Connectomic analysis reveals an interneuron with an integral role in the retinal circuit for night vision
Figures
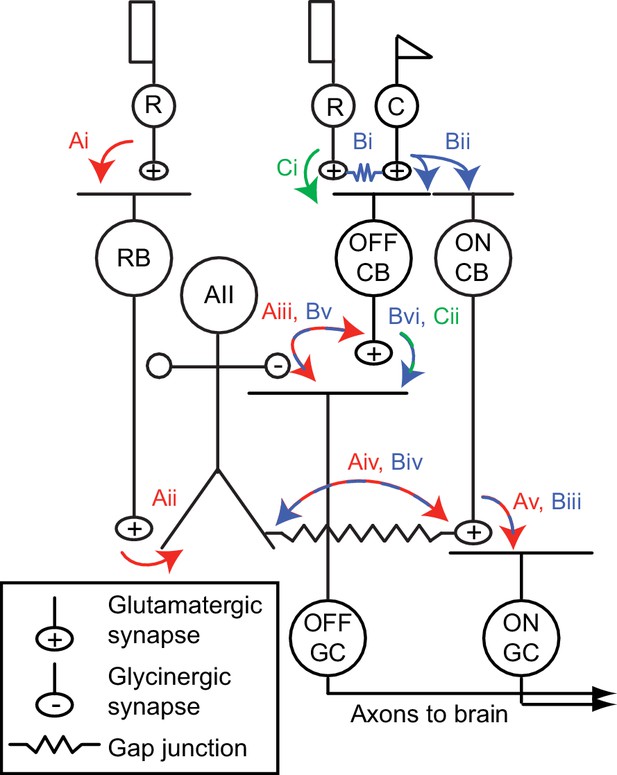
The mammalian retinal pathway for night vision.
(Ai-Av) In red: the rod bipolar (RB) pathway of mammalian retina. Rods make synapses onto RBs (Ai), which make synapses onto the AII. Signaling from AIIs to ON and OFF pathways is compartmentalized by the morphology of the AII, a bistratified cell with distinct neurites in the ON and OFF sublaminae of the inner plexiform layer (IPL). (Aii). Proximal (OFF-layer) dendrites provide glycinergic synapses (Aiii) onto the terminals of some OFF cone bipolar (CB) cells [primarily type 2 CBs; (Graydon et al., 2018)] and onto the dendrites of some OFF ganglion cells (GCs), including OFF α and δ GCs as well as suppressed-by-contrast GCs (Beaudoin et al., 2019; Demb and Singer, 2012; Jacoby et al., 2015). At their distal (ON-layer) dendrites, AIIs are coupled by electrical synapses to the terminals of ON CBs (Aiv) (particularly type 6 CBs presynaptic to ON α GCs); depolarization of AIIs drives excitatory transmission to GCs, including ON α GCs (Schwartz et al., 2012) (Av). Thus, the AII mediates so-called ‘cross-over’ inhibition, whereby one pathway (ON, in this case) suppresses the other (OFF) and thereby decorrelates their outputs (Demb and Singer, 2015). (Bi-Bvi) In blue: rods are coupled electrically to cones by gap junctions (Bi), and cones make synapses onto ON and OFF CBs (Bii). Depolarization of the ON CB by the cone not only drives glutamatergic transmission to ON GCs (Biii), it also depolarizes AIIs via the electrical synapses (Biv) and thereby elicits glycinergic transmission to OFF GCs and perhaps OFF CBs (Bv). (Ci-Cii) In green: rods make direct chemical synapses onto some types of OFF CB (Ci), which in turn contact OFF GCs (Cii).
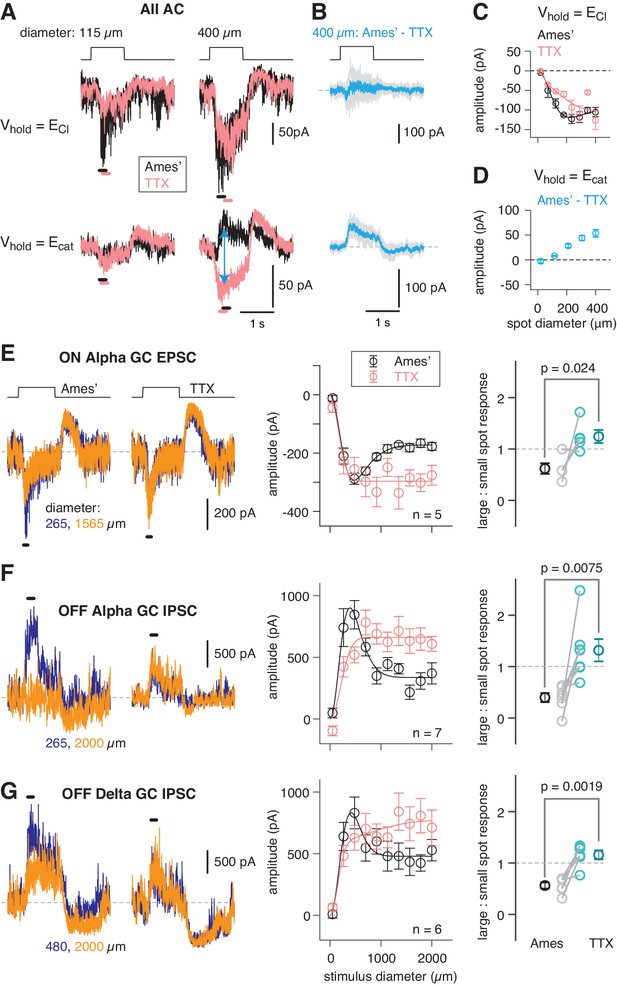
An inhibitory surround recorded in mouse AII ACs.
