Compartment-specific opioid receptor signaling is selectively modulated by different dynorphin peptides
Figures
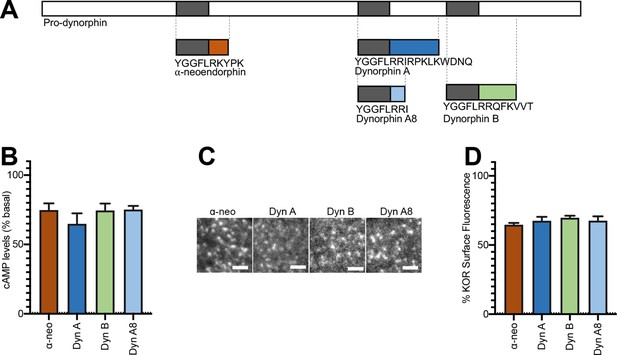
Initial activation and internalization of KOR by Dynorphins are comparable.
(A) Schematic of the regions of Pro-dynorphin from which Dynorphin A8 (Dyn A8), Dynorphin A (Dyn A), Dynorphin B (Dyn B), and α-neoendorphin (α-neo) peptides are generated, showing that Dyn A and Dyn B are processed from adjacent regions. (B) Dyn A8, Dyn A, Dyn B, and α-neo (1 μM) inhibit intracellular cAMP levels to a similar extent in PC12 cells stably expressing SpH-KOR. Values were normalized to basal cAMP measurements in the absence of peptide, which were set as 100% (mean ± SEM shown). (C) Representative TIR-FM images of PC12 cells stably expressing SpH-KOR treated with Dyn A8, Dyn A, Dyn B, and α-neo (1 μM) show roughly similar agonist-mediated receptor clustering at the cell surface following 1 min treatment. Scale bar = 2 µm. (D) Quantitation of the loss of surface SpH-KOR fluorescence, as an index of internalization, after 5 min of treatment with each peptide (1 μM), normalized to surface fluorescence before agonist treatment, show similar levels of internalization for all four peptides (mean ± SEM shown, Dyn A: n = 10 cells; Dyn B: n = 10 cells; Dyn A8: n = 10 cells; ɑ-neo: n = 11 cells).
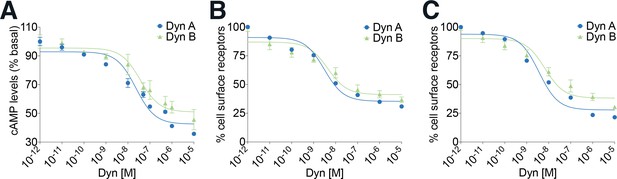
Ligand-mediated decreases in intracellular cAMP levels and endocytosis of KOR saturates at 1 µM for Dyn A and Dyn B.
(A) Dyn A- and Dyn B-mediated decreases in intracellular cAMP levels were measured by carrying out doseresponse curves (0–10 µM) in PC12 cells stably expressing SpH-KOR treated for 30 min at 37°C. (B) Dyn A- and Dyn B-mediated changes in surface receptor levels were measured by ELISA by carrying out dose–response curves (0–10 µM) in PC12 cells stably expressing SpH-KOR treated for 30 min at 37°C. (C) Dyn A- and Dyn B-mediated changes in surface receptor levels were measured by ELISA by carrying out dose–response curves (0–10 µM) in CHO cells stably expressing Flag-KOR treated for 60 min at 37 °C.
