Loss of cortical control over the descending pain modulatory system determines the development of the neuropathic pain state in rats
Figures
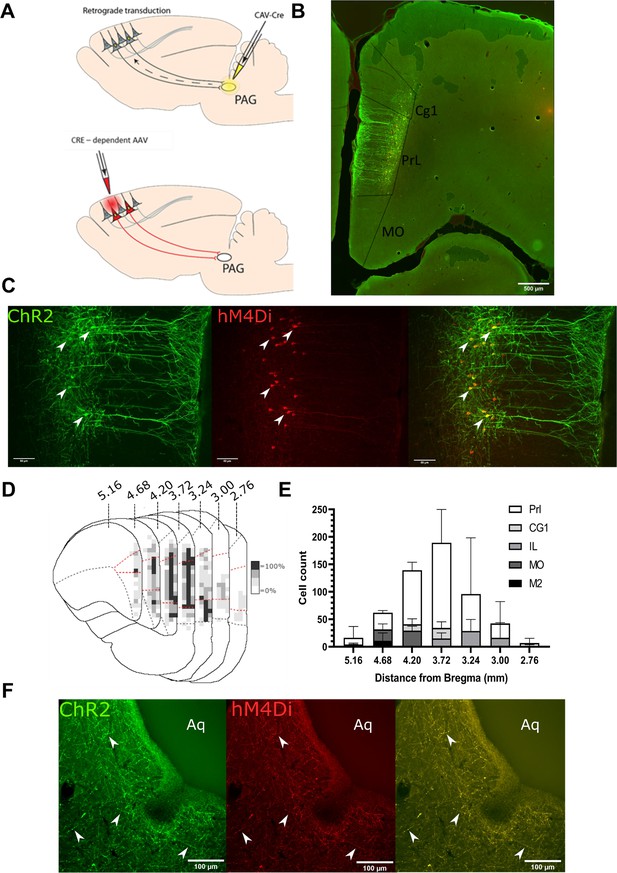
Transfected mPFC→PAG neurons arise mainly from the pre-limbic (PrL) cortex.
(A) Intersectional viral vector strategy. We used a retrograde canine adenovirus and Cre-dependent adeno-associated viral vectors to express genetically encoded actuators (both channelrhodopsin-2 [ChR2] and hMD4i) within medial prefrontal cortex (mPFC) neurons that project to the periaqueductal grey (PAG). (B) Photomicrograph of mPFC showing labelled neurons residing mainly in the PrL cortex. (C) PrL cortex with colocalisation of mCherry (hM4Di) and EYFP (ChR2) in neurons projecting to PAG (many examples but several marked with white arrows). (D) Conjunction plot illustrating location of mPFC→PAG neurons throughout the mPFC (n=3 rats). Darker shading indicates positional overlap of positively labelled (hM4Di) neurons from more than one animal (light=1 animal, mid=2, and dark=3). Dotted red line demarks the PrL cortex. (E) Comparative distribution of mPFC→PAG neurons throughout the cortex (mean ± SEM). (F) Photomicrograph showing ChR2-EYFP and hM4Di-mCherry containing fibres from mPFC projecting to the ventrolateral region of PAG (many examples but several marked with white arrows).
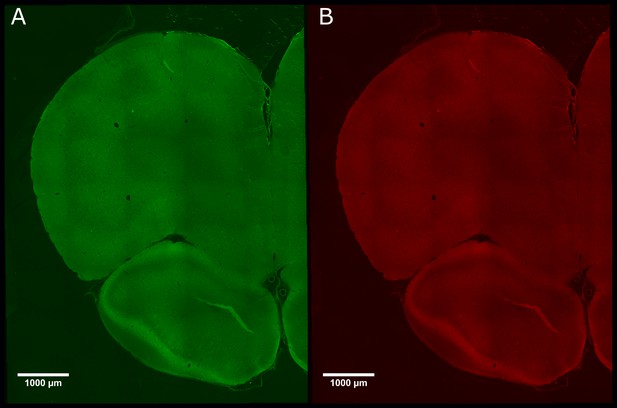
There was negligible expression of channelrhodopsin-2 (ChR2)-EYP and hM4Di-mCherry in control animals that did not receive CAV–CMV–CRE into the periaqueductal grey.
