Dissociable control of unconditioned responses and associative fear learning by parabrachial CGRP neurons
Figures
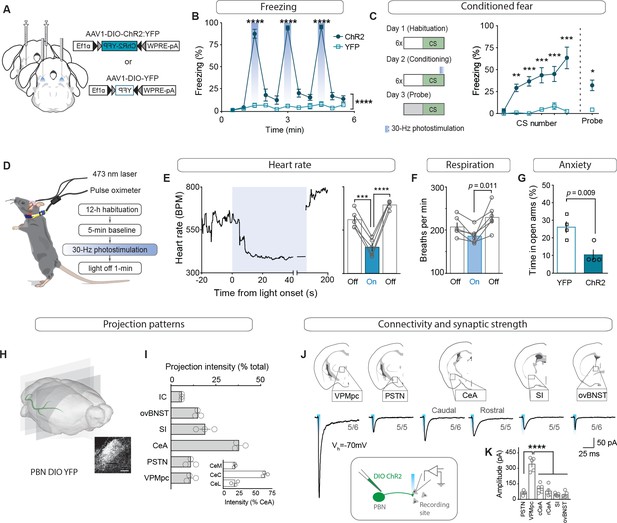
CGRPPBN neurons potentiate fear behavior, drive associative learning and robustly activate forebrain targets.
(A) Bilateral injections of AAV1-DIO-ChR2:YFP or AAV1-DIO-YFP and fiberoptic cannula implants above the PBN of CalcaCre/+ mice. (B) Photostimulation (30 Hz) of CGRPPBN neurons generated robust freezing behavior (n = 8,6 (n = ChR2, YFP); significant group x time interaction in a two-way ANOVA, F10,120 = 83.53, p<0.0001; subsequent Sidak pairwise comparisons, ****p<0.0001). (C) Optogenetic stimulation of CGRPPBN neurons conditioned freezing behavior when preceded by a 10 kHz auditory CS (n = 4,4 (n = ChR2, YFP); significant group x time interaction in a two-way ANOVA, F5,36 = 5.62, p=0.0006; subsequent Sidak pairwise comparisons, **p<0.01; ***p<0.001; and Welch’s unpaired t-test for probe trial, t(3.47) = 5.62, *p=0.016). (D) Schematic and timeline for pulse-oximetry measurements of autonomic responses to optogenetic stimulation. (E) Representative and mean bradycardia caused by 30 Hz photostimulation of CGRPPBN neurons (n = 5, one-way ANOVA, F2,12 = 39.66, p<0.0001; subsequent Dunnett correction for multiple comparisons). (F) Respiratory rate was also reduced during photostimulation (n = 6, one-way ANOVA, F2,15 = 5.12, p=0.0196; subsequent Dunnett correction for multiple comparisons, p=0.011). (G) Stimulation of CGRPPBN neurons was anxiogenic (n = 4,4, Welch’s unpaired t-test, t(5.93) = 3.78, p=0.009). (H) Expression of a fluorescent protein in CGRPPBN neurons to identify efferent projections. Scale bar: 100 µm. (I) Fluorescence in downstream targets relative to cumulative projection intensity; inset is fluorescence in CeA subnuclei relative to total CeA fluorescence. (J) Representative light-evoked EPSCs from cells downstream of CGRPPBN neurons; figures below traces (e.g. 5/6) indicate proportion of recorded cells that responded within each region. (K) Average amplitudes of EPSCs from responsive cells (5 cells for each site from four mice; 30/33 cells responded, significance for one-way ANOVA, F5,24 = 38.75, p<0.0001; subsequent Tukey correction for multiple comparisons). Data are represented as mean ± SEM. For full statistical information see Supplementary file 1.
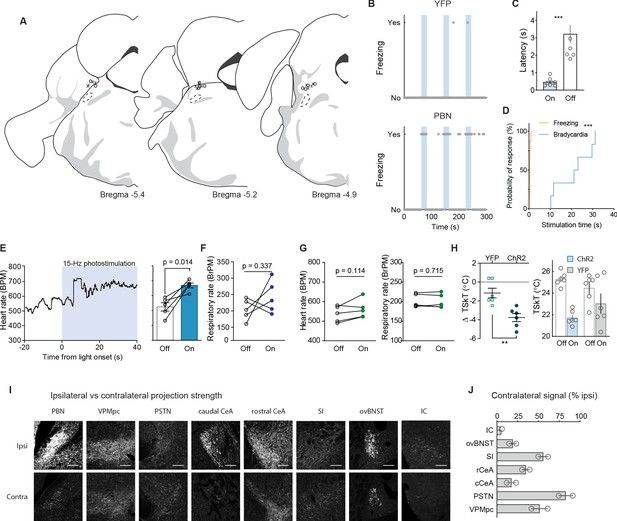
Fiber placement, autonomic measurements, and contralateral projection strength.
(A) Position of fiberoptic cannula tips for CGRPPBN neuron stimulation. (B) Example of freezing behavior in response to repeated 30 Hz photostimulation of CGRPPBN neurons. (C) Latency to freeze during stimulation and post-stimulation epochs (some data points outside axis limits; n = 8, paired t-test, t(7) = 5.681, p=0.0007). (D) Survival curve comparing latencies of freezing and bradycardia responses to 30 Hz photostimulation of CGRPPBN neurons (n = 8 freezing, n = 6 heartrate, Mantel-Cox Logrank test Chi square = 13.13, p=0.0003). (E) Effect of 15 Hz photostimulation of CGRPPBN neurons on heart rate (n = 5, paired t-test, t(4) = 4.173, p=0.014). (F) Effect of 15 Hz photostimulation of CGRPPBN neurons respiratory rate (n = 5, paired t-test, t(4) = 1.09, p=0.34). (G) Effect of 15 Hz photostimulation in YFP-expressing control animals on heart rate (left), and respiratory rate (right) (n = 5, paired t-tests, heart rate t(4) = 2.02, p=0.12; respiration t(4) = 0.39, p=0.71). (H) Vasoconstriction elicited by 30 Hz photostimulation of CGRPPBN neurons, change in tail-skin temperature (left), and absolute tail-skin temperature (right) (n = 6.6; ChR2, YFP, Welch’s unpaired t-test, t(9.64) = 3.92, **p=0.0031). (I) Ipsi- and contralateral fluorescent images of CGRPPBN-neuron projection targets from mouse unilaterally expressing DIO-YFP in CGRPPBN neurons. Scale bar: 100 µm. (J) Contralateral projection strength relative to ipsilateral fluorescent signal in each projection target structure (n = 2). Data are represented as mean ± SEM. For full statistical information see Supplementary file 1.
