Different CFTR modulator combinations downregulate inflammation differently in cystic fibrosis
Figures
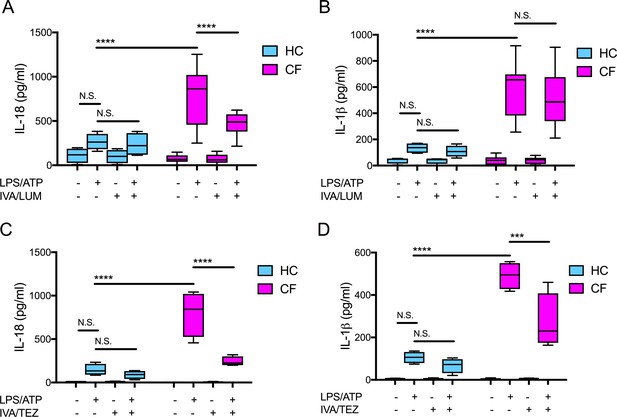
Cytokine secretion in NLRP3-stimulated monocytes with a differential response between CF (homozygous Phe508del) and HC (healthy controls) following in vitro exposure to either IVA/LUM or IVA/TEZ.
ELISA assays were used to detect A, C IL-18; B, D IL-1β secretion in monocytes from patients with CF-associated mutations (n = 10 IVA/LUM; n = 4 IVA/TEZ) vs HC (n = 4). Monocytes were treated with IVA/LUM (A, B) for 24 hr or IVA/TEZ (C, D) for 24 hr, then stimulated with LPS (10 ng/mL, 4 hr), and ATP (5 mM) for the final 30 min. A two-way ANOVA statistical test with Tukey’s multiple comparison was performed (p values * = ≤ 0.05, ** = ≤ 0.01, *** = ≤ 0.001 and **** = ≤ 0.0001). N.S. not-significant. IL-18 levels for HC (no treatment 10.84 pg/ml; IVA/TEZ 11.71 pg/ml); IL-1β levels for HC (no treatment 5.91 pg/ml; IVA/TEZ 5.27 pg/ml).
-
Figure 1—source data 1
Cytokine secretion in NLRP3-stimulated (LPS/ATP) monocytes isolated from CF (homozygous Phe508del) and HC (healthy controls) following in vitro exposure to either IVA/LUM or IVA/TEZ.
ELISA assays were used to detect IL-18 or IL-1β secretion (n = 10 IVA/LUM; n = 4 IVA/TEZ) vs HC (n = 4).
- https://cdn.elifesciences.org/articles/54556/elife-54556-fig1-data1-v2.xlsx
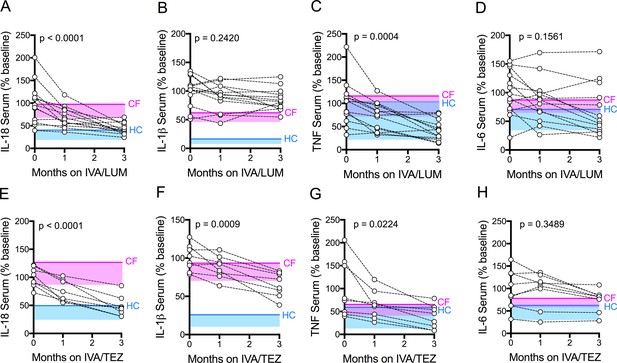
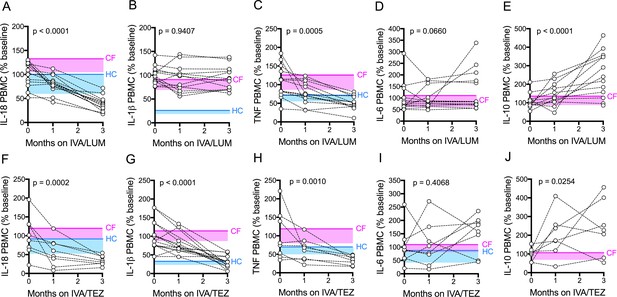
Serum cytokine levels in patients with CF (homozygous Phe508del), following treatment with IVA/LUM or IVA/TEZ.
