Disynaptic cerebrocerebellar pathways originating from multiple functionally distinct cortical areas
Figures
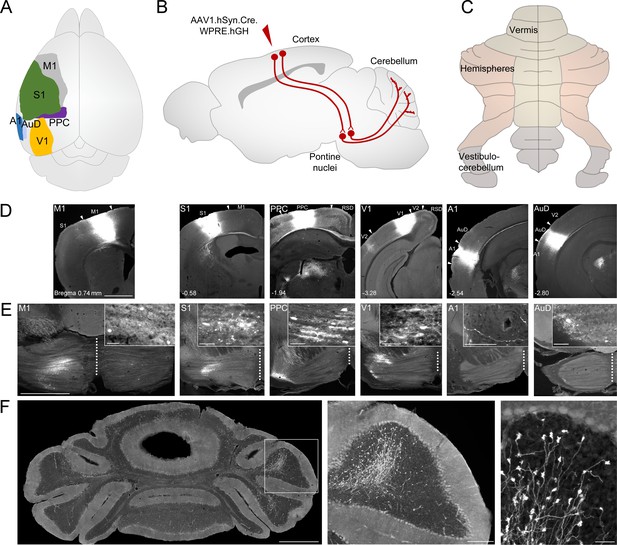
Anterograde tracing of indirect cerebrocerebellar pathways using a mono-trans-synaptic adeno-associated virus (AAV).
(A) Schematic outlining cortical target areas for mono-trans-synaptic anterograde tracer injections: primary motor (M1), primary somatosensory (S1), posterior parietal association cortex (PPC), primary visual (V1), primary auditory (A1), and dorsal auditory (AuD) cortex. (B) Principle of mono-trans-synaptic anterograde tracing (e.g cortico-pontine-cerebellar pathway) using a specific adeno-associated virus (AAV1.cre). (C) Schematic of gross anatomical divisions of the unfolded mouse cerebellum (according to Marani and Voogd, 1979). (D) Images of coronal sections showing representative injection sites into M1, S1, PPC, V1, A1, and AuD (from left to right). Arrowheads indicate regional borders. Distance from bregma is indicated based on the mouse stereotaxic atlas (Franklin and Paxinos, 2007). (E) Images of coronal sections illustrating the mono-trans-synaptic labeling in the pontine nuclei (i.e. corticopontine fibers, postsynaptic pontine cells, pontine fibers) following injections into M1, S1, PPC, V1, A1, and AuD. Note that the medial-lateral topography of pontine labeling correlates with the rostral-caudal localization of these cortical regions. Injections into A1 resulted in labeled fibers within the pons, but no labeled cells. (F) Images of coronal section showing representative mossy fiber labeling following an injection into M1 at different magnifications. Scale bars 1 mm (D, E, F left), 20 µm (insets in E), 200 µm (F middle), 50 µm (F right). Retrosplenial Cortex (RSD); secondary visual cortex (V2).
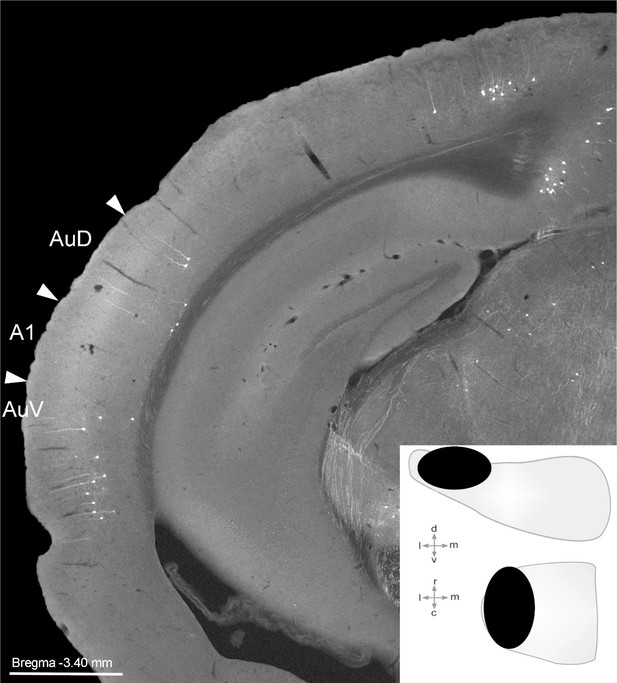
Retrogradely labelled cells following injection in pontine nuclei.
Image of coronal section after injection of retrograde virus (CAV.cre) in the pontine nuclei (injection location shown in schematic inset). Image shows representative labeling in the ventral (AuV) and dorsal (AuD) auditory fields and the lack of labeling in primary auditory cortex (A1). Distance from bregma is indicated according to the mouse stereotaxic atlas (Franklin and Paxinos, 2007). Scale bar 1 mm.