Freezing behavior generated by activating CGRPPBNneurons.
Supplement to Figure 1.
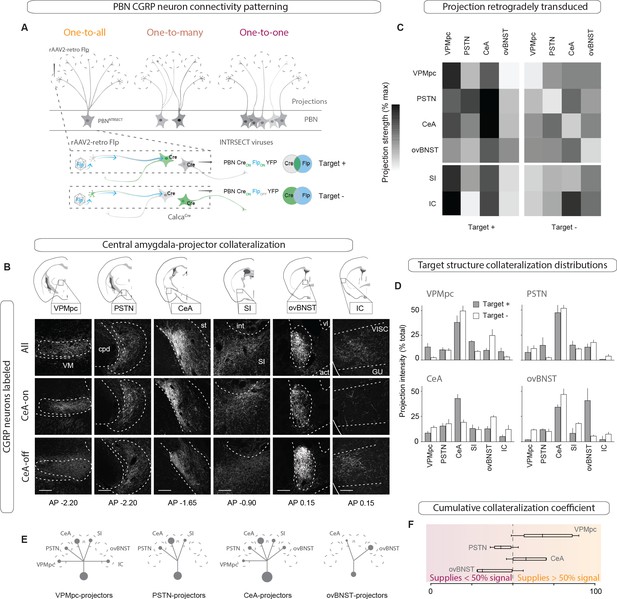
CGRPPBN neurons broadly collateralize to forebrain targets.
(A) Injections of rAAV2-retro-Flp into projection targets and INTRSECT viruses into the PBN of CalcaCre/+ mice to isolate target-projecting (Target +, Cre-on Flp-on) or non-projecting (Target -, Cre-on Flp-off) populations. (B) Fluorescent images of projection targets in mice expressing tracer in either all CGRPPBN neurons, CeA-projectors (CeA-on), or non-CeA-projectors (CeA-off). Scale bar: 100 µm.(C) Heat maps of averaged fluorescent intensity in downstream sites for Target + or Target - viral expression conditions for the VPMpc, PSTN, CeA, and ovBNST; values normalized to maximal target projection intensity given by expression of DIO-YFP (n = 3 per condition). (D) Overview of target-projecting projection distributions for VPMpc, PSTN, CeA, and ovBNST in Target + and Target – conditions (mean ± SEM). (E) Schematic of relative population size and collateralization distribution from each target-projecting subset. Collaterals were indicated if collateralization coefficient was >50% (see below), or if structure made up >35% projection distribution in (D) from Target + condition. (F) Collateralization coefficient calculated as difference between normalized fluorescence intensity in projection site in Flp-on condition – Flp-off condition, averaged across all sites, scaled by 50% and forced through 0 for y-intercept. Example calculation for VPMpc-projector to CeA collateralization coefficient: (([CeA fluorescence]VPMpc-ON – [CeA fluorescence]VPMpcOFF)/[CeA fluorescence]DIOYFP)x 50% + 50%. Center line, mean; box limits, upper and lower quartiles; whiskers, min to max.
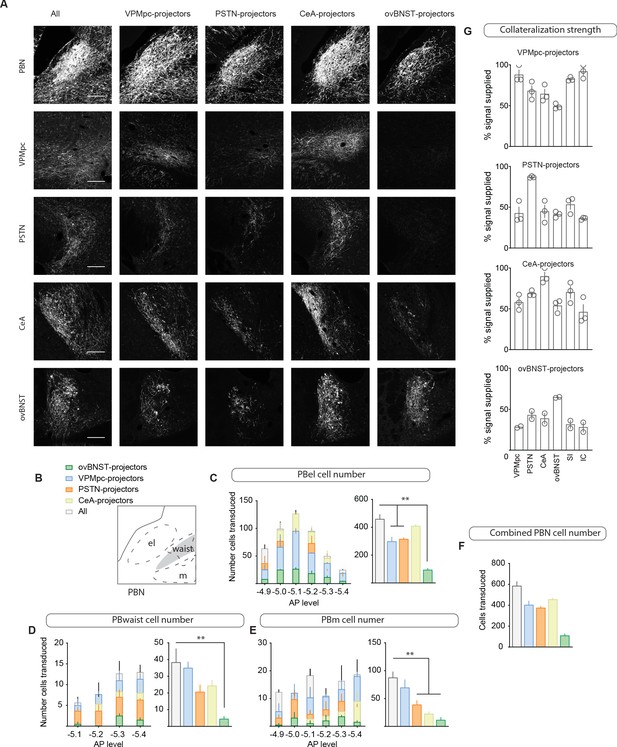
Collateralization to forebrain targets by CGRPPBN neurons.
(A) Fluorescent images of CGRPPBN neurons and their projection targets from mice expressing YFP in either all CGRPPBN neurons or those projecting to the VPMpc, PSTN, CeA, or ovBNST. Scale bar: 100 µm. (B–F) Cell counts across the AP-axis in various PBN subnuclei of CGRPPBN neurons transduced with the help of retrogradely transported Flp injected into the VPMpc, PSTN, CeA, or ovBNST (n = 3 per condition). Significance for ordinary one-way ANOVAs with subsequent Dunnett correction for multiple comparisons ((C) F4,9 = 31.17, p<0.0001; (D) F4,9 = 10.81, p=0.0017; (E) F4,9 = 6.05, p=0.012; pairwise comparisons **p<0.01). (G) Collateralization coefficients for each projection target calculated for each projection-specific subset of CGRPPBN neurons. Data are represented as mean ± SEM. For full statistical information see Supplementary file 1.