Sera were collected at baseline, one month and three months of treatment from patients homozygous for Phe508del CFTR mutations receiving compassionate use IVA/LUM (n = 13) or IVA/TEZ (n = 8) therapy. ELISA assays were used to detect levels of A, IL-18; B, IL-1β; C, TNF and D, IL-6 in serum from the IVA/LUM treated group. ELISA assays were used to detect levels of E, IL-18; F, IL-1β; G,TNF; H, IL-6; in serum from the IVA/TEZ treated group. A one-way ANOVA statistical test with Tukey’s multiple comparison was performed. P value for baseline to three months shown on each graph. Baseline ranges were established for each cytokine from HC and clinically stable drug-naïve CF patients (Figure 2—figure supplement 3). Upper 95% confidence interval for baseline HC (solid blue line) or CF (solid pink line) with block colour shading (HC, blue; CF, pink) to lower 5% confidence interval is displayed for each cytokine.
-
Figure 2—source data 1
Serum cytokine levels in patients with CF (homozygous Phe508del), following treatment with IVA/LUM or IVA/TEZ.
The baseline (pre-therapy, zero month) values for each patient were calculated as a percentage of the average baseline within each patient group (IVA/LUM or IVA/TEZ). The one month and three month samples were calculated as a percentage of the baseline average. ELISA assays were used to detect IL-18, IL-1β, TNF or IL-6 secretion (n = 13 IVA/LUM; n = 8 IVA/TEZ).
- https://cdn.elifesciences.org/articles/54556/elife-54556-fig2-data1-v2.xlsx
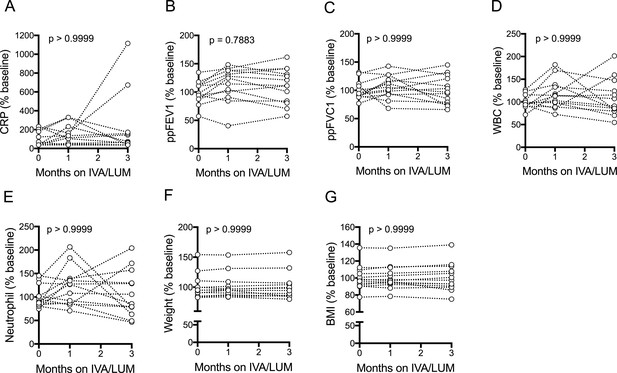
Clinical measurements of patients with CF (homozygous Phe508del) following IVA/LUM treatment.
Clinical measurements were taken at baseline, one month and three months from patients homozygous for Phe508del CFTR mutations receiving compassionate use IVA/LUM (n = 13). Measurements were plotted as % baseline. A, CRP; B, ppFEV1; C, ppFVC1; D, WBC; E, Neutrophil; F, Weight; G, BMI. A non-parametric Kruskal-Wallis statistical test with Dunn’s multiple comparison was performed. P value for baseline to three months is shown on each graph.
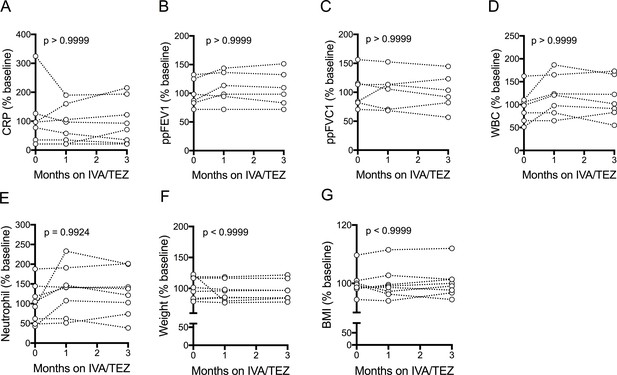
Clinical measurements of patients with CF (homozygous Phe508del) following IVA/TEZ treatment.
Clinical measurements were taken at baseline, one month and three months from patients homozygous for Phe508del CFTR mutations receiving compassionate use IVA/TEZ therapy. Measurements were plotted as % baseline. A, CRP (n = 8); B, ppFEV1 (n = 6); C, ppFVC1 (n = 6); D, WBC (n = 7); E, Neutrophil (n = 8); F, Weight (n = 8); G, BMI (n = 8). A non-parametric Kruskal-Wallis statistical test with Dunn’s multiple comparison was performed. P value for baseline to three months is shown on each graph.
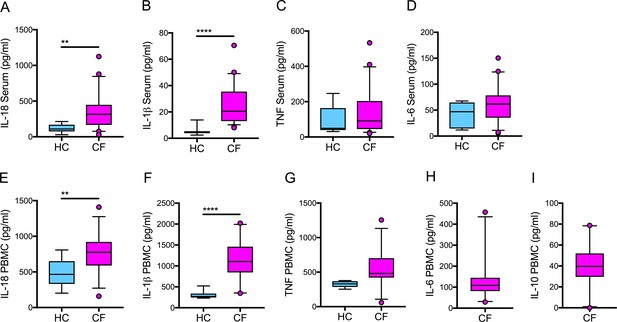
Baseline serum cytokine levels in HC and CF (homozygous Phe508del) and corresponding baseline cytokine levels in NLRP3-stimulated HC and CF immune cells.
