A gradient of Wnt activity positions the neurosensory domains of the inner ear
Figures
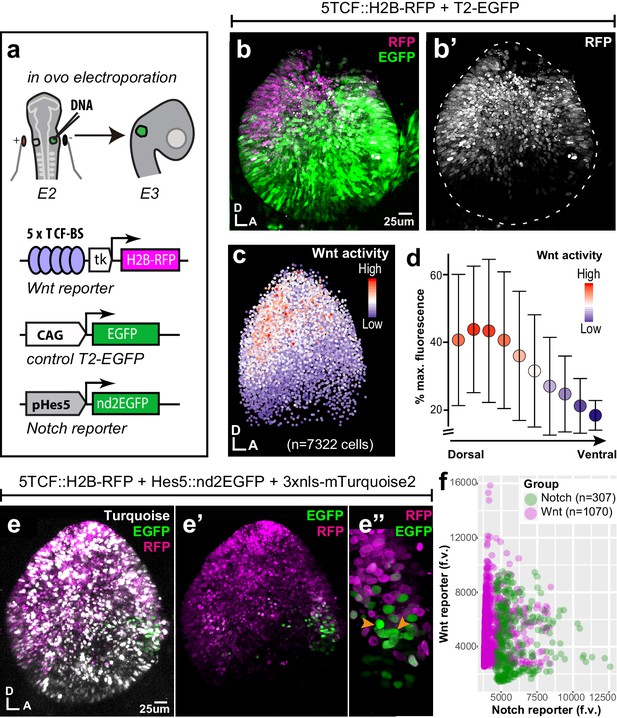
Spatial pattern of Wnt activity in the E3 chicken otocyst.
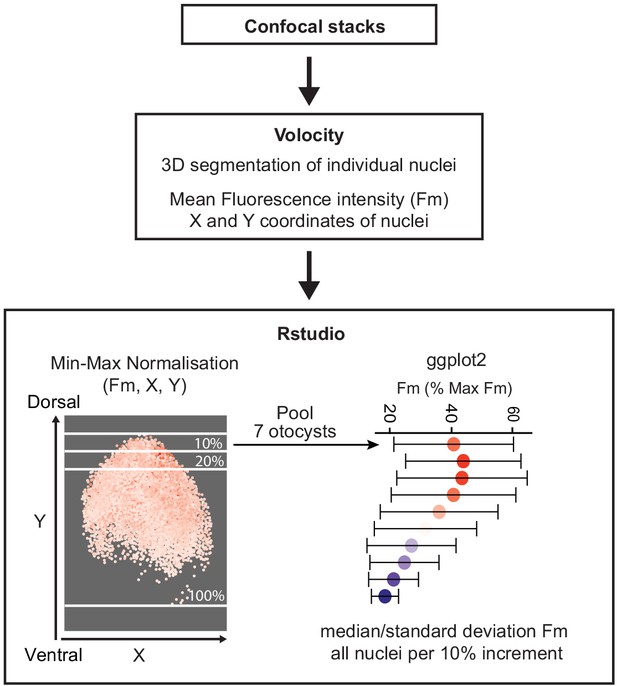
In all panels, dorsal (D) is up and anterior (A) is right. (a) E2 chicken embryos were co-electroporated either with Wnt reporter and a control plasmid T2-EGFP or Wnt reporter together with a Notch reporter and collected at E3. The Wnt reporter (5TCF::H2B-RFP) contains 5 TCF/LEF binding sites regulating an H2B-RFP fusion protein. In the Notch reporter (T2-Hes5::nd2EGFP), the mouse Hes5 promoter regulates expression of a nuclear destabilised EGFP. The control vector drives constitutive expression of EGFP. (b–b') Whole-mount view of an E3 otocyst electroporated with the Wnt reporter and a control plasmid. Wnt-responsive cells (b') are detected in the dorsal 2/3 of the otocyst. (c) Quantification of Wnt reporter fluorescent levels in individual cells from seven otocysts transfected with the Wnt reporter (see Materials and methods). A decreasing gradient of Wnt reporter fluorescence is observed along the dorso-ventral and postero-anterior axis of the otocyst. (d) Plot of the normalised median fluorescence levels of cells as a function of their position along the dorso-ventral axis of the otocyst. The standard deviation bars reflect variability in fluorescent intensity along the anterio-posterior axis. (e–e”) E3 chicken otocyst co-electroporated with the Wnt and Notch reporters and a control plasmid. The Notch reporter marks the prosensory cells in the antero-ventral prosensory domain (e”). (f) A representative scatter plot of the mean fluorescence values (f.v.) for Wnt (5TCF::H2B-RFP) and Notch (T2-Hes5::nd2EGFP) reporters in individual cells of the anterior prosensory domain. The two groups correspond to cells segmented using either the Notch (green) or Wnt (magenta) reporter fluorescence signal. The cells with high Notch activity tend to have low levels of Wnt activity, and cells with high Wnt activity have low levels of Notch activity, but there is no inverse correlation between the reporters activities at intermediate fluorescence intensity values.
