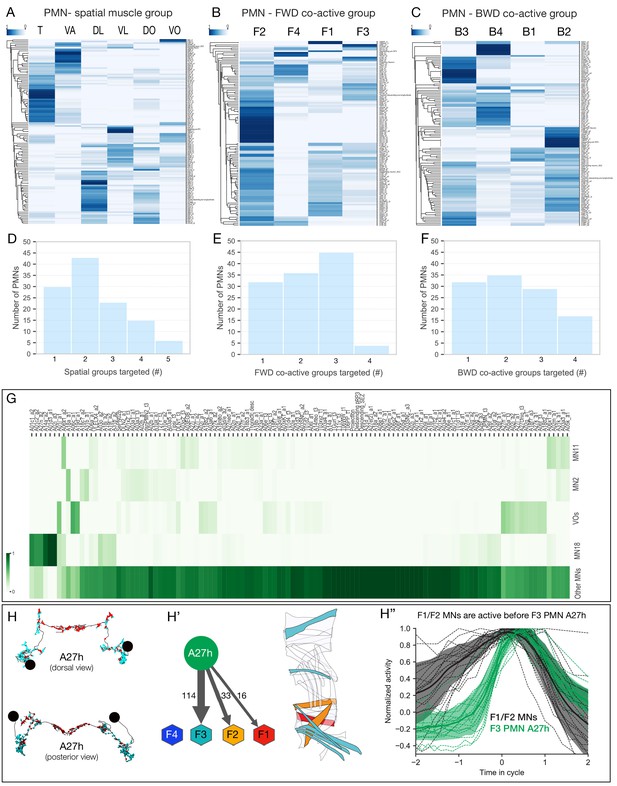
PMN pools preferentially connected to individual spatial muscle groups and co-activated muscle groups.
(A–C) Hierarchical clustering of PMNs based on their connectivity to type Ib MNs of the same spatial muscle group (A), forward co-activated muscle group (B), or backward co-activated muscle group (C). Heat maps represent the sum of normalized weighted-synaptic output of a given left/right pair of PMNs onto left/right pair of MNs grouped in each panel. Values in each row were normalized to sum to 1. (D–F) Quantification of connectivity between PMNs and type Ib MNs innervating spatial muscles (D), forward co-active (E), and backward co-active groups. PMN-MN connections with total weighted synapses of less than 1% were excluded from these analyses. (D) X-axis shows binned number of spatial muscle groups which receive inputs from PMNs. Y-axis shows number of PMNs in each bin. While 30 PMNs connect to only one spatial muscle group, the rest of the PMNs connect to more than one groups. (E) X-axis shows binned number of forward co-active groups which receive inputs from PMNs. Y-axis shows number of PMNs in each bin. While 32 PMNs connect to only one forward co-active group, the rest of the PMNs connect to more than one groups. (F) X-axis shows binned number of backward co-active groups which receive inputs from PMNs. Y-axis shows number of PMNs in each bin. While 32 PMNs connect to only one backward co-active group, the rest of the PMNs connect to more than one groups. (G) Connectivity pattern of PMNs to differentially recruited MNs (11, 2, 18, and VOs) versus other type Ib MNs (Other MNs). Heat maps represent the sum of normalized weighted-synaptic output of a given left/right pair of PMNs onto left/right pair of MNs in each group. Values in each column were normalized to sum to 1. (H–H’’) A27h is active following MNs in forward co-active groups F1/F2. (H) Morphology of the reconstructed A27h in segment A1 left and right, showing presynapses (red) and postsynapses (cyan). (H’) Pattern of A27h connectivity showing preferential output to MNs active in co-active group F3. Hexagons represent MNs preferentially active in the F1-F4 co-active groups. Muscles innervated by MNs targeted by A27h are shown. (H’’) Dual color calcium imaging of jRCaMP1b in A27h (green) and GCaMP6m in U1-U5 MNs (black: MN2, MN3, MN4, MN9, MN10). Consistent with predictions from the connectome, U1-U5 MNs (co-activated muscle group F1/2) are activated before A27h (co-activated muscle group F3). Green and dark error bars (ribbons) represent the standard deviation of the average neuronal activity. Genotype: CQ-lexA/+; lexAop-GCaMP6m/R36G02-Gal4 UAS-jRCaMP1b.
