Autophagy in T cells from aged donors is maintained by spermidine and correlates with function and vaccine responses
Figures
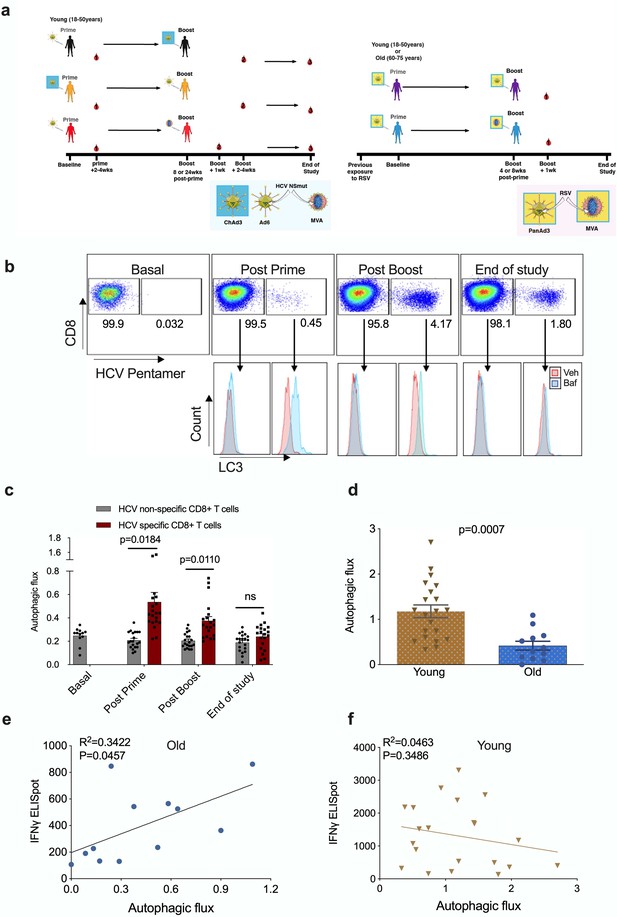
Autophagy is induced by vaccination in antigen-specific T cells and correlates with donor age.
PBMCs were isolated from blood samples of vaccinated healthy donors. LC3-II was measured in CD8+ cells using flow cytometry after 2 hr treatment with 10 nM bafilomycin A1 (BafA1) or vehicle. Autophagic flux was calculated as LC3-II mean fluorescence intensity (BafA1-Vehicle)/Vehicle. (a) Vaccine regimen for HCV and RSV trials. (b) Representative plots showing BafA1 in light blue and vehicle pink. (c) Quantification in HCV non-specific CD8+ T cells and HCV-specific CD8+ T cells detected by HCV pentamers from 10 vaccinees (includling duplicates) using different HCV vaccine regimens, priming with ChimAd and boosting with MVA or AD6 vectors. Autophagy was measured at the peak of the T cell response post prime vaccination, peak of the T cell response post boost vaccination and at the end of the study. (d) Autophagic flux was measured in CD8+ cells from young donors (N = 12,<65 years) and old donors (N = 21,>65 years) vaccinated with respiratory syncytial virus (RSV) 7 days after last boost, quantification calculated as mentioned above. Data represented as mean ± SEM. (e, f) Correlation of autophagic flux with total response to peptide pools specific T cell IFNγ response to RSV exposure measured by ELISpot in CD8+ cells from old donors (e) and young donors (f), donors as in (d). Linear regression with 95% confidence intervals from old and young donors. The goodness of fit was assessed by R2. The p value of the slope is calculated by F test.
