A novel mechanosensitive channel controls osmoregulation, differentiation, and infectivity in Trypanosoma cruzi
Figures
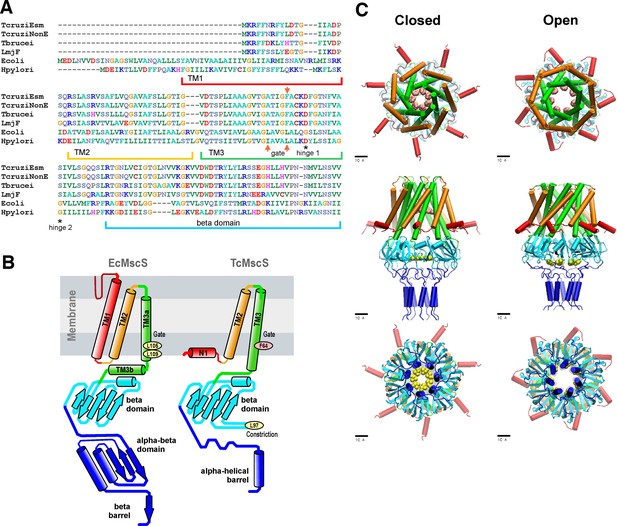
Sequence alignment and predictions of TcMscS structure by homology to EcMscS.
(A) Partial protein sequence alignment of four small-conductance mechanosensitive (MscS)-type channels from Trypanosoma cruzi, T. cruzi Esmeraldo-like (TcCLB.504171.40) and non-Esmeraldo-like (TcCLB.509795.40) haplotypes of the CL Brener reference strain, Trypanosoma brucei (Tb427.10.9030) and Leishmania major (LmjF.36.5770), with two bacterial MscS channels from Escherichia coli (WP_000389819) and Helicobacter pylori (WP_000343449.1). The positions of the transmembrane domains TM1, TM2, and TM3 are underlined, the positions of the putative gate residues are indicated by red arrows, and conserved residues forming the hinges of TM3 are indicated with asterisks. The full alignment can be found in Figure 1—figure supplement 1. (B) Arrangements of transmembrane helices of EcMscS (PDB ID 2OAU) and in the proposed homology model of TcMscS. The positions of the putative gate and secondary constriction residues for TcMscS (F64 and L97) are indicated. (C) Full-homology TcMscS model in the closed and open states. N-terminal domain 1 (red), TM2 (gold), and TM3 (green) are followed by the cytoplasmic cage (cyan) and the C-terminal bundle (blue). The gate residues are indicated in pink and yellow.
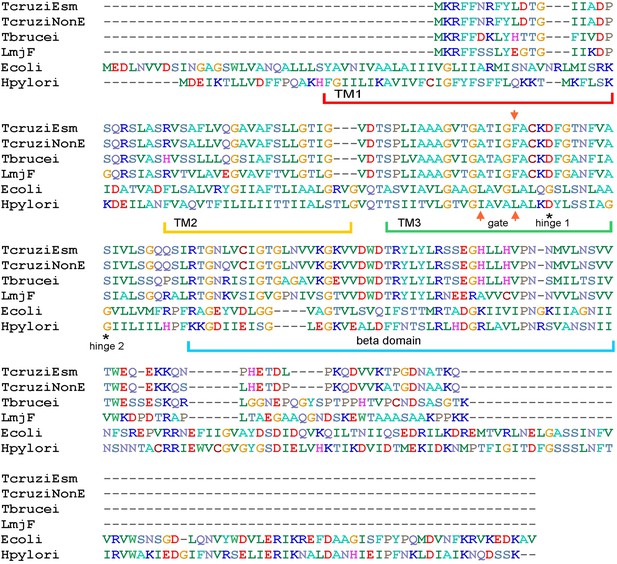
Sequence alignment of TcMscS.
Full protein sequence alignment of four small-conductance mechanosensitive (MscS)-type channels from Trypanosoma cruzi, T. cruzi Esmeraldo-like (TcCLB.504171.40) and non-Esmeraldo-like (TcCLB.509795.40) haplotypes of the CL Brener reference strain, Trypanosoma brucei (Tb427.10.9030) and Leishmania major (LmjF.36.5770), with two bacterial MscS channels from Escherichia coli (WP_000389819) and Helicobacter pylori (WP_000343449.1). The positions of the transmembrane domains TM1, TM2, and TM3 are underlined, the position of the putative gate residues is indicated by red arrows, and conserved residues forming the hinges of TM3 are indicated by asterisks.