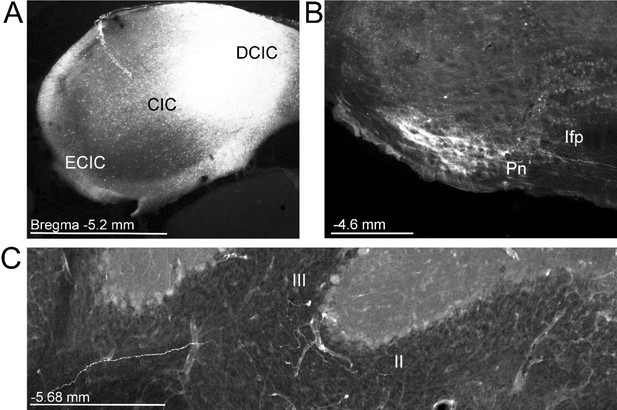
Resulting anterograde labeling in the pontine nuclei and mossy fiber terminals following injection of mono-trans-synaptic adeno-associated virus (AAV) into the inferior colliculus.
(A) Coronal section shows the injection site of AAV1.cre centered in the dorsal cortex of the inferior colliculus (DCIC). (B) Mono-trans-synaptic anterograde labeling in the pontine nuclei (Pn) and (C) labeled mossy fiber terminals in lobule II and III in the cerebellar cortex. For all, distance from bregma is indicated based on the mouse stereotaxic atlas (Franklin and Paxinos, 2007). Scale bar 1 mm (A), 500 µm (B, C). Central inferior colliculus (CIC), external cortex of the inferior colliculus (ECIC), longitudinal fasciculus of the pons (lfp).
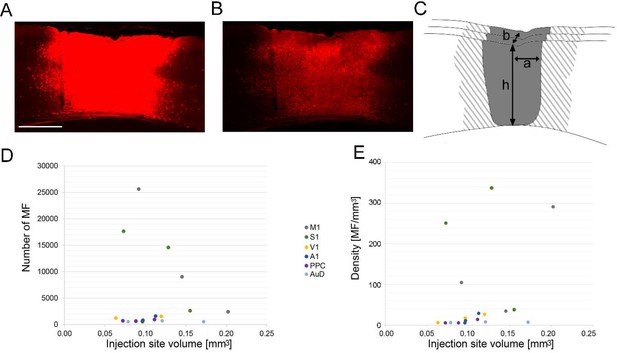
Injection site quantification method and relationship between the injection site volume and the resulting mossy fiber labeling in the cerebellar cortex.
Images of coronal sections showing a representative injection site with high (A) and low (B) exposure time. (C) Schematic of the core (solid grey) and halo (grey striped) designations of the injection site are shown. For injection site volume calculations (), low-exposure images such as (B) were used to measure h, a and b, where h corresponds to the maximal extent of the injection site across the cortical layers seen in all sections (dorsoventral direction), a corresponds to the radius of the maximal extent of the injection site parallel to the cortical layers seen in all sections (mediolateral direction), and b corresponds to the radius of the maximal extent of the injection site across coronal sections (rostrocaudal direction). Scale bar 500 µm (applies to A and B). (D) Correlation between the number of mossy fibers (MF) and the injections volume for each case (R = −0.103, p=0.714, n = 18, Pearson’s correlation). (E) Correlation between the density of MF terminals in the contralateral cerebellum and the injections volume for each case (R = 0.314, p=0.204, n = 18, Pearson’s correlation). Cortical target location for injection sites are indicated: primary motor (M1), primary somatosensory (S1), posterior parietal association (PPC), primary visual (V1), primary auditory (A1), or dorsal auditory (AuD) cortex.
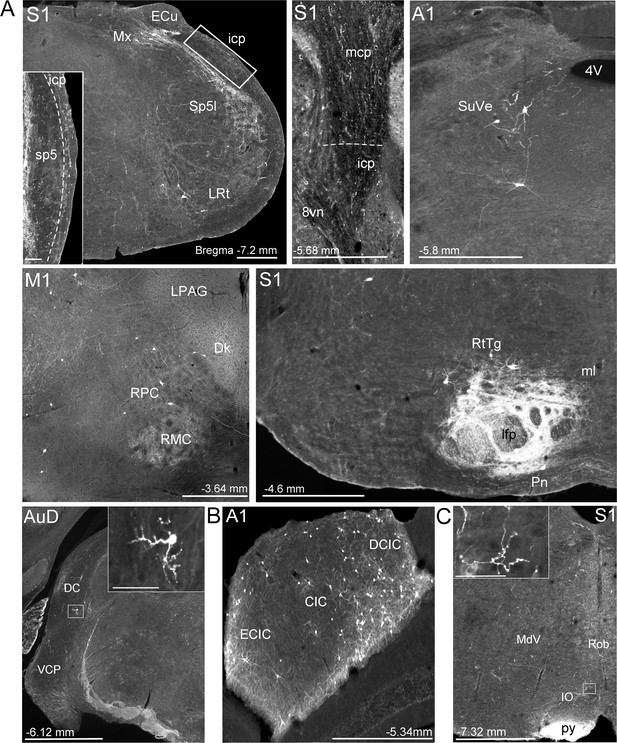
Extra-pontine labeling in key precerebellar nuclei following mono-trans-synaptic adeno-associated virus (AAV) injections in target cerebral cortical regions.