(A) AII responses to spots of light (10 R*/rod/s, 1 s duration) with the indicated diameter in control condition (Ames’ medium) and after applying TTX (1 µM). Responses were recorded at holding potentials (Vhold) near ECl (−70 mV; top row) and near Ecat (+5 mV; bottom row). (B) Difference current at each Vhold for the 400 µm spot (average of n = 5 cells; shaded areas are ± SEM across cells as a function of time). (C) Response amplitude (measured over a 200 ms window: horizontal bars in A) to spot of variable diameter at Vhold = ECl (n = 5 cells). Error bars are ± SEM across cells. (D) Amplitude of difference current at Vhold = Ecat averaged across cells. Conventions as in (C). (E) Left, light flash (dark background, two spot sizes, 4 or 40 R*/rod/ s across cells)-evoked EPSCs in an ON α RGC. Under control conditions, the response is smaller for the larger diameter, illustrating the surround effect, which is blocked by TTX (1 µM). Middle, Spot stimuli of varying diameter (Ames’ and TTX) elicit EPSCs; peak response amplitude measured in a time window (100–200 ms; horizontal line at left). Right, ratio of response to large (averaged over the three largest diameters) and small (chosen as the optimal spot size for each cell in the control condition) spots. A ratio <1 indicates a surround effect. The ratio increased significantly in TTX. (F) Same as (E) for IPSCs in OFF α RGCs (spot intensity, 4 R*/rod/s). (G) Same as (F) for OFF δ RGCs.
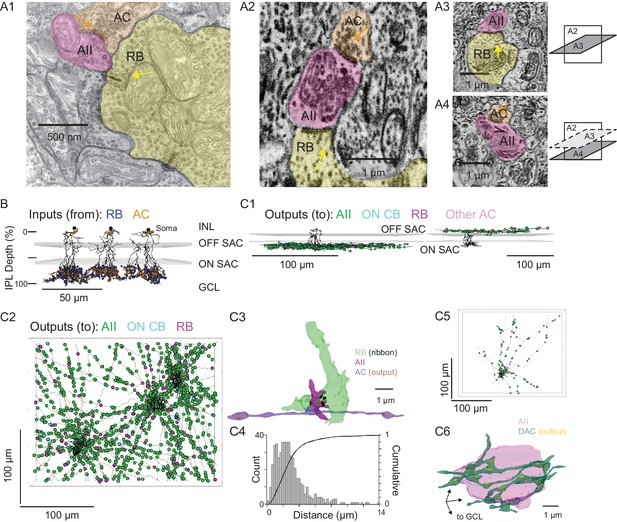
Anatomical characterization of AC axons presynaptic to the AII.