-
Figure 1—source data 1
Wnt reporter activity in the E3 chicken otocyst.
- https://cdn.elifesciences.org/articles/59540/elife-59540-fig1-data1-v3.xlsx
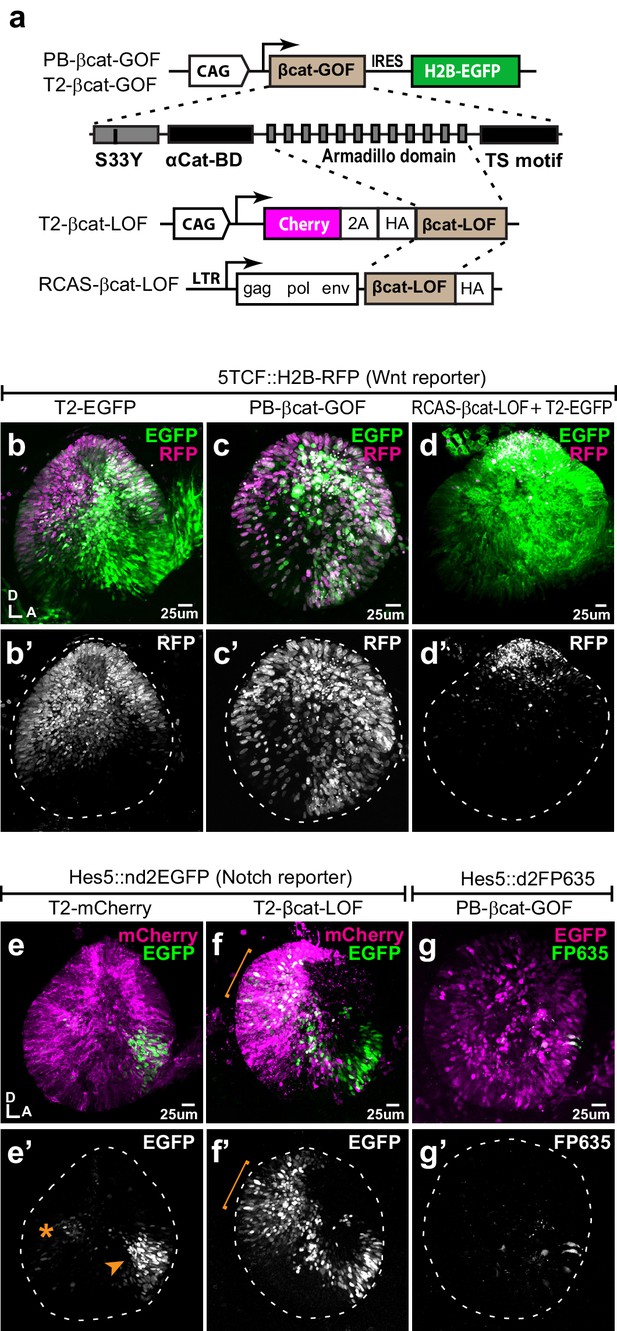
Wnt signalling antagonises Notch activity.
(a–d’) Schematic representation of the Piggybac, Tol2, and RCAS constructs used for β-catenin gain- (GOF) and loss-of-function (LOF) experiments. The PB-βcat-GOF and T2-βcat-GOF contain the full-length β-catenin including the α-catenin binding domain (αCat-BD), 12 Armadillo domains, the transactivator (TS) motif, and the S33Y mutation preventing phosphorylation and degradation. The RCAS-βcat-LOF and T2-βcat-LOF constructs drive expression of a truncated form of β-catenin comprising the Armadillo repeats only. (b–d’) Activity of the Wnt reporter in E3 otocysts co-electroporated with either T2-EGFP (control; b–b’), PB-βcat-GOF (c–c’), or RCAS-βcat-LOF (d–d’). Note the ventral expansion of the Wnt reporter fluorescence in (c–c’) and its restriction to the most dorsal part of the otocyst in (d–d’). (e–g’) Activity of the Notch reporters T2-Hes5::nd2EGFP or Hes5::d2FP635 in E3 otocysts co-electroporated with either T2-mCherry (control, e–e’), T2-βcat-LOF (f–f’), or PB-βcat-GOF (g–g’). The Notch reporter is normally activated in the anterior (arrowhead) and to a lesser extent posterior (asterisk) prosensory domains of the otocyst (e–e’). It is strongly upregulated in dorsal regions transfected with the T2-βcat-LOF construct (brackets in f–f’), but barely detectable in otocysts co-electroporated with PB-βcat-GOF (g–g’). On the other hand, manipulation of Notch activity had no discernible effect on the activity of the Wnt reporter (Figure 2—figure supplement 1).