(A) Representative labeling in precerebellar nuclei listed in Supplementary file 1B, after injections of AAV1.cre into primary motor (M1), primary somatosensory (S1), primary auditory (A1), and dorsal auditory (AuD) cortex. Images show labeled cells in interpolar part of the spinal trigeminal nucleus (Sp5I), lateral reticular nucleus (LRt), matrix region x (Mx), superior vestibular nucleus (SuVe), red nucleus (RPC, RMC), pontine reticulotegmental nucleus (RtTg), dorsal cochlear nucleus (DC) and ventral cochlear nucleus, posterior part (VCP), which provide alternative extra-pontine mossy fiber pathways for indirect cortical input to the cerebellum. Descending labeled fiber tracts can also be observed in the longitudinal fasciculus of the pons (lfp) and the pyramidal tracts (py). Further, fibers travelling to the cerebellar cortex in the middle and inferior cerebellar peduncles (mcp and icp, respectively) after S1 injections. (B) Labeled cells in the inferior colliculus (central [CIC], external cortex [ECIC] and dorsal cortex [DCIC] inferior colliculus) following A1 injections. (C) Labeled fibers in the ipsilateral inferior olive (IO) following S1 injections. For all, distance from bregma is indicated based on the mouse stereotaxic atlas (Franklin and Paxinos, 2007). Scale Bars 500 µm (A), 1 mm (B, C), 100 µm all insets. 4th ventricle (4V), vestibular nerve (8vn), nucleus of Darkschewitsch (Dk), lateral periaqueductal gray (LPAG), medial lemniscus (ml), medullary reticular nucleus, ventral (MdV), raphe obscurus nucleus (Rob).

Organization of indirect cerebrocerebellar mossy fiber terminals in the cerebellum from target cerebral cortical regions.
(A) Schematic of cortical target areas as outlined on the cortical surface (according to Franklin and Paxinos, 2007; Kirkcaldie, 2012) including core (black) and halo (grey) regions of mono-trans-synaptic anterograde tracer injections: primary motor (M1), primary somatosensory (S1), posterior parietal association cortex (PPC), primary visual (V1), primary auditory (A1), and dorsal auditory (AuD) cortex. Scale bar is 1 mm. (B) Schematic illustrating the topographical location of pontine labeling following injections. Colored lines indicate regions where labeled pontine neurons were found and dots (following A1 injections) indicate regions where terminals were observed. (C) Percentage of mossy fiber labeling in the ipsilateral and contralateral cerebellum (left) and in its gross divisions (right): vermis, hemispheres and vestibulocerebellum (VbC). (D) Percentage of labeled mossy fiber terminals observed in the anatomical lobules (simplex lobule [Sim], crus I/II ansiform lobule [crus I/II], paramedian lobule [PM], copula pyramis [Cop], paraflocculus [PFI], flocculus [FL], vermal cerebellar lobules [I-X]) of the ipsilateral (left) and contralateral (right) cerebellum (% of the total cerebellar input).
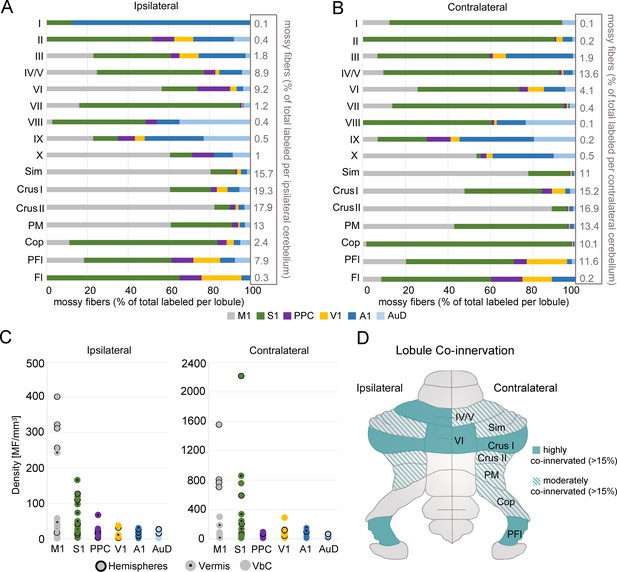
Regional convergence of cerebrocerebellar terminals originating across functionally distinct regions of cerebral cortex.
(A–B) Proportion of labeled mossy fiber terminals in the ipsilateral (A) and contralateral (B) cerebellum following AAV injections into primary motor (M1), primary somatosensory (S1), posterior parietal association cortex (PPC), primary visual (V1), primary auditory (A1), or dorsal auditory (AuD) cortex. Values were normalized to the total number of labeled mossy fibers found within each lobule (simplex lobule [Sim], crus I/II ansiform lobule [crus I/II], paramedian lobule [PM], copula pyramis [Cop], paraflocculus [PFI], flocculus [FL], vermal lobules [I-X]) across all animals (right x-axis). (C) Density of mossy fiber (MF) terminals in the ipsilateral (left) and contralateral (right) cerebellum. Each dot represents the average density (number of MFs/mm3) after each cortical injection M1, S1, PPC, V1, A1 and AuD. See also Supplementary file 1C. (D) Schematic of the unfolded mouse cerebellum summarizing the topography of mossy fiber inputs. Multimodal lobules receive at least 4% of the total mossy fiber input for that side (see A-B) and are highly (solid color) or moderately (striped color) multimodal based on input from at least two different functional cortical regions in addition to the dominant one (>15% and<15%, respectively).