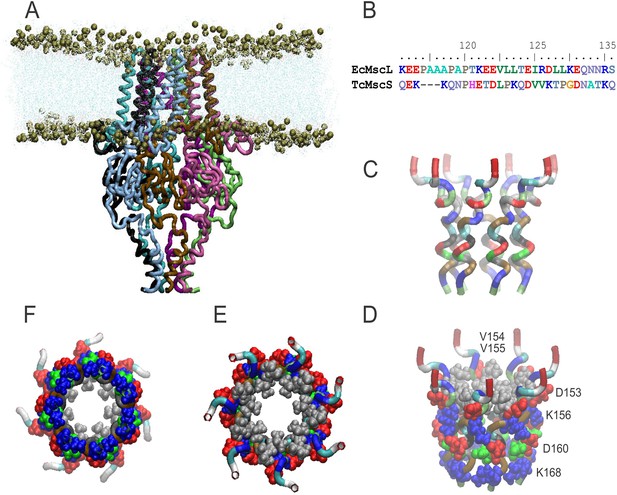
Molecular dynamic simulations of the TcMscS model.
Panel (A) shows the model after a 10 nS unrestrained molecular dynamic (MD) simulation in the POPC (1-palmitoyl-2-oleoyl-sn-glycero-3-phosphocholine) bilayer followed by symmetrization. The transmembrane barrel of TcMscS is considerably shorter than that of EcMscS (Reddy et al., 2019). Yet, it is sufficient to form a stable transmembrane pore made out of 14 transmembrane (TM) helices with short N-terminal helices oriented flat on the inner surface of the membrane. Panel (B) presents sequence alignment of the C-terminal segments of EcMscL and TcMscS. The two structures show only slight sequence resemblance apart from the fact that both can form amphipathic structures. In contrast to MscL, which is α-helical, the C-terminal segment of TcMscS carries one glycine and three prolines and lacks the canonical heptad motif typically mediating coiled-coil assemblies. The energy-minimized model (C–F) indicates the possibility of a bundle-like arrangement of the TcMscS termini with hydrophobic chains inside and polar and charged groups on the periphery. In this model, K156 salt-bridges to D160. In MD simulations, the C-terminal segments are quite dynamic and, after a 10 ns run and symmetrization (A), they arrived at a slightly different conformation compared to the initial energy-minimized model, still retaining its hydrophobic interior. We have additionally tested the stability of the predicted C-terminal bundle of TcMscS by manually re-orienting the helical segments in random directions in every subunit, followed by a 5-ns unrestrained simulation and symmetry annealing. The termini showed the tendency to spontaneously re-assemble back into a helical bundle similar to the starting conformation, with hydrophobic sides of the helices forming the core (not shown). While the modeled structure is hypothetical and requires a thorough experimental testing, these simulations increase our confidence in the model.
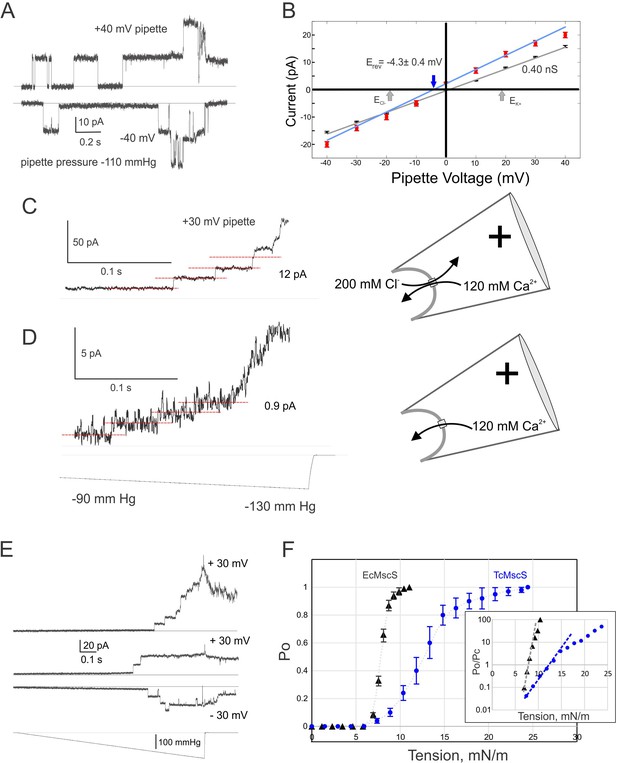
Electrophysiological properties of TcMscS.
(A) Two single-channel traces recorded sequentially under identical steps of a negative pipette pressure of 110 mmHg at pipette voltages +40 mV and –40 mV. (B) Current-to-voltage relationships measured in symmetric (200 mM/200 mM KCl, black symbols, n = 11) and asymmetric conditions (425 mM/200 mM KCl, red symbols, n = 4). The theoretical reversal potentials for Cl- and K+ (gray arrows) and the experimental reversal potential measured for TcMscS (red arrow) are indicated. (C) Unitary conductance of TcMscS recorded with 100 mM Ca gluconate, 20 mM CaCl2 in the pipette, and 200 mM KCl in the bath, at +30 mV in the pipette. (D) The trace recorded from the same patch after perfusion of the bath with 100 mM Ca gluconate. The pressure stimuli for traces in (C) and (D) were identical ramps shown at the bottom of panel (D). In the latter configuration (shown on the right), Ca2+ is the dominant permeable ion. (E) Examples of traces recorded with identical 1 s linear ramps on three separate patches bearing 11, 2, and 3 active channels at +30 mV or −30 mV pipette potential. (F) Multiple ramp responses of TcMscS populations (E) were normalized to the current at the saturating pressure and converted to dose-response curves. The aggregated TcMscS curve (blue symbols, n = 11) is compared with EcMscS dose-response curves (black symbols, n = 54). The inset in panel (F) shows the same dose-response curves plotted as log (Po/Pc) and fitted with the Boltzmann equation. Po and Pc are the population-normalized open and closed probabilities. The shallower slope of the TcMscS dose-response curve indicates a smaller lateral expansion of the protein complex.