(A1) A RB dyad synapse, visualized by TEM. Note the synaptic ribbon (yellow arrow) and the nearby AC input to the postsynaptic AII (orange arrow). (A2) A similar dyad synapse observed by SBEM. Again, the RB ribbon and AC input to the postsynaptic AII are marked with yellow and orange arrows, respectively. (A3, A4) Orthogonal views of the synapse illustrated in A2 showing the RB dyad (top) and the AC input to the postsynaptic AII (bottom). A3 and A4 are from different sections to illustrate the appearance of both ribbon and AC inputs in the section orthogonal to A2. This is illustrated schematically by the squares at right. (B) Three AIIs, skeletonized and annotated with either RB ribbon synapse locations or AC synapse locations. The view is from the side of the volume, representing a transverse section through the retina. The planes containing ON and OFF SAC dendrites are represented by gray rectangles. Note that the majority of AC inputs to the AII are found in the same IPL sublaminae as the RB inputs, the vitreal side of the ON SAC dendrites (from Ding et al., 2016). As well, AIIs receive no synaptic input from ACs in the IPL sublaminae between the ON and OFF SAC dendrites. AII skeletons and annotations are contained within Source data 1 and downloadable in Knossos XML format. SAC skeletons are downloadable as Source data 3. (C1) Side view of a single AII and presynaptic neurites with output synapses annotated (right: presynaptic to distal dendrites; left: presynaptic to soma and proximal dendrites; different AIIs are shown in the two panels). (C2) An en face view, visualized from the GCL, of 3 AIIs (black) and 61 presynaptic axons. Note the preponderance of green circles, indicating synapses to the three reconstructed AIIs and other AIIs within the volume. Virtually every synapse that was not made with an AII was onto a RB. We observed two synapses onto these axons. Neurite annotations are contained within Source data 1 and downloadable in Knossos XML format. (C3) Segmentation of the RB-AII-AC complex. The AC axon is thin, with occasional large varicosities containing clusters of vesicles. It makes synapses with AIIs, usually quite close to RB→AII ribbon synapses. In this example, the AII dendritic segment receives four ribbon inputs from the axon terminal varicosity of a RB and one conventional synaptic input from an AC. The other synapses made by this AC axon segment also are with AIIs. (C4) Conventional and normalized cumulative histograms of distribution of nearest-neighbor distances between AC→AII and RB→AII synapses. (C5) An en face (from GCL) view of a single AII and neurites presynaptic to its soma and proximal dendrites. (C6) Segmentation of an AII soma and presynaptic neurites, with presynaptic active zones annotated. The image is a tilted side view; the orientation axis (lower left) indicates the relative position of the GCL.
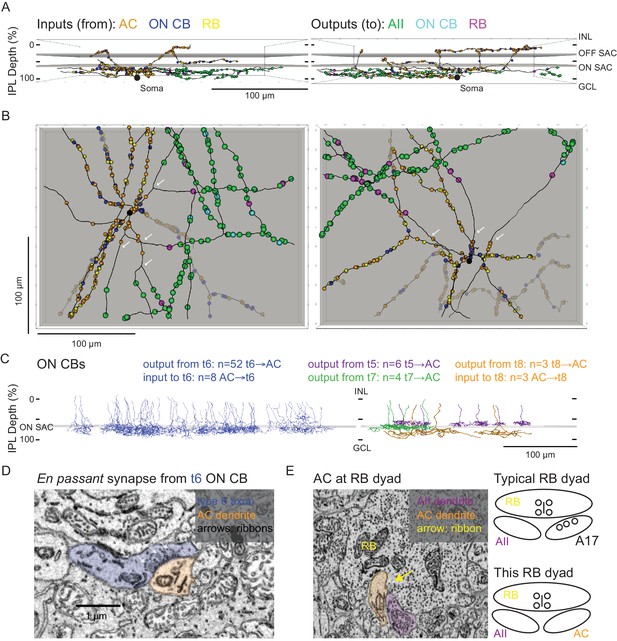
Anatomical characterization of an AC presynaptic to the AII.
(A) Skeletonization and annotation of two ACs (left and right panels) with similar morphologies and patterns of synaptic connectivity. Note that both cells, viewed from the side, as in a transverse section through the retina, have similar neurite branching patterns and receive synaptic input from ACs and from ON CBs on dendrites in the OFF laminae of the IPL, on the outer side of the OFF SAC dendrites (i.e. close to 0% IPL depth; here, SAC dendrites are represented as gray rectangles). AC skeletons and annotations are contained within Source data 1 and downloadable in Knossos XML format. (B) An en face view (viewed from the GCL; the gray represents the layer of ON SAC dendrites) of the two ACs illustrated in (A). Note that their synaptic inputs and outputs are segregated to different sections of their processes; the area receiving input is dendritic, and the area making output is axonal. White arrows indicate areas where dendrites become axons (inputs are proximal to the arrow, closest to the soma; outputs are distal to the arrow, farther from the soma). AC skeletons and annotations are contained within Source data 1 and downloadable in Knossos XML format. (C) Side (transverse) view illustrating all ON CBs pre- or postsynaptic to the two ACs illustrated in (A) and (B). ON CBs were classified based on axon branching pattern and stratification depth relative to the ON SAC dendrites (Helmstaedter et al., 2013). CB skeletons and annotations are contained within Source data 2 and downloadable in Knossos XML format. (D) Example en passant ribbon-type synapses in a type 6 ON CB axon. Note three ribbons clustered together and presynaptic to the same AC process. See Figure 4—video 1 for a larger image stack. (E) Example of RB dyad at which the AC type shown in (A) and (B) replaces the A17 as one of the two postsynaptic cells (see schematic at right). See Figure 4—video 2 for a larger image stack.