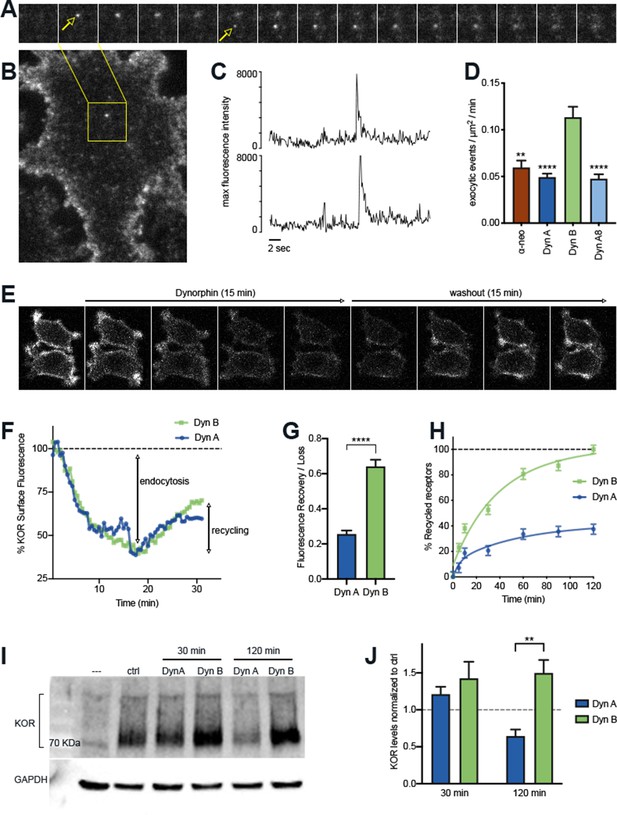
The post-endocytic fate of KOR is determined by the specific Dynorphin that activates it.
(A) Frames from a time lapse movie of a representative region of a PC12 cell stably expressing SpH-KOR (SpH-KOR cells) shown in (B), treated for 5 min with Dyn B (1 μM) and imaged in TIR-FM, showing two examples of exocytic events (white arrows in A) associated with KOR recycling. (C) Fluorescence traces of the two exocytic events, arbitrary units, showing a characteristic abrupt increase in maximum fluorescence intensity followed by exponential decay. (D) Quantitation of the number of exocytic events/µm2/min showing a significant increase for Dyn B compared to the other peptides (mean ± SEM, **p<0.01, ****p<0.0001 in multiple comparisons after ANOVA, n = 14, 39, 52, and 33 cells for α-neo, Dyn A, Dyn B, and Dyn A8, respectively). (E) Ensemble SpH-KOR surface fluorescence measured over time using confocal microscopy shows a decrease in fluorescence upon agonist addition because of quenching of internalized SpH-KOR, and an increase upon peptide washout as receptors recycle to the surface and SpH-KOR is dequenched. (F) Quantification of change in ensemble surface fluorescence over 30 min following treatment with Dyn A or Dyn B (1 μM), normalized to fluorescence before agonist addition, showing the loss during endocytosis and increase during recycling. (G) Quantitation of the amount of SpH-KOR recycled, normalized to the amount endocytosed after Dyn A or Dyn B treatment B (1 μM), shows that a higher amount of receptor is recycled after Dyn B washout (mean ± SEM, ****p<0.0001 by Mann–Whitney; n = 33 and 30 fields for Dyn A and Dyn B, respectively). (H) Recycling of SpH-KOR to the cell surface after treatment with 100 nM Dyn A or Dyn B measured by ELISA shows a much higher rate and extent of recycling after Dyn B washout. (I) Representative immunoblot of total receptor levels in SpH-KOR cells treated with cycloheximide (3 µg/ml) for 2 hr prior to Dyn A or Dyn B treatment (1 μM) for the indicated times show receptor loss after 120 min of Dyn A but not Dyn B treatment. GAPDH is used as a control. (J) Quantification of total receptor levels normalized to untreated control cells under each condition (**p<0.01 by post hoc comparison after two-way ANOVA; n = 5).
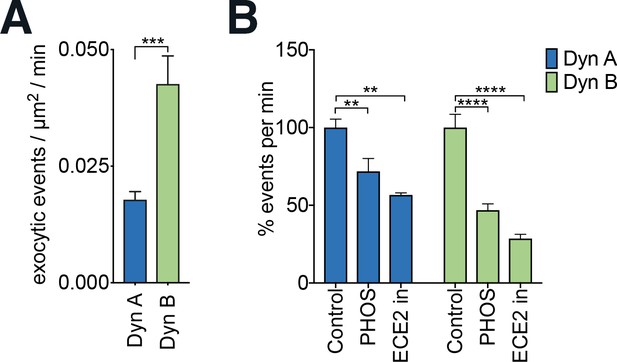
The differences in KOR recycling between Dyn A and Dyn B cannot be explained entirely by peptide degradation.