Serum collected and PBMCs isolated at baseline from patients homozygous for Phe508del CFTR mutations (HC, n = 10; CF, n = 51). ELISA assays were used to detect levels of A, IL-18; B, IL-1β; C, TNF and D, IL-6 in serum. ELISA assays were used to detect levels of E, IL-18; F, IL-1β; G,TNF; H, IL-6; I, IL-10 secretion in PBMCs. PBMCs were stimulated with LPS (10 ng/mL, 4 hr), and ATP (5 mM) for the final 30 min. For PBMC data IL-18, IL-1β (HC n = 11, CF n = 32); TNF (HC n = 6, CF n = 27); IL-6 and IL-10 (CF n = 20) A non-parametric Mann Whitney test was performed. Error bars displayed as 5–95% percentile range with median (p values * = ≤ 0.05, ** = ≤ 0.01, *** = ≤ 0.001 and **** = ≤ 0.0001).
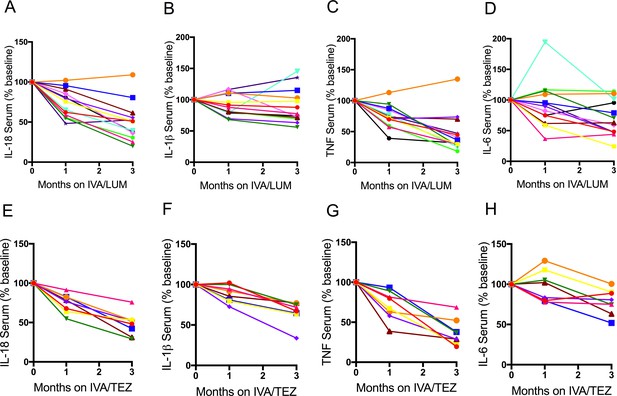
Individual therapy serum cytokine response in patients with CF (homozygous Phe508del) following IVA/LUM or IVA/TEZ treatment.
Serum collected at baseline, one month and three months from patients homozygous for Phe508del CFTR mutations receiving compassionate use IVA/LUM (n = 13) or IVA/TEZ (n = 8) therapy. ELISA assays were used to detect A, IL-18; B, IL-1β; C, TNF and D, IL-6 in serum from the IVA/LUM treated group. ELISA assays were used to detect levels of E, IL-18; F, IL-1β; G,TNF; H, IL-6; in serum from the IVA/TEZ treated group. Baseline for each patient set at 100% and proportional change at one and three months plotted.
Cytokine secretion in NLRP3-stimulated CF immune cells isolated from patients with CF (homozygous Phe508del), following treatment with IVA/LUM or IVA/TEZ.
PBMCs isolated at baseline, one month and three months from patients homozygous for Phe508del CFTR mutations receiving compassionate use IVA/LUM (n = 12/13) or IVA/TEZ (n = 8) therapy. Following isolation, PBMCs were immediately stimulated with LPS (10 ng/mL, 4 hr), and ATP (5 mM) for the final 30 min. ELISA assays were used to detect levels of A, IL-18; B, IL-1β; C, TNF; D, IL-6 and E, IL-10 secretion from PBMCs in the IVA/LUM treated group. ELISA assays were used to detect levels of F, IL-18; G, IL-1β; H,TNF; I, IL-6; J, IL-10 secretion from PBMCs in the IVA/TEZ treated group. For IVA/LUM: IL-18 (n = 13); IL-1β (n = 13); TNF (n = 12); IL-6 (n = 13); IL-10 (n = 12). A two-way ANOVA statistical test with Tukey’s multiple comparison was performed. P value for baseline to three months is shown on each graph. For IL-6 (IVA/LUM), a non-parametric Kruskal-Wallis statistical test with Dunn’s multiple comparison was performed. Baseline ranges were established for each cytokine from HC and clinically stable drug-naïve CF patients (Figure 2—figure supplement 3). Upper 95% confidence interval for baseline HC (solid blue line) or CF (solid pink line) with block colour shading (HC, blue; CF, pink) to lower 5% confidence interval is displayed for each cytokine.