Image stack containing en passant type 6 CB→AC synapses illustrated in Figure 4D.
Coordinates X: 805 Y: 1598 Z: 2832–51 at https://webknossos.org/datasets/Demo_Organization/110629_k0725/view.
Image stack containing RB→AII, AC dyad synapse illustrated in Figure 4E.
Coordinates X: 3273 Y: 14171 Z: 6421–38 at https://webknossos.org/datasets/Demo_Organization/110629_k0725/view.
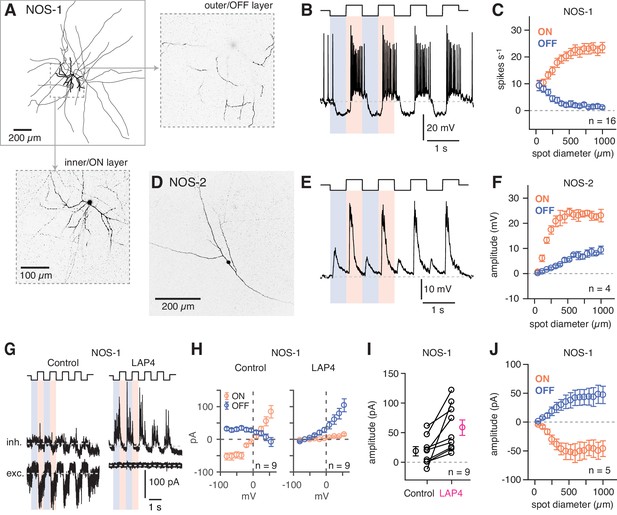
NOS-1 ACs are spiking ON-center cells that can be distinguished from NOS-2 ACs.
(A) Morphology of a NOS-1 AC. Dendrites (thick) and axons (thin) were drawn from confocal images of a NOS-1 AC filled by Lucifer Yellow during whole-cell recording. Single confocal sections are shown for the inner/ON and outer/OFF layers of processes for the region indicated (dashed, boxed region). The cell is bistratified in the region proximal to the cell body; only the ON-layer processes are shown in the drawing. (B) Membrane potential recording for the cell in (A). The cell had a baseline firing rate at mean luminance (~104 R*/cone/s) that modulated above and below baseline during positive and negative contrast periods, respectively (spot diameter, 600 µm; 100% contrast). (C) Population (n = 16 cells) changes in firing rate during positive contrast (ON response) and negative contrast (OFF response) as a function of spot size (100% contrast). Firing rate was computed over a 500 ms time window for each contrast. Error bars indicated ± SEM across cells. (D) Collapsed confocal stack (maximum projection image) of a filled NOS-2 cell. (E) The NOS-2 cell in (D) responds with depolarization at both positive and negative contrast, an ON-OFF response (spot diameter, 600 µm; 100% contrast). (F) Population (n = 4 cells) changes in membrane potential as a function of spot size (measured over a 100 ms time window). Conventions are the same as in (C). (G) Excitatory and inhibitory current measured in a NOS-1 cell to a spot stimulus (diameter, 400 µm). After adding L-AP4 (20 µM) to suppress the ON pathway, the excitatory current is blocked and the inhibitory current increases and is OFF responding. (H) Current-voltage (I–V) plots for ON and OFF responses for data in (G) averaged across cells (measured over a 100- to 200-ms time window). Error bars indicate ± SEM across cells. (I) Population change in the OFF inhibitory current after adding L-AP4. Individual cell data are connected by lines. Population data indicate mean ± SEM across cells. (J) Excitatory current amplitude for a population of NOS-1 cells (n = 5 cells) as a function of spot diameter (100% contrast; measured over a 100 ms time window). Error bars indicate ± SEM across cells.
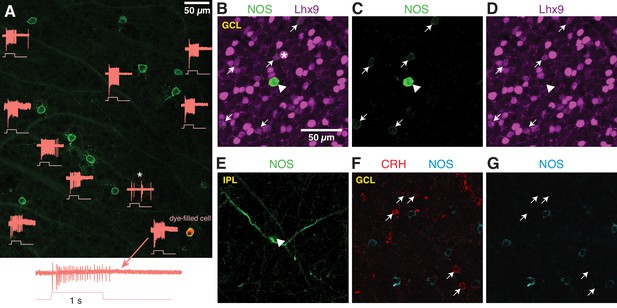
NOS-expressing ACs in the ganglion cell layer are primarily NOS-1 cells.