(A) Quantitation of the number of exocytic events/μm2/min in SpH-KOR PC12 cells shows a significantly higher number of events for Dyn B compared to Dyn A (1 µM) in the continued presence of protease inhibitors (***p<0.001, in unpaired t-test n = 9 and 6 cells for Dyn B and Dyn A, respectively). (B) Quantitation of the percent change of exocytic events in PC12 SpH-KOR cells treated with Dyn A or Dyn B in the presence or absence of 10 µM phosphoramidon (PHOS), a neprilysin/ECE inhibitor, or 20 μM S136492, an ECE2 inhibitor (ECE2 inh). Both inhibitors decreased % events/minute for both Dyn A and Dyn B, suggesting that ECE2 inhibition on its own cannot explain the differences between the peptides.
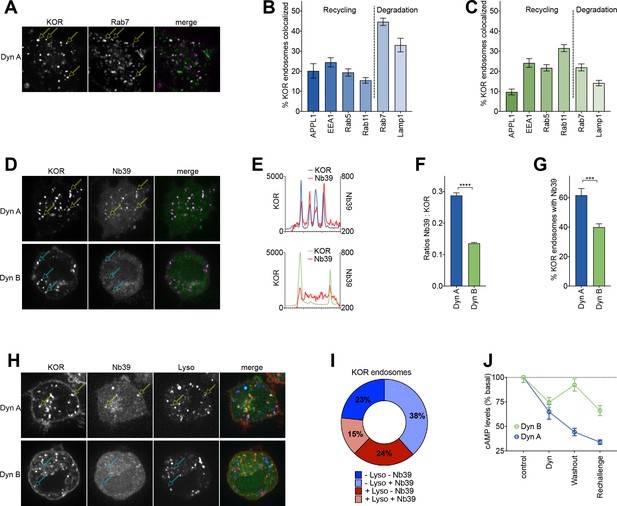
Dyn A selectively drives KOR signaling from late endosomal compartments.
(A) Representative image of a PC12 cell expressing FLAG-KOR and Rab7-GFP, treated with 1 µM Dyn A for 20 min. Yellow arrows denote KOR endosomes that colocalize with Rab7. (B) SpH-KOR cells treated with 1 μM Dyn A for 20 min were fixed and processed for immunofluorescence with the noted markers. Quantitation, across multiple cells, of the percentage of KOR containing endosomes that colocalize with each of the endosomal markers is noted. KOR primarily localizes in Rab7 and Lamp1 positive late endosomes after Dyn A (n = 8, 10, 9, 11, 20, and 17 cells for APPL1, EEA1, Rab5, Rab11, Rab7, and Lamp1, respectively). (C) A similar quantitation of immunofluorescence images after Dyn B treatment (1 μM for 20 min) shows that KOR localizes less with late endosomes, and more with markers of early/recycling endosomes (n = 18, 16, 15, 18, 23, and 29 cells for APPL1, EEA1, Rab5, Rab11, Rab7, and Lamp1, respectively). (D) Representative images of PC12 cells expressing FLAG-KOR and Nb39, imaged live after treatment with 1 µM Dyn A or Dyn B for 20 min. Yellow arrows in Dyn A show KOR endosomes that recruited Nb39, while cyan arrows in Dyn