A reduction in voluntary physical activity in early pregnancy in mice is mediated by prolactin
Figures
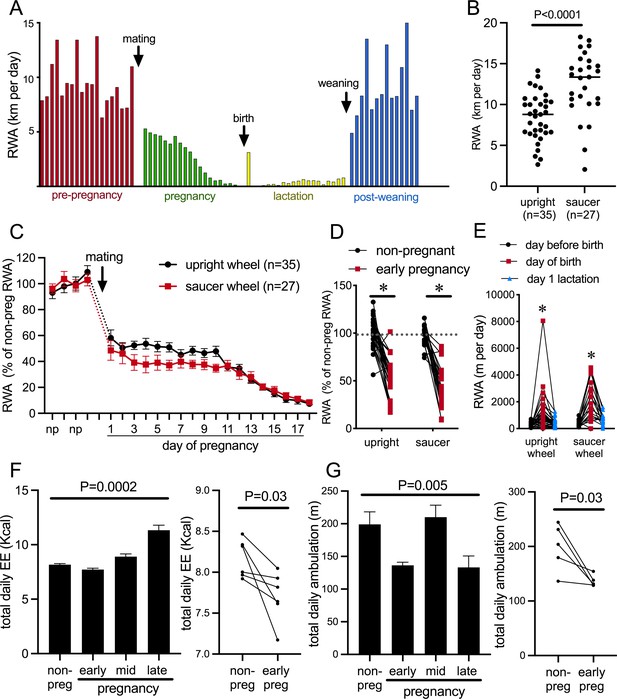
Pregnancy rapidly decreases voluntary running wheel activity (RWA).
(A) Daily RWA of one representative mouse through one cycle of reproduction (pre-pregnancy estrous cycles, pregnancy, lactation, and after the weaning of her pups). Each bar represents RWA on 1 day. (B) RWA in non-pregnant, non-lactating (control state, termed ‘non-pregnant’) female mice with access to a traditional upright wheel or a low-profile saucer wheel. (C) RWA in female mice, with either traditional upright wheel or a low-profile saucer wheel, before and after successful mating. RWA activity for each mouse is expressed as a percentage of their average daily levels pre-pregnancy. (D) Daily RWA for the first three days of pregnancy expressed as a percentage of each mouse’s RWA level pre-pregnancy. Dotted line indicates mean RWA level of non-pregnant groups, and * indicates a significant difference between non-pregnant RWA and early pregnancy RWA (effect of physiological state p<0.0001, post hoc non-pregnant vs. early pregnancy upright wheel: p<0.0001, saucer wheel: p<0.0001), n values same as C. (E) RWA for the 24 hr before giving birth, the 24 hr period that included birth, and the first day of lactation for mice housed with either an upright or saucer running wheel (effect of time p<0.0001, post hoc test: * indicates significantly different to other time points) (F, G) (Left) Bars represent total daily energy expenditure (EE; F) and total daily home cage ambulation (G) in the non-pregnant state and different time points during pregnancy (early = days 2–7, mid = days 8–13, late = days 14–18; repeated measures one-way ANOVA, n = 6). (Right) Points and lines represent the change in total daily EE (F) and total daily home cage ambulation (G) for each individual mouse between the non-pregnant state and early pregnancy (days 2–7; t-test, F: n = 6, G: n = 5).
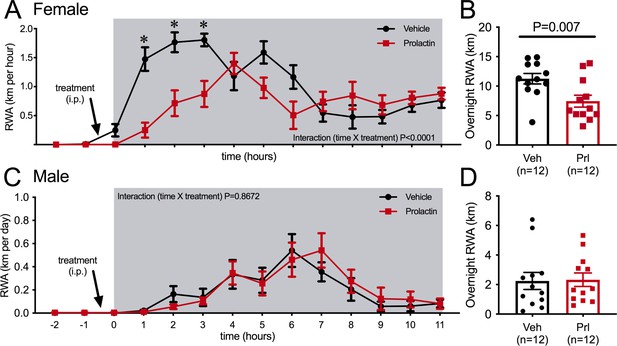
Acute effects of exogenous prolactin on physical activity.