-
Figure 1—source data 1
Autophagy is induced by vaccination in antigen-specific T cells and correlates with donor age.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig1-data1-v2.xlsx
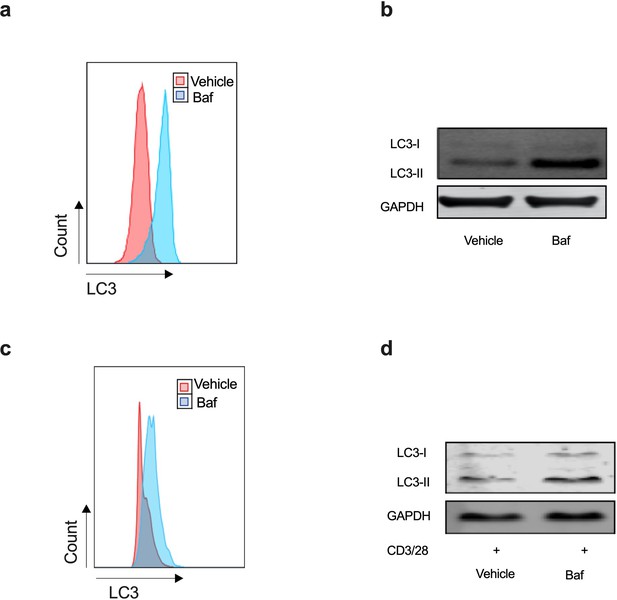
Autophagy levels by flow cytometry-based assay and conventional LC3 western blot in Jurkat cell line and PBMCs.
(a–b) Human Jurkat T cell line was cultured for 24 hr and treated with or without Bafilomycin A1 for the last 2 hr. Cells were split into two aliquots, representative flow cytometry-based assay (a), representative western blot for LC3-II and GAPDH for the same sample (b). (c–d) PBMCs from young human donors were cultured with anti-CD3/CD28 for 3 days in the absence/presence of Bafilomycin A1 for the last 2 hr. Cells were split into two aliquots, representative flow cytometry-based assay (c), representative western blot for LC3-II and GAPDH for the same sample (d).

LC3-II detection by flow cytometry is a reliable and reproducible technique in immune cells over several blood draws.
PBMCs were generated from blood taken at 3 weeks intervals (samples 1, 2, 3) from young human donors and were cultured for 24 hr, in the abence/presence of Bafilomycin A1 for the last 2 hr. Here basal autophagic flux was calculated as LC3-II mean fluorescence intensity (treatment-basal)/basal. Monocytes gated on CD14+ treated with IFNγ or LPS (a), B cells gated on CD19+ treated with anti-CD40L and anti-IgM (b), CD4+ T cells gated on CD3+CD4+ treated with anti-CD3/CD28 (c), CD8+ T cells gated on CD3+ CD8+ treated with anti-CD3/CD28 (d).
-
Figure 1—figure supplement 2—source data 1
LC3-II detection by flow cytometry is a reliable and reproducible technique in immune cells over several blood draws.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig1-figsupp2-data1-v2.xlsx

Regimen of immunizations and blood sampling for HCV trail.
HCV = Hepatitis C virus, ChAd = Chimpanzee Adenoviral Vector, MVA = Modified Ankara Virus vector.
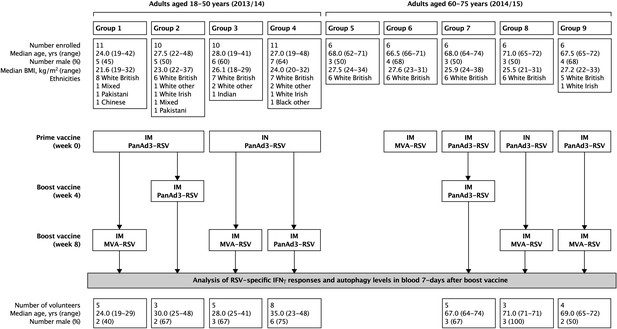
Regimen of immunizations and blood sampling for RSV trail.
RSV = respiratory syncitial virus, ChAd = Chimpanzee Adenoviral Vector, MVA = Modified Ankara Virus vector, Unlike for HCV, the adults in the RSV study will have prior immune responses that have been boosted by natural exposure throughout life. In the context of RSV, we still use the term ‘prime’ to mean the first dose of vaccine. Similarly, the term ‘boost’ means the second dose of vaccine and not exposure.