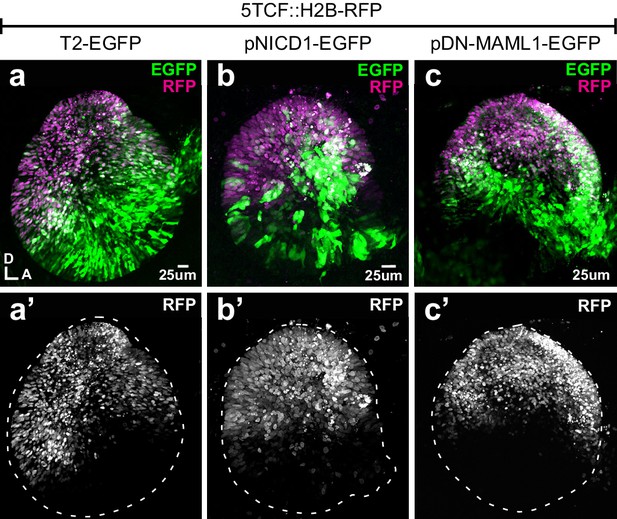
Manipulating Notch activity does not affect Wnt signalling.
(a–c’) Whole mounts of E3 chicken otocysts co-electroporated with Wnt reporter 5TCF::H2B-RFP and a control plasmid T2-EGFP or constructs activating (pNICD1-EGFP) and blocking (pDN-MAML1-EGFP) Notch signalling. There are no major changes in the dorso-ventral pattern of activation of the Wnt reporter in response to gain- and loss-of-Notch function (a’, b’, c’).
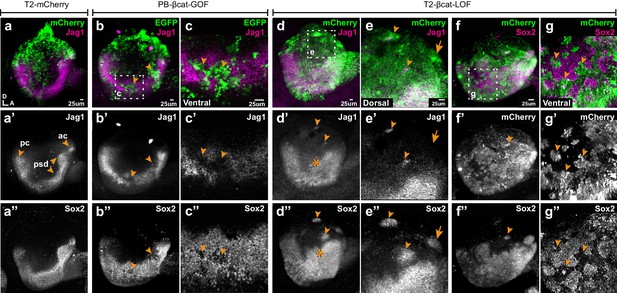
Wnt signalling antagonises prosensory specification.
Whole-mount views of E4 chicken otocysts electroporated at E2 and immunostained for Jag1 and Sox2 expression. (a–a”) Control sample electroporated with T2-mCherry. Jag1 and Sox2 are expressed in a U-shaped ventral common prosensory-competent domain (psd) and prospective prosensory domains (pc = posterior crista; ac=anterior crista). (b–c”) βcat-GOF overexpression induces a mosaic down-regulation of Sox2 and Jag1 expression (arrowheads) in the ventral half of the otocyst. High magnification views of transfected cells (arrowheads in c–c’’) and analysis of mean fluorescence values of Sox2 and βcat-GOF (Figure 3—figure supplement 1a–e) show that this effect is cell-autonomous. (d–g”) Otocysts transfected with T2-βcat-LOF exhibit a dorsal expansion of Jag1 and Sox2 expression (star in d’–d’’) and ectopic prosensory patches dorsally (arrowheads in d’–d’’, f’’) and high magnification views in (e–e’’). Note that some ectopic Sox2-positive patches are Jag1-negative (arrows in e’–e’’). The prosensory effects of βcat-LOF were dependent on Notch activity (Figure 3—figure supplement 2a–b’”). In contrast, in the ventral-most aspect of the otocyst, βcat-LOF overexpressing cells exhibit reduced Sox2 expression (arrowheads in high magnification views g–g’’). Overexpression of βcat-LOF elicits the formation of ectopic Islet-1 expressing otic neurons in the posterior and dorsal aspect of the otocyst (Figure 3—figure supplement 3).