(A) Acute prolactin treatment significantly reduced running wheel activity (RWA) in female mice (A, B) but not in male mice (C, D). (A, C) Lines indicate RWA per hour in non-pregnant female mice (metestrous phase of estrous cycle for female mice) or male mice treated with either vehicle or prolactin (5 mg/kg intraperitoneally [i.p.]) 30 min before the start of the dark phase (dark phase indicated by gray shading). All mice (n = 12 per sex) received both treatments in a randomized order. * indicates time points that showed a significant difference (post hoc analysis p<0.05). (B, D) Bars represent total 12 hr RWA following either vehicle or prolactin (5 mg/kg i.p.) as described above.
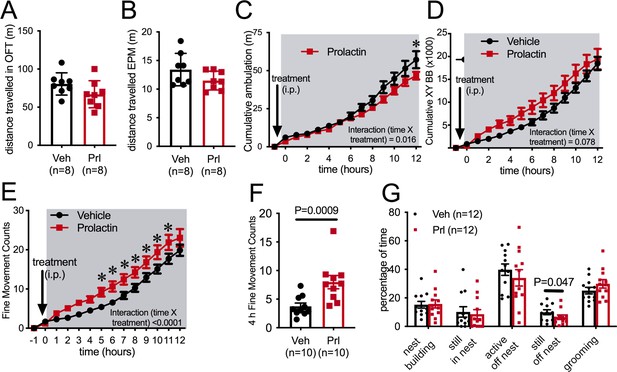
Prolactin does not acutely decrease ambulation.
Bars represent total distanced traveled in an open field test (OFT) of 30 min duration (A) or elevated plus maze (EPM) of 5 min duration (B) carried out in the dark phase of the light cycle under dark conditions following either vehicle or prolactin (5 mg/kg intraperitoneally [i.p.]) treatment in female mice in the metestrous stage of the estrous cycle (n = 8 per group; t-test). (C–F) Acute prolactin had a subtle effect on ambulation in home cage. Lines represent cumulative ambulation (C), total X + Y beam breaks (D) and fine movement (activity that is not ambulation) (E) of female mice in the metestrous phase of the estrous cycle in home cage during the dark phase of the light cycle after vehicle or prolactin (5 mg/kg i.p.) treatment (repeated measures ANOVA). (F) Bars represent total fine movement counts in the first 4 hr following the start of the dark phase (paired t-test). (G) Acute prolactin did not greatly impact on time mice spent engaging in different behaviors in their home cages (paired t-test: ‘still, off nest’ vehicle vs. prolactin treatment p = 0.047 [n = 11 due to exclusion of data due to outlier; ROUT outlier test, outlier much higher value than the rest of the group], all other behaviors p>0.05). Bars represent percentage of time mice were engaged in various activities in their home cages (total test time 40 min) following vehicle or prolactin (5 mg/kg i.p.) treatment (n = 12 mice, all mice received both vehicle and prolactin in a randomized order, paired t-test).
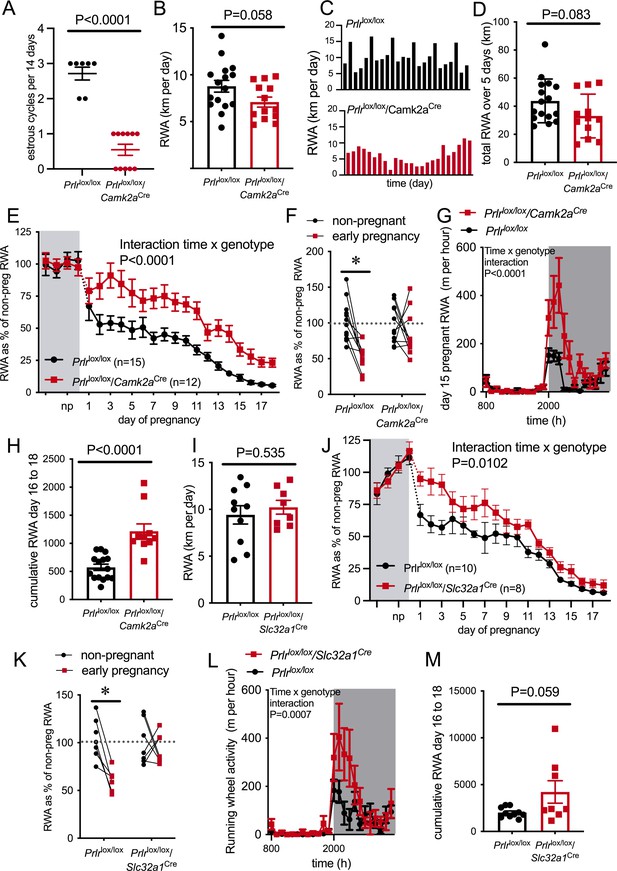
Pregnancy-induced suppression of running wheel activity (RWA) is attenuated in mice lacking Prlr in forebrain neurons.