Correlation of age with total and peptide-pool specific T cell IFNγ response to RSV exposure measured by ELISpot in CD8+ cells, donors as in Figure 1e.
Linear regression with 95% confidence intervals from old and young donors. The goodness of fit was assessed by R2. The p value of the slope is calculated by F test.
-
Figure 1—figure supplement 5—source data 1
Correlation of age with total and peptide-pool specific T cell IFNγ response to RSV.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig1-figsupp5-data1-v2.xlsx
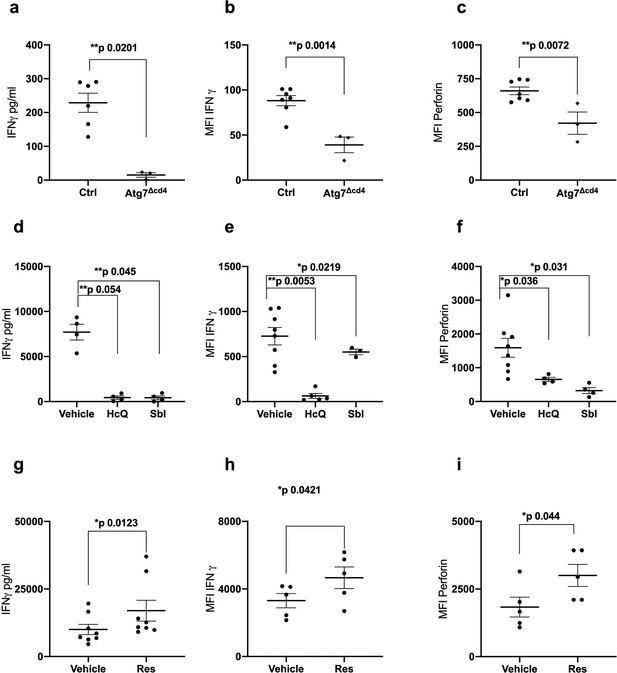
Autophagy is required for CD8+ T cell function.
(a–c) Splenocytes from control mice (Ctrl: CD4-cre;Atg7+/+) and Atg7 knockout mice (Atg7Δcd4: CD4-cre;Atg7-/-) were cultured with anti-CD3/CD28 for 4 days and IFNγ assessed by ELISA in tissue culture supernatant (a), intracellular IFNγ by flow cytometry (b), intracellular perforin by flow cytometry (c), all gated on CD8+ T cells. (d–i) PBMCs from human donors (>65 years) were cultured with anti-CD3/CD28 for 4 days and where indicated treated with 10 µM Hydroxychloroquine (HcQ), 10 µM BSI-0206965 (SbI), 10 µM Resveratrol (Res) and IFNγ assessed by ELISA in tissue culture supernatant (d, g), intracellular IFNγ by flow cytometry (e, h), intracellular perforin by flow cytometry (f, i), all gated on CD8+ cells. Data represented as mean ± SEM, MFI = mean fluorescence intensity. Statistics by paired t-test for (d–i).