-
Figure 3—source data 1
Cytokine secretion in NLRP3-stimulated (LPS/ATP) CF immune cells isolated from patients with CF (homozygous Phe508del), following treatment with IVA/LUM or IVA/TEZ.
The baseline (pre-therapy, zero month) values for each patient were calculated as a percentage of the average baseline within each patient group (IVA/LUM or IVA/TEZ). The one month and three month samples were calculated as a percentage of the baseline average. ELISA assays were used to detect IL-18, IL-1β, TNF, IL-6 or IL-10 secretion (n = 13 IVA/LUM; n = 8 IVA/TEZ).
- https://cdn.elifesciences.org/articles/54556/elife-54556-fig3-data1-v2.xlsx
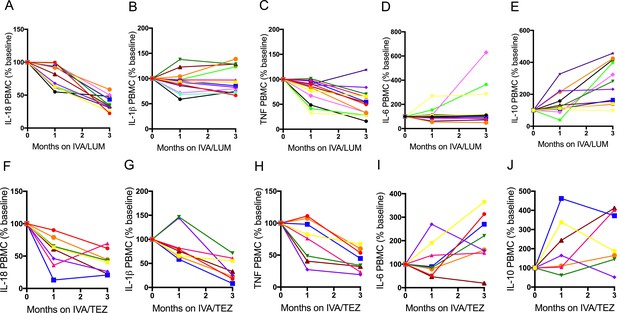
Individual therapy PBMC cytokine response in NLRP3- stimulated CF immune cells isolated from patients with CF (homozygous Phe508del) following treatment with IVA/LUM or IVA/TEZ.
PBMCs isolated at baseline, one month and three months from patients homozygous for Phe508del CFTR mutations receiving compassionate use IVA/LUM (n = 12/13) or IVA/TEZ (n = 8) therapy. Following isolation, PBMCs were immediately stimulated with LPS (10 ng/mL, 4 hr), and ATP (5 mM) for the final 30 min. ELISA assays were used to detect levels of A, IL-18; B, IL-1β; C, TNF; D, IL-6 and E, IL-10 secretion from PBMCs in the IVA/LUM treated group. ELISA assays were used to detect levels of F, IL-18; G, IL-1β; H,TNF; I, IL-6; J, IL-10 secretion from PBMCs in the IVA/TEZ treated group. Baseline for each patient set at 100% and proportional change at one and three months plotted. For IVA/LUM: IL-18 (n = 13); IL-1β (n = 13); TNF (n = 12); IL-6 (n = 13); IL-10 (n = 12).
NLRP3-inflammasome activation in patients with CF (homozygous Phe508del) receiving IVA/LUM or IVA/TEZ treatment.
PBMCs isolated at baseline, one month and three months from patients homozygous for Phe508del CFTR mutations receiving compassionate use IVA/LUM or IVA/TEZ therapy. Following isolation, PBMCs were immediately stimulated with LPS (10 ng/mL, 4 hr), and ATP (5 mM) for the final 30 min. Caspase-1 activity was detected in stimulated PBMCs at each time point for A, IVA/LUM (n = 4) and D, IVA/TEZ (n = 8). qPCR analysis was used to asses a fold change in mRNA expression of B, E NLRP3, and C, F pro-IL-1β for IVA/LUM (n = 4) (B–C) or IVA/TEZ (n = 7) (E–F). A one-way ANOVA with Tukey’s multiple comparison was performed on caspase-1 activity data and a non-parametric Kruskal-Wallis statistical test with Dunn’s multiple comparison was performed on the mRNA data. P value for baseline to three months indicated on each graph.
mRNA expression in NLRP3-stimulated PBMCs isolated from patients with CF (homozygous Phe508del) following IVA/LUM or IVA/TEZ treatment.
PBMCs isolated at baseline, one month and three months from patients homozygous for Phe508del CFTR mutations receiving compassionate IVA/LUM or IVA/TEZ treatment. Following isolation, PBMCs were immediately stimulated with LPS (10 ng/mL, 4 hr), and ATP (5 mM) for the final 30 min. qPCR analysis was used to asses a fold change in mRNA expression of A, E NLRC4, B, F TNF, C, G IL-6, D, H IL-10 for IVA/LUM (n = 4) (A–D) or IVA/TEZ (n = 7) (E–H). A non-parametric Kruskal-Wallis statistical test with Dunn’s multiple comparison was performed on mRNA data. P value for baseline to three months indicated on each graph.