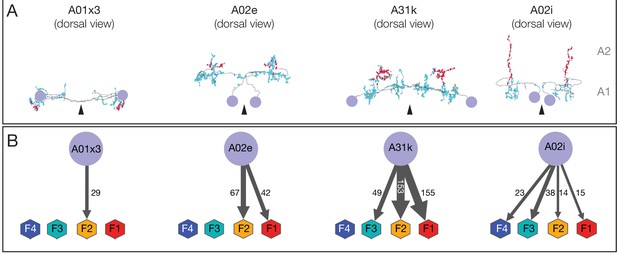
Morphology and connectivity of premotor neurons innervating one or more co-active motor neurons (F1–F4).
(A) Dorsal view of the indicated premotor neurons. In all panels, neuron names are at the top; red and cyan dots indicate pre-synaptic and postsynaptic sites respectively. (B) Number of presynapses on motor neurons in each co-active group are shown next to the black arrows, whose width is proportional to synapse number. Arrowhead, midline.
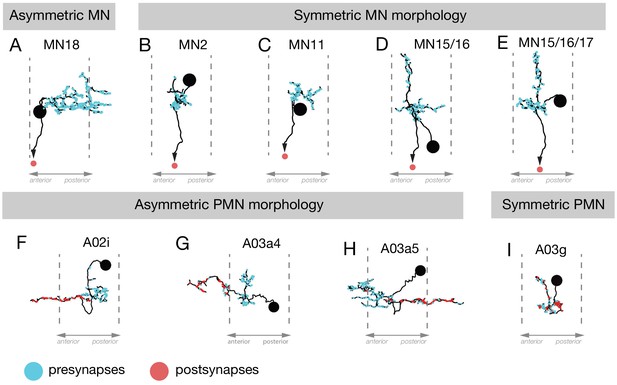
Neuronal asymmetry along the anterior-posterior axis.
(A–E) MN18 has asymmetric dendrites extending to the next posterior segment, but the dendritic arbors of other differentially recruited MNs (2, 11, 15/16, 15/16/17) were not asymmetric along this axis. (F–I) The PMNs A02i, A03a4 and A03a5 have asymmetric dendrite projections to the anterior (F,G) or posterior (H) of their cell body and presynaptic domain, whereas A03g is an example of a PMN that is symmetric along the anteroposterior axis.
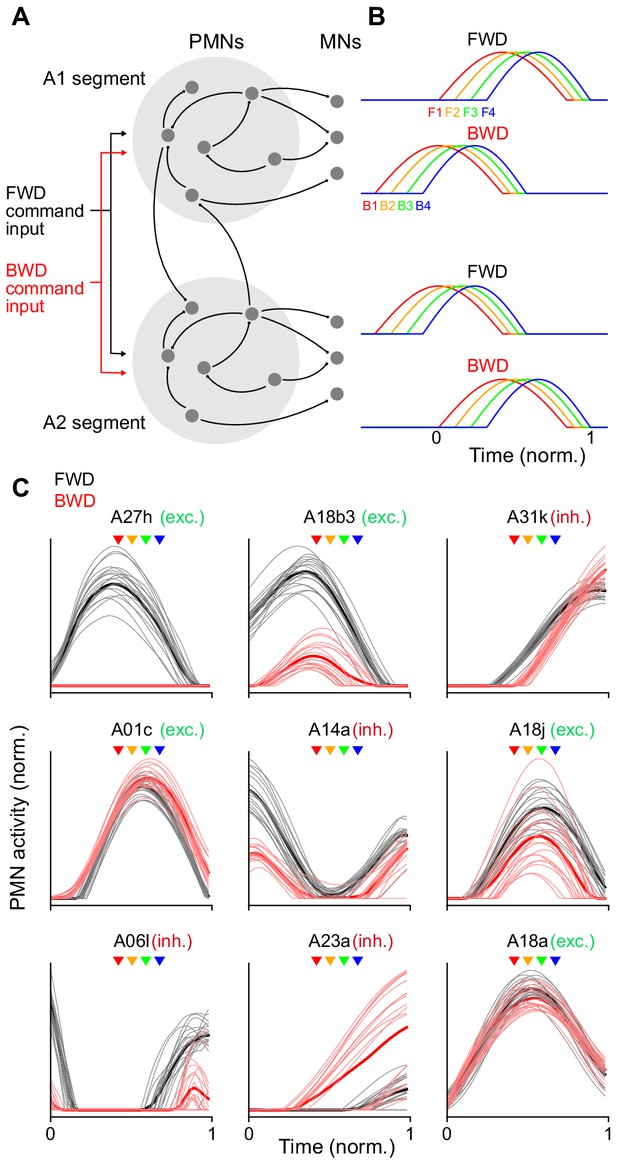
Recurrent network model generating sequential MN activity.