(A) Number of estrous cycles in a continuous 12-day period in Prlrlox/lox/Camk2aCre mice (n = 7–11). (B) Average daily RWA in non-pregnant state (Prlrlox/lox/Camk2aCre n = 14, Prlrlox/lox, n = 15). (C) Representative examples of daily RWA in one control (Prlrlox/lox) and one Prlrlox/lox/Camk2aCre mouse over a 24-day period. (D) Cumulative RWA over the 5 days prior to mating in Prlrlox/lox/Camk2aCre (n = 14) and controls (Prlrlox/lox, n = 15). (E) RWA activity before and after successful mating in Prlrlox/lox/Camk2aCre mice. RWA activity for each mouse is expressed as a percentage of their individual average daily levels in the non-pregnant state (Prlrlox/lox/Camk2aCre n = 12, Prlrlox/lox, n = 15, total daily RWA per animal shown in Figure 4—source data 1). (F) Change in RWA from non-pregnant state to early pregnancy in Prlrlox/lox/Camk2aCre mice. Dotted line indicates mean non-pregnant RWA for each group. (G) 24 hr RWA profile from day 15 pregnant Prlrlox/lox/Camk2aCre mice (n = 13) and controls (n = 15), RWA per animal shown in Figure 4—source data 3. Gray indicates dark phase of light cycle. (H) Cumulative RWA over days 16–18 pregnancy (Prlrlox/lox/Camk2aCre n = 10, Prlrlox/lox, n = 15). (I) Average daily RWA in non-pregnant state (Prlrlox/lox/Slc32a1Cre n = 8, Prlrlox/lox, n = 10). (J) RWA activity before and after successful mating in Prlrlox/lox/Slc32a1Cre mice. RWA activity for each mouse is expressed as a percentage of their individual average daily levels in the non-pregnant state (Prlrlox/lox/Slc32a1Cre, n = 8,Prlrlox/lox, n = 10, total daily RWA per animal shown in Figure 4—source data 2). (K) Change in RWA from non-pregnant state to early pregnancy in Prlrlox/lox/Slc32a1Cre mice. Dotted line indicates mean non-pregnant RWA for each group. (L) 24 hr RWA profile from day 15 pregnant Prlrlox/lox/Slc32a1Cre mice (n = 8) and controls (n = 10), RWA per animal shown in Figure 4—source data 3. Gray indicates dark phase of light cycle. (M) Cumulative RWA over days 16–18 pregnancy (Prlrlox/lox/Slc32a1Cre, n = 8, Prlrlox/lox, n = 10).
-
Figure 4—source data 1
Each line (gray = control, red = Prlrlox/lox/Camk2aCre) represents total daily running wheel activity (RWA) from each individual mouse in the non-pregnant state (4 days before mating) and during pregnancy (days 1–18).
Black lines show the mean ± SEM for the group.
- https://cdn.elifesciences.org/articles/62260/elife-62260-fig4-data1-v2.tif
-
Figure 4—source data 2
Each line (gray = control, red = Prlrlox/lox/Slc32a1Cre) represents total daily running wheel activity (RWA) from each individual mouse in the non-pregnant state (4 days before mating) and during pregnancy (days 1–18).
Black lines show the mean ± SEM for the group.