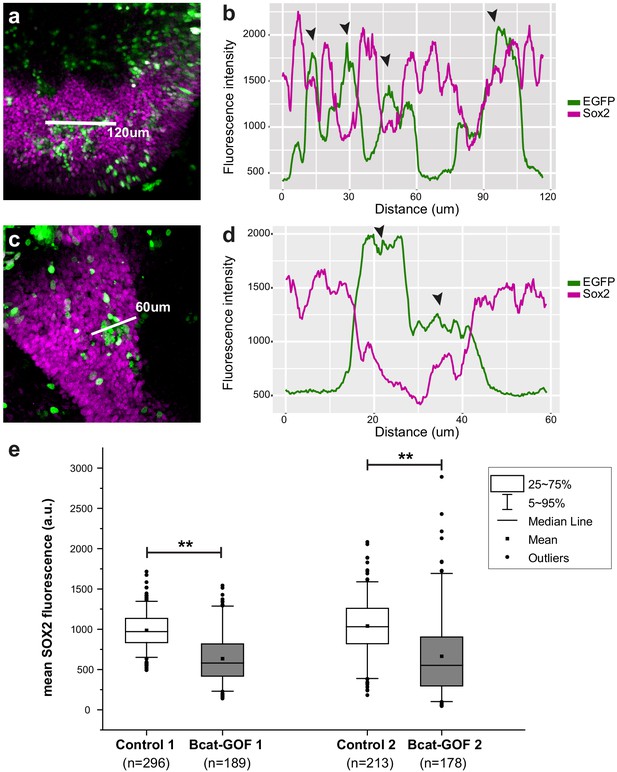
Analyses of Sox2 (magenta) and βcat-GOF (EGFP, green) fluorescence intensity levels in transfected prosensory regions.
In figures (a) and (c) the white line indicates the line selected for the profile plots shown in (b) and (d). The line profile plots (b and d) show that transfected cells with high levels of EGFP fluorescence (black arrowhead) have in general lower levels of Sox2 expression than untransfected cells. (e) Box plots of the average Sox2 fluorescence intensity within individual nuclei of untransfected (control) versus βcat-GOF transfected cells selected from two samples. Statistical analyses show a significant reduction in the levels of Sox2 expression in βcat-GOF transfected cells (p<0.01; Mann–Whitney U = 46,503 and U = 28,973 for respectively samples 1 and 2).
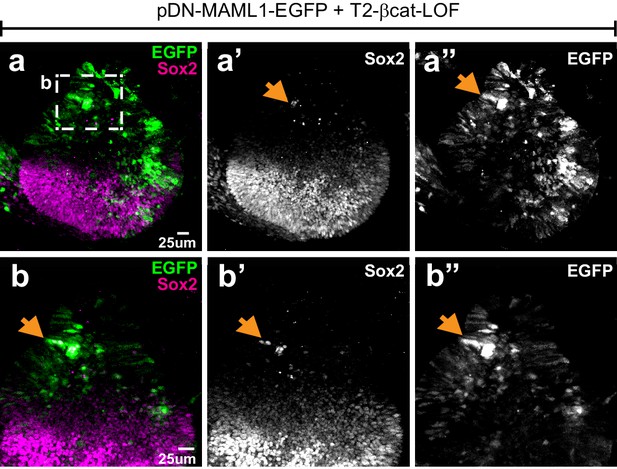
Effects of simultaneous loss of Wnt and Notch activity on prosensory specification.
Whole mount of an E4 otocyst co-electroporated with T2-βcat-LOF and a dominant-negative form of Maml1 (pDN-MAML1-EGFP) and immunostained for Sox2. (a–a’’) Sox2-expressing cells occupy the ventral half of the otocyst. There is no noticeable dorsal expansion of Sox2 expression (compare with Figure 3) and only a very limited number of EGFP-positive cells with ectopic Sox2 expression (arrows and high magnification views in b–b’”) are present in the dorsal half of the otocyst.
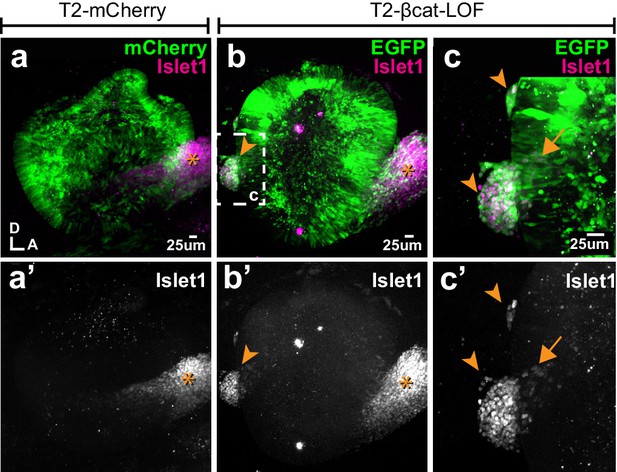
Blocking Wnt signalling triggers ectopic neurogenesis.