B show KOR endosomes that do not show obvious recruitment of Nb39. (E) Linear profile plots of fluorescence of KOR and Nb39, measured along lines drawn across regions of the cell with KOR endosomes after treatment with 1 μM Dyn A or Dyn B for 20 min, show that Nb39 fluorescence increases along with KOR in Dyn A, but less noticeably with Dyn B. (F) Ratios of integrated fluorescence of Nb39:KOR in endosomes identified by 3D object analysis show higher amounts of Nb39 relative to KOR in Dyn A-treated cells (****p<0.0001 by Mann–Whitney, n = 766 and 800 endosomes for Dyn A and Dyn B, respectively). (G) Quantitation of the percentage of KOR endosomes per cell with a noticeable increase in Nb39 fluorescence above background shows a higher fraction of KOR endosomes recruited Nb39 in 1 µM Dyn A-treated cells (***p<0.001 by Mann–Whitney, n = 11 and 14 cells for Dyn A and Dyn B, respectively). (H) Representative images of PC12 cells expressing FLAG-KOR and Nb39 and labeled with LysoTracker imaged live after treatment with 1 μM Dyn A or Dyn B for 20 min. Yellow arrows in Dyn A show KOR endosomes that recruited Nb39 that were also labeled with Lysotracker, while cyan arrows in Dyn B show KOR endosomes that do not show obvious labeling with Nb39 and Lysotracker. (I) The average composition of total KOR endosomes that are positive for ±Nb39 and ±Lysotracker after 20 min treatment with Dyn A (1 μM). n = 10 cells. −Lyso/−Nb39 = 23.4 ± 8.1%; −Lyso/+Nb39 = 38.4% ± 17.4%; +Lyso/−Nb39 = 23.7 ± 11.4%; +Lyso/+Nb39 = 14.6 ± 5.1%. (J) cAMP levels after initial Dynorphin treatment (1 μM) for 5 min, washout for 25 min, or a Dynorphin rechallenge (1 μM) at end of the washout, show comparable initial cAMP inhibition by both Dyn A and Dyn B, but persistent signaling by Dyn A after agonist washout.
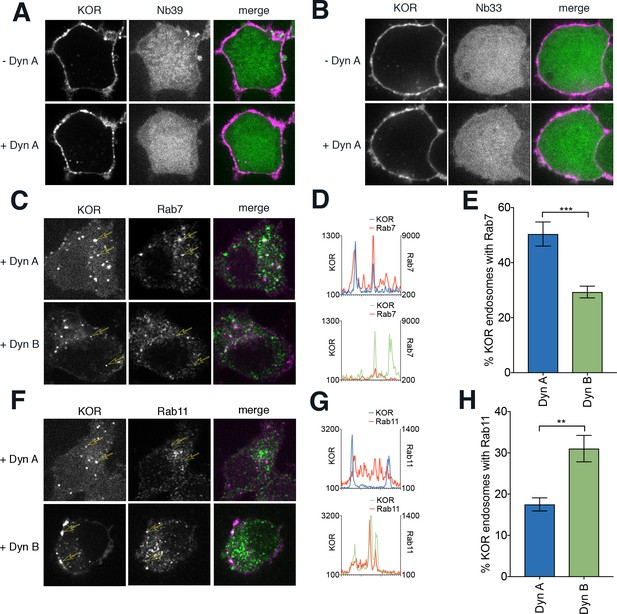
Differential receptor sorting between Dyn A and Dyn B persists even after agonists are washed out from the surface.