- https://cdn.elifesciences.org/articles/62260/elife-62260-fig4-data2-v2.tif
-
Figure 4—source data 3
Each line black = control, red = Prlrlox/lox/Camk2aCre (top) or Prlrlox/lox/Slc32a1Cre (bottom) represents hourly running wheel activity (RWA) from each individual mouse on day 15 of pregnancy.
Shaded area represents dark phase of the light cycle.
- https://cdn.elifesciences.org/articles/62260/elife-62260-fig4-data3-v2.tif
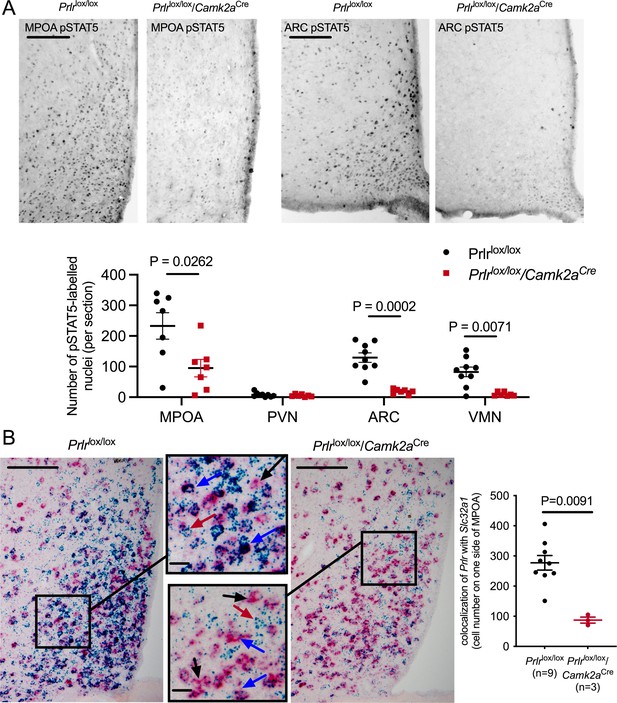
Conditional deletion of Prlr in forebrain neurons.
(A) Representative images of prolactin-induced pSTAT5 (black nuclear staining) from the medial preoptic area (MPOA) and arcuate nucleus of Prlrlox/lox/Camk2aCre and control (Prlrlox/lox) mice (scale bar = 50 µm). Data show quantification of the mean number of prolactin-induced pSTAT5 (black nuclear staining) per section per animal in various hypothalamic regions, showing functional loss of Prlr signaling in Prlrlox/lox/Camk2aCre, n = 7–10 per group. (B) Data show mean number of Slc32a1 mRNA/Prlr mRNA-positive cells per section per animal in the MPOA of Prlrlox/lox/Camk2aCre and control (Prlrlox/lox) female mice. Images show representative dual label RNAscope in situ hybridization for Prlr mRNA (blue) and Slc32a1 mRNA (red/pink) in the MPOA of Prlrlox/lox/Camk2aCre and control (Prlrlox/lox) female mice. Arrows indicate: black: Slc32a1 mRNA positive, Prlr mRNA negative; blue: Slc32a1 mRNA positive, Prlr mRNA positive; red: Slc32a1 mRNA negative, Prlr mRNA positive (scale bar = 50 µm, inset scale bar = 10 µm).