(a–a’) Whole-mount views of an E4 otocyst electroporated at E2 with a control T2-mCherry vector and immunostained for the otic neuronal marker Islet1. The cochleo-vestibular ganglion (star) is on the anterior side of the otocyst. (b–c’) In the T2-βcat-LOF transfected otocyst, ganglion-like clusters of Islet1-expressing cells are present in posterior and dorsal locations (arrowheads). (c–c’) A higher magnification view of the posterior region of the otocyst shown in (b). Note the presence of Islet1-positive cells within the epithelial lining of the otocyst itself (arrow).
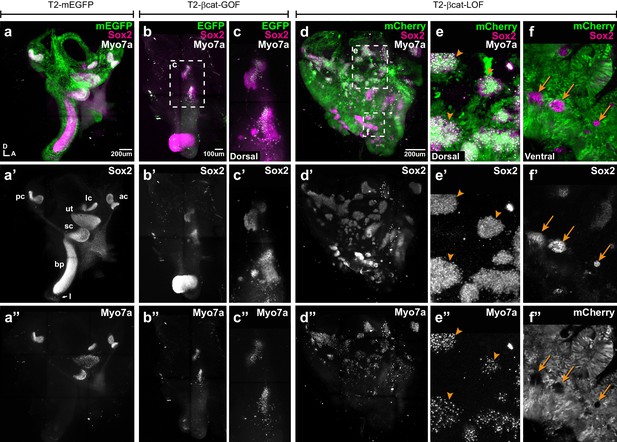
Manipulating Wnt activity alters inner ear sensory organ formation.
(a–a”) Whole-mount (tiled maximum projection) views of an E7 chicken inner ear electroporated at E2 with a control vector (T2-mEGFP) and immunostained for Sox2 (a’) and two hair cell markers, Myo7a and HCA (‘HC’ in all panels). (a”) All sensory organs are properly formed: posterior (pc), anterior (ac) and lateral (lc) cristae, saccule (sc), utricle (ut), basilar papilla (bp), and lagena (l). (b–c”) An inner ear transfected with T2-βcat-GOF. Note the absence of EGFP expression and severe defects in overall morphology of the vestibular system and basilar papilla; the remaining sensory patches are small and abnormally shaped (b’). (c–c”) Higher magnification of the vestibular Sox2-positive patches containing Myo7a and HCA-expressing hair cells. (d–f”) An inner ear transfected with T2-βcat-LOF. (d–d’’) Whole-mount (tiled maximum projection) views demonstrating the presence of numerous ectopic sensory patches with hair cells, and severe defects in inner ear morphology. (e-e’) Higher magnification of the dorsal region, where transfected cells form ectopic sensory patches positive for Sox2 (e’) and populated with Myo7a and HCA-expressing hair cells (arrowheads). (f–f”) In contrast, in ventral domains, EGFP-positive patches are devoid of Sox2 and hair cell markers expression. The only remaining Sox2-expressing patches are not transfected (arrows).
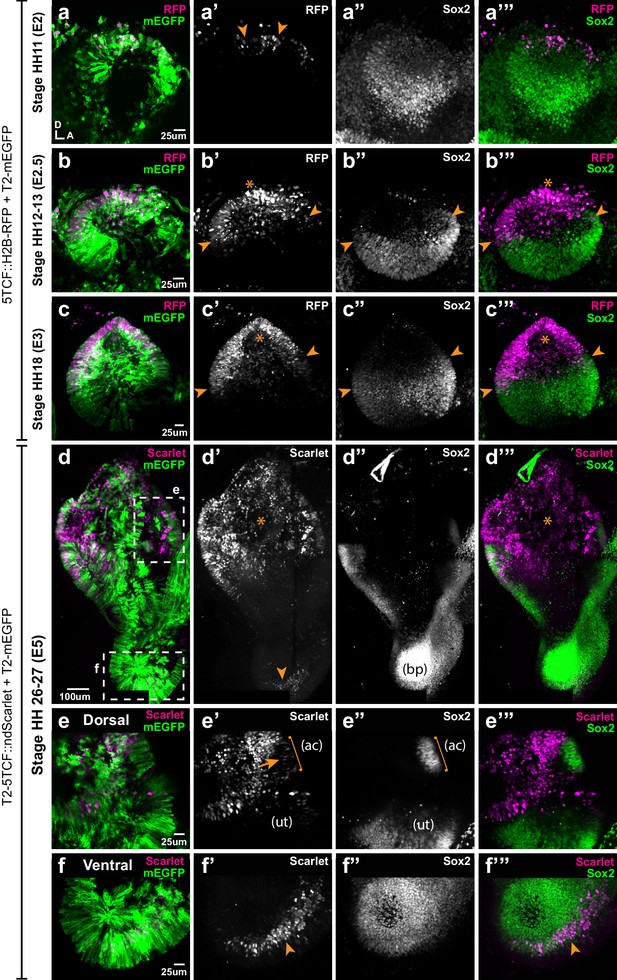
Spatial pattern of Wnt activity in the developing chicken inner ear.