(A) Loose-patch spike recording from a region of retina with cells labeled in the nNOS-CreER x Ai32 mouse. The response to a light flash (800-µm diameter, ~104 R*/cone/s) is shown next to each soma that was recorded. The majority of cells showed a sustained ON response in the spike rate during the light flash. In one case, the response differed (*) and showed a transient ON-OFF response. The cell at lower right was subsequently studied by whole-cell recording and was filled with dye; for this cell, the spike response is shown at an expanded scale below the image. (B–E) P12 retinas (C57/B6 wild-type) stained with antibodies to label NOS and Lhx9 expressing cells in the GCL. Most NOS-expressing cells showed dim labeling for Lhx9 antibody (arrows). A well-stained NOS-expressing cell in the center (arrow-head) did not show Lhx9 labeling. Laser power was increased for Lhx9 imaging, such that fluorescence of strongly-labeled cells was saturated (*, example cell), making it easier to visualize weakly-labeled cells. NOS labeling alone (C) and Lhx9 labeling alone (D). (E) Same as (C) with the image plane shifted to the inner plexiform layer (IPL). The NOS-expressing cell that lacked Lhx9 expression (arrowhead) had thick dendrites that could be followed into the IPL, with the characteristic properties and stratification of a NOS-2 cell. (F–G) P12 retina (C57/B6) stained with antibodies to label corticotropin releasing hormone (CRH) and NOS. NOS-expressing cells do not overlap with CRH-expressing cells. NOS labeling alone (G). Scale bar in (B) applies to (B) – (G).
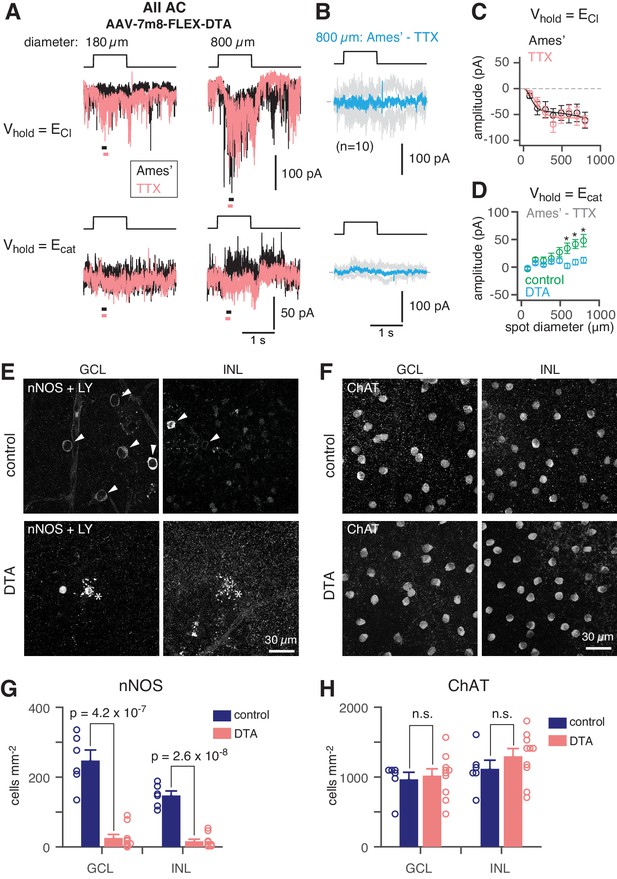
NOS+ ACs generate the TTX-sensitive receptive field surround of the AII.
(A) AII responses to spots (diameter indicated; 40 R*/rod/s, 1 s duration) in control (Ames’) and in TTX (1 µM) recorded at ECl (−70 mV; top) and near Ecat (+5 mV; bottom) in a nNOS-CreER retina injected with Cre-dependent DTA virus (AAV-7m8-FLEX-DTA). (B) Difference current at each Vhold for the 800 µm diameter spot (average of n = 10 cells; shaded areas are ± SEM across cells as a function of time). (C) Spot (variable diameter) response amplitudes [measured over a 150 ms time window, indicated by horizontal bars in A] at Vhold = ECl (n = 10 cells). Error bars are ± SEM across cells. (D) Difference current amplitude at Vhold = Ecat averaged across cells. Same conventions as in (C). Recordings from control retinas are superimposed (n = 6 cells). Control mice had the same AAV and tamoxifen injections but were Cre-negative. Responses were smaller in the DTA group compared to the control group (one-tailed t-tests, *): 590 µm, t = 3.16, p=3.51×10−3; 695 µm, t = 3.31, p=2.12×10−3; 800 µm, t = 2.93, p=5.45×10−3). (E) nNOS immunolabeling in GCL and INL, centered on a region with a recorded AII (visible in some images, marked by *). (F) Same format as (E) for ChAT immunolabeling of starburst ACs. (G) NOS+ cell density over a square region (0.64 × 0.64 mm) centered on a recorded AII and visualized by nNOS immunolabeling: DTA (n = 10 retinas) vs. control (n = 6 retinas). Virus-injected retinas had significantly fewer cells (one-tailed t-test): GCL, t = 8.35, p=4.2×10−7; INL, t = 10.5, p=2.6×10−8. (H) Same format as (G) for ChAT immunolabeling. Cell density assessed over a square region (0.16 × 0.16 mm) centered on a recorded AII. Starburst AC density in DTA virus-injected retinas (n = 10) was not significantly (n.s.) different than in control retinas (n = 6): GCL, t = −0.35; INL, t = −1.05.