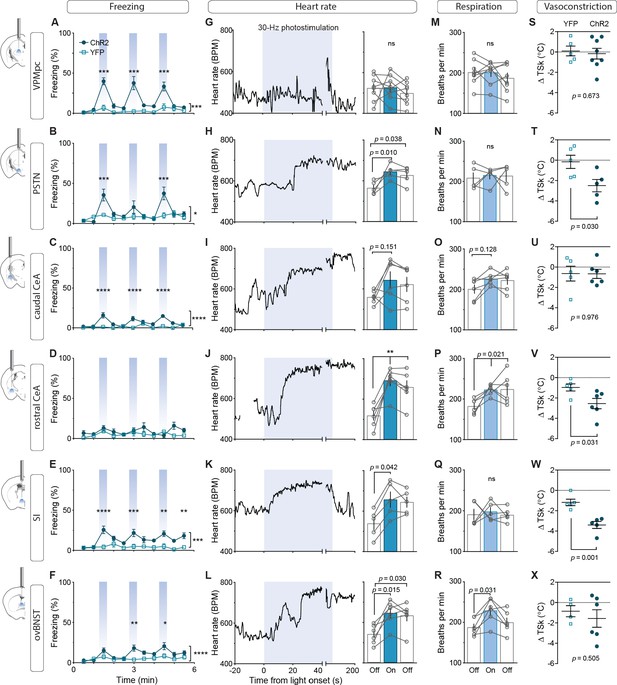
Photostimulation of CGRPPBN neuron terminals in individual downstream targets exerts diverse effects on physiology and behavior.
(A) Activating terminals in the VPMpc (n = 8,5) (ChR2, YFP) elicited freezing behavior but had no effect on (G) heart rate (M) respiration or (S) vasoconstriction. (B) Photostimulating terminals in the PSTN (n = 6,5) elicited freezing behavior, (H) caused mild tachycardia, (N) had no effect on respiration but (T) caused vasoconstriction. (C) Photostimulating terminals in the cCeA (n = 6,5) increased freezing behavior but had no effect on (I) heart rate (O) respiration or (U) vasoconstriction. (D) Photostimulating terminals in the rCeA (n = 6,5) had no effect on freezing behavior (J) elicited robust tachycardia (P) hyperventilation and (V) vasoconstriction. (E) Photostimulating terminals in the SI (n = 8,6) increased freezing behavior, (K) caused tachycardia (n = 5), (Q) had no effect on respiration and (W) caused vasoconstriction. (F) Photostimulating terminals in the ovBNST (n = 9,5) increased freezing behavior, (L) caused tachycardia and (R) hyperventilation but (X) did not affect vasoconstriction. (A–F) Significance for effect of group in a two-way ANOVA with subsequent Sidak pairwise comparisons. (G–R) Significance for one-way ANOVA with subsequent Dunnett correction for multiple comparisons. (S–X) Significance for Welch’s unpaired t-test. Data are represented as mean ± SEM. *p<0.05; **p<0.01; ***p<0.001; ****p<0.0001. For full statistical information see Supplementary file 1.
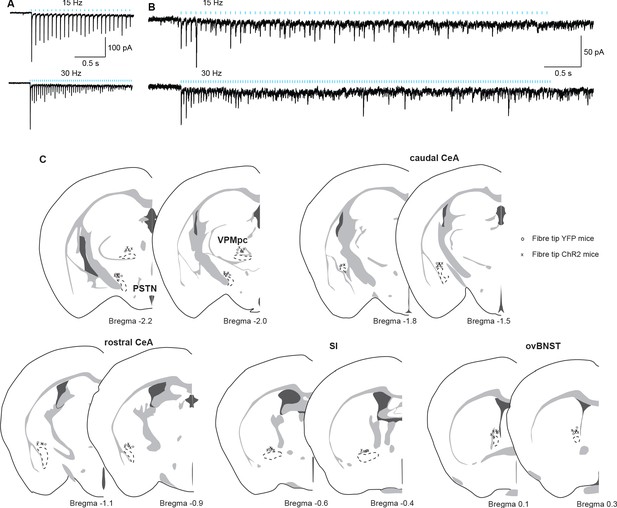
Verification of terminal stimulation of CGRPPBN neuron projections.
(A–B) Postsynaptic neurons are reliably activated by 15- (top) and 30 Hz (bottom) photostimulation of CGRPPBN neuron terminals (five cells from two mice, two cells represented). (C) Position of fiber-optic cannula tips for projection-specific terminal photostimulation in control (o) and experimental (x) groups. Related to Figures 3–5.
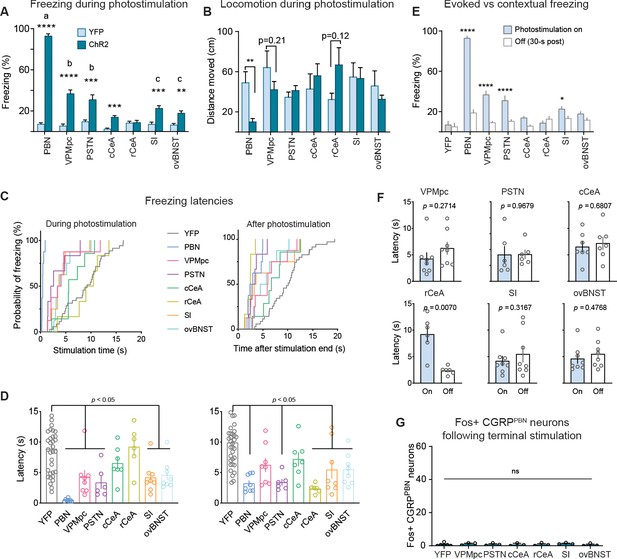
Freezing behavior elicited by photostimulation of CGRPPBN neuron terminals.