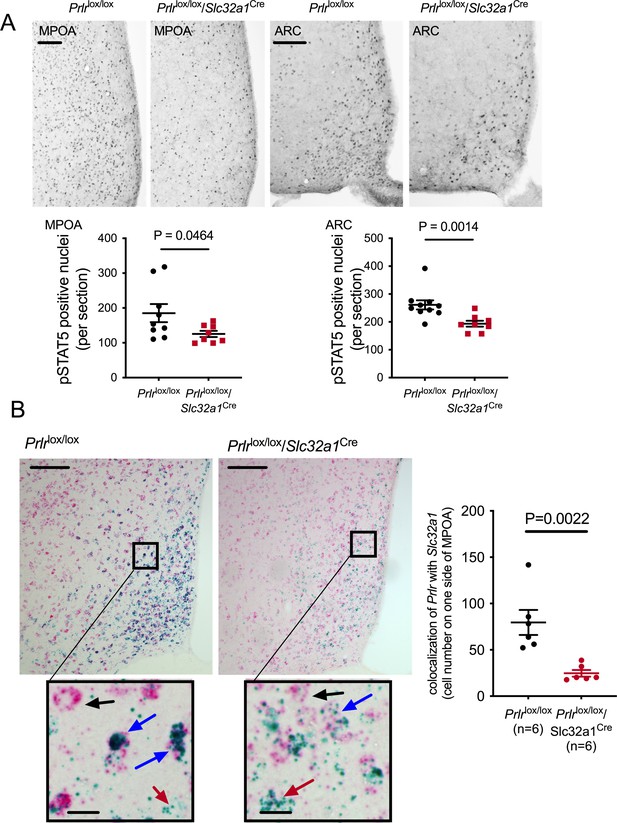
Conditional deletion of Prlr in Slc32a1-expressing neurons.
(A) Representative images of prolactin-induced pSTAT5 (black nuclear staining) from the medial preoptic area (MPOA) and arcuate nucleus of Prlrlox/lox/Slc32a1Cre and control (Prlrlox/lox) mice (scale bar = 50 µm). Data show quantification of the mean number of prolactin-induced pSTAT5 (black nuclear staining) per section per animal in various hypothalamic regions showing functional loss of Prlr signaling in Prlrlox/lox/Slc32a1Cre female mice alongside representative images, n = 7–10 per group. Note the degree of knockout is not as pronounced as seen in the Prlrlox/lox/Camk2aCre, indicative of significant populations of non-GABA neurons in these nuclei that also express the Prlr. (B) Images show dual label RNAscope in situ hybridization for Prlr mRNA (blue) and Slc32a1C mRNA (red/pink) in the MPOA of Prlrlox/lox/Slc32a1Cre and control (Prlrlox/lox) female mice. Arrows indicate: black: Slc32a1 mRNA positive, Prlr mRNA negative; blue: Slc32a1 mRNA positive, Prlr mRNA positive; red: Slc32a1 mRNA negative, Prlr mRNA positive (scale bar = 50 µm, inset scale bar = 10 µm). Data show quantification of mean number of Prlrlox/lox/Slc32a1Cre mRNA-positive cells per section per animal in the MPOA of Prlrlox/lox/Slc32a1Cre and control (Prlrlox/lox) female mice.

Pregnancy parameters of mice with conditional deletion of Prlr in most forebrain neurons or in GABA neurons.
(A) Graphs show representative examples of estrous cycle stages over a continuous 12-day period from one control (Prlrlox/lox) (left) and one Prlrlox/lox/Camk2aCre (right) female mouse. (B) Non-pregnant body weight (BW) in Prlrlox/lox/Camk2aCre (n = 14) and control (Prlrlox/lox) female mice (n = 17) of the same age. (C) Number of pups alive on day 4 of lactation in Prlrlox/lox/Camk2aCre and control mice. Note that most animals displayed adequate maternal behavior to raise their pups. (D) Uterus weight on day 15/16 of pregnancy in Prlrlox/lox/Camk2aCre (n = 7–8 per genotype). (E) Fetus number on 15/16 of pregnancy Prlrlox/lox/Camk2aCre (n = 7–8 per genotype). (F) Cumulative weight gain in pregnancy for Prlrlox/lox/Camk2aCre and controls. (G) BW of day 17 pregnant Prlrlox/lox/Camk2aCre and controls (n = 14–17). (H) Daily food intake in pregnancy in Prlrlox/lox/Camk2aCre and controls (n = 14–17). (I) Percentage time spent in open arms of an elevated plus maze (EPM) on day 15/16 of pregnancy in Prlrlox/lox/Camk2aCre and controls (n = 7–8). (J) Non-pregnant BW in Prlrlox/lox/Slc32a1Cre (n = 8) and control (Prlrlox/lox) female mice (n = 10) of the same age. (K) Number of pups alive on day 4 of lactation in Prlrlox/lox/Slc32a1Cre (n = 9) and control mice (n = 10). (L) Percentage time spent in open arms of an EPM on day 15/16 of pregnancy in Prlrlox/lox/Slc32a1Cre (n = 11) and controls (n = 13). (M) Cumulative weight gain in pregnancy for Prlrlox/lox/Slc32a1Cre and controls (n = 9–10). (N) Daily food intake in pregnancy in Prlrlox/lox/Slc32a1Cre and controls (n = 9–10). (O) BW of day 17 pregnant Prlrlox/lox/Slc32a1Cre and controls (n = 9–10).