-
Figure 2—source data 1
Autophagy is required for CD8+ T cell function.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig2-data1-v2.xlsx
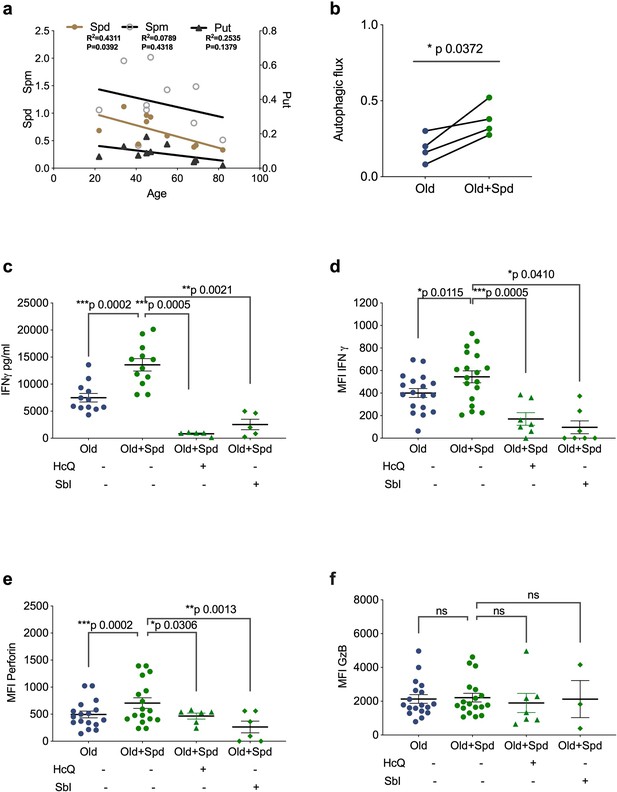
Spermidine declines with age and supplementing spermidine improves autophagy and CD8+ T cell function in old donors.
(a) Spermidine (Spd), spermine (Spm), and putrescine (Put) content of PBMCs collected from healthy donors were measured by GC-MS. Linear regression with 95% confidence intervals. The goodness of fit was assessed by R (Lurie et al., 2020). The p value of the slope is calculated by F test. (b–f) PBMCs from old human donors (>65 years) were cultured with anti-CD3/CD28 for 4 days and where indicated treated with 10 µM spermidine alone or with 10 µM Hydroxychloroquine (HcQ), 10 µM SBI-0206965 (SbI), and autophagic flux measured by flow cytometry (b), IFNγ assessed by ELISA in tissue culture supernatant (c), intracellular IFNγ by flow cytometry (d), intracellular perforin by flow cytometry (e), intracellular granzyme B (f), all gated on CD8+ cells. Data represented as mean ± SEM, MFI = mean fluorescence intensity. Statistics by paired t-test for (b–f).
-
Figure 3—source data 1
Spermidine declines with age and supplementing spermidine improves autophagy and CD8+ T cell function in old donors.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig3-data1-v2.xlsx

Spermine does not improve function of CD8+ T cell from old donors.
PBMCs from old human donors (>65 years) were cultured with anti-CD3/CD28 for 4 days and where indicated treated with 10 µM spermidine or 10 µM spermine and intracellular IFNγ assessed by flow cytometry (a), intracellular perforin by flow cytometry (b), intracellular granzyme B (d), all gated on CD8+ cells. Data represented as mean ± SEM, MFI = mean fluorescence intensity. Statistics by paired t-test for (a–c).
-
Figure 3—figure supplement 1—source data 1
Spermine does not improve function of CD8+ T cell from old donors.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig3-figsupp1-data1-v2.xlsx

Spermidine reduces mitochondrial mass in CD8+ T cell from old donors.
PBMCs from old human donors (>65 years) were cultured with anti-CD3/CD28 for 4 days and where indicated treated with 10 µM spermidine and quantified for mitochondrial mass by flow cytometry using MitoTracker Green (MTG) (a) or nonylacridine orange (NAO) (b). Mitochondrial membrane potential was assessed by flow cytometry using TMRM dye (c) and mitochondrial ROS (mtROS) by mitoSOX staining (d). All gated on CD8+ cells. Data represented as mean ± SEM, MFI = mean fluorescence intensity. MFI normalized to Old untreated group. Statistics by paired t-test for (a–d).