(A) The PMN and MN network of the A1 and A2 segments was modeled using connectivity taken from the TEM reconstruction. Connections within each segment (light gray circles) are identical. The network was optimized using gradient descent to produce a sequential pattern of activity in the MNs when a tonic external command input for forward (FWD, black) or backward (BWD, red) locomotion was applied. (B) The network in A was optimized to produce an appropriate sequential activity pattern of co-activated muscle groups during forward and backward crawling. The direction of propagation from the posterior (A2) to anterior (A1) segment or vice versa differs for forward and backward crawling. To compare PMN activity relative to MN activation, time is measured in units normalized to the onset and offset of MN activity in a segment (bottom right). (C) Y-Axis is the normalized activity of a subset of PMNs in the model during forward and backward crawling. Thick lines denote averages over the ensemble of models generated. X-axis (time) is measured relative to A1 MN onset and offset as in B. Arrowheads denote the peak activation onset time for the MNs innervating different co-activated muscle groups (color key as in panel B); exc, excitatory; inh, inhibitory.

Recurrent network model of PMNs activity aligned to onset and offset of A1 MNs during locomotion.
Y-Axis is the normalized activity of PMNs in the model during forward (black traces) and backward (red traces) crawling. Thick lines denote averages over the ensemble of models generated. X-axis (time) is measured relative to A1 MN onset and offset as in Figure 10B. e, excitatory;(e), presumed excitatory based on lineage; i, inhibitory; (i), presumed inhibitory based on lineage.
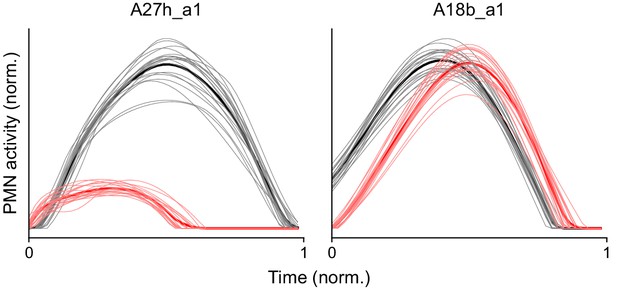
Models constructed without constraints on the activity of A27h/A18b.
Similar to Figure 9C, but without the penalty term (see Materials and methods) that constrains the activity of the A27h/A18b PMNs. While A27h retains its selectivity, A18b does not.
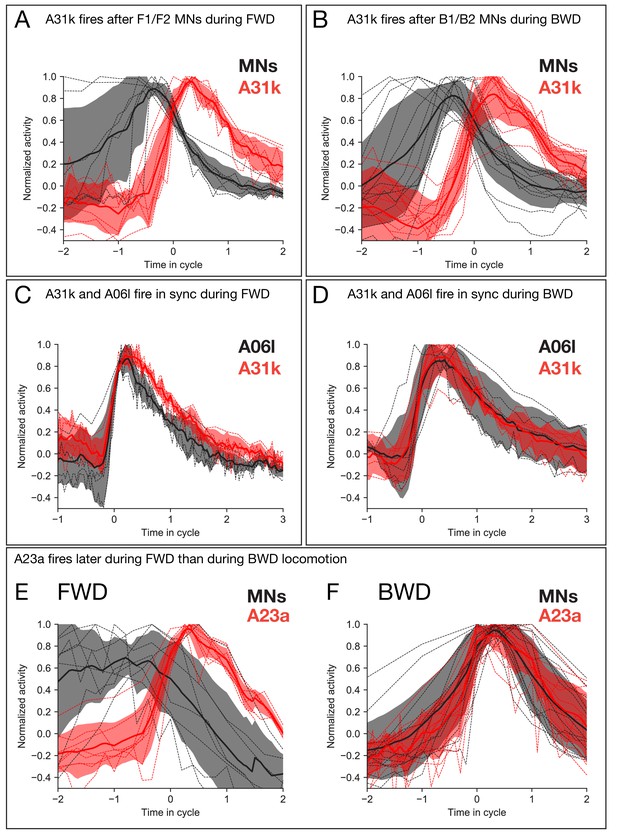
Calcium imaging of A31k/A06l/A23a PMNs and their target MNs validates the activity pattern predicted by recurrent modeling.