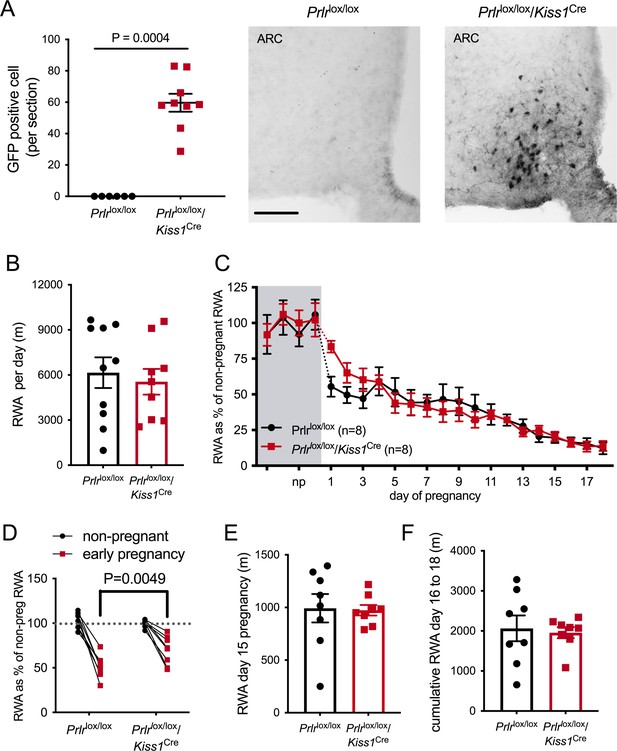
Effect of Prlr deletion from arcuate nucleus kisspeptin neurons on running wheel activity (RWA) during pregnancy.
(A) Data represent number of green fluorescent protein (GFP)-positive cells (black staining), indicative of Prlr gene deletion, in the arcuate nucleus of Prlrlox/lox/Kiss1Cre female mice alongside representative images, n = 6–9 per group (scale bar = 50 µm). (B) Average daily RWA in non-pregnant state (Prlrlox/lox/Kiss1Cre, n = 9, Prlrlox/lox, n = 10). (C) RWA activity before and after successful mating in Prlrlox/lox/Kiss1Cre mice. RWA activity for each mouse is expressed as a percentage of their individual average daily levels in the non-pregnant state (Prlrlox/lox/Kiss1Cre n = 8, Prlrlox/lox n = 8, total daily RWA per animal shown in Figure 5—source data 1). (D) Change in RWA from non-pregnant state to early pregnancy in Prlrlox/lox/Kiss1Cre mice. Dotted line indicates mean non-pregnant RWA for each group. (E) RWA on day 15 pregnancy for Prlrlox/lox/Kiss1Cre mice (n = 8) and controls (n = 8). (F) Cumulative RWA over days 16–18 pregnancy (Prlrlox/lox/Kiss1Cre, n = 8, Prlrlox/lox, n = 8). Note that in contrast to Prlrlox/lox/Camk2aCre and Prlrlox/lox/Slc32a1Cre models, above, there was no sustained difference in levels of RWA in mid and late pregnancy.
-
Figure 5—source data 1
Each line (gray = control, red = Prlrlox/lox/Kiss1Cre) represents total daily running wheel activity (RWA) from each individual mouse in the non-pregnant state (4 days before mating) and during pregnancy (days 1–18).
Black lines show the mean ± SEM for the group.