Sustainability of oral drug therapy (IVA/LUM or IVA/TEZ) on NLRP3-stimulated cytokine production in PBMCs isolated from patients on therapy for three months.
PBMCs were isolated from patients receiving IVA/LUM or IVA/TEZ treatment for three months or longer. PBMCs were stimulated with LPS (10 ng/mL, 4 hr), and ATP (5 mM) for the final 30 min at 12 hr post oral therapy and at 36 hr post oral therapy with or without pre-treatment with IVA/LUM or IVA/TEZ as shown in schematic A. ELISA assays were used to detect levels of (B, E), IL-18; (C, F), IL-1β; secretion in media from PBMCs (IVA/LUM, n = 5; IVA/TEZ. n = 8). (D, G), Caspase-1 activity was detected in stimulated PBMCs at each time point, (IVA/LUM, n = 5; IVA/TEZ, n = 5). A one-way ANOVA with Tukey’s multiple comparison was performed on caspase-1 activity data and a non-parametric Kruskal-Wallis statistical test with Dunn’s multiple comparison was performed on IL-18 and IL-1β data (p values * = ≤ 0.05, ** = ≤ 0.01, *** = ≤ 0.001 and **** = ≤ 0.0001).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Biological sample (Homo sapiens) | Human Blood Samples | St James's University Hospital | Health Research Authority REC reference 17/YH/0084 | |
| Chemical compound, drug | Lymphoprep | Axis Shield | Cat# 1114544 | |
| Chemical compound, drug | Pan Monocyte Isolation Kit, human | Miltenyi Biotec | Cat# 130-096-537 | |
| Chemical compound, drug | Lipopolysacchride Ultrapure EK | InvivoGen | Cat# tlrl-eklps | 10ng/ml |
| Chemical compound, drug | ATP | InvivoGen | Cat# tlrl-atpl | 5 mM, 30 min |
| Chemical compound, drug | Lumacaftor (LUM) | AdooQ Bioscience | Cat#A10986 | 3 μM, 24 hr |
| Chemical compound, drug | Ivacaftor (IVA) | Cayman chemicals | Cat#15145 | 5 μM, 24 hr |
| Chemical compound, drug | Tezacaftor (TEZ) | TargetMol | Cat#T2263 | 5 μM, 24 hr |
| Commercial assay or kit | IL-1 beta Human Matched Antibody Pair | ThermoFisher Scientific | Cat# CHC1213 | Assay sensitivity < 31.2 pg/mL |
| Commercial assay or kit | IL-18 Human Matched Antibody Pair | ThermoFisher Scientific | Cat# BMS267/2MST | Assay sensitivity 78 pg/mL |
| Commercial assay or kit | IL-6 Human Matched Antibody Pair | ThermoFisher Scientific | Cat# CHC1263 | Assay sensitivity 15.6 pg/mL |
| Commercial assay or kit | TNF alpha Human Matched Antibody Pair | ThermoFisher Scientific | Cat# CHC1753 | Assay sensitivity < 15.6 pg/mL |
| Commercial assay or kit | IL1RA Human Matched Antibody Pair | ThermoFisher Scientific | Cat# CHC1183 | Assay sensitivity < 31.2 pg/mL |
| Chemical compound, drug | (TMB) substrate solution | Sigma | Cat# T0440 | |
| Commercial assay or kit | Caspase-1 Colorimetrix Assay | R and D Systems | Cat# BF15100 | |
| Commercial assay or kit | High-Capacity cDNA Reverse Transcription Kit | ThermoFisher Scientific | Cat# 4368814 | |
| Software, algorithm | GraphPad Prism7 | Graphpad software |
Additional files
-
Supplementary file 1
Demographic and clinical characteristics for CF patients on ivacaftor/lumacaftor (IVA/LUM) and ivacaftor/tezacaftor (IVA/TEZ).
Data are expressed as median and range. BMI: Body Mass Index; ppFEV: percent predicted forced expiratory volume, ppFVC: forced vital capacity, CRP: C-reactive protein. WBC: white blood count.
- https://cdn.elifesciences.org/articles/54556/elife-54556-supp1-v2.docx
-
Supplementary file 2
Cytokine secretion in unstimulated CF PBMCs following IVA/LUM (n = 13) or IVA/TEZ (n = 8) treatment.
ELISA assays were used to detect IL-18, IL-1β, TNF, IL-6 and IL-10 secretion in PBMCs.
- https://cdn.elifesciences.org/articles/54556/elife-54556-supp2-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/54556/elife-54556-transrepform-v2.pdf