-
Figure 2—source data 1
Electrophysiological data analysis.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig2-data1-v2.xlsx
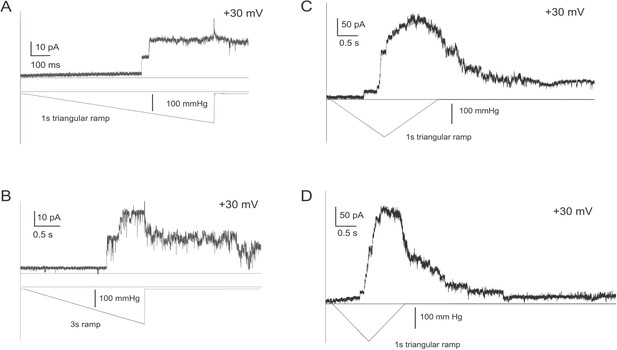
Examples of traces showing slow closure of TcMscS and a ‘lingering’ channel activity lasting for seconds after the end of the mechanical stimulus.
Traces (A) and (B) were recorded with abruptly ending linear ramps, whereas traces (C) and (D) were recorded with symmetric triangular ramps. The closing transition does not recapitulate the opening trajectory and often proceeds through a sequence of subconductive states, which suggests that the closing is less cooperative and may involve an asynchronous action of two gates.
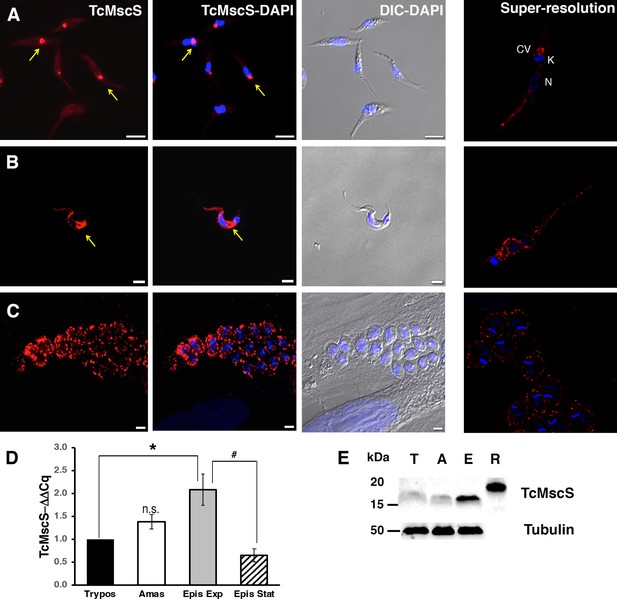
Localization and expression of TcMscS in T.cruzi.
Immunolocalization of TcMscS (red) with specific polyclonal antibodies in epimastigotes (A), trypomastigotes (B), and intracellular amastigotes (C). The right panels correspond to images of the same life stages obtained by super-resolution microscopy. Cellular structures are indicated as CV (contractile vacuole), K (kinetoplast), and N (nucleus). DNA was 4′,6-diamidino-2-phenylindole (DAPI) stained. Bar sizes: (A) 5 µm; (B) and (C) 5 µm. (D) Expression of TcMscS in different life stages of the parasite, quantified by quantitative reverse transcription PCR (RT-qPCR). Trypomastigotes (Tryp) and amastigotes (Amast) were obtained from HEK-293 cells. Epimastigotes were analyzed at 4 days (Epis Exp) and 10 days of growth (Epis Stat). The values are indicated as ΔΔCq with respect to the expression in trypomastigotes and normalized against GAPDH as a housekeeping gene. The values are mean ± SEM of three independent experiments in triplicate (*p = 0.006, #p = 0.009). (E) Western blot analysis of TcMscS in trypomastigotes (T), amastigotes (A), and epimastigotes (E). Purified recombinant protein (R) was used as the positive control. The whole-cell lysates were probed with polyclonal antibodies against TcMscS and monoclonal anti-tubulin was used as the loading control.