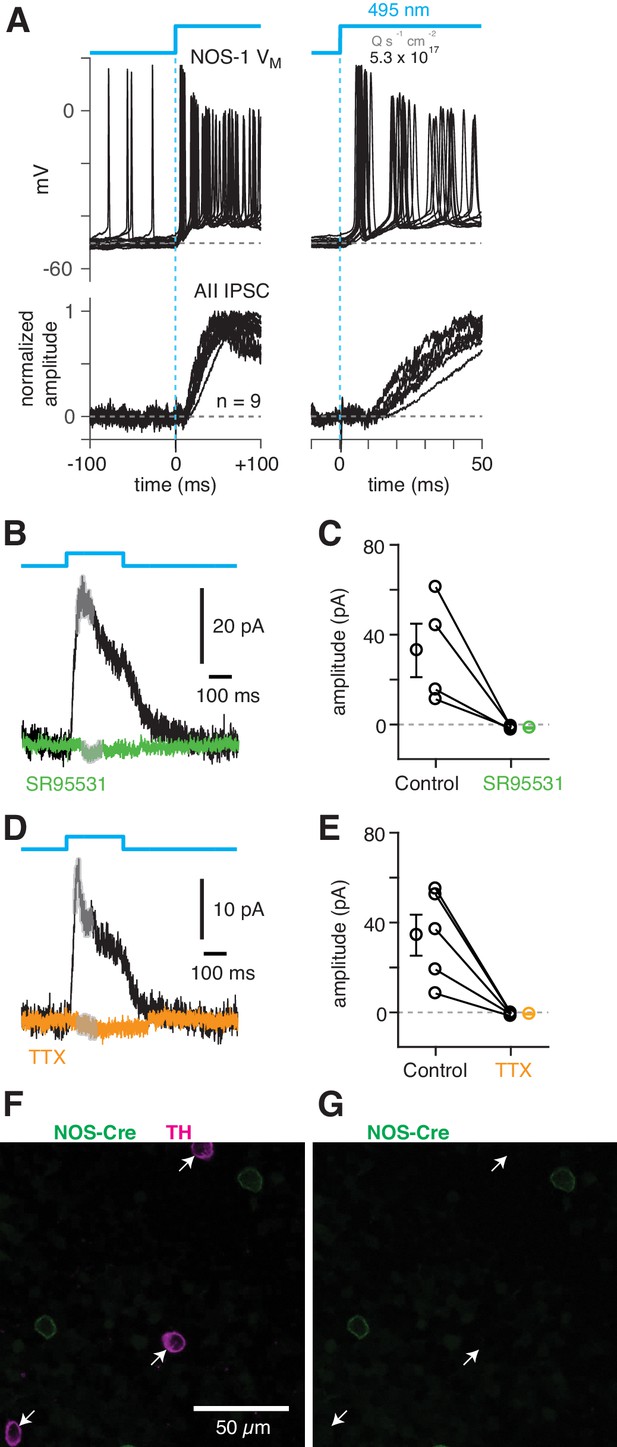
NOS-1 cells make synapses with AII amacrine cells.
(A) Top/Left, optogenetic stimulation of a ChR2-expressing NOS-1 cell (of n = 3 total) in the nNOS-CreER::Ai32 retina responded with increased spike firing to blue light (n = 12 trials overlaid). Response was recorded in whole-mount retina in the presence of drugs to block photoreceptor-mediated inputs to retinal circuitry: DNQX (50 µM), D-AP5 (50 µM), L-AP4 (2 µM), and ACET (1 µM). Bottom/Left, the optogenetic stimulus evoked IPSCs (Vhold = Ecat) in AII ACs (n = 9 cells). Responses are normalized to the maximum amplitude, 39 ± 6 pA (measured over a 60–70 ms time window). Right, expanded version of traces at left. The initial spike in the NOS-1 cell occurred a few milliseconds after optogenetic stimulation (top), followed a few milliseconds later by the onset of IPSCs in the AII. (B) In AII ACs recorded under the conditions in (A), inhibitory current (measured over the gray region) was blocked by the GABA-A receptor antagonist SR95531 (50 µM). (C) Effect of SR95531 in a sample of AII ACs (n = 4 cells). Error bars indicate ± SEM across cells. (D) Same format as (B) with the sodium channel blocker TTX (1 µM). (E) Same format as (C) with TTX (n = 5 cells). (F) Confocal image of the inner nuclear layer of a retina from the nNOS-creER::Ai32 mouse. A tyrosine-hydroxylase (TH) antibody was used to label dopaminergic ACs (arrows), which did not overlap with Cre-expressing NOS+ ACs. (G) Same image as (F) without the TH labeling. None of the cells with TH immunolabeling (arrows) were eYFP+.