Samples co-electroporated at the early otic placode stage with a Wnt reporter (5TCF::H2B-RFP or T2-5TCF::nd2Scarlet for long-term integration) and a control plasmid (T2-mEGFP in a–d) were collected 6 hr (stage HH11), 12 hr (HH12-13), 24 hr (HH18), and 3 days (E5, HH26-27) post-electroporation and immunostained for Sox2 expression. (a–a’”) Only a few cells on the dorso-medial wall of the otic placode are positive for Wnt reporter 5TCF::H2B-RFP (arrowheads in a’), whilst most otic cells express Sox2 (a’’). (b–c’”) Wnt activity increases gradually in the dorsal aspect of the otic cup and otocyst (stars in b’ and c’), concomitant to a dorsal decrease in Sox2 expression (b’’–c’’). Note the overlap between the signals of Wnt reporter and Sox2 (arrowheads in b’–b’ and c’–c’) at the dorsal edges of the prosensory domains. (d–d’’’) The Wnt reporter T2-5TCF::nd2Scarlet is strongly active in the dorsal half of the E5 inner ear (stars in d’–d’’’) and a weaker signal is also detected at the tip of developing basilar papilla (arrowhead). (e–e’”) Higher magnification views of the anterior vestibular organs. Note the high levels of Wnt activity in the non-sensory territories. In comparison, transfected prosensory cells located within the anterior crista (ac) and utricle (ut) have lower levels of fluorescence (arrow in e’). (f–f’”) Higher magnification views of the ventral (distal) tip of the basilar papilla, which also contains Wnt-active prosensory cells (arrowheads).
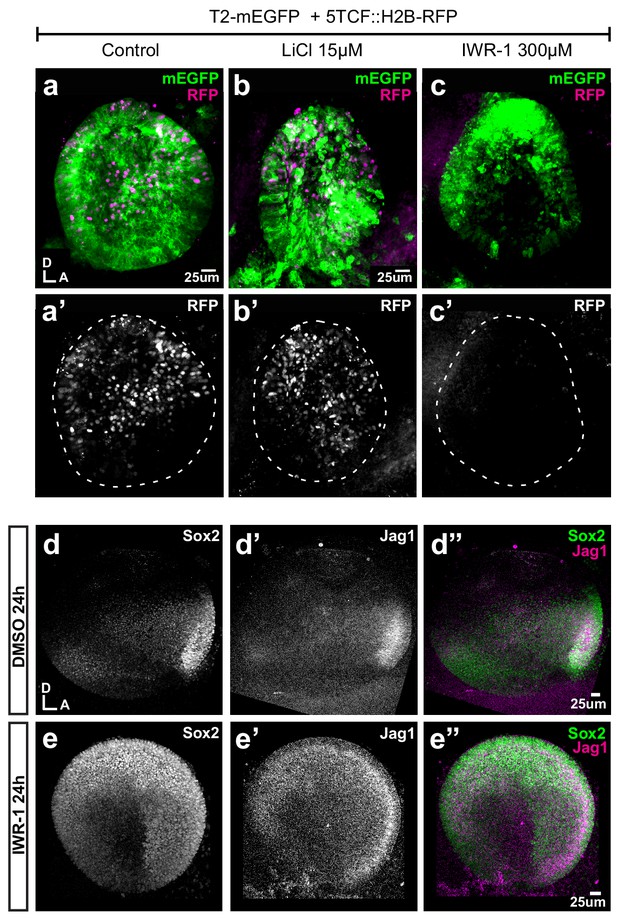
Pharmacological modulation of Wnt activity in explanted E3 otocysts.