Photomicrograph of medial prefrontal cortex showing negligible expression of ChR2-EYFP (A) or hM4Di-mCherry (B) in PrL.Control rats.
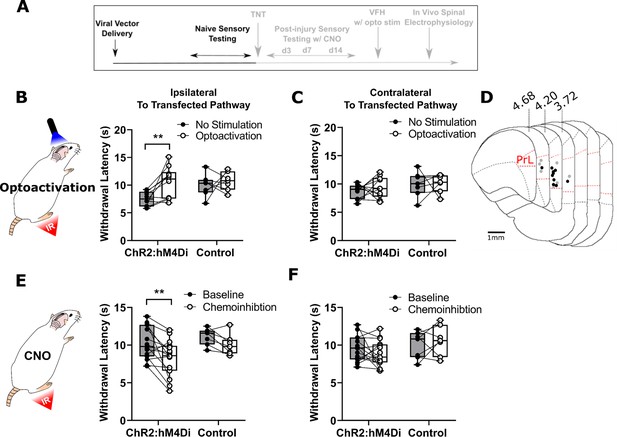
PrL-P neurons bidirectionally regulate nociception in naive rats.
(A) Experimental timeline. (B) Illumination of PrL (445 nm, 20 Hz, 10–15 mW, 10 ms pulse, concomitant with hind-paw heating) in NaivePrL.ChR2:hM4Di rats increased thermal withdrawal latencies of the ipsilateral hindpaw but not in NaivePrL.Control rats that did not express channelrhodopsin-2 (ChR2; paired t-test, t(9)=3.37, p=0.008, n=10 for ChR2:hM4Di group; t(6)=0.63, p=0.55, n=7 for control group). (C) Equivalent illumination of PrL had no effect on the thermal withdrawal latency of the contralateral hindpaw in either NaivePrL.ChR2:hM4Di or NaivePrL.Control rats (paired t-test, t(9)=0.86, p=0.40, n=10 for NaivePrL.ChR2:hM4Di rats; t(6)=0.14, p=0.90, n=7 for NaivePrL.Control rats). (D) Optic fibre tip locations in the medial prefrontal cortex from NaivePrL.ChR2:hM4Di (●) and NaivePrL.Control (○) rats. For simplicity, fibre placements are depicted in a single hemisphere. (E) Systemic CNO (2.5 mg·kg−1 i.p.) in NaivePrL.ChR2:hM4Di rats decreased withdrawal latencies (mean value at 20–40 min post-injection) of the ipsilateral paw but not in NaivePrL.Control rats (paired t-test, t(14)=3.26, p=0.006, n=15 for NaivePrL.ChR2:hM4Di rats; t(6)=0.63, p=0.55, n=7 for NaivePrL.Control rats). (F) CNO had no effect on the thermal withdrawal latency of the contralateral hindpaw in either NaivePrL.ChR2:hM4Di rats or NaivePrL.Control rats (paired t-test, t(14)=1.22, p=0.24, n=15 for NaivePrL.ChR2:hM4Di rats; t(6)=0.43 p=0.68, n=7 for NaivePrL.Control rats).
-
Figure 2—source data 1
Numerical data to support graphs in Figure 2.
- https://cdn.elifesciences.org/articles/65156/elife-65156-fig2-data1-v2.xlsx
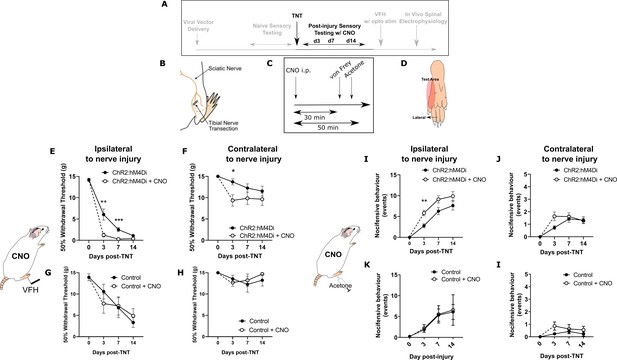
Inhibition of PrL-P neurons unmasks hypersensitivity in neuropathic rats.