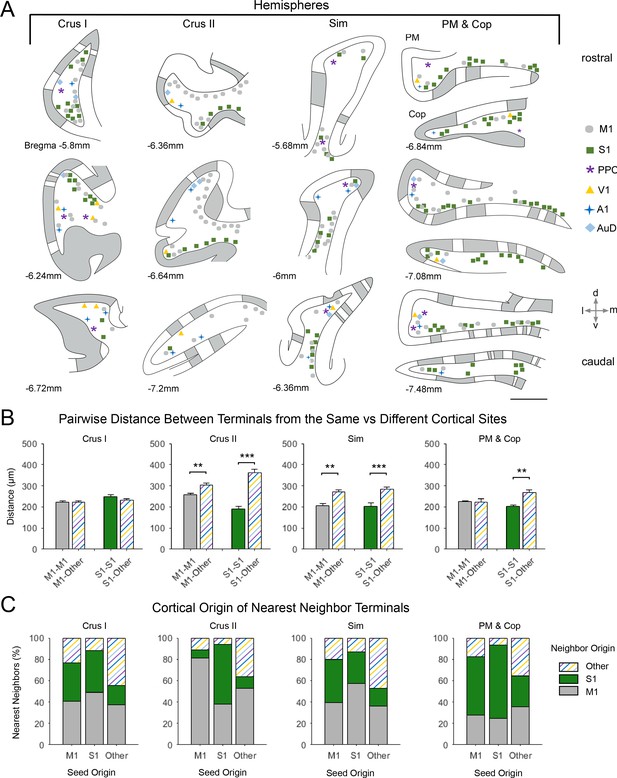
Spatial distribution of mossy fiber terminals within divisions of the cerebellar hemispheres with multimodal regional convergence.
(A) Schematic reconstruction of the parasagittal distribution of labeled mossy fiber terminals in regions of the contralateral cerebellar hemispheres, after injections of mono-trans-synaptic AAV into target cortical regions: primary motor (M1), primary somatosensory (S1), posterior parietal association cortex (PPC), primary visual (V1), primary auditory (A1), or dorsal auditory (AuD) cortex. Data taken from 120 μm thick representative sections (three coronal sections) for rostral (top) to caudal (bottom) regions of the indicated lobules; distance from bregma is indicated based on the mouse stereotaxic atlas (Franklin and Paxinos, 2007). Data were aligned across animals using the parasagittal pattern of Purkinje cell molecular marker aldolase C (grey represents high expression, AldoC+; white represents low expression, AldoC-; see also Figure 5—figure supplement 1). For high density projections, M1 and S1, each dot represents 5–10 mossy fiber terminals, for all other lower density projections each dot represents 1–5 mossy fiber terminals. Simplex lobule (Sim), crus I/II ansiform lobule (crus I/II), paramedian lobule (PM), and copula pyramis (Cop). (B) Average pairwise distance between all terminals originating from the same cortical injection site (e.g. M1–M1) or from different cortical injection sites (e.g. M1-Other, where ‘Other’ is represented by all injected sites except M1 and S1). Statistical significance represents terminals being closer to like-terminals than to terminals from other cortical regions (**p<0.01, ***p<0.001, for exact p-values see Supplementary file 1E). (C) Proportion of the five nearest neighbors of each terminal location following each cortical injection site (i.e. seed origin: M1, S1 and Other; indicated on x-axis) that originate from M1, S1 or other cortical regions (i.e. neighbor origin: indicated on legend).
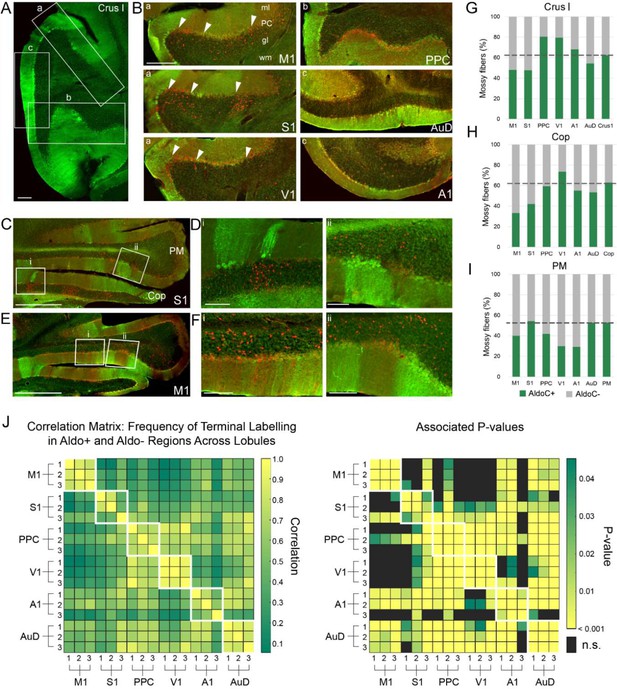
Parasagittal distribution of mossy fiber terminals and aldolase c (AldoC) expression.