-
Figure 3—figure supplement 2—source data 1
Spermidine reduces mitochondrial mass in CD8+ T cell from old donors.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig3-figsupp2-data1-v2.xlsx
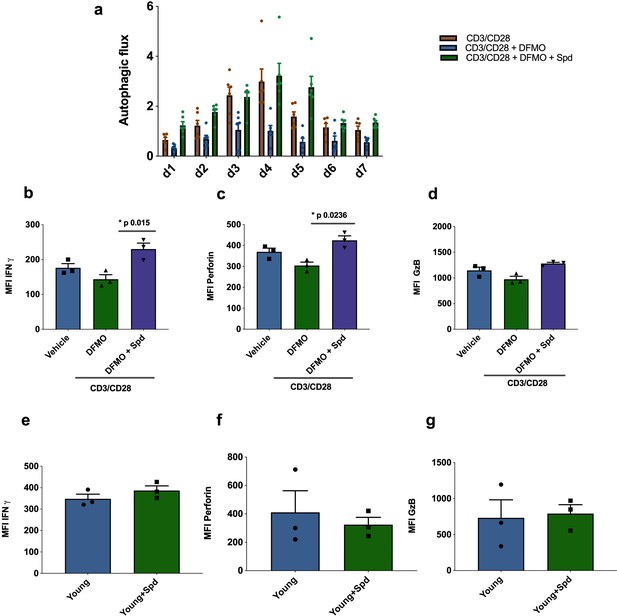
Endogenous spermidine maintains levels of autophagy and T cell function.
(a–d) PBMCs cells from young human donors (<65 years) were activated with anti-CD3/CD28 for 7 days and treated with spermidine synthesis inhibitor 1 mM DFMO alone or together with 10 µM spermidine (Spd). Autophagic flux (a) was assessed each day and IFNγ (b), Perforin (c), Granzyme B (d) were measured by flow cytometry in CD8+ cells on day 4. (e–f) PBMCs cells from young human donors (<65 years) were cultured with anti-CD3/CD28 for 4 days and streated with 10 µM spermidine. (e) Intracellular IFNγ, (f) intracellular perforin, (g) and intracellular granzyme B were measured in CD8+ cells by flow cytometry. Data represented as mean ± SEM.
-
Figure 4—source data 1
Endogenous spermidine maintains levels of autophagy and T cell function.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig4-data1-v2.xlsx
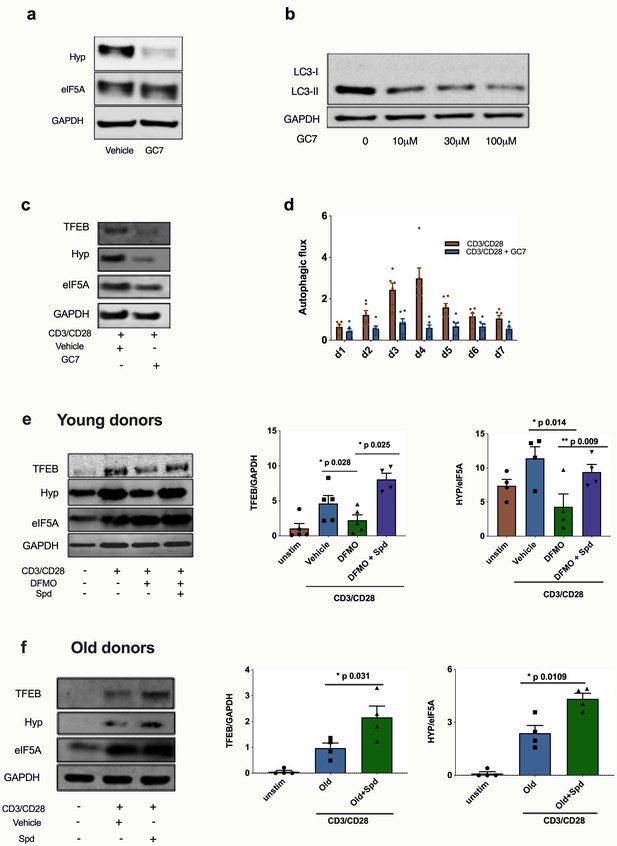
Spermidine’s mode of action is via eIF5A and TFEB in human CD8+ T cells.