(a–c') Whole-mount views of otic cups co-electroporated with the Wnt reporter and a control EGFP vector and incubated for 24 hr in control medium (DMSO) (a–a’), or media supplemented with either the Wnt agonist LiCl (b–b’) or the antagonist IWR-1 (c–c’). (d–e’’) E3 otocysts cultured for 24 hr in IWR-1 or DMSO (vehicle) as a control. IWR-1 treatment results in a dorsal expansion of Sox2 and Jag1 staining. The effects of increasing concentration of LiCl on Sox2 expression are shown in Figure 6—figure supplement 1a–e.
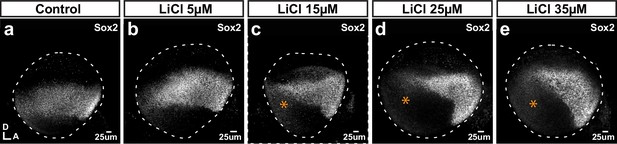
Effects of the Wnt agonist LiCl on Sox2 expression.
(a–e) Whole mounts of E3 chicken otocysts incubated for 24 hr in control medium or media enriched with increasing doses of LiCl. (a) In control condition, Sox2 marks a medial band of sensory-competent cells stretching along the antero-posterior axis of the otocyst. (b–e) With increasing doses of LiCl, there is a decrease of Sox2 expression in the posterior side (stars in c–e) as well as a noticeable shift of the orientation of the anterior Sox2-positive domain towards the ventral side.
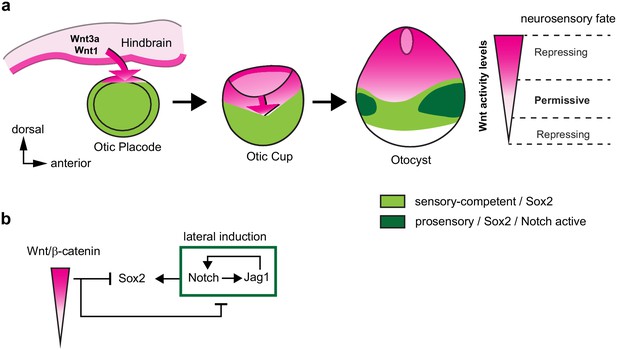
A schematic model of the effects of canonical Wnt activity on the patterning of inner ear neurosensory-competent domains.
(a) The hindbrain produces Wnt1 and Wnt3a ligands activating Wnt signalling in the dorsal aspect of the otic placode. Over time, a dorso-ventral gradient of Wnt activity forms in the otic cup and otocyst and regulates in a dose-dependent manner neural and prosensory specification. At intermediate levels, Wnt activity is permissive for the maintenance of Sox2 expression and Jag1/Notch signalling, which reinforces Sox2 expression and promotes acquisition of a prosensory fate by lateral induction. Hence, the dorso-ventral gradient of Wnt activity confines Sox2 expression to a middle region of neurosensory-competence from where the individual sensory organs will originate. (b) Schematic representation of the hypothetical regulatory interactions between Wnt and Notch signalling and their impact on Sox2 expression. The connectors do not imply direct interactions and intermediary factors are likely to contribute to the feedback loops.
Videos
E3 chicken otocyst electroporated with Wnt reporter 5TCF::H2B-RFP and control plasmid T2-EGFP.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Gene (Mus musculus) | B-catenin (Ctnnb1) | GenBank | ||
| Software, algorithm | R (RRID:SCR_001905) | https://www.r-project.org/ | Used for quantification and visualisation | |
| Software, algorithm | Volocity (RRID:SCR_002668) | https://quorumtechnologies.com/index.php/component/content/category/31-volocity-software | Used for quantification | |
| Software, algorithm | ImageJ (RRID:SCR_003070) | https://www.imagej.net | Used for quantification and visualisation | |
| Software, algorithm | OriginPro 2020 | OriginLab Corporation | Used for statistical analysis and visualisation | |
| Recombinant DNA reagent | 5TCF::H2B-RFP (plasmid) | PMID:24942669 | Wnt reporter | |
| Recombinant DNA reagent | T2-5TCF::nd2Scarlet (plasmid) | This paper and PMID:27869816 | Wnt reporter cloned into Tol2 transposon system, Daudet lab | |
| Recombinant DNA reagent | T2-Hes5::nd2EGFP (plasmid) | PMID:22991441 | Notch reporter | |
| Recombinant DNA reagent | Hes5::d2FP635 (plasmid) | PMID:22991441 | Notch reporter | |