(A) Image of AldoC immunostaining (green) of cerebellar crus I, illustrating AldoC+ and AldoC- stripes. (B–F). Images of coronal sections of crus I (B), paramedian lobule (PM) and copula pyramis (Cop; C–F), illustrating the distribution of mossy fiber terminals (red, in the granule cell layer [gl]) following AAV1.cre injections into various cortical regions indicated on images (primary motor [M1], primary somatosensory [S1], posterior parietal association cortex [PPC], primary visual [V1], primary auditory [A1], and dorsal auditory [AuD] cortex) in relation to the parasagittal expression pattern of AldoC (green). Note that red fluorescence in the Purkinje cell layer (PC) results from background fluorescence from the Ai9 mouse line, not AAV1.cre-induced tdTomato expression (see Materials and methods). (G–I) The overall mossy fiber distribution corresponding to AldoC+ (green) or AldoC- (grey) compartments in crus I, Cop and PM, respectively, from the different cortical targets as well as the baseline proportion of AldoC+ or AldoC- compartments in each lobule (dashed line; right column); indicating the likelihood of mossy fiber terminals to be found more or less often in AldoC+ compartments (green above or below dashed line, respectively). (J) The frequency of mossy fiber terminals in AldoC+ and AldoC- regions of each lobule were correlated across animals (Pearson’s correlation: R values, left; p-values, right). Significant correlations between animals with the same cortical injection sites (outlined white boxes; e.g. M1- animal 1, 2, and 3) provide validation that AldoC alignment is consistent across animals. Scale bars 100 µm (A, B), 500 µm (C, E), 50 µm (D, F).
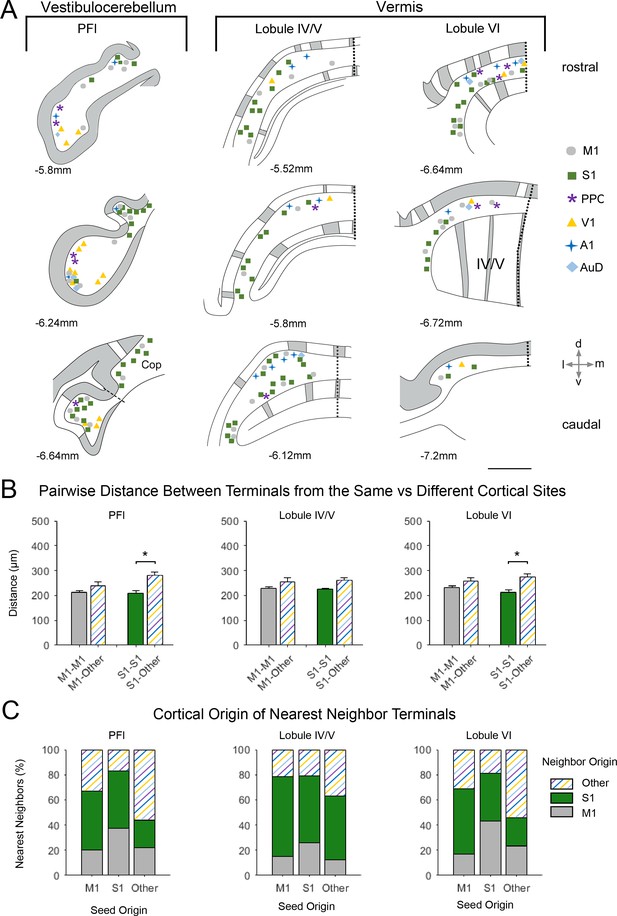
Spatial distribution of mossy fiber terminals within divisions of the vestibulocerebellum and vermis with multimodal regional convergence.
(A) Schematic reconstruction of the parasagittal distribution of labeled mossy fiber terminals in contralateral cerebellar lobules spanning the vestibulocerebellum (VbC) and the vermis, after injections of mono-trans-synaptic AAV into target cortical regions: primary motor (M1), primary somatosensory (S1), posterior parietal association cortex (PPC), primary visual (V1), primary auditory (A1), or dorsal auditory (AuD) cortex. Data taken from 120 μm thick representative sections (three coronal sections) for rostral (top) to caudal (bottom) regions of the indicated lobules; distance from bregma is indicated based on the mouse stereotaxic atlas (Franklin and Paxinos, 2007). Data were aligned across animals using the parasagittal pattern of Purkinje cell molecular marker aldolase C (grey represents high expression, AldoC+; white represents low expression, AldoC-; see also Figure 5—figure supplement 1). For high density projections, M1 and S1, each dot represents 5–10 mossy fiber terminals, for all other lower density projections each dot represents 1–5 mossy fiber terminals. Dotted lines represent midline and dashed line represents division between paraflocculus (PFl) and copula pyramis (Cop). Vermal cerebellar lobules: lobule IV/V and lobule IV). (B) Average pairwise distance between all terminals originating from the same cortical injection site (e.g. M1–M1) or from different cortical injection sites (e.g. M1-Other, where ‘Other’ is represented by all injected sites except M1 and S1). Statistical significance represents terminals being closer to like-terminals than to terminals from other cortical regions (*p<0.05, for exact p-values see Supplementary file 1E). (C) Proportion of the five nearest neighbors of each terminal location following each cortical injection site (i.e. seed origin: M1, S1 and Other; indicated on x-axis) that originate from M1, S1 or other cortical regions (i.e. neighbor origin: indicated on legend).