(a–d) Human T cell line Jurkat was cultured for 24 hr with 100 µM GC7, then eIF5A and hypusinated eIF5A were measured by WB (a). Jurkat cell line was stimulated with increasing concentrations of GC7 and cell lysates blotted for LC3B (b). (c–d) PBMCs from young human donors were cultured with anti-CD3/CD28 for 7 days and treated with GC7. The protein levels of TFEB and eIF5A hypusination were measured in CD8+ cells by Western blot on day 4 (c) and autophagic flux was determined as in Figure 1 (d). PBMCs from young human donors were cultured with anti-CD3/CD28 for 4 days and treated with spermidine synthesis inhibitor DFMO alone or together with 10 µM spermidine and the protein levels of TFEB and eIF5A hypusination were measured in CD8+ cells by wWestern blot (e), representative images (left) and quantified (right). PBMCs from old human donors (>65 years) were cultured with anti-CD3/CD28 for 4 days and where indicated treated with 10 µM spermidine and the protein levels of TFEB and eIF5A hypusination were measured in CD8+ cells by wWestern blot (f), representative images (left) and quantified (right). Target band intensity was normalized to eIF5A (for Hyp) or GAPDH (for TFEB). Data represented as mean ± SEM.
-
Figure 5—source data 1
Dpermidine’s mode of action is via eIF5A and TFEB in human CD8+ T cells.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig5-data1-v2.xlsx

Spermidine does not improve eIF5A and TFEB in young donors.
PBMCs from young human donors were cultured with anti-CD3/CD28 for 4 days and treated with 10 µM spermidine for 4 days. The protein levels of TFEB and eIF5A hypusination were measured in sorted CD8+ cells by wWestern blot, representative images (left) and quantified (right). Target band intensity was normalized to eIF5A (for Hyp) or GAPDH (for TFEB). Data represented as mean ± SEM.
-
Figure 5—figure supplement 1—source data 1
Spermidine does not improve eIF5A and TFEB in young donors.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig5-figsupp1-data1-v2.xlsx
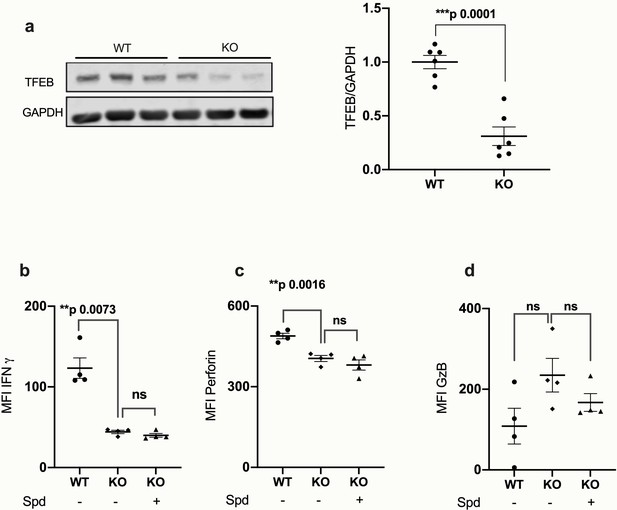
TFEB is required for CD8+ T cell function.
Splenic T cells from wildtype C57BL/6 mice (WT) or tamoxifen-inducible Tfeb knockout mice (KO: CAG-Cre;Esr1+;Tfeb-/-) were cultured with anti-CD3/CD28 and 4-Hydroxytamoxifen (4-OHT) for 4 days. The protein levels of TFEB and GAPDH were measured in sorted CD8+ cells by wWestern blot (a) representative images (left) and quantified (right). Target band intensity was normalized to GAPDH. Intracellular IFNγ by flow cytometry (b), intracellular perforin by flow cytometry (c), intracellular granzyme B (d). All cells were gated on CD8+ T cells. Data represented as mean ± SEM, MFI = mean fluorescence intensity. Statistics by unpaired t-test.