(A) Freezing behavior from each animal collapsed across stimulation epochs. Photostimulation of CGRPPBN neurons resulted in robust freezing behavior not replicated by activation of any individual projection (significant group effect in two-way ANOVA, F6,253 = 75.73, p<0.0001; subsequent Sidak pairwise comparisons, **p<0.01; ***p<0.001). (B) Average distance moved for each animal during stimulation epochs; only stimulation of CGRPPBN neurons significantly reduced locomotion (Welch’s unpaired t-tests, PBN n = 7,6 (ChR2, YFP), t(5.91) = 3.37, p=0.0153; rCeA n = 6,5, t(6.30) = 1.88, p=0.1066). (C) Survival curves comparing average latencies of freezing responses during (left) and after (right) photostimulation of CGRPPBN neuron terminals or cell bodies (YFP control n = 35, PBN n = 8, VPMpc n = 8, PSTN n = 6, cCeA n = 7, rCeA n = 6, SI n = 8, ovBNST n = 8; Mantel-Cox Log-rank test, Chi-squared = 168.1, p<0.0001). (D) Average freezing response latencies during (left) and after (right) photostimulation (n same as in C). Significance for ordinary one-way ANOVAs with subsequent Tukey’s correction for multiple comparisons. Left: F7,78 = 9.00, p<0.0001; right: F7,78 = 8.73, p<0.0001. (E) Comparison of freezing behavior from each stimulation/post-stimulation epoch collapsed for each animal (three values per animal) (significant treatment effect in two-way ANOVA, F7,345 = 116.4, p<0.0001; subsequent Sidak pairwise comparisons, *p<0.05; ****p<0.0001) (F) Comparison of freezing-bout response latencies during and after photostimulation of CGRPPBN neuron-terminals (paired t-tests, VPMpc n = 8, t(7) = 1.19, p>0.05; PSTN n = 6, t(5) = 0.04, p>0.05; cCeA n = 7, t(6) = 0.43, p>0.05; rCeA n = 6, t(5) = 4.41, p=0.007; SI n = 8, t(7) = 1.08, p>0.05; ovBNST n = 8, t(7) = 0.75, p>0.05). (G) Photostimulation of CGRPPBN-neuron terminals did not induce Fos in CGRPPBN neurons (ordinary one-way ANOVA, F6,17 = 0.37, p=0.89).

Physiological responses to photostimulation of CGRPPBN neuron terminals.
(A) Autonomic responses to 15 Hz photostimulation of CGRPPBN neuron terminals in the VPMpc (n = 8, paired t-test, heart rate t(7) = 1.47, p=0.19; respiration t(7) = 0.03, p>0.05). (B) Autonomic responses to 15 Hz photostimulation of CGRPPBN neuron terminals in the PSTN (n = 7, paired t-test, heart rate t(6) = 1.37, p>0.05; respiration t(6) = 1.32, p>0.05). (C) Autonomic responses to 15 Hz photostimulation of CGRPPBN neuron terminals in the cCeA (n = 6, paired t-test, heart rate t(5) = 0.90, p>0.05; respiration t(5) = 0.31, p>0.05). (D) Autonomic responses to 15 Hz photostimulation of CGRPPBN neuron terminals in the rCeA (n = 9, paired t-test, heart rate t(8) = 7.65, p<0.0001; respiration t(8) = 1.94, p>0.05). (E) Autonomic responses to 15 Hz photostimulation of CGRPPBN neuron terminals in the SI (n = 7, paired t-test, heart rate t(6) = 2.48, p=0.048; respiration t(6) = 1.66, p>0.05). (F) Autonomic responses to 15 Hz photostimulation of CGRPPBN neuron terminals in the ovBNST (n = 6, paired t-test, heart rate t(5) = 4.35, p=0.0074; respiration t(5) = 0.36, p>0.05). (G–L) Autonomic responses to 15 Hz light delivery in control animals. Data represented as mean ± SEM. For full statistical information see Supplementary file 1.
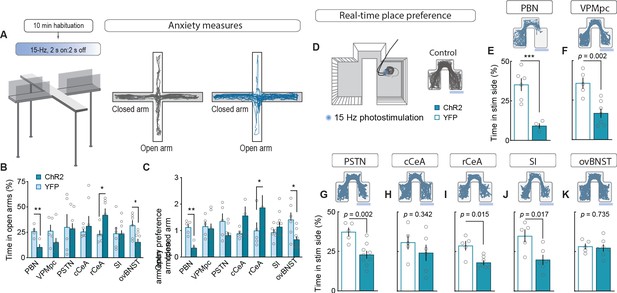
Stimulating CGRPPBN neuron terminals in ovBNST is anxiogenic while stimulating most other projections is aversive.
(A) Experimental timeline and example responses to stimulation of CGRPPBN neuron terminals or somata during measurements of anxiety-like behavior. (B) Activation of CGRPPBN neurons reduced time spent in open arms, as did stimulation of terminals in the ovBNST. Activation of terminals in the rCeA increased open-arm exploration time. Significance for Welch’s unpaired t-test (PBN t(5.93) = 3.77, **p=0.009, n = 4.4 (ChR2, YFP); rCeA t(9.42) = 2.59, *p=0.028, n = 7.5; ovBNST t(8.85) = 2.65, *p=0.034, n = 9.6). (C) Activation of CGRPPBN neurons or their projection to the ovBNST reduced open-arm entry preference; activation of the projection to the rCeA increased open-arm entries. Significance for Welch’s unpaired t-test (PBN t(6.90) = 4.87, **p=0.002, n = 4.4; rCeA t(5.59) = 2.51, *p=0.049, n = 7.5; ovBNST t(6.87) = 2.89, *p=0.018, n = 9.6). (D) Illustration of RTPP paradigm and example trace of control mouse maze exploration. (E) Activation of CGRPPBN neurons led to avoidance of light-paired side (Welch’s unpaired t-test, t(6.31) = 6.27, ***p<0.001, n = 6.4). (F) Mice avoid photostimulation of CGRPPBN neuron terminals in the VPMpc (Welch’s unpaired t-test, t(8.75) = 4.28, p=0.002, n = 7.5). (G) Mice avoid photostimulation of CGRPPBN neuron terminals in the PSTN (Welch’s unpaired t-test, t(9.71) = 4.11, p=0.002, n = 9.5). (H) Photostimulation of CGRPPBN neuron terminals in the cCeA does not affect place-preference (Welch’s unpaired t-test, t(8.99) = 1.00, p>0.05, n = 6.5). (I) Mice avoid photostimulation of CGRPPBN neuron terminals in the rCeA (Welch’s unpaired t-test, t(5.92) = 3.38, p=0.015, n = 7.5). (J) Mice avoid photostimulation of CGRPPBN neuron terminals in the SI (Welch’s unpaired t-test, t(7.87) = 3.02, p=0.017, n = 6.5). (K) Photostimulation of CGRPPBN neuron terminals in the ovBNST does not affect place-preference (Welch’s unpaired t-test, t(9.99) = 0.35, p>0.05, n = 7.5). Data are represented as mean ± SEM. For full statistical information see Supplementary file 1.