-
Figure 3—source data 1
Data source for Figure 3 provided as Figure 3—source data 1.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig3-data1-v2.xlsx
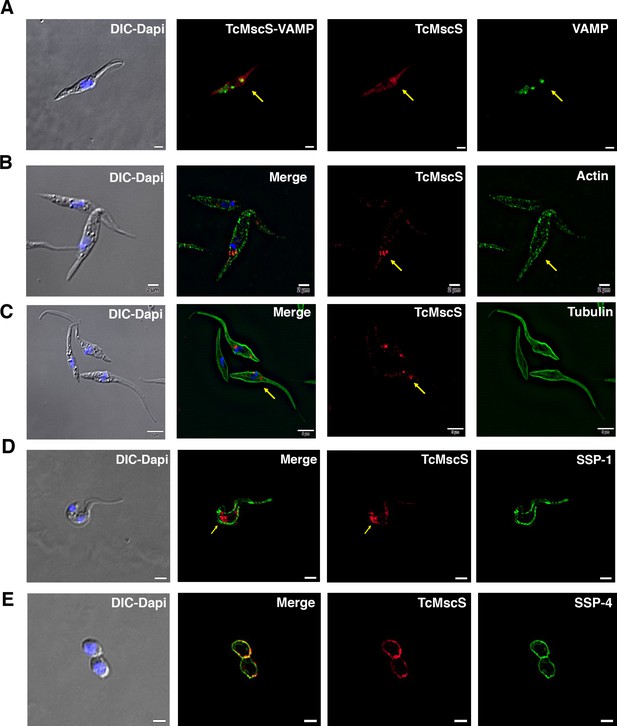
TcMscS colocalization with organelle markers.
(A) Immunofluorescence analysis of TcMscS (red) localization in epimastigotes overexpressing TcVAMP-GFP (green). (B) Labeling of TcMscS (red) in wild-type Y strain (WT) epimastigotes partially colocalizes with anti-actin antibodies (green). (C) Localization of TcMscS (red) and tubulin (green) in Y strain epimastigotes. No significant colocalization was observed. (D) Immunolocalization of TcMscS (red) and membrane marker SSP-1 (green) in Y strain tissue-derived trypomastigotes. (E) Colocalization of TcMscS (red) and membrane marker SSP-4 (green) in extracellular amastigotes obtained by host cell lysis. Nuclei and kinetoplasts were 4′,6-diamidino-2-phenylindole (DAPI) stained. Arrows indicate the position of the contractile vacuole. Bar size: 5 µm.
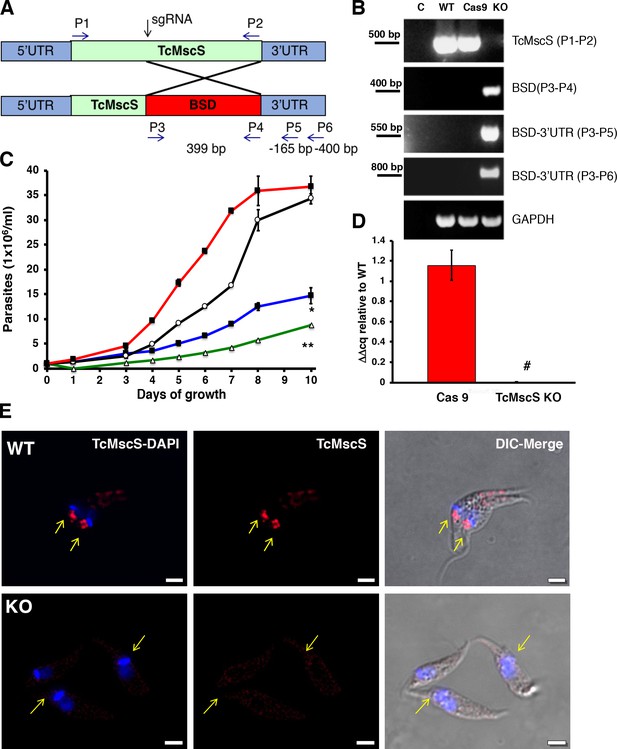
Phenotypic characterization of TcMscS-KO.
(A) Schematic representation of TcMcS gene targeting by CRISPR/Cas9. A double-strand DNA cut was induced at position +185 and repaired by homologous recombination with a construct containing the Bsd gene. The primers used for screening are listed in Supplementary file 1-Table 1. (B) Genomic DNA screening of TcMscS-KO epimastigotes by PCR. Negative controls (C) without DNA are in lane 1 for all reactions. The ORF for TcMscS was amplified in wild-type Y strain (WT) and Cas9 controls but absent in TcMscS-KO parasites. The correct insertion of the blasticidin cassette (BSD) was verified by amplification with primers annealing in ORF and the 3’UTR. GAPDH was used as a housekeeping gene for the loading control. (C) Growth curve of epimastigotes WT (black line), Cas9 (red line), TcMscS-KD (blue line), and TcMscS-KO (green line). All the experiments were started at 1 x 106 cells/ml and counted daily. The values are mean ± SEM of three independent experiments in triplicate (*, **p<0.01). (D) Quantitative reverse transcription PCR (RT-qPCR) quantifying the expression of TcMscS in epimastigotes control (Cas9) vs TcMscS-KO. The values are indicated as ΔΔCq with respect to the expression in WT epimastigotes and normalized against GAPDH as the housekeeping gene. All the samples were collected from parasites at 4 days of growth. The values are mean ± SEM of three independent experiments in triplicate (#p = 0.0001). (E) Immunofluorescence analysis of TcMscS (red) in epimastigotes Y strain (WT), TcMscS-KO. Nuclei and kinetoplasts were 4′,6-diamidino-2-phenylindole (DAPI) stained. The position of the contractile vacuole is indicated with yellow arrows. Bar size = 2 µm.