-
Figure 5—figure supplement 2—source data 1
TFEB is required for CD8+ T cell function.
- https://cdn.elifesciences.org/articles/57950/elife-57950-fig5-figsupp2-data1-v2.xlsx
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Genetic reagent (M. musculus) | Atg7flox | PMID:15866887 | MGI:3587769 | Dr. Masaaki Komatsu (Juntendo University) |
| Genetic reagent (M. musculus) | TFEBflox | PMID:23604321 | Prof. Andrea Ballabio (Telethon Institute of Genetics and Medicine (TIGEM)). | |
| Cell line (H. sapiens) | Jurkat E6.1 | ATCC | RRID:CVCL_0367 | |
| Antibody | Rabbit anti-GAPDH | Millipore | MAB374, RRID:AB_2107445 | WB (1:5,000) |
| Antibody | Rabbit anti-LC3 | Sigma | L8918, RRID:AB_1079382 | WB (1:1500) |
| Antibody | Mouse anti-eIFA5 | BD Biosciences | 611976, RRID:AB_399397 | WB (1:3000) |
| Antibody | Rabbit anti-hypusine | Millipore | ABS1064, RRID:AB_2631138 | WB (1:1500) |
| Antibody | Rabbit anti-TFEB | Bethyl | A303-673A, RID:AB_11204751 | WB (1:1500) |
| Antibody | Mouse anti-Actin | Cell Signaling | 3700, RRID:AB_2242334 | WB (1:10,000) |
| Antibody | IRDye 800CW Donkey Anti-Rabbit IgG (H+L) | LI-COR | 926–32213, RRID:AB_621848 | WB (1:10,000) |
| Antibody | IRDye 680RD Donkey Anti-Mouse IgG (H+L) | LI-COR | 926–68022, RRID:AB_10715072 | WB (1:10,000) |
| Antibody | BV605 anti-human CD14 | BioLegend | 301834, Clone: M5E2 | FACS (1:200) |
| Antibody | PE anti-human Granzyme B | eBioscience | 12-8899-41, Clone: GB11 | FACS (1:100) |
| Antibody | PE/Cy7 anti-human CD8a | BioLegend | 344712, Clone: SK1 | FACS (1:100) |
| Antibody | PE/Cy5 anti-human CD19 | BioLegend | 302210, Clone:HIB19 | FACS (1:100) |
| Antibody | BV711anti-human CD19 | BioLegend | 302245, Clone:HIB19 | FACS (1:200) |
| Antibody | APC anti-human CD3 | BioLegend | 300312, Clone:HIT3a | FACS (1:200) |
| Antibody | APC anti-human Perforin | BioLegend | 353311, Clone:B-D48 | FACS (1:100) |
| Antibody | APC anti-human CD4 | BioLegend | 317416, Clone:OKT4 | FACS (1:200) |
| Antibody | Alexa Fluor 700 anti-human IFNγ | BioLegend | 506515, Clone:B27 | FACS (1:100) |
| Antibody | PE/Cy7 anti-mouse CD8a Antibody | BioLegend | 100722, RRID:AB_312761 | FACS (1:200) |
| Antibody | BV605 anti-mouse CD4 Antibody | BioLegend | 100451, Clone: GK1.5 | FACS (1:200) |
| Antibody | Alexa Fluor 700 anti-mouse IFNγ | BioLegend | 505823, Clone: XMG1.2 | FACS (1:100) |
| Antibody | APC anti-mouse Perforin | BioLegend | 154303, Clone: S16009A | FACS (1:100) |
| Antibody | PE anti-mouse Granzyme B | BioLegend | 372207, Clone: QA16A02 | FACS (1:100) |
| Antibody | APC anti-human CD19 | BioLegend | 302212, RRID:AB_314242 | FACS (1:200) |
| Antibody | Alexa Fluor 700 anti-mouse CD8a | BioLegend | 100730, RRID:AB_493703 | FACS (1:100) |