(A–B) Dual color calcium imaging of jRCaMP1b in A31k (red) and GCaMP6m in MNs (black). Consistent with the recurrent model predictions, A31k fires with a delay after its postsynaptic MNs in both forward (A) and backward (B) waves. Red and dark error bars (ribbons) represent the standard deviation of the average neuronal activity. Genotype: CQ-lexA/+; lexAop-GCaMP6m/R87H09-Gal4 UAS-jRCaMP1b. (C–D) Single color calcium imaging of jRCaMP1b in A31k (red) and A06l (black). Consistent with the recurrent model predictions, A31k and A06l show synchronous activity patterns during forward (C) and backward waves (D). Red and dark error bars (ribbons) represent the standard deviation of the average neuronal activity. Genotype: R87H09-Gal4 UAS-jRCaMP1b. (E,F) A23a fires later during forward locomotion than during backward locomotion. Dual color calcium imaging of jRCaMP1b in A23a (red) and GCaMP6m in MNs (black). Red and dark error bars (ribbons) represent the standard deviation of the average neuronal activity. Genotype: CQ-lexA/+; lexAop-GCaMP6m/R78F07-Gal4 UAS-jRCaMP1b.
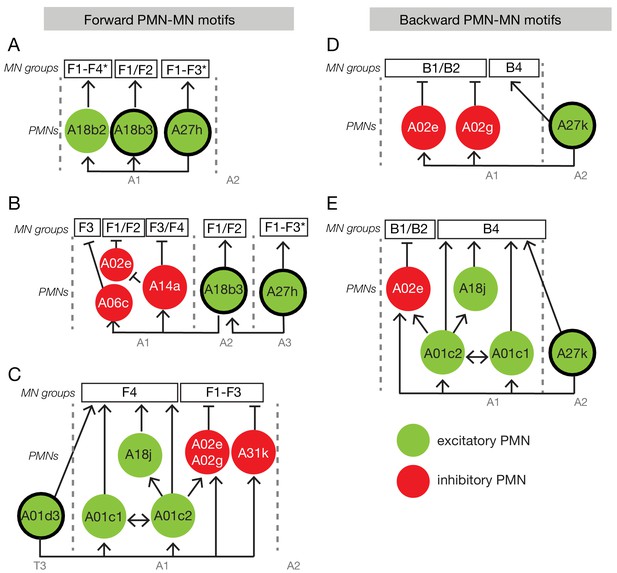
Neural circuit motifs specific for forward or backward locomotion.
Circuit motifs composed of forward-specific PMNs (A–C) and backward specific PMNs (D–E). See text for details. Arrow/green, excitatory connection; T-bar/red, inhibitory connection; F1-F4, forward co-active group; B1-B4, backward co-active group. Thick outlines indicate forward-specific or backward-specific PMNs.
Videos
GCaM6f muscle activation during forward locomotion in a Drosophila second instar larva.
Dorsolateral view; anterior left.
GCaM6f muscle activation during backward locomotion in a Drosophila second instar larva.
Dorsolateral view; anterior left.
Tables
Motor neurons present in the CATMAID reconstruction.
All MNs were identified in the first abdominal segment on both left and right sides, with the exception of MN25 which is not present in A1 and thus annotated in A2. See text for abbreviations.