-
Figure 5—source data 1
Phenotype analysis of TcMscS KO epimastigotes.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig5-data1-v2.xlsx
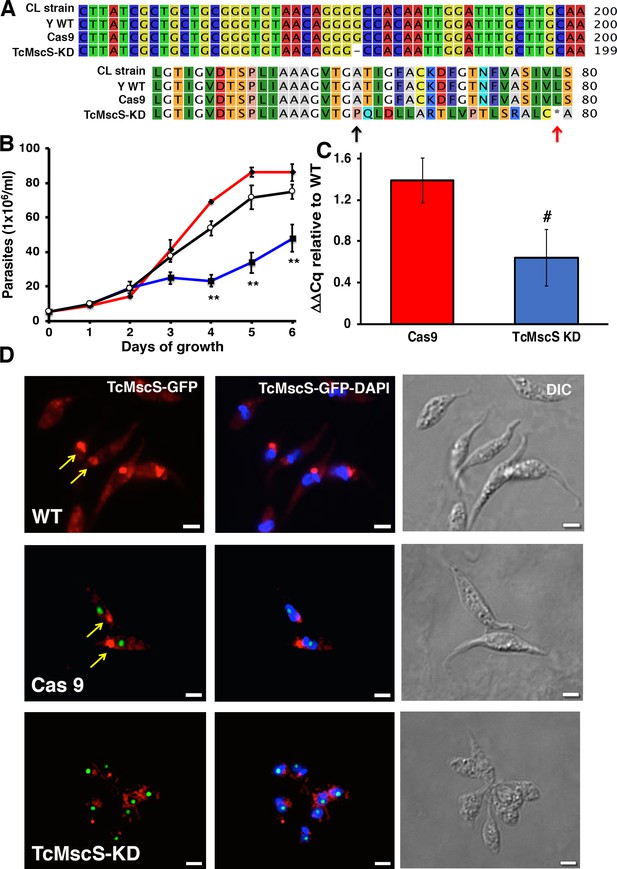
TcMscS targeting by CRISPR/Cas9 produced a knockdown effect.
(A) Partial alignment of TcMscS nucleotide sequencing (top panel) and predicted protein (bottom panel) obtained from wild-type epimastigotes CL Brener and Y strains, parasites expressing a scrambled single guide RNA (sgRNA) and Cas9, and those in which TcMscS-specific sgRNA2 was transfected (TcMcsS-KD). A single-nucleotide deletion in position 178 results in a shift in the ORF (black arrow) and a premature stop codon (red arrow). (B) Growth curve of epimastigotes wild-type Y strain (WT) (black line), Cas9 (red line), and TcMscS-KD (blue line). Mean ± SEM of three independent experiments in triplicate (**p<0.01). (C) Quantitative reverse transcription PCR (RT-qPCR) quantifying the expression of TcMscS in epimastigotes control (Cas9) vs TcMscS-KD. The values are indicated as ΔΔCq with respect to the expression in WT epimastigotes and normalized against GAPDH as a housekeeping gene. All the samples were collected from parasites at 4 days of growth. The values are mean ± SEM of three independent experiments in triplicate (#p = 0.040). (D) Immunofluorescence analysis of TcMscS (red) in epimastigotes Y strain (WT), Cas9, and TcMscS-KD. The expression of Cas9 was detected with anti-GFP antibodies. Nuclei and kinetoplasts were 4′,6-diamidino-2-phenylindole (DAPI) stained. The position of the contractile vacuole is indicated with yellow arrows. Bar size = 5 µm.
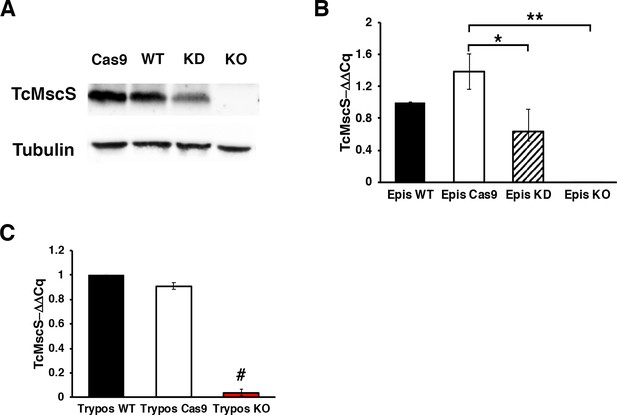
TcMscS expression levels in epimastigotes and trypomastigotes.