- https://cdn.elifesciences.org/articles/62260/elife-62260-fig5-data1-v2.tif
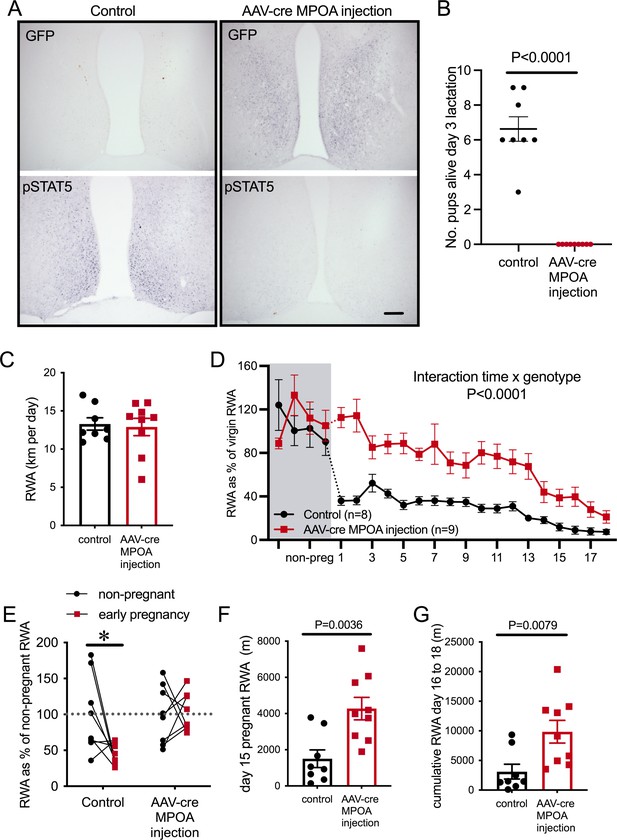
Prolactin-sensitive neurons in the medial preoptic area (MPOA) mediate pregnancy-induced suppression of running wheel activity (RWA).
(A) Representative images of coronal sections through the MPOA showing immunohistochemical staining for green fluorescence protein (GFP) (top) and prolactin-induced pSTAT5 (bottom). Control injection Prlrlox/lox mice showed an absence of GFP and extensive pSTAT5 while AAV-cre-injected Prlrlox/lox had extensive GFP and absence of pSTAT5 in the MPOA, indicative of loss of Prlr. (B) As reported previously (Brown et al., 2017), AAV-cre-mediated deletion of Prlr from the MPOA resulted in failure of maternal behavior, and no pups from MPOA AAV-cre injection Prlrlox/lox mice survived to day 3 of lactation. (C) Average daily RWA in non-pregnant state. (D) RWA activity before and after successful mating in mice lacking Prlr in the MPOA following AAV-cre injection (AAV-cre MPOA injection). RWA activity for each mouse is expressed as a percentage of their individual average daily levels in the non-pregnant state (control injection, n = 8, AAV-cre injection, n = 9, total daily RWA per animal shown in Figure 6—source data 1). (E) Change in RWA from non-pregnant state to early pregnancy in AAV-cre-injected and control-injected mice. Dotted line indicates mean non-pregnant RWA for each group. (F) RWA on day 15 pregnancy for AAV-cre-injected mice (n = 9) and controls (n = 8). (G) Cumulative RWA over days 16–18 pregnancy for AAV-cre-injected mice (n = 9) and controls (n = 8).
-
Figure 6—source data 1
Each line (gray = control, red = AAV medial preoptic area[ MPOA] injection of cre) represents total daily running wheel activity (RWA) from each individual mouse in the non-pregnant state (4 days before mating) and during pregnancy (days 1–18).
Black lines show the mean ± SEM for the group.