(A) Representative images of PC12 cells expressing FLAG KOR treated with Dyn A in the presence of 40 µM Dyngo4A for 30 min to block endocytosis. KOR (magenta in merge) fluorescence is restricted to the surface with little endosomal KOR after 20 min Dyn A (1 µM), and no recruitment of Nb39 (green) to internal endosomes. (B) A similar Dyngo4a treatment blocked recruitment of Nb33 to internal endosomes after 20 min Dyn A (1 µM). (C) Representative images of cells labeled live with anti-FLAG antibodies for surface KOR, treated for 5 min with 1 µM Dyn A or Dyn B followed by a 25 min washout, then fixed and stained for Rab7 to mark late endosomes. (D) Linear profile plots of fluorescence for KOR and Rab7, measured along lines drawn across regions of the cells in C, show that Rab7 fluorescence spikes correlate with KOR spikes in Dyn A, but less with Dyn B. (E) Quantitation of the percentage of KOR endosomes/cell colocalizing with Rab7 shows a higher fraction of KOR endosomes recruited Rab7 in Dyn A-treated cells (***p<0.001 by Mann–Whitney, n = 10 and 11 cells for Dyn A and Dyn B, respectively). (F) Representative images of cells labeled live with anti-FLAG antibodies for surface KOR, treated for 5 min with 1 µM Dyn A or Dyn B followed by a 25 min washout, then fixed and stained for Rab11 to mark recycling endosomes. (G) Linear profile plots of fluorescence for KOR and Rab11, measured along lines drawn across regions of the cells in F, show that Rab11 fluorescence increases along with KOR in Dyn B, but less noticeably with Dyn A. (H) Quantitation of the percentage of KOR endosomes/cell colocalizing with Rab11 shows a higher fraction of KOR endosomes recruited Rab11 in Dyn B-treated cells (**p<0.01 by Mann–Whitney, n = 9 and 10 cells for Dyn A and Dyn B, respectively).
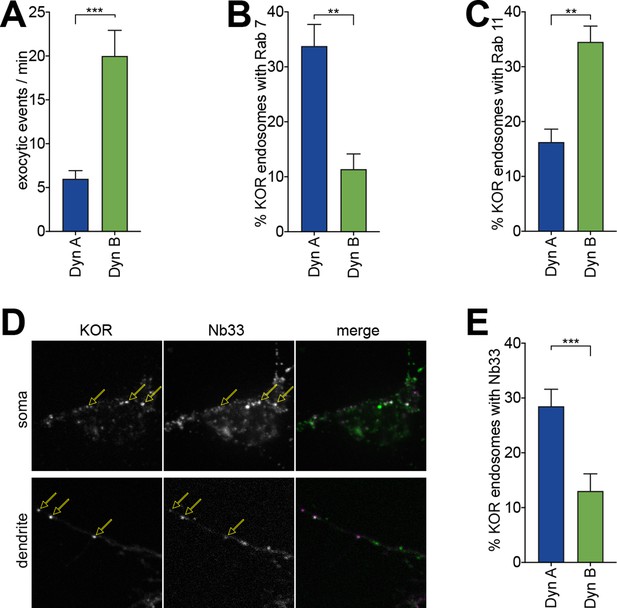
Dyn A-specific late endosomal localization and signaling is conserved in striatal neurons.
(A) The number of discrete exocytic events quantitated in rat medium spiny neuron (MSN) expressing SpH-KOR shows increased recycling for 1 μM Dyn B compared to Dyn A (***p<0.001, n = 8 cells). (B) Quantification of the percentage of KOR endosomes colocalized with Rab7 in MSN expressing SpH-KOR treated with 1 μM Dyn A or Dyn B for 30 min (**p<0.01, n = 5 cells for both). (C) Quantification of the percentage of KOR endosomes colocalized with Rab11 in MSN expressing SpH-KOR treated with 1 μM Dyn A or Dyn B for 30 min (**p<0.01, n = 5 and 9 cells for Dyn A and Dyn B, respectively). (D) Colocalization of FLAG-KOR and Nb33-GFP in the soma and in dendrites of MSNs treated with 1 μM Dyn A for 30 min, seen by confocal microscopy. Yellow arrows show KOR endosomes that recruit Nb33. (E) Quantitation of the percentage of KOR endosomes/cell with a noticeable increase in Nb33 fluorescence above background shows that a higher fraction of KOR endosomes recruited Nb33 in Dyn A-treated cells (1 μM for 30 min; ***p=0 < 0.001, n = 10 cells). All p-values were from non-parametric Mann–Whitney tests.