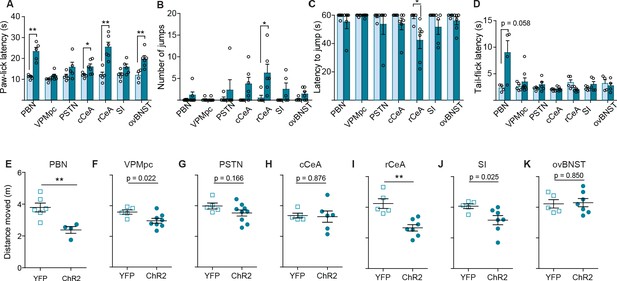
Activation of CGRPPBN terminals in the rCeA potentiates nocifensive responses.
(A) Response latency on 52°C hot plate increased by stimulating CGRPPBN neurons or projections to the cCeA, rCeA, or ovBNST prior to exposure, consistent with stress-induced analgesia (significance for Welch’s unpaired t-test; PBN n = 6,5 (ChR2, YFP); t(4.31) = 6.22, **p=0.0027; cCeA n = 6.5; t(6.21) = 2.76, *p=0.0315; rCeA n = 6.5; t(8.00) = 5.00, **p=0.0011; ovBNST n = 6.4; t=(7.91)=3.57, **p=0.0074). (B) Number of jumps in 1-min exposure to 52°C hot plate increased by stimulating CGRPPBN neuron terminals in the rCeA (Welch’s unpaired t-test, t(5.85) = 2.69, *p=0.0369; n = 6,5). (C) Latency to jump was also reduced by rCeA-terminal activation (Welch’s unpaired t-test, t(5.73) = 2.80, *p=0.0329; n = 6.5). (D) Tail-flick latency upon tail submersion in 52.5°C water bath was not significantly affected by either somata or terminal photostimulation of CGRPPBN neurons (Welch’s unpaired t-test, p>0.05). (E–K) Distance moved during RTPP assay pairing one side of a novel chamber with photostimulation of CGRPPBN-neuron terminals or somata (significance for Welch’s unpaired t-test). Data are represented as mean ± SEM. For full statistical information see Supplementary file 1.
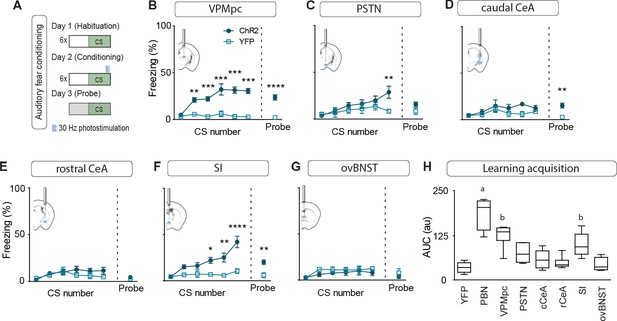
Photostimulating terminals in the VPMpc or SI can promote associative fear learning.
(A) Illustration of experimental paradigm for cue-dependent optogenetic conditioning. (B) Conditioned-freezing responses to CS paired with CGRPPBN terminal stimulation in the VPMpc during training (n = 8.5; ChR2, YFP; significant effect of group in two-way ANOVA, F1,66 = 115.4, p<0.0001; subsequent Sidak pairwise comparisons, **p<0.01; ***p<0.001) and in probe test 24 hr following conditioning (Welch’s unpaired t-test, t(8.93) = 7.29, ****p<0.0001). (C) Conditioned freezing responses to PSTN (n = 5.4) terminal stimulation (significant effect of group in two-way ANOVA, F1,42 = 6.99, p=0.012; subsequent Sidak pairwise comparisons). (D) Conditioned freezing responses to cCeA (n = 7.5) terminal stimulation (significant group effect in two-way ANOVA during training, F1,60 = 4.69, p=0.0343; and probe test, Welch’s unpaired t-test, t(8.15) = 4.40, **p=0.0022). (E) Conditioned freezing responses to rCeA (n = 8.5) terminal stimulation (two-way ANOVA effect of group, F1,60 = 2.74, p=0.1032). (F) Conditioned freezing responses to SI (n = 8.6) terminal stimulation. Significant group effect in two-way ANOVA during training, F1,60 = 23.45, p=0.0004; subsequent Sidak pairwise comparisons; and in probe test 24 hr following conditioning (Welch’s unpaired t-test, t(11.15) = 3.86, **p=0.0026). (G) Conditioned freezing responses to ovBNST (n = 5.4) terminal stimulation (two-way ANOVA effect of group, F1,66 = 2.764, p=0.1011). (H) Area under the curve for conditioning in each ChR2 fiber-placement group, including PBN-stimulation (n = 8) and control groups (n = 6, averaged for each YFP fiber-placement group). Significance for one-way ANOVA, F7,40 = 19.44, p<0.0001; subsequent Tukey correction for multiple comparisons, differences indicated by dissimilar letters above data columns. Bar graphs are represented as mean ± SEM. For full statistical information see Supplementary file 1.