(A) Western blot analysis of epimastigotes with anti-TcMscS antibodies. Whole-cell lysates were obtained from Cas9-expressing parasites, wild-type Y strain (WT), TcMscS-KD (KD), and TcMscS-KO (KO). The purified recombinant protein was used as a positive control. Tubulin was used as the loading control for all the samples. (B) Quantification of TcMscS expression levels in epimastigotes by quantitative reverse transcription PCR (RT-qPCR). All the samples were collected from parasites at 4 days of growth. (C) TcMscS expression levels in trypomastigotes were quantified by RT-qPCR. The values are indicated as ΔΔCq with respect to the expression in WT and normalized against GAPDH as a housekeeping gene. Mean ± SEM of three independent experiments in triplicate (*p = 0.040, **p = 0.0001, #p = 6.43E-07).
-
Figure 5—figure supplement 2—source data 1
Quantification of TcMscS expression.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig5-figsupp2-data1-v2.xlsx
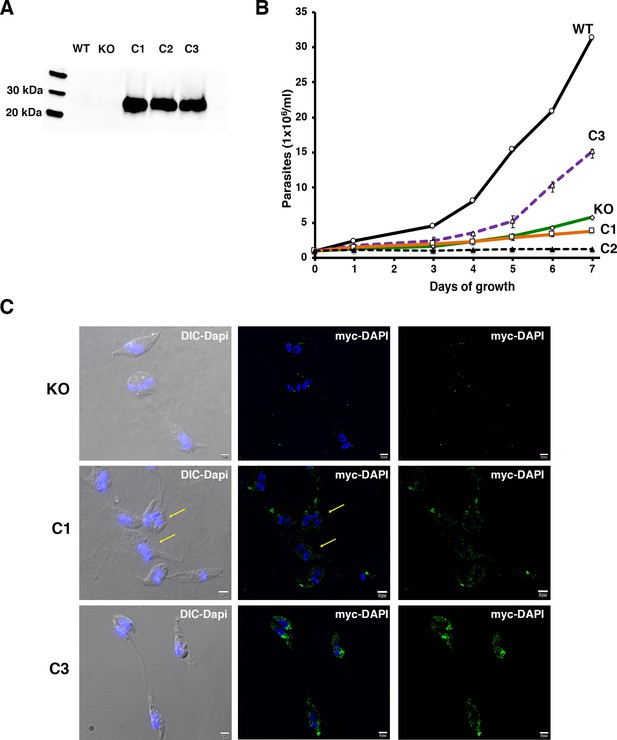
Complementation of TcMscS-KO strains.
(A) Western blot analysis. Total homogenates of wild-type Y strain (WT), KO, and complemented clonal strains C1 and C2 carrying TcMscS-PAM-mutated myc and C3 expressing TbMscS-myc were analyzed with anti-myc antibodies. (B) Growth curve of epimastigotes WT (solid black line), KO (green line), and complemented strains C1 (orange line), C2 (dashed black line), and C3 (purple line). The values are mean ± SD of three independent experiments in triplicate. (C) Immunofluorescence analysis of complemented strains. Morphology and localization of myc-tagged TcMcsS (C1 and C2) and TbMscS (C3) were analyzed with anti-myc antibodies (green). TcMscS-KO cells were used as controls. Nuclei and kinetoplasts were 4′,6-diamidino-2-phenylindole (DAPI) stained. Bar size: 2 µm.
-
Figure 5—figure supplement 3—source data 1
Complementation of TcMscS KO strains.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig5-figsupp3-data1-v2.xlsx
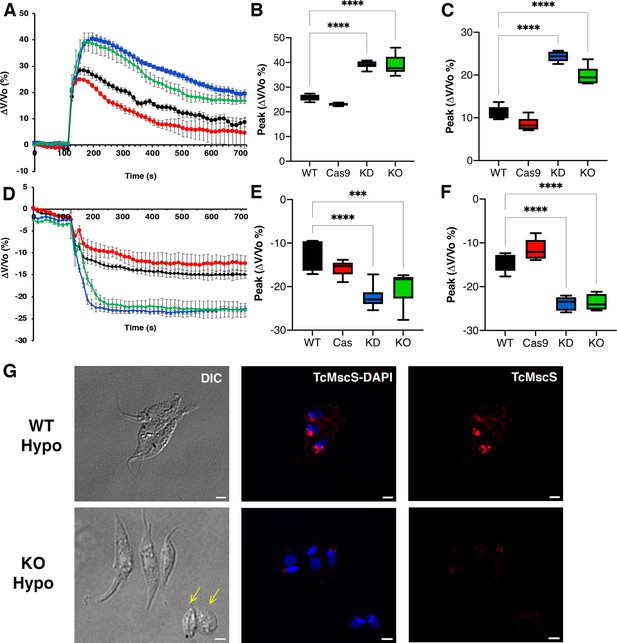
Osmotic stress responses in TcMscS mutants.