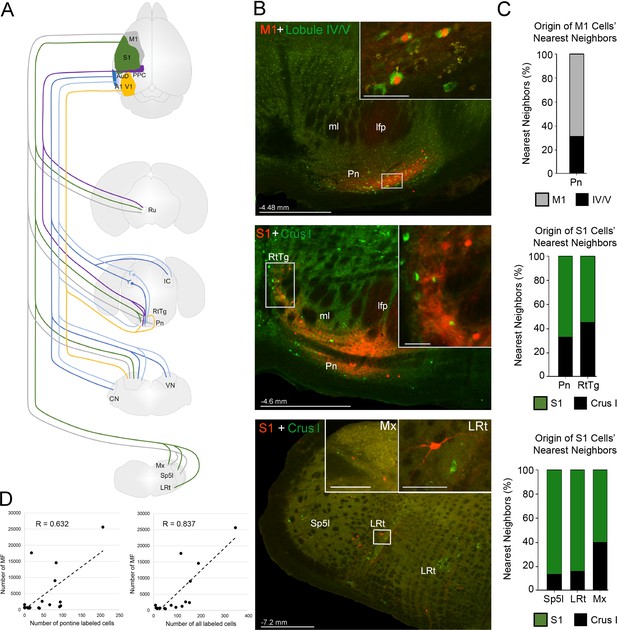
Summary of intermediate brainstem nuclei potentially supporting disynaptic cerebrocerebellar pathways.
(A) Schematic summary of pathways to precerebellar nuclei from target regions of cerebral cortex: primary motor (M1), primary somatosensory (S1), posterior parietal association cortex (PPC), primary visual (V1), primary auditory (A1), or dorsal auditory (AuD) cortex. (B) Representative labeling in pontine (top) and precerebellar nuclei (middle, bottom) after combined injections of AAV1.cre (red) into cortical regions and CTB (green) into cerebellar regions (M1 + lobule lV/V; S1 + Crus I). Note the double labeled cells within the pontine nuclei (Pn; total 7 double labelled/52 anterogradely labeled cells from M1), and spatially overlapping cells within the reticulotegmental nucleus (RtTg) and lateral reticular nucleus (LRt). Distance from bregma is indicated based on the mouse stereotaxic atlas (Franklin and Paxinos, 2007). Scale bars 500 µm and 50 µm for insets. (C) Proportion of the five nearest neighboring cells for the cortical injection sites in (B) that are anterogradely labeled cells originating from the same cortical injection site (e.g. M1) or retrogradely labeled cells originating from injections in the cerebellar cortex (e.g. lobule IV/V; indicated by legend) within the indicated precerebellar nuclei. (D) Correlation of anterogradely labeled precerebellar cells with the total number of mossy fiber terminals per animal for either pontine only cells (left; R = 0.632, p=0.012, n = 18, Pearson’s correlation) or pontine plus extra-pontine precerebellar cells (right; R = 0.837, p<0.001, n = 18, Pearson’s correlation). Cochlear nuclei (CN), longitudinal fasciculus of the pons (lfp), matrix region x (Mx), medial lemniscus (ml), interpolar part of the spinal trigeminal nucleus (Sp5I), red nucleus (Ru), and vestibular nuclei (VN).
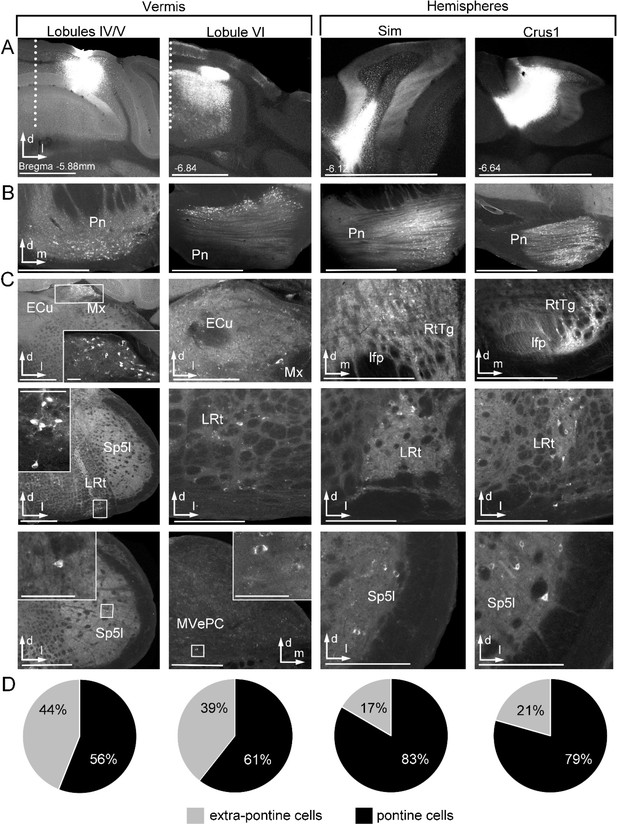
Retrograde tracing of cerebellar pathways from cerebral cortical receiving precerebellar target nuclei.