| Spatial muscle group | Nerve | Motor neurons (synonyms) | Target muscles (synonyms) | Synapse Type |
|---|---|---|---|---|
| DL | ISNDM | MN1 (aCC) | 1 (DA1) | Ib |
| DL | ISNDM | MN2 (U3) | 2 (DA2) | Ib |
| DL | ISNDM | MN3 (U4) | 3 (DA3) | Ib |
| DL | ISNDM | MN4 (U5) | 4 (LL1) | Ib |
| DL | ISNDM | MN9 (U1) | 9 (DO1) | Ib |
| DL | ISNDM | MN10 (U2) | 10 (DO2) | Ib |
| DO | ISNL | MN11 | 11 (DO3) | Ib |
| DO | ISNL | MN19 | 19 (DO4) | Ib |
| DO | ISNL | MN20 | 20 (DO5) | Ib |
| DO | SNa | MN5 (LO1) | 5 (LO1) | Ib |
| VL | ISNb | MN6/7 (RP3) | 6/7 (VL3/VL4) | Ib |
| VL | ISNb | MN12 (V-MN) | 12 (VL1) | III |
| VL | ISNb | MN13 (MN-VL2) | 13 (VL2) | Ib |
| VL | ISNb | MN14 (RP1) | 14 (VO2) | Ib |
| VL | ISNb | MN30 (RP4) | 30 (VO1) | Ib |
| VA | SNc | MN26 | 26 (VA1) | Ib |
| VA | SNc | MN27 | 27 (VA2) | Ib |
| VA | SNc | MN29 | 29 (VA3) | Ib |
| VO | ISNd | MN15/16 (MN-VO4/5) | 15/16 (VO4/VO5) | Ib |
| VO | ISNd | MN15/16/17 (MN-VO4-6) | 15/16/17 (VO4/VO5/VO6) | Ib |
| VO | ISNb | MN28 | 28 (VO3) | Ib |
| T | SNa | MN8 (SBM) | 8 (SBM) | Ib |
| T | SNa | MN21/22 (LT1/LT2) | 21/22 (LT1/LT2) | Ib |
| T | SNa | MN22/23 (LT2/LT3) | 22/23 (LT2/LT3) | Ib |
| T | SNa | MN23/24 (LT3/LT4) | 23/24 (LT3/LT4) | Ib |
| T | ISNL | MN18 | 18 (DT1) | Ib |
| T | TN | MN25 (VT1) | 25 (VT1) | Ib |
| Broad | ISNDM | MNISN (RP2) | 1/2/3/4/9/10/11/[18]/19/20 (DA/DO) | Is |
| Broad | ISNb | MNISNb/d (RP5) | 6/7/12/13/14/15/16/30 (VL/VO) | Is |
| Broad | SNa | MNSNa-II (VUM) | 21/22/[23/24/25] (LT) | II |
| Broad | ISNDM | MNISN-II (VUM) | 1/2/3/4/9/10/11/18/19/20 (DA/DO) | II |
| Broad | ISNb | MNISNb/d-II (VUM) | 12/13/14/15/16/17/30 (VL/VO) | II |
Co-activated muscle groups during forward or backward locomotion.
There are four co-activated muscle groups during backward and forward locomotion, but the muscles in each group differ in forward versus backward locomotion. Note that backward locomotion is not simple a reverse of the pattern seen in forward locomotion. This represents the most common activation sequences, although there is some variation, particularly during the fastest locomotor velocities.
| Forward | Co-activated muscles |
|---|---|
| F1 | 2,6,10,11,14,30 |
| F2 | 3,4,5,9,12,13,18,19,25,26,29 |
| F3 | 1,8,15,16,17,20,28 |
| F4 | 21,22,23 |
| Backward | Co-activated muscles |
| B1 | 10,15,16,17 |
| B2 | 1,3,4,6,9,12,13,28 |
| B3 | 2,5,8,19,20,26,29 |
| B4 | 11,18,21,22,23,24 |
Premotor neurons innervating type Ib MNs Left column, spatial muscle groups named as in Figure 1.
Middle column, type Ib MNs innervating 1–3 muscles in each muscle group (synonym, parentheses); the immature neuromodulatory VUMs are not shown. Right column, premotor interneurons innervating the indicated MNs. Premotor connectivity uncertain, parentheses.