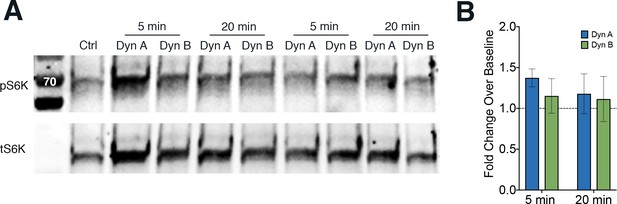
mTOR signaling does not show significant differences between Dyn A and Dyn B.
(A) Representative blots showing phosphorylated (pS6K) and total (tS6K) S6K levels in PC12 cells stably expressing SpH-KOR treated with 1 µM Dyn A or Dyn B for 5 and 20 min. (B) Quantitation of the fold change over Ctrl baseline to measure mTOR activation: pS6K signal divided by tS6K signal in cells treated with Dyn A or Dyn B for 5 or 20 min. n = 5 biological replicates.
BAM22 drives noticeably less KOR recycling than DynB, even though they are both sensitive to ECE2 inhibition.
(A) Recycling of HA-KOR to the cell surface after 30 min treatment with 100 nM Dyn B or BAM22 and washout for 0-120 min. Cell surface receptors were quantified by ELISA as described in Methods. Levels of cell surface receptors before agonist treatment were taken as 100% for each individual experiment. % recycled receptors were calculated by subtracting surface receptors at t = 0 (30 min internalization) from each recycling time point. (B) Cell surface receptors quantified after peptides were washed out for 120 min in media without or with 20 μm or the ECE2 inhibitor (S136492). The data represent mean ± SEM from three independent experiments carried out in triplicate.
Dyn B is dominant over Dyn A for KOR endocytic trafficking.
(A) Quantitation of the number of exocytic events/μm2/min, as in Figure 2, in SpH-KOR PC12 cells co-treated with 1μM of Dyn A and 1μM of Dyn B, compared to either on its own. The co-treatment mimics Dyn B. (B) SpH-KOR PC12 cells were treated with 100nM Dyn A, Dyn B or a combination of both for 30 min. Peptides were washed out and cells incubated for 60 min in media without the agonist. Surface receptors were measured by ELISA as described in Methods. Percentage of recycled receptors were calculated by subtracting surface receptors at t = 0 (30 min internalization; 0% recycling) from each recycling time point. Co-treatmentcauses KOR recycling comparable to Dyn B alone. The data represent mean ± SEM from threindependent experiments carried out in triplicate.
Tables
Displacement binding parameters for Dynorphin peptides at PC12 SPH-KOR and CHO-KOR cells.
| PC12 SpH-KOR cells | CHO-KOR cells | |||||||
|---|---|---|---|---|---|---|---|---|
| Ligand | Low Ki (nM) | High Ki (nM) | % Bmax at 10 μM | nH | Low Ki (nM) | High Ki (nM) | % Bmax at 10 μM | nH |
| Dyn A8 | 56.9 ± 0.3 | 0.020 ± 1.90 | 18.30 ± 1.48 | 36.9% | 563 ± 0.1 | 0.41 ± 0.15 | 21.51 ± 1.54 | 41.6% |
| Dyn A17 (Dyn A) | 52.4 ± 0.3 | 0.016 ± 0.47 | 15.22 ± 1.56 | 39.1% | 119 ± 0.2 | 0.26 ± 0.54 | 18.72 ± 1.09 | 29.4% |
| Dyn B13 (Dyn B) | 37.8 ± 0.2 | 0.010 ± 0.31 | 11.13 ± 1.07 | 33.4% | 355 ± 0.1 | 0.38 ± 0.11 | 17.16 ± 0.82 | 42.2% |
| a-neo-endorphin | 39.1 ± 0.1 | 0.011 ± 0.20 | 13.32 ± 1.84 | 38.7% | 591 ± 0.1 | 0.18 ± 0.34 | 20.55 ± 1.57 | 21.4% |
Additional files
-
Source data 1
Numerical data.
- https://cdn.elifesciences.org/articles/60270/elife-60270-data1-v2.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/60270/elife-60270-transrepform-v2.docx