(A) Experimental timeline. (B) Tibial nerve transection (TNT) was used to produce the neuropathic injury. (C)- Sensory testing was conducted at 30 min after systemic delivery of CNO and (D) testing was conducted on the lateral plantar surface of the hindpaw in a receptive field adjacent to injured tibial nerve. (E) In TNTPrL.ChR2:hM4Di rats, CNO (2.5 mg·kg−1 i.p.) reduced the mechanical withdrawal threshold at 3 and 7 days post nerve injury on the ipsilateral (injured) hindpaw (two-way ANOVA, main effect CNO, F(1,30)=20.09, p=0.0001; timexCNO, F(2, 60)=6.892, p=0.002; Sidak’s post-test day 3, p=0.008; day 7, p=0.001, n = 16) and (F) on the contralateral paw at 3 days post-injury (two-way ANOVA, CNO F(1,30)=5.77, p=0.02; Sidak’s post-test, p=0.02, n = 16). (G and H) In TNTPrL.Control rats, the same dose of CNO did not alter mechanical withdrawal thresholds on either the ipsilateral or contralateral hindpaw (two-way ANOVA, main effect; ipsilateral CNO, F(1,14)=0.02, p=0.90, n=8 and contralateral CNO, F(1,14)=0.15, p=0.71, n=8, respectively). (I and J) In TNTPrL.ChR2:hM4Di rats, CNO increased acetone-evoked nocifensive events at 3 days post-injury on the ipsilateral paw (two-way ANOVA, main effect CNO, F(1,30)=9.6, p=0.004; Sidak’s post-test, p=0.003, n=16) but not contralaterally (two-way ANOVA, main effect CNO, F(1,29)=1.3, p=0.26, n=16). (K and I) In TNTPrL.Control rats, CNO did not alter acetone-evoked nocicfensive behaviour (two-way ANOVA, main effect CNO ipsilateral, F(1,12)=0.02, p=0.89, n=7 and main effect CNO contralateral, F(1,12)=2.2, p=0.16, n=7).
-
Figure 3—source data 1
Numerical data to support graphs in Figure 3.
- https://cdn.elifesciences.org/articles/65156/elife-65156-fig3-data1-v2.xlsx
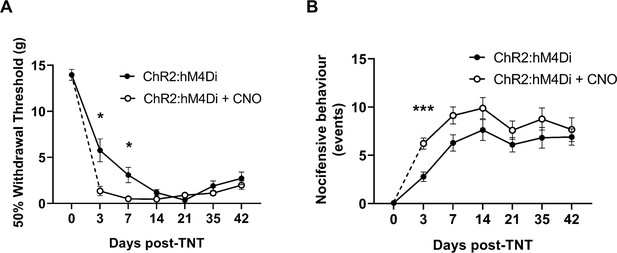
Chemo-inhibition of PrL-P neurons affects nocicfensive behaviour in early but not late timepoints post-injury in neuropathic animals.
(A) In TNTPrL.ChR2:hM4Di rats, systemic delivery of CNO (2.5 mg·kg−1 i.p.) significantly reduced the mechanical withdrawal threshold at 3 and 7 days post-injury on the ipsilateral (injured) hindpaw (mixed model [REML], fixed effects CNO, F(1,28)=7.26, p=0.002; timexCNO, F(5,95)=4.92, p=0.0005; Sidak’s post-test, *p<0.05, n=16). (B) In TNTPrL.ChR2:hM4Di rats, systemic delivery of CNO (2.5 mg·kg−1 i.p.) significantly increased the cold (acetone)-evoked nocicfensive events at 3 days post-injury on the ipsilateral (injured) hindpaw (mixed model [REML], fixed effects CNO, F(1,30)=6.3, p=0.02; timexCNO, F(5,98)=0.6, p=0.70; Sidak’s post-test, ***p=0.0006, n=16).
-
Figure 3—figure supplement 1—source data 1
Numerical data to support graphs in Figure 3—figure supplement 1.