| Recombinant DNA reagent | RCAS-βcat-LOF (plasmid) | PMID:12941626 PMID:7876319 | β-catenin LOF | |
| Recombinant DNA reagent | T2-βcat-LOF (plasmid) | This study | β-catenin LOF cloned into Tol2 transposon system, Daudet lab | |
| Recombinant DNA reagent | PB-βcat-GOF (plasmid) | PMID:24942669 | β-catenin GOF | |
| Recombinant DNA reagent | T2-βcat-GOF (plasmid) | This study | β-catenin GOF cloned into Tol2 transposon system, Daudet lab | |
| Recombinant DNA reagent | pNICD1-EGFP (plasmid) | PMID:15634704 | Notch GOF | |
| Recombinant DNA reagent | pDN-MAML1-EGFP (plasmid) | PMID:27218451 | Notch LOF | |
| Recombinant DNA reagent | T2-EGFP (plasmid) | PMID:17362912 | Control plasmid | |
| Recombinant DNA reagent | T2-mEGFP (plasmid) | This study | Control plasmid, mEGFP cloned into Tol2 transposon system, Daudet lab | |
| Recombinant DNA reagent | T2-mRFP (plasmid) | This study | Control plasmid, mRFP cloned into Tol2 transposon system, Daudet lab | |
| Recombinant DNA reagent | pTurquoise (plasmid) (RRID:Addgene_98817) | Addgene | Addgene No: 98817 | Control plasmid |
| Recombinant DNA reagent | mPB (plasmid) | PMID:19755504 | PiggyBac transposase | |
| Recombinant DNA reagent | pCAGGS-T2-TP (plasmid) | PMID:17362912 | Tol2 Transposase | |
| Commercial assay or kit | In-Fusion HD Cloning | Takarabio | No: 638916 | |
| Commercial assay or kit | RNAqueous-Micro Total RNA Isolation Kit | Life Technologies | No: AM1931 | |
| Antibody | Rabbit polyclonal anti-Jagged 1 (RRID:AB_649685) | Santa-Cruz Biotechnology | No: sc-8303 | IF (1:200) |
| Antibody | Rabbit polyclonal anti-Sox2 (RRID:AB_2341193) | Abcam | No: 97959 | IF (1:500) |
| Antibody | Mouse IgG1 monoclonal anti-Sox2 (RRID:AB_10694256) | BD Biosciences | No: 561469 | IF (1:500) |
| Antibody | Mouse monoclonal IgG1 anti-Islet1 (RRID:AB_1157901) | Developmental Studies Hybridoma Bank | Clone 39.3F7 | IF (1:250) |
| Antibody | Mouse monoclonal IgG1 anti-HA-tag (RRID:AB_291262) | Babco Inc | No: MMS-101R | IF (1:500) |
| Antibody | Mouse monoclonal IgG1 anti-Myo7a (RRID:AB_2282417) | Developmental Studies Hybridoma Bank | Clone 138–1 | IF (1:500) |
| Antibody | Mouse monoclonal IgG1 anti-HCA (RRID:AB_2314626) | Guy Richardson | IF (1:1000) | |
| Chemical compound, drug | LiCl | Sigma-Aldrich | No: L7026 | Concentrations: 5 µM, 15 µM, 25 µM, 35 µM |
| Chemical compound, drug | IWR-1 | Sigma-Aldrich | No: I0161 | Concentration 300 µM |
| Chemical compound, drug | Leibovitz’s | Gibco | No: 21083–027 | |
| Chemical compound, drug | Matrigel | Corning | No: 354230 | |
| Chemical compound, drug | DMEM/F12 | Gibco | No: 21041–025 | |
| Chemical compound, drug | HEPES | Sigma-Aldrich | No: SRE 0065 | Concentration 1% |
| Chemical compound, drug | Ciprofloxacin | Fluka | No: 17850–5 G-F | Concentration 0.1% |
| Sequence-based reagent | 5xTCF-BS_F | This paper | PCR primers | ATGGGCCCTCGTCGAACGACGTTGTAAAACGACGG |
| Sequence-based reagent | 5xTCF-BS_R | This paper | PCR primers | TGGTGGCgAGATCTGCGGCACGCTG |
| Sequence-based reagent | Bcat_GOF_F | This paper | PCR primers | TTTTGGCAAAGAATTGCCACCATGGCTACTCAAGC |
| Sequence-based reagent | Bcat_GOF_R | This paper | PCR primers | TAGACTCGAGGAATTtcacctattatcacggccgcc |
| Sequence-based reagent | Bcat_LOF_F | This paper | PCR primers | gattacgctgctcgagcaatccccgagc |
| Sequence-based reagent | Bcat_LOF_R | This paper | PCR primers | ctagagtgaagcagctcagtaagag |