(A) Regulatory volume decreases in epimastigotes. Cells suspended in isosmotic buffer recorded for 120 s and diluted to a final osmolarity of 115 mOsm/l under constant ionic conditions. Relative changes in cell volume were monitored by determining absorbance at 550 nm over time, in wild-type Y strain (WT) (black), Cas9 (red), TcMscS-KD (blue), and TcMscS-KO (green). The absorbance values were normalized by the initial volume under isosmotic conditions and expressed as percentage of volume change. (B) Analysis of the maximum volume change under hypoosmotic conditions. The peak area was calculated between 150 and 250 s for all the experiments. (C) Final volume recovery calculated between 600 and 700 s. (D) Volume decrease in epimastigotes after a hyperosmotic shock. After 3 min under isosmotic conditions, cells were placed under hyperosmotic stress at 650 mOsm/l. Volume changes were monitored and calculated as explained in (A). Peak analysis (E) and final volumes were calculated at the same times as that in (B) and (C). The values for (A–F) are mean ± SEM of six independent experiments in triplicate (p<0.001). Statistical analysis details are listed in Supplementary files 2 and 3. (G) Representative immunofluorescence images of WT and TcMscS-KO epimastigotes at 2 min post hypoosmotic stress. TcMscS (red) was detected with specific polyclonal antibodies against the channel. Nuclei and kinetoplasts were 4′,6-diamidino-2-phenylindole (DAPI) stained. Yellow arrows indicate the presence of cells with an abnormal morphology, observed in the TcMscS-KO strain. Bar size = 2 µm.
-
Figure 6—source data 1
Analysis of osmotic stress responses.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig6-data1-v2.xlsx
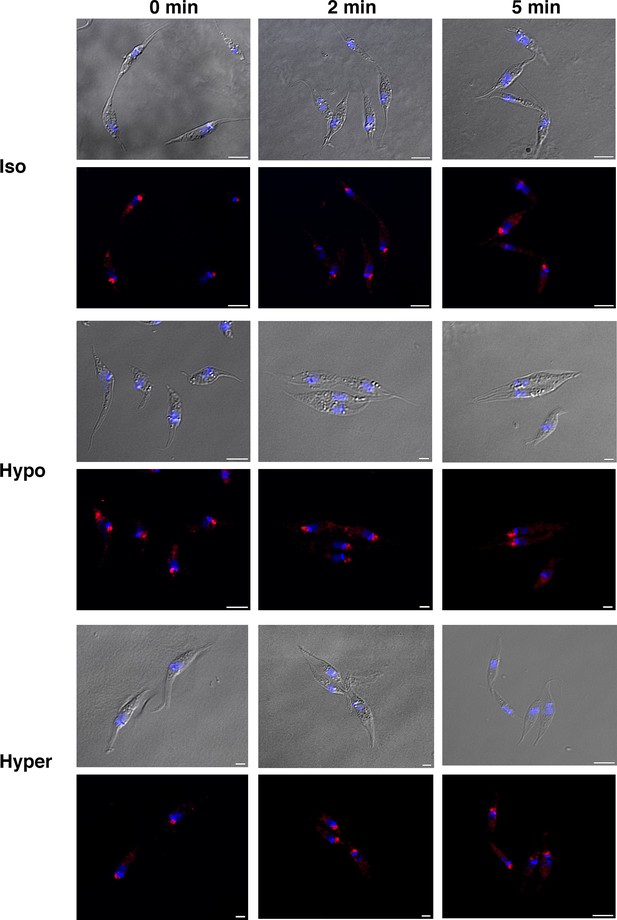
Localization of TcMscS in epimastigotes under osmotic stress conditions.
Representative images showing the localization of TcMscS in epimastigotes under isosmotic, hypoosmotic, or hyperosmotic conditions and different time points after stress. TcMscS was detected with specific antibodies against the channel. Nuclei and kinetoplasts were 4′,6-diamidino-2-phenylindole (DAPI) stained. Bar size: 5 µm.
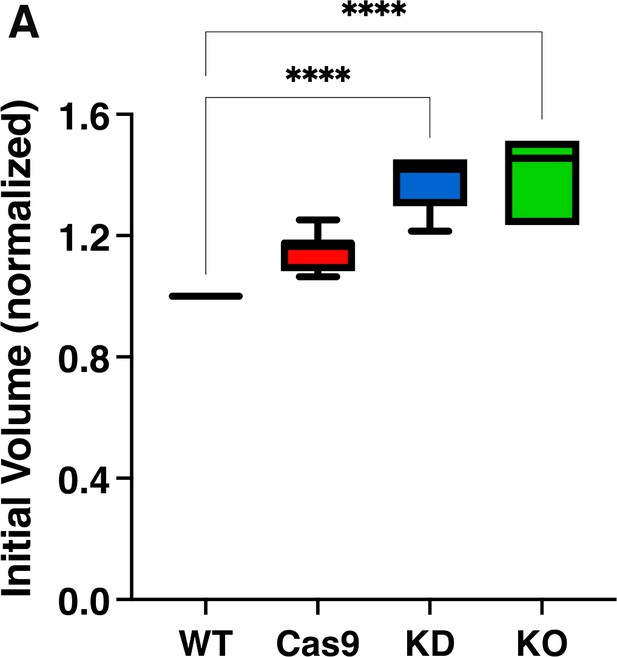
Volume of TcMscS mutants under isosmotic conditions.