- https://cdn.elifesciences.org/articles/65156/elife-65156-fig3-figsupp1-data1-v2.xlsx
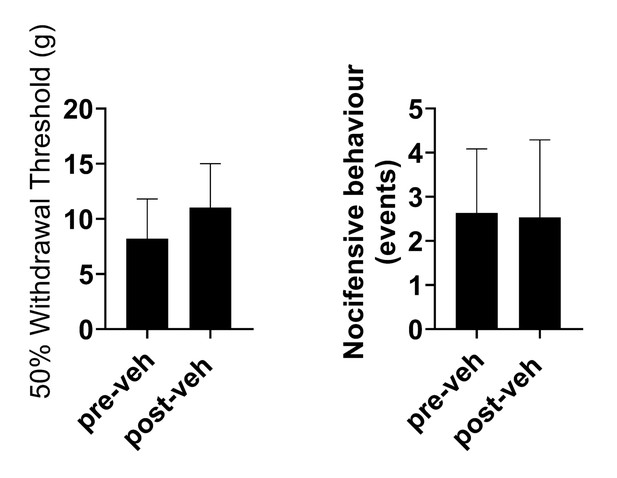
Delivery of vehicle does not affect sensitisation in TNTPrL.ChR2:hM4Di rats at 7 days post-TNT.
Delivery of vehicle (sterile saline with 5% DMSO, i.p.) didnot alter mechanical withdrawal thresholds (paired t-test, t=0.4, n=3) (A) or cold-evoked nocicfensive behaviour (paired t-test, t=0.86, (B) in TNTPrL.ChR2:hM4Di rats [n = 3]).
-
Figure 3—figure supplement 2—source data 1
Numerical data to support graphs in Figure 3—figure supplement 2.
- https://cdn.elifesciences.org/articles/65156/elife-65156-fig3-figsupp2-data1-v2.xlsx
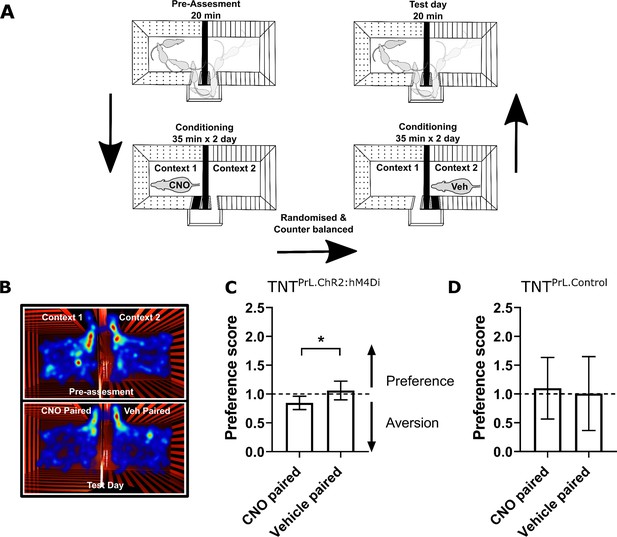
Inhibition of PrL-P neurons produces aversion in neuropathic animals.
(A) Conditioned place aversion protocol. (B) Example heatmap visualisation of the time spent within the testing chambers prior (top) and following conditioning with CNO or vehicle. (C) Group data showing conditioning with CNO in TNTPrL.ChR2:hM4Di rats 2–5 days after tibial nerve transection (TNT) produced place aversion (paired t-test, t(7)=2.43, p=0.04, n=8). (D) CNO administration to TNTPrL.Control rats did not produce place aversion (paired t-test, t(8)=0.25, p=0.81, n=9).
-
Figure 4—source data 1
Numerical data to support graphs in Figure 4.
- https://cdn.elifesciences.org/articles/65156/elife-65156-fig4-data1-v2.xlsx
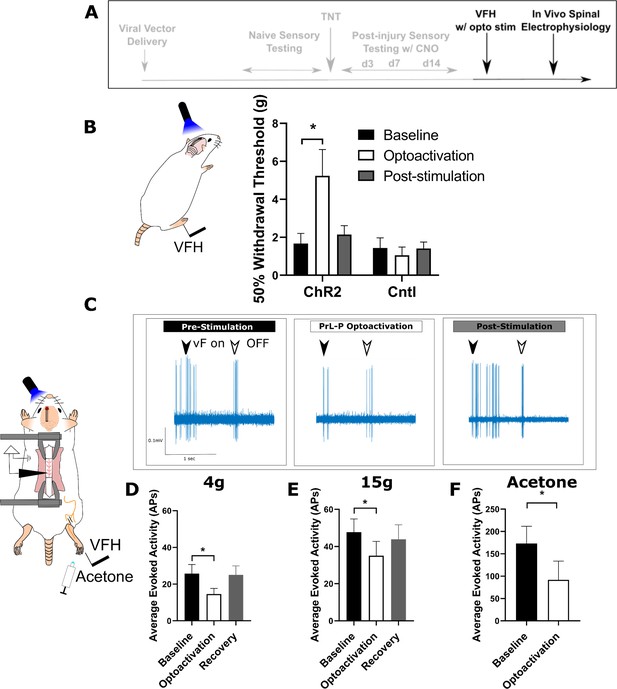
Activation of PrL-P neurons produces antinociception in neuropathic rats by inhibition at a spinal level.