| Muscle position | Motor Neurons | Pre-Motor Neurons |
|---|---|---|
| DL | MN1-Ib (aCC) | A27h, A18a, A18b, A03g, A31k, A31b, A06e, A23a, A02h, A10e, A03a1, A03a3, A05k, A07f2, DLN2, TJPMNThoracic descending pre-longitudinals, T27Y, dsnPMN2, DLN1, A18neo. |
| DL | MN2-Ib (U3) | A01x2, A18a, A03a5, A31k, A31b, A23a, A02h, A03a3, A03a1, A10e, A10a , T27Y, dsnPMN2. |
| DL | MN3-Ib (U4) | A18a, A03a5 A03g, A31k, A31b, A06e, A02h, A02e, A02f, A03a3, A03a6, A03d/e, A03x-eghb, A07f2, A10a, A18neo. |
| DL | MN4-Ib (U5) | A03a5, A03g, A31k, A27l, A06l, A06m, A06g2, A02e, A02f, A03a6, A03a1, A03x-eghb, SePN02b, DLN2, Descending, pre RP3, A18neo. |
| DL | MN9-Ib (U1) | A01x2, A18a, A31k, A31b, A06x1, A27l, A23a, A02m, A02n, A02h, A03a1, A03a3, A03x-eghb, A03xyz, A05k, DLN2, |
| DLN2, TJPMN, Tipsi, T27Y, dsnPMN2, DLN1, A18neo. | ||
| DL | MN10-Ib (U2) | A01x2, A18b, A08e1, A31k, A27j, A23a, A06a, A06x1, A02h, A02e, A02g, A10e, A03a1, A03a3, A03x, A03a4, |
| A03d/e, A03x-eghb, VLELX4, Tipsi, dsnPMN2, DLN2, DLN1, A18neo, A18c. | ||
| DO | MN11-Ib | A31k, A06x1, A23a, A06a, A27l, T03g2, A03a1, A03a3, A03x-eghb. |
| DO | MN19-Ib | A27k, A18j, A18b, A18b3, T01d2, A31k, A27j, A23a, A06a, A06l, A06x1, A02f, A03a1, A03a3, T27Y, dsnPMN2, A27neo. |
| DO | MN20-Ib | A27h, A18j, A01c1, T01d2, T01d4, A19l, A06e, A03d/e, A27neo, a14neo, A03xyz, A26f. |
| DO | MN5-Ib (LO1) | A18b3, A18b2, A23a, A03a1, A03a3, A03a4, VLELX4, T27Y. |
| VL | MN6/7-Ib (RP3) | A18b3, A03a5, A27l, A06l, A06e, A02g, A02e, A03a4, T06WW, T06PP, Descending pre RP3. |
| VL | MN12-III (V-MN) | A27h, A03a5, A03g, A02g, A02e A27l, A06l,, A06e A03a6, A03a4, A03d/e, DLN1, Descending pre RP3. |
| VL | MN13-Ib (MN-VL2) | A27k, A03a5, A03g, A01d3, T01d4, A06l, A06a, A06e, A02g, A02e, A27l, A03a6, A03a4, A03x-eghb, A03d/e. |
| VL | MN14-Ib (RP1) | A27h, A18b2, A18b3, A27l, A06l, A02i, A03a4, A03a1, DLN1. |
| VL | MN30-Ib (RP4) | A18b3, A03a5, A01x2, A01d3,A01d4, A06e , A27l, A06l, A02g, A02e, A03a4, A03a6, A03x-eghb, A03d/e, A03SNC, A10a, A27Uniq, DLN1, A03xyz, SePN02b |
| VA | MN26-Ib | A27h, A01x3, A18f, A02j, A06e, A06l, A27l, T03g2, A03x-eghb, Descending neuron_SEZ, A03SNC, A03xKT, A03d/e, T11v, T27Y. |
| VA | MN27-Ib | A27h, A27k, A03g, A18j, A18f, A01x3, A01c1, A01c2, T01d2, T01d4, A06e, A06f, A19l, A14a, A31b, T03g2, A27n, A27neo, A03xKT, T11v, A26f. |
| VA | MN29-Ib | A01x3, A01x2, A01x3, T01d2, T01d4, A27l, A02g, A06e, T03g2, A27e2, A03a6, A03d/e, A10a, A27neo, T11v, A03SNC. |
| VO | MN15/16-Ib | A27h, A27k, A18b2, A06c, A06l, A06e, A02g, A02i, A03a6, DLN1. |
| VO | MN15/16/17-Ib | A27h, A03g, A06c, A06e, A27l, A02g, A02i, A01j, A27Uniq. |
| VO | MN28-Ib | A01x2, A27h, A18b2, A06c, A06l, A06e, A02g, A02i, A03a6. |
| T | MN8-1b (SBM) | A01c1, A01c2, A01d3, A27k, A03g, T01d1, A18j, A19l, A14a, A27n, A27e2, A27neo, A26f. |
| T | MN18-Ib | A01c1, A01c2, A01d3, A03g, A03o, A18j, A06a, A23a, A19l, A14a, A06x1, A02i, A01j, A27n, A10a, A10b, A27neo, T27Y, A26f. |