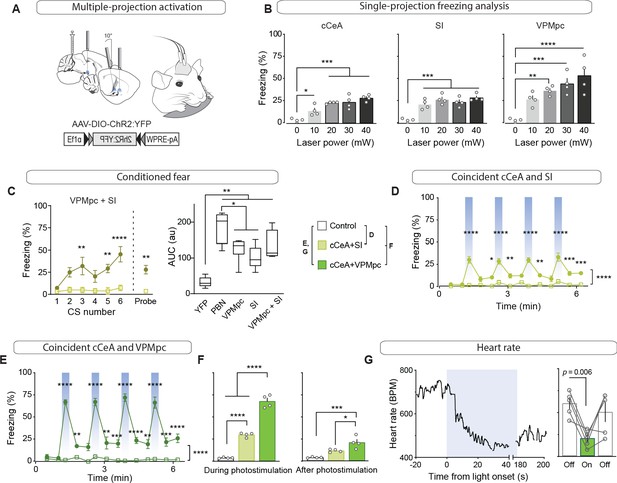
Combined activation of CGRPPBN neuron terminals in the VPMpc and cCeA scales freezing responses and produces bradycardia.
(A) Schematic showing configuration for implantation of 3 fiberoptic cannulae into one hemisphere allowing simultaneous photostimulation of multiple CGRPPBN-neuron terminal fields. (B) Freezing behavior during 30 Hz photostimulation with increasing power of CGRPPBN neuron terminal fields in the cCeA (n = 4, one-way ANOVA, F4,15 = 18.08, p<0.0001), SI (n = 4, F4,15 = 19.21, p<0.0001), or VPMpc (n = 4, F4,15 = 12.09, p=0.0001). Subsequent Tukey correction for multiple comparisons, *p<0.05; **p<0.01; ***p<0.01; ****p<0.0001. (C) Freezing behavior to auditory CS co-terminating with simultaneous photostimulation of terminals in the VPMpc and SI (left) (significant group effect in two-way ANOVA, F1,6 = 21.57, p=0.0035; subsequent Sidak pairwise comparisons) or probe test with CS presented in novel context 24 hr after conditioning (Welch’s unpaired t-test, t(3.414) = 4.90, p=0.012). Comparison of area-under-curve for associative learning generated by CS paired with CGRPPBN neuron or terminal activation (right) (one-way ANOVA, F4,27 = 19.73, p<0.0001); subsequent Tukey correction for multiple comparisons. Center line, mean; box limits, upper and lower quartiles; whiskers, min to max. (D) Freezing behavior in response to simultaneous activation of CGRPPBN neuron terminals in the cCeA and SI (n = 4.4 (ChR2, control), significant group effect in two-way ANOVA, F1,78 = 213.5, p<0.0001; subsequent Sidak pairwise comparisons). (E) Freezing behavior in response to simultaneous activation of CGRPPBN neuron terminals in the caudal CeA and VPMpc (n = 4.4; significant group effect in two-way ANOVA, F1,84 = 631.5, p<0.0001; subsequent Sidak pairwise comparisons). (F) Comparison of averaged freezing behavior for each stimulation combination during the stimulation epoch (left) (n = 4.4; one-way ANOVA, F2,9 = 218.9, p<0.0001), and during the post-stimulation epoch (right) (n = 4,4; one-way ANOVA, F2,9 = 17,67, p=0.0008; subsequent Tukey correction for multiple comparisons). (G) Representative (left) and mean bradycardia elicited by simultaneous photostimulation of CGRPPBN-neuron terminals in the cCeA and VPMpc (n = 5; one-way ANOVA, F2,12 = 7.38, p=0.0081; subsequent Dunnett correction for multiple comparisons, p=0.0058). Bar graphs represented as mean ± SEM. See also Figure 6—videos 1 and 2. For full statistical information see Supplementary file 1.
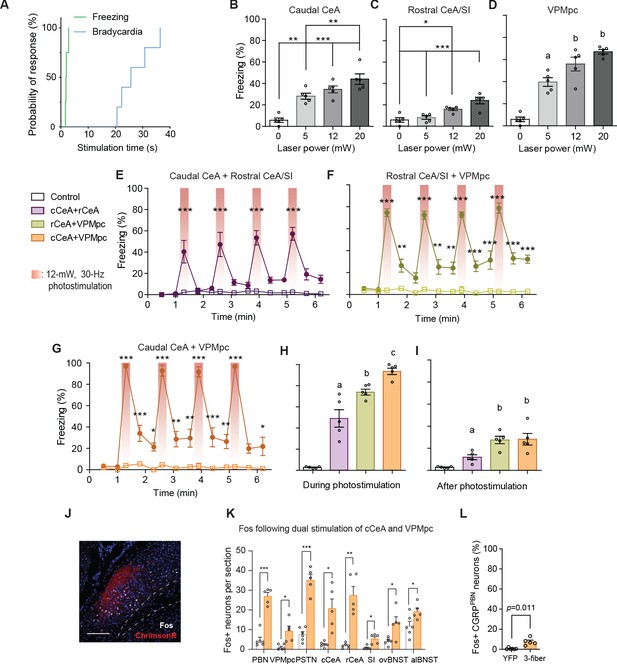
Coincident activation of CGRPPBN neuron projections using ChrimsonR causes profound freezing responses.