(A) Experimental timeline. (B) Delivery of blue light (445 nm, 20 Hz, 10–15 mW, 10 ms pulse, concomitant with hind-paw stimulation) produced a significant increase in mechanical withdrawal threshold of the injured (ipsilateral) hind-paw in TNTPrL.ChR2:hM4Di rats (repeated-measures [RM] ANOVA, treatment, F(1.12, 8.96)=8.07, p=0.02; Sidak’s post-test, *p<0.05, n=9) but not in TNTPrL.Controlrats (RM ANOVA, treatment, F(1, 2)=0.35, p=0.61, n=3). (C) Example raw data trace illustrating suppression of von Frey hair evoked spinal dorsal horn neuron activity during blue light (420 nm, 10–15 mW, 10 ms duration, concomitant with hind-paw stimulation) delivery to the PrL in TNTPrL.ChR2:hM4Di rats (arrows demark beginning and end of stimulus). (D) Group data illustrating suppression of 4 g evoked spinal dorsal horn neuronal activity by illumination of the PrL in TNTPrL.ChR2:hM4Di rats (mixed model [REML], fixed effect opto-activation, F[1.99, 13.98]=7.18, p=0.007; Dunnet’s post-test baseline vs opto-activation, p=0.009, n=10). (E) Illumination of the PrL in TNTPrL.ChR2:hM4Di rats also supressed 15 g evoked spinal dorsal horn neuronal activity (mixed model [REML], fixed effect opto-activation, F(1.52, 10.64)=2.94, p=0.10, Dunnet’s post-test baseline vs opto-activation, p=0.046, n=10.) (F) Delivery of blue light to the Prl in TNTPrL.ChR2:hM4Di rats decreased acetone-evoked spinal dorsal horn neuronal activity (paired t-test, t(3)=3.58, p=0.04, n=4).
-
Figure 5—source data 1
Numerical data to support graphs in Figure 5.
- https://cdn.elifesciences.org/articles/65156/elife-65156-fig5-data1-v2.xlsx
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background | Wistar (rat) male | Envigo, NL | RCCHan | |
| Transfected construct | Canine adenoviralvector | IGMM, FR | CAV2-CMV-CRE | |
| Transfected construct | Adeno-associatedviral vector | UNC Vector Core, USA | AAV2-EF1a-DIO- hChR2-EYFP | |
| Transfected construct | Adeno-associatedviral vector | Addgene, USA | AAV2-hSyn-DIO- hM4Di-mCherry | |
| Chemical compound | Clozepine-N-oxide | Tocris Bioscience, UK | 4936 | |
| Antibody | Anti-GFP (chicken polyclonal) | Abcam, USA | Ab13970 | (1:5000) |
| Antibody | Anti-mCherry (rabbit polyclonal) | BioVision | 5993 | (1:2000) |
| Other | Optic fibre | Optogenix, IT | Lambda-B | NA 0.66, length 4.4 mm, and light-emitting length 2 mm |
| Other | Q-probes | Neuronexus, USA | Q1 × 1-tet-10mm-121-Q4 | |
| Other | Headstage chip | Intan Technologies, USA | RHD2132 | |
| Other | Diode laser | Omricon Laserage, DE | LuxX445-100 | 445 nm/100 mW |
| Other | OpenEphys Acquisition System | OpenEphys, USA | ||
| Software, algorithm | OpenEphys GUI | OpenEphys, USA | ||
| Software, algorithm | EthovisionXT | Noldus, NL | ||
| Software, algorithm | Prism | GraphPad, USA | ||
| Software, algorithm | GNU Image Manipulation Program | GIMP, USA | ||
| Software, algorithm | BORIS event logging software | BORIS, IT |