| T | MN21/22-Ib (LT1/LT2) | A01c1, A01c2, A27k, A03g, A18j, A18b2, T01d1, T01d2, A19l, A14a, A02i,A02f, A03xKT, T27Y, TGun, A27n, A27neo, A26f. |
| T | MN22/23-1b (LT2/LT3) | A01x, A01c1, A01c2, A27k, A03g, A09l, A18j, T01d1, T01d2, A01d3, A19l, A14a, A02f, A27n, A27neo, A27e2, T27Y, A26f. |
| T | MN23/24-1b (LT3/LT4) | A27k, A18j, A03g, A01c1, A01c2, T01d1, T01d2, A01d3, A19l, A27n, A27neo, A26f. |
| T | MN25-Ib (MN-VT1) | A01c1, A18a, A18b2, A18j, A18f, A27l, A14a, A19l, A02i, A31d, A03xKT, A05a. |
| DL/DO | MNISN (RP2) | A01x2, A18b, A03g, A31k, A27j, A27l, A02m, A02n, A02b, A06a, A23a, A03a1, A03a3, A03d/e, A03x-eghb, A05k, A10a, DLN2, DLN1, A18neo, dsnPMN2, SePN02b, T27Y, TJPMN, Projection neuron, A18c. |
| VL/VO | MSNISNb/d(RP5) | A27h, A03a5, A06l, A06c, A06f, A02g, A02e, A02b, A03a4, A03a6, A03x-eghb, A03d/e, A19d, A27Uniq, DLN1, SePN02b. |
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (Drosophila melanogaster) | R36G02-Gal4 | BDSC | # 49939 | A27h line |
| Genetic reagent (Drosophila melanogaster) | R87H09-Gal4 | BDSC | #40507 | A31k and A06l line |
| Genetic reagent (Drosophila melanogaster) | R78F07-Gal4 | BDSC | #47409 | A23a line |
| Genetic reagent (Drosophila melanogaster) | R44H10-LexA | BDSC | # 61543 | Muscle line |
| Genetic reagent (Drosophila melanogaster) | CQ2-lexA | this paper | U1-U5 motor neurons | |
| Genetic reagent (Drosophila melanogaster) | UAS-jRCaMP1b | BDSC | # 63793 | calcium indicator |
| Genetic reagent (Drosophila melanogaster) | lexAop-GCaMP6m | BDSC | #44275 | calcium indicator |
| Genetic reagent (Drosophila melanogaster) | 13XLexAop2-6XmCherry-HA | BDSC | #52271 | Used for ratiometric muscle calcium imaging |
Additional files
-
Supplementary file 1
CATMAID. json file of all reconstructed MNs in segment A1 as of 17 February 2019.
- https://cdn.elifesciences.org/articles/51781/elife-51781-supp1-v2.zip
-
Supplementary file 2
List of all PMN names and published synonyms.
- https://cdn.elifesciences.org/articles/51781/elife-51781-supp2-v2.xlsx
-
Supplementary file 3
Excel file of PMN neurotransmitter identity as of 31 August 2019.
Data from our own work plus previous studies (Kohsaka et al., 2014; Heckscher et al., 2015; Fushiki et al., 2016; Hasegawa et al., 2016; MacNamee et al., 2016; Zwart et al., 2016; Takagi et al., 2017; Yoshino et al., 2017; Burgos et al., 2018; Carreira-Rosario et al., 2018; Kohsaka et al., 2019).
- https://cdn.elifesciences.org/articles/51781/elife-51781-supp3-v2.xlsx
-
Supplementary file 4
CATMAID. json file of all reconstructed PMNs in segment A1 as of 21 August 2019.
- https://cdn.elifesciences.org/articles/51781/elife-51781-supp4-v2.zip
-
Supplementary file 5
Excel file of PMN to MN connectivity as of 31 August 2019.
- https://cdn.elifesciences.org/articles/51781/elife-51781-supp5-v2.xlsx
-
Supplementary file 6
Excel file of PMN to PMN connectivity as of 31 August 2019.
- https://cdn.elifesciences.org/articles/51781/elife-51781-supp6-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/51781/elife-51781-transrepform-v2.pdf