(A) Survival curve comparing latencies of freezing and bradycardia responses to simultaneous 30 Hz photostimulation of CGRPPBN neuron terminals in the VPMpc and cCeA (n = 4 freezing, n = 5 heart rate, Mantel-Cox Log-rank test, Chi-square = 9.03, p=0.0027). (B–H) Freezing responses to simultaneous optogenetic activation of multiple CGRPPBN-neuron terminal fields using ChrimsonR. (B–D) Freezing behavior during 30 Hz photostimulation of increasing power of CGRP terminal fields in (B) the cCeA (n = 5), (C) rCeA (n = 5), or (D) the VPMpc (n = 5). Significance for one-way ANOVA with subsequent Tukey correction for multiple comparisons. (E) Freezing behavior in response to simultaneous activation of CGRP terminals in the rostral and caudal CeA (n = 5.4 (ChR2, control), significant effect of group in two-way repeated-measure ANOVA, F1,7 = 41.27, p=0.0004; subsequent Sidak pairwise comparisons, ****p<0.0001). (F) Freezing behavior in response to simultaneous activation of CGRPPBN terminals in the rostral CeA and VPMpc (n = 5.4; significant effect of group in two-way RM ANOVA, F1,7 = 173.5, p<0.0001; subsequent Sidak pairwise comparisons, **p<0.01; ***p<0.001). (G) Freezing behavior in response to simultaneous activation of CGRPPBN terminals in the caudal CeA and VPMpc (n = 5.4, significant effect of group in two-way RM ANOVA, F1,7 = 118.3, p<0.0001; subsequent Sidak pairwise comparisons, *p<0.05). (H) Comparison of averaged freezing behavior for each stimulation combination during the stimulation epoch (n = 5.4, one-way ANOVA, F3,16 = 72.18, p<0.0001; subsequent Tukey correction for multiple comparisons, dissimilar letters above columns of data indicate statistical differences between groups). (I) Comparison of averaged freezing behavior for each stimulation combination during the post-stimulation epoch (n = 5.4, one-way ANOVA, F3,16 = 14.04, p<0.0001; subsequent Tukey correction for multiple comparisons). (J) Representative image showing ChrimsonR:tdTomato expression in CGRPPBN neurons (red) and Fos expression (white) following activation of terminals in the cCeA and VPMpc. Scale bar: 100 µm.(K) Quantification of number of Fos-positive neurons in the PBN and forebrain targets following simultaneous activation of CGRPPBN neuron terminals in the cCeA and VPMpc (n = 5), or light delivery in a YFP-expressing control (n = 6). Significance for Welch’s unpaired t-tests. (L) Quantification of the number of CGRPPBN neurons expressing Fos following simultaneous activation of terminals in the cCeA and VPMpc (n = 5), or light delivery in a YFP-expressing control (n = 6) (Welch’s unpaired t-test, t(4.40) = 4.26, p=0.0106). Data represented as mean ± SEM. For full statistical information see Supplementary file 1.
Freezing behavior generated by activating the SI and caudal CeA simultaneously supplement to Figure 6.
Freezing behavior generated by activating the caudal CeA and VPMpc simultaneously supplement to Figure 6.
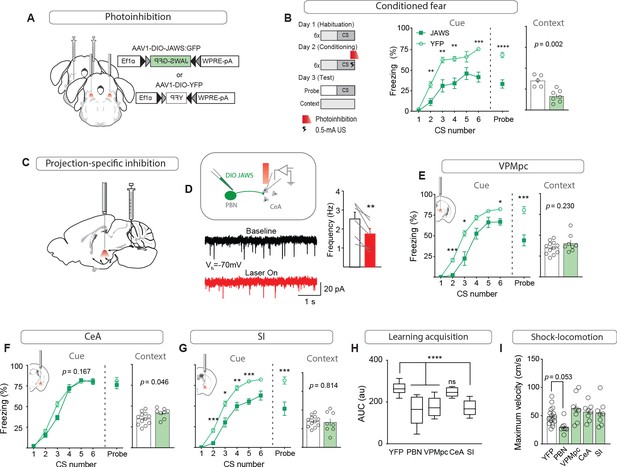
Foot shock-induced activation of the VPMpc and SI by CGRPPBN neurons contributes to associative fear learning.
(A) Bilateral injections of AAV1-DIO-JAWS:GFP or AAV1-DIO-YFP and fiber-optic cannula implants above the PBN of CalcaCre/+ mice for photoinhibition of CGRPPBN neurons. (B) Photoinhibition of CGRPPBN neurons (n = 8,5; JAWS, GFP) during foot shock delivery attenuated freezing responses both to CS and context (significant group effect in two-way ANOVA for training, F1,55 = 21.66, p=0.0007; subsequent Sidak pairwise comparisons, *p<0.05; **p<0.01; Welch’s unpaired t-test for probe and context, probe t(10.88) = 6.45, ****p<0.0001; context t(8.93) = 4.43, p=0.002). (C) Placement of fiber-optics over projection sites for projection-specific photoinhibition. (D) Representative recording of EPSCs in a CeA neuron surrounded by JAWS:GFP-positive fibers from CGRPPBN neurons. Red light decreased frequency of EPSCs in downstream cells (6 cells from two mice, paired t-test, t(5) = 4.84, **p=0.0047). (E) Photoinhibition of CGRPPBN neuron terminals in the VPMpc (n = 8,12) during footshock attenuated freezing responses to CS (significant group effect in two-way ANOVA for training, F1,18 = 28.78, p<0.0001; subsequent Sidak pairwise comparisons; probe test Welch’s unpaired t-test, t(14.41) = 4.58, ***p=0.0004) but not context (Welch’s unpaired t-test, t(11.72) = 1.27, p>0.05). (F) Effect of photoinhibition of CGRPPBN neuron terminals in the CeA (n = 8,12) during foot shock on conditioned freezing responses to cue (two-way ANOVA for training, group effect F1,18 = 2.08, p=0.167; Welch’s unpaired t-test for probe, t(16.69) = 0.76, p=0.46) or context (Welch’s unpaired t-test, t(17.17) = 2.15, p=0.046). (G) Photoinhibition of CGRPPBN neuron terminals in the SI (n = 8.12) during shock attenuated freezing responses to CS (significant group effect in two-way ANOVA for training, F1,18 = 40.52, p<0.0001; subsequent Sidak pairwise comparisons; probe test Welch’s unpaired t-test, t(11.06) = 3.70, **p=0.0035) but not the context (Welch’s unpaired t-test, t(9.83) = 0.24, p>0.05). (H) Photoinhibition of CGRPPBN neuron projections to either the VPMpc or SI during footshock attenuated associative learning (area under learning curve) as efficiently as silencing the entire population. Center line, mean; box limits, upper and lower quartiles; whiskers, min to max. Significance for one-way ANOVA, F4,45 = 15.35, p<0.0001; subsequent Sidak pairwise comparisons found no difference between PBN, VPMpc, and SI fiber-placement groups. (I) Locomotion during foot shock was not significantly affected by photoinhibition of CGRPPBN neurons (one-way ANOVA, F4,44 = 4.13, p=0.0063; subsequent Dunnett pairwise comparisons p>0.05). Bar graphs are represented as mean ± SEM. For full statistical information see Supplementary file 1.