(A) Coronal sections show representative injection sites after injection of cholera-toxin-B (CTB) into vermal lobules IV/V, VI and cerebellar hemispheres simplex lobule (Sim) and crus I ansiform lobule (crus I). Dotted line represents midline. Distance from bregma is indicated based on the mouse stereotaxic atlas (Franklin and Paxinos, 2007). (B) Resulting retrograde labeling from the above corresponding injections in the pontine nuclei (Pn). (C) Representative retrograde labeling in precerebellar target nuclei (in the lateral reticular nucleus (LRt), matrix region x (Mx), the reticulotegmental nucleus (RtTg), spinal trigeminal nucleus (Sp5I) and the medial vestibular nucleus (MVePC)) from the corresponding injections above. (D) Percentage of retrograde labeled cells found in pontine (black) versus extra-pontine (grey) precerebellar nuclei (total cells in pontine: lobules IV/V = 825; lobule VI = 1011; sim = 1978; crus I = 2615; total cells in extra-pontine: lobules IV/V = 648; lobule VI = 659; sim = 393; crus I = 678); all cell counts are from n = 3 mice for each injected cerebellar region. Scale bars 1 mm (A); 500 µm (B; C first column); 250 µm (C); inset 50 µm (C); external cuneatus (ECu), longitudinal fasciculus of the pons (Ifp).
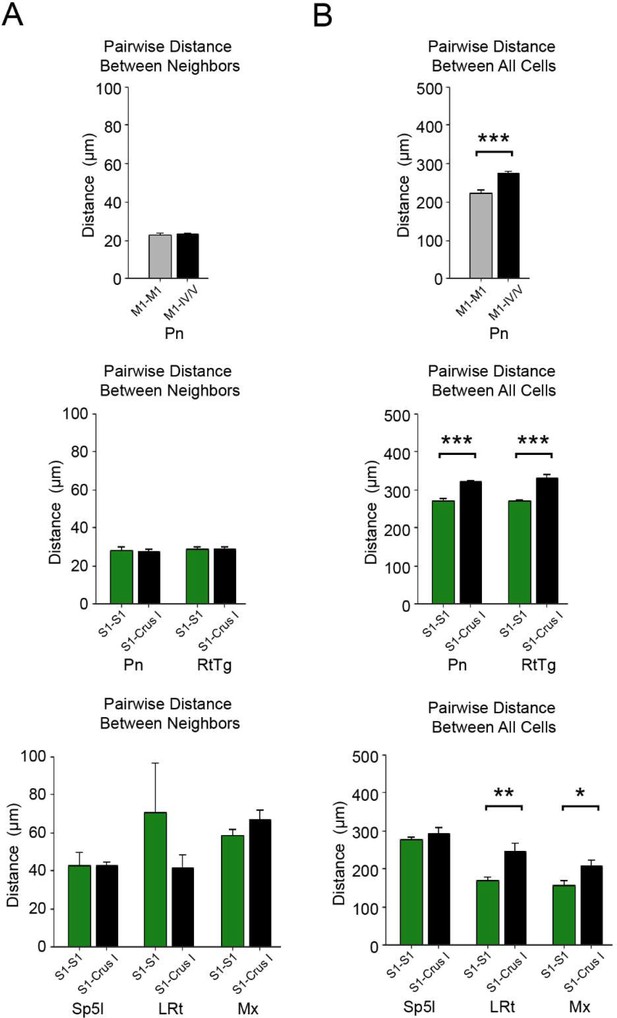
Pairwise distance between anterogradely and retrogradely labeled cells within pontine (Pn) and extra-pontine precerebellar nuclei.
(A) Average pairwise distance between the five nearest neighbors originating from anterogradely labeled cells from the same cortical injection site (e.g. M1–M1) or between anterogradely (from cerebral cortex) and retrogradely (from cerebellar cortex) labeled cells (e.g. M1-IV/V), within specified pre-cerebellar nuclei. No statistically significant differences were found in relation to the average difference between categories (for exact p-values see Supplementary file 1E), i.e., anterogradely labeled cells from the cerebral cortex where the same distance away from their own nearest neighbors as they were to their nearest neighbors that were retrogradely labeled cells from the cerebellum (B) Average pairwise distance between all anterogradely labeled cells originating from the same cortical injection site (e.g. M1–M1) or between all anterogradely (from cerebral cortex) and retrogradely (from cerebellar cortex) labeled cells (e.g. M1-IV/V) within specified pre-cerebellar nuclei. Statistical significance represents cells being closer to other anterogradely labeled cells from the same cortical injections site as themselves, compared to retrogradely labeled cells from cerebellar injection sites (*p<0.05, **p<0.01, ***p<0.001, for exact p-values see Supplementary file 1E). Primary motor cortex (M1), primary somatosensory cortex (S1), reticulotegmental nucleus (RtTg), interpolar part of the spinal trigeminal nucleus (Sp5I), matrix region x (Mx), lateral reticular nucleus (LRt).