- https://cdn.elifesciences.org/articles/62260/elife-62260-fig6-data1-v2.tif
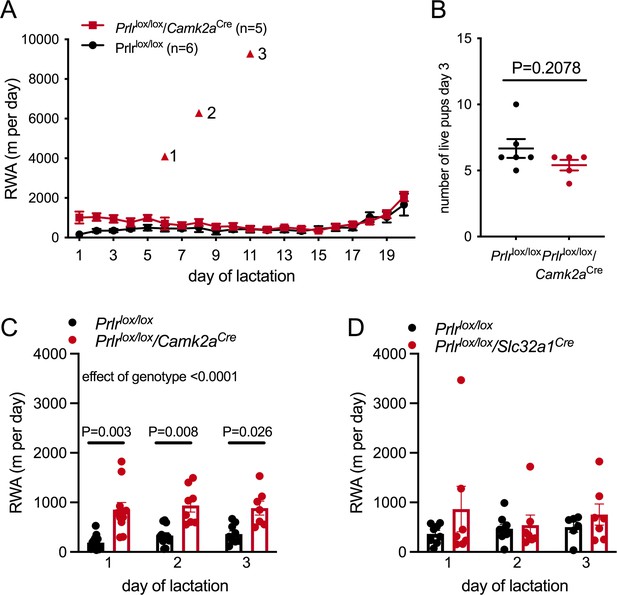
Prolactin contributes to early lactation attenuation in running wheel activity (RWA) but not through GABA neurons.
(A) Daily RWA across lactation in a small cohort of Prlrlox/lox/Camk2aCre and control littermates (Prlrlox/lox). Triangles 1–3 each indicate RWA for an individual mouse on a single day of excessive RWA in mid lactation. (B) Number of pups alive per litter on the morning of day 3 of lactation for each dams in (A), no pups died across lactation, and so this graph also represents the number of pups for each litter at weaning. (C, D) Bars represent average daily RWA in control (Prlrlox/lox) and mice lacking Prlr in forebrain neurons (C) (Prlrlox/lox/Camk2aCre, n = 13, Prlrlox/lox, n = 11, p-value from post hoc test between genotype at each time point) or GABA neurons (D) (Prlrlox/lox/Slc32a1Cre, n = 8, Prlrlox/lox, n = 9).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus, female and male) | C57BL/6J | Jackson Laboratory | Stock #:000664; RRID:IMSR_JAX:000664 | |
| Genetic reagent (M. musculus) | Prlrlox/lox | PMID:27581458 | MGI:6196142 | Prof. Dave Grattan (University of Otago, New Zealand) |
| Genetic reagent (M. musculus) | Camk2aCre | PMID:11668676 | RRID:IMSR_EM:01153 | PMID:11668676 |
| Genetic reagent (M. musculus) | Slc32a1Cre | Jackson Laboratory | Stock #:028862; RRID:IMSR_JAX:028862 | PMID:21745644 |
| Genetic reagent (M. musculus) | Kiss1Cre | PMID:21149719 | MGI:4878876 | Prof. Ulrich Boehm (Saarland University Germany) |
| Antibody | GFP, polyclonal rabbit | Life Technologies | Cat #:A6455; RRID:AB_221570 | (1:20,000) |
| Antibody | Phospho STAT5, polyclonal rabbit | Cell Signaling Technology | Cat #:9351; RRID:AB_2491009 | (1:1000) |
| Sequence-based reagent | Slc32a-probe, Mm-Slc32a1-C2 | Advanced Cell Diagnostic | Cat #:319191_C2 | Targeting the nucleotide sequence in the region 894–2037 of NM_009508.2 |
| Sequence-based reagent | Prlr probe, Mm-Prlr-01 | Advanced Cell Diagnostic | Cat #:ADV588621 | Targeting the nucleotide sequence in the region 1107–2147 of NM_011169.5 |
| Recombinant DNA reagent | AAV/DJ-CMV-mCherry-iCre | Vector Biosystems | Cat #:VB7600 | |
| Recombinant DNA reagent | AAV/DJ-CMV-mCherry | Vector Biosystems | Cat #:VB7777 | |
| Peptide, recombinant protein | Prolactin, ovine | Sigma-Aldrich | Cat #:L6520 | |
| Peptide, recombinant protein | Prolactin, ovine | National Hormone and Pituitary Program, NIDDK | Cat #:AFP-10692C | |
| Commercial assay or kit | RNAscope 2.5HD Duplex assay | Advanced Cell Diagnostic | Cat #:ADV322430 |
Additional files
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/62260/elife-62260-transrepform1-v2.docx
-
Source data 1
Running wheel activity levels across pregnancy.
- https://cdn.elifesciences.org/articles/62260/elife-62260-supp1-v2.xlsx