| Antibody | PE-labeled MHC class I pentamer (HLA-A*02:01, HCV peptide KLSGLGINAV) | ProImmune | (HLA-A*02:01, HCV peptide KLSGLGINAV) | FACS (1:50) |
| Commercial assay or kit | FlowCellect Autophagy LC3 Antibody-based Detection Kit | Merck Millipore | FCCH100171 | FACS (1:20) |
| Commercial assay or kit | LIVE/DEAD Fixable Near-IR Dead Cell Stain Kit | Invitrogen ThermoFisher | L10119 | FACS (1:1000) |
| Commercial assay or kit | LIVE/DEAD Fixable Aqua Dead Cell Stain Kit | Invitrogen ThermoFisher | L34957 | FACS (1:1000) |
| Commercial assay or kit | CellTrace Violet Cell Proliferation Kit | Invitrogen,ThermoFisher | C34557 | FACS (1:1000) |
| Commercial assay or kit | MitoTracker Green | Invitrogen,ThermoFisher | M7514 | FACS (150 nM) |
| Commercial assay or kit | MitoSox | Invitrogen,ThermoFisher | M36008 | FACS (5 uM) |
| Commercial assay or kit | CD3/CD28 activation Dynabeads | Thermo Fisher | 11161D | |
| Commercial assay or kit | Human IFN-γ ELISpotPLUS kit (ALP) | Mabtech | 3420-4APT-2 | |
| Commercial assay or kit | IFNγ ELISA Kit | Life Technologies Ltd | 88-7316-22 | |
| Commercial assay or kit | EasySep Human CD8+ T Cell Isolation Kit | Stemcell | 17953 | |
| Commercial assay or kit | BCA Assay | Thermo Fisher | 23227 | 100 μl/sample |
| Chemical compound, drug | Spermidine | Cayman Chemical | 14918 | 10 μM |
| Chemical compound, drug | Spermine | Cayman Chemical | 18041 | 10 μM |
| Chemical compound, drug | Hydroxychloroquine Sulfate | Stratech Scientific Ltd | B4874-APE-10mM | 10 μM |
| Chemical compound, drug | Bafilomycin A1 | Cayman Chemical | 11038 | 10 μM |
| Chemical compound, drug | Resveratrol | Stratech Scientific Ltd | A4182-APE | 10 μM |
| Chemical compound, drug | BSI-0206965 | Stratech Scientific Ltd | A8715-APE | 10 μM |
| Chemical compound, drug | AICR | Stratech Scientific Ltd | A8184-APE | 10 μM |
| Chemical compound, drug | NAO | Thermo Fisher Scientific | A1372 | 100 nM |
| Chemical compound, drug | TRMR | Thermo Fisher Scientific | T668 | FACS (100 nM) |
| Chemical compound, drug | Difluoromethylornithine (DFMO) | Enzo Life Sciences | ALX-270–283 M010 | 1 mM |
| Chemical compound, drug | GC7 | Millipore | 259545–10 MG | 10 μM |
| Chemical compound, drug | 4-Hydroxytamoxifen | Sigma | H7904 | 100 nM |
| Chemical compound, drug | LPS | Santa Cruz | sc-3535 | 10 µg/ml |
| Chemical compound, drug | Aanti-IgM | Jackson Immuno Research | 109-005-043 | 5 µg/ml |
| Chemical compound, drug | Aanti-CD40L | Enzo Life science | ALX-522–015 C010 | 100 ng/ml, |
| Peptide, recombinant protein | Human IFNγ | Enzo Life science | ENZ-PRT141-0100 | 20 ng/ml |
| Software, algorithm | Image Studio Lite | LI-COR | ||
| Software, algorithm | Prism | GraphPad |