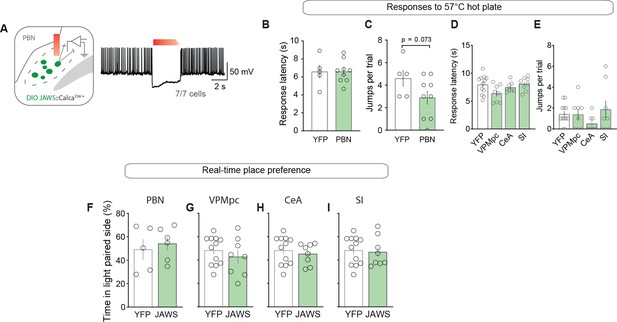
Photoinhibition of CGRPPBN neurons or projections does not affect nocifensive responses or alter place preference.
(A) Representative recording of action potentials from a CGRPPBN neuron. Red-light photostimulation (3 s on and 1 s ramp-down) effectively suppressed firing rate of CGRPPBN neurons with minimal rebound excitation (seven cells from two mice). (B) Nociceptive response latency to 57°C hot plate with photoinhibition of CGRPPBN neurons (2 s on, 1 s ramp, 1 s off for 30 s trial) (Welch’s unpaired t-test, t(7.28) = 0.07, p>0.05; n = 8.5). (C) Number of jumps on 57°C hot plate during 30 s trial with photoinhibition of CGRPPBN neurons (Welch’s unpaired t-test, t(9.02) = 2.03, p=0.0734; n = 8.5). (D) Nociceptive response latency to 57°C hot plate with photoinhibition of CGRPPBN terminals in the VPMpc, CeA, or SI (n = 8 per group) relative to controls (n = 12) (one-way ANOVA, F3.32 = 2.46, p=0.0808). (E) Number of jumps on 57°C hot plate during 30 s trial with photoinhibition of CGRPPBN terminals in the VPMpc, CeA, or SI (n = 8 per group) relative to controls (n = 12) (one-way ANOVA, F3.32 = 1.26, p=0.3057). (F–I) Photoinhibition of CGRPPBN neurons or individual projections in one side of chamber did not influence place preference (Welch’s unpaired t test; PBN t(7.22) = 0.48, p>0.05, n = 8.5); VPMpc t(12.17) = 0.81, p>0.05, n = 8.12; CeA t(17.72) = 0.62, p>0.05, n = 8.12; SI t(14.64) = 0.24, p>0.05, n = 8.12. Data represented as mean ± SEM. For full statistical information see Supplementary file 1.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus) | Calca-Cre, C57BL6/J | Carter et al., 2013 | RRID:IMSR_JAX:033168 | |
| Strain, strain background (AAV1) | pAAV1-Ef1alpha-DIO hChR2(H134R) eYFP | Carter et al., 2013 | Addgene Plasmid #20298 RRID:Addgene_20298 | |
| Strain, strain background (AAV1) | pAAV1-Ef1alpha-DIO YFP | Carter et al., 2013 | Addgene Plasmid #27056 RRID:Addgene_27056 | |
| Strain, strain background (AAV1) | pAAV1-Ef1alpha-DIO JAWS GFP | Jo et al., 2018 | RRID:Addgene_78174 | |
| Strain, strain background (AAV1) | pAAV1-nEF-Con/Fon-ChR2-mCherry | Fenno et al., 2014 | Addgene Plasmid #137142 RRID:Addgene_137142 | |
| Strain, strain background (AAV1) | pAAV1-nEF-Con/Foff 2.0-ChR2-mCherry | Fenno et al., 2014 | Addgene Plasmid #137143 RRID:Addgene_137143 | |
| Strain, strain background (rAAV2-retro) | AAV2-retro-CBA-Flippase-dsRed | This paper | N/A | palmiter@uw.edu |
| Strain, strain background (AAV9) | AAV9-Syn-ChrimsonR-tdTomato | UNC Vector Core | Cat# AV6556B A RRID:Addgene_62723 | |
| Antibody | Anti-c-Fos (Rabbit polyclonal) | Abcam | Cat#: ab190289 RRID:AB_2737414 | (1:1000) |
| Antibody | Anti-GFP (Chicken polyclonal) | Abcam | Cat#: ab13970 RRID:AB_300798 | (1:10,000) |
| Antibody | Anti-dsRed (Rabbit monoclonal) | Takara | Cat#: 632496 RRID:AB_10013483 | (1:1000) |
| Antibody | Alexa Fluor 488 anti-Chicken (Donkey monoclonal) | Jackson ImmunoResearch | Cat#: 703-545-155 RRID:AB_2340375 | (1:500) |
| Antibody | Alexa Fluor 594 anti-Rabbit (Donkey monoclonal) | Jackson ImmunoResearch | Cat#: 711-585-152 RRID:AB_2340621 | (1:500) |
| Antibody | Cy5 anti-rabbit (Donkey monoclonal) | Jackson ImmunoResearch | Cat#: 711-175-152 RRID:AB_2340607 | (1:500) |
| Other | Normal donkey serum | Jackson ImmunoResearch | Cat#:017-000-121 RRID:AB_2337258 | |
| Other | Pulse oximeter | STARR Life Sciences | Part#: 015000 | |
| Other | Pulse oximeter collar sensor | STARR Life Sciences | Part#: 015021 | |
| Software, algorithm | MouseOxPlus conscious applications module | STARR Life Sciences | Part#: 015002 | |
| Software, algorithm | Ethovision XT 10 | Noldus Technology | www.noldus.com RRID:SCR_000441 |
Additional files
-
Supplementary file 1
Full statistical information for all data figures.
- https://cdn.elifesciences.org/articles/59799/elife-59799-supp1-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/59799/elife-59799-transrepform-v2.pdf