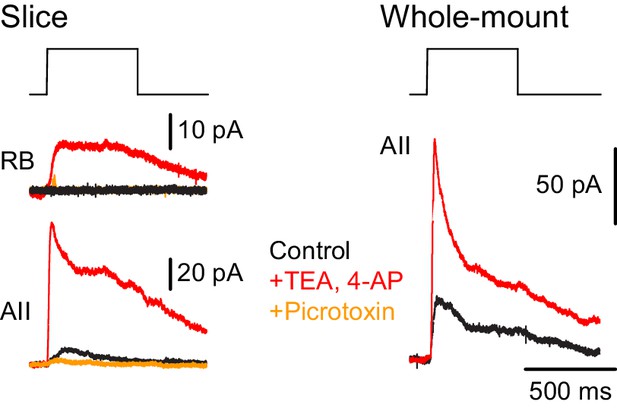
NOS-1 ACs make synapses with RBs.
At left, recordings from a RB (top) and an AII (bottom) in a retinal slice demonstrate that optogenetic stimulation of cre-expressing cells in the nNOS-CreER::Ai32 retina evoked inhibitory currents (Vhold = Ecat). Potentiation of presynaptic depolarization with K channel blockers was necessary to elicit responses in RBs owing to the small number of presynaptic axons preserved in the 200 µm thin slice. At right, K channel blockers potentiate larger inhibitory currents (Vhold = Ecat) recorded in an AII and evoked by optogenetic stimulation of cre-expressing cells in a whole-mount preparation of nNOS-CreER::Ai32 retina.
Tables
Inputs to AII amacrine cells.
| AII #1 | AII #2 | AII #3 | Mean ±SD | |
|---|---|---|---|---|
| Inputs (from) | ||||
| RB | 173 | 171 | 176 | 173 ± 3 |
| AC (Total) | 178 | 177 | 176 | 177 ± 1 |
| AC (ON layer) | 161 (96%) | 157 (89%) | 161 (91%) | 160 ± 2 (92 ± 4%) |
| AC (Soma) | 17 (4%) | 20 (11%) | 15 (9%) | 17 ± 3 (8 ± 4%) |
Connetivity of ACs presynaptic to AIIs.
| Axons (Figure 3C2) | Cell 1 (Figure 4) | Cell 2 (Figure 4) | |
|---|---|---|---|
| Inputs (from) | Total: 2 | Total: 314 | Total: 297 |
| AC | 200 (63.5%) | 183 (61%) | |
| RB | 1 | 47 (15%) | 56 (19%) |
| ON CB | 1 | 66 (21%) | 55 (18.5%) |
| Unidentified | 2 (<1%) | 3 (1%) | |
| Outputs (to) | Total: 1425 | Total: 115 | Total: 106 |
| AII | 1212 (85%) | 93 (81%) | 86 (81%) |
| RB | 173 (12%) | 11 (9.5%) | 18 (17%) |
| ON CB | 35 (2%) | 11 (9.5%) | 2 (2%) |
| Unidentified | 5 (<1%) |
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (Mus musculus) | C57BL/6J | Jackson Laboratory Stock #000664 | IMSR Cat# JAX000664, RRID:IMSR_JAX:000664 | Wild type mouse |
| Genetic reagent (M. musculus) | nNOS-CreER | Jackson Laboratory Stock #014541 | IMSR Cat# JAX014541, RRID:IMSR_JAX:014541 | Transgenic mouse: cre-driver line |
| Genetic reagent (M. musculus) | Ai14 | Jackson Laboratory Stock #007914 | IMSR Cat# JAX007914 RRID:IMSR_JAX:007914 | Transgenic mouse: cre-dependent tdTomato expression |
| Genetic reagent (M. musculus) | Ai32 | Jackson Laboratory Stock #024109 | IMSR Cat# JAX024109 RRID:IMSR_JAX:024109 | Transgenic mouse: cre-dependent ChR2/eYFP expression |
| Recombinant DNA reagent | AAV2/7m8-CAG-FLEX-DTA-WPRE-SC40pA | This paper | Adeno-associated virus; 1013 GC/ml | |
| Recombinant DNA reagent | pAAV-Syn-FLEX-rc[CoChR-GFP] | PMID:24509633 | Addgene #62724; RRID:Addgene_62724 | Plasmid |
| Recombinant DNA reagent | pAAV-CAG-fDIO-oG-WPRE-SV40pA | PMID:27149846 | Addgene #74291; RRID:Addgene_74291 | Plasmid |
| Recombinant DNA reagent | PGKdtabpA | PMID:9226440 | Addgene #13440; RRID:Addgene_13440 | Plasmid |
| Recombinant DNA reagent | AAV2/7m8 capsid | Gift of Dr. John Flannery, UC Berkeley; PMID:23761039 | Addgene #64839; RRID:Addgene_64839 | Plasmid |
| Antibody | anti-ChAT (Goat polyclonal) | EMD Millipore | Cat# AB144P; RRID:AB_2079751 | 1:200 dilution |