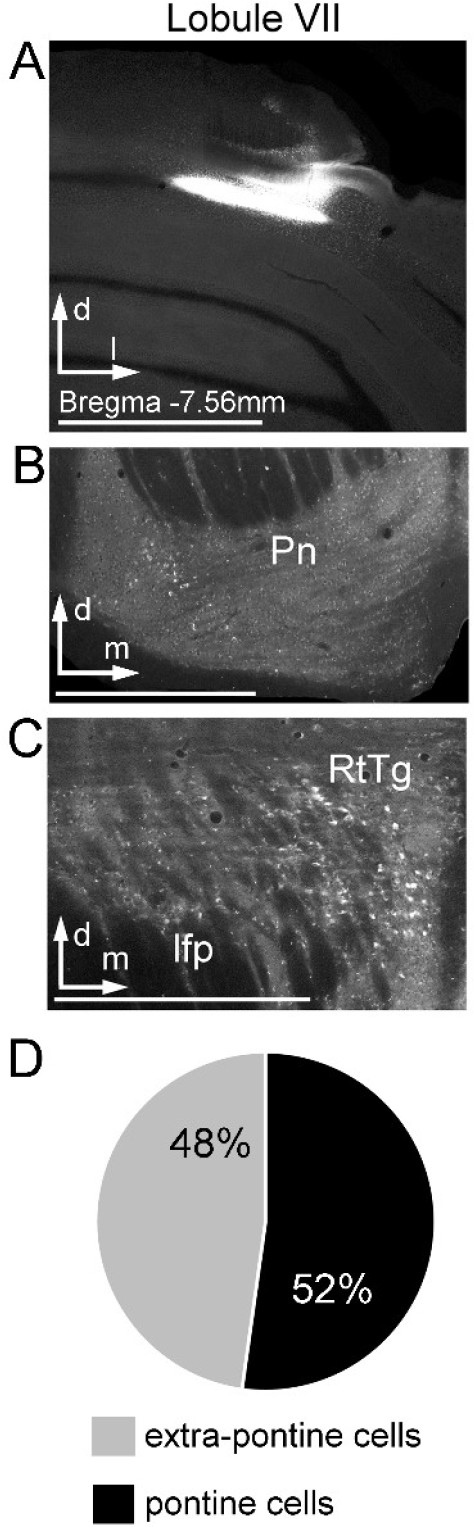
Retrograde tracing of cerebellar pathways to lobule VII from cerebral cortical receiving precerebellar target nuclei.
(A) Coronal section showing representative injection site for retrograde tracer cholera-toxin-B (CTB) into vermal lobule VII. Distance from bregma is indicated based on the mouse stereotaxic atlas (Franklin and Paxinos, 2007). (B) Resulting retrograde labeling in the pontine nuclei (Pn) after injection in lobule VII. (C) Representative retrograde labeling in precerebellar target nuclei in the reticulotegmental nucleus (RtTG; total cells = 127; n = 2 mice), which was the only precerebellar target region with a substantial number of retrogradely labelled cells. (D) Percentage of retrograde labeled cells found in the pontine nuclei (total cells = 153; n = 2 mice) and all precerebellar extra-pontine nuclei (total cells = 140; n = 2 mice). Scale bars 1 mm (A); 500 µm (B; C first column); 250 µm (C).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background Mus musculus | Strain: Ai9; Rosa26-CAG-LSL-tdTomato Cre reporter mice line Background:C57BL/6J | The Jackson Laboratory | RRID:IMSR_JAX:007909 | male and female |
| Recombinant DNA reagent | AAV1.hSyn.Cre.WPRE.hGH, | Addgene, | RRID:Addgene_105553 | titer 3.0 × 10 13 GC/ml |
| Recombinant DNA reagent | CAV.cre | PVM IGMM Montpellier, | titer 5.5 × 10 12 GC/ml | |
| Peptide, recombinant protein | Cholera-toxin-B 488 | ThermoFisher Scientific | Cat no. C34775 | |
| Antibody | anti-Aldolase C (H-11) (mouse monoclonal) | Santa Cruz Biotechnoloy Host species: mouse | RRID:AB_10659113 | 1:100 |
| Antibody | anti-RFP (polyclonal) | Rockland Host species: rabbit | RRID:AB_2209751 | 1:2000 |
| Antibody | Anti-Mouse IgG (H+L) Secondary, Alexa Fluor 488 Conjugated (polyclonal) | ThermoFisher Scientific Host species: donkey | RRID:AB_141607 | 1:200 |
| Antibody | Cy3-AffiniPure Goat Anti-Rabbit IgG (H+L) (polyclonal) | Jackson Immuno-Research Host species: goat | RRID:AB_2338006 | 1:200 |
| Software, algorithm | MATLAB 2013/2017a | Mathworks | RRID:SCR_001622 | |
| Software, algorithm | ImageJ (Fiji) | NIH – public domain | http://fiji.sc; RRID:SCR_002285 |
Additional files
-
Supplementary file 1
Associated data quantification and Tables.
(A) Table listing the location and size of mono-trans-synaptic adeno-associated virus (AAV1.cre) injection sites per animal. (B) Table showing the quantification of resulting anterograde labeling in precerebellar nuclei following injection of AAV1.cre into target cortical regions. (C) Table showing the density of mossy fibers within each cerebellar region following injection of AAV1.cre into target cortical regions. (D) Table showing the quantification of resulting retrograde labeling following injection of Cholera-toxin B retrograde tracer into target cerebellar regions. (E) Exact p-values associated with Figure 5, Figure 6, and Figure 7—figure supplement 3.
- https://cdn.elifesciences.org/articles/59148/elife-59148-supp1-v1.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/59148/elife-59148-transrepform-v1.pdf