| Antibody | anti-LuciferYellow (Rabbit polyclonal) | ThermoFisher Scientific | Cat# A-5750; RRID:AB_2536190 | 1:2000 dilution |
| Antibody | anti-nNOS (Rabbit polyclonal) | ThermoFisher Scientific | Cat# 61–7000; RRID:AB_2313734 | 1:500 dilution |
| Antibody | anti-nNOS (Guinea pig polyclonal) | Frontier Institute | Cat# Af740; RRID:AB_2571816 | 1:2000 dilution |
| Antibody | anti-TH (Rabbit polyclonal) | EMD Millipore | Cat# AB152; RRID:AB_ 390204 | 1:1000 dilution |
| Antibody | anti-human/rat CRF serum (Rabbit polyclonal) | gift of Dr. Paul Sawchenko, Salk Institute | Code# PBL rC68 | 1:40,000 dilution |
| Antibody | anti-LHX9 (Guinea pig polyclonal) | gift of Dr. Jane Dodd, Columbia University | 1:20,000 dilution | |
| Antibody | Donkey Anti-Goat AlexaFluor 633 secondary | ThermoFisher Scientific | Cat # A-21082; RRID:AB_2535739 | 1:500 dilution |
| Antibody | Donkey Anti-Rabbit AlexaFluor 488 secondary | Jackson ImmunoResearch | Cat# 711-545-152; RRID:AB_2313584 | 1:500 dilution |
| Antibody | Donkey Anti-Rabbit Cy3 secondary | Jackson ImmunoResearch | Cat# 711-165-152; RRID:AB_2307443 | 1:500 dilution |
| Antibody | Donkey Anti-Rabbit Cy5 secondary | Jackson ImmunoResearch | Cat# 711-175-152; RRID:AB_2340607 | 1:500 dilution |
| Antibody | Donkey Anti-Guinea pig secondary | Jackson ImmunoResearch | Cat# 706-175-148; RRID:AB_2340462 | 1:500 dilution |
| Software, algorithm | ScanImage | http://scanimage.vidriotechnologies.com/ PMID:12801419 | RRID:SCR_014307 | Software for 2PLSM |
| Software, algorithm | ParaView | https://www.paraview.org/ | RRID:SCR_002516 | Software for visualization |
| Software, algorithm | IgorPro | https://www.wavemetrics.com/ | RRID:SCR_000325 | Software for data acquisition and analysis |
| Software, algorithm | PClamp | https://www.moleculardevices.com/ | RRID:SCR_011323 | Software for data acquisition and analysis |
| Software, algorithm | Knossos | https://knossos.app/ PMID:21743472 | RRID:SCR_003582 | Software for volumetric imaging analysis |
| Software, algorithm | ImageJ | https://ImageJ.net | RRID:SCR_003070 | Software for cell counting and image projection |
Additional files
-
Source code 1
A compressed file containing Python scripts used to parse Knossos XML files containing skeletons and annotations: see ‘tutorial.py’ for descriptions.
- https://cdn.elifesciences.org/articles/56077/elife-56077-code1-v1.zip
-
Source data 1
Skeletons of AIIs and other ACs.
- https://cdn.elifesciences.org/articles/56077/elife-56077-data1-v1.k.zip
-
Source data 2
Skeletons of ON CBs, NOS-1 ACs, and one ON SAC.
- https://cdn.elifesciences.org/articles/56077/elife-56077-data2-v1.k.zip
-
Source data 3
Skeletons of ON and OFF SACs.
- https://cdn.elifesciences.org/articles/56077/elife-56077-data3-v1.k.zip
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/56077/elife-56077-transrepform-v1.docx