Epimastigotes’ cell volume under isosmotic conditions was calculated based on the absorbance at 550 nm. The values were normalized with respect to the values of wild-type parasites (WT) and expressed as relative units. Values are expressed as mean ± SEM of seven independent experiments (*p<0.001).
-
Figure 6—figure supplement 2—source data 1
Cell volume analysis under normotonic conditions.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig6-figsupp2-data1-v2.xlsx
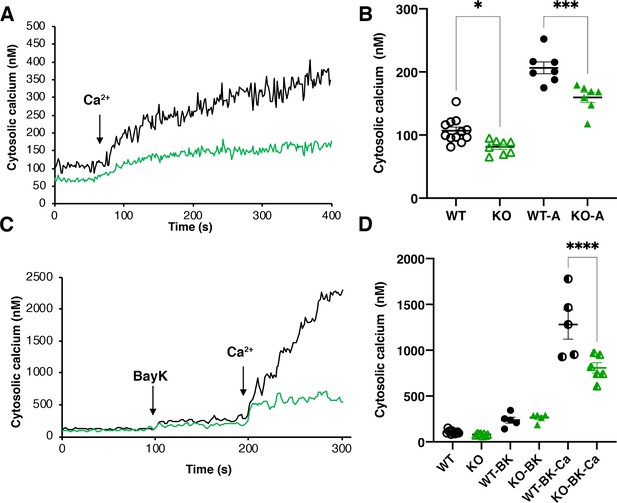
Intracellular calcium measurements.
(A) and (C) are representative traces of intracellular calcium level in wild-type Y strain (WT) (black line) or TcMscS-KO (green line) epimastigotes loaded with Fura 2-AM at baseline or after the addition of 1.8 mM CaCl2 and 10 μM of Bay K8644 (BK). (B) and (D) show the quantification of baseline Ca2+ concentration in the first 75 s WT (black) and TcMscS-KO (green) and in the first 100 s after addition of the stimuli for multiple experiments. In (B), WT-A and KO-A indicate the values after calcium addition. In (D), quantifications were done after addition of Bay K8644 (BK and Bay K plus calcium (BK-Ca)). The values are mean ± SE of n = 7 (B) and n = 5 (D) independent experiments (*p<0.01, ***p<0.001, ****p< 0.0001).
-
Figure 7—source data 1
Intracellular calcium measurements data analysis.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig7-data1-v2.xlsx
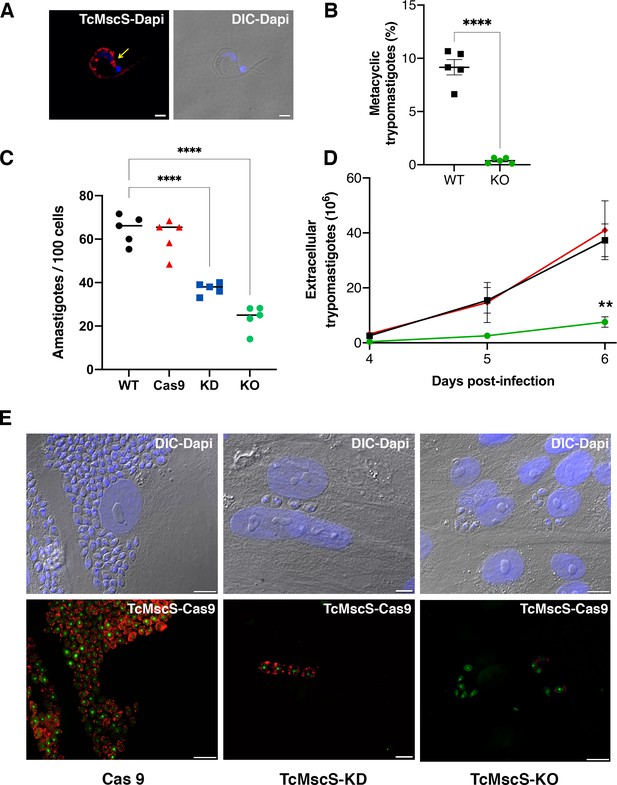
Infectivity defects in TcMscS mutants.
(A) Representative immunofluorescence images showing the localization of TcMscS in metacyclic trypomastigotes labeled with anti-TcMscS antibodies (red). The position of the contractile vacuole is indicated by yellow arrows. Nuclei and kinetoplasts were 4′, 6-diamidino-2-phenylindole (DAPI) stained. Bar size = 2 µm. (B) Percentage of metacyclic forms obtained in vitro from wild-type Y strain (WT) and TcMscS-KO parasites incubated for 72 hr in TAU 3AAG media. Values are mean ± SEM of five independent experiments. (C) Quantification of intracellular amastigotes at 48 hr post-infection. HEK-293 cells were infected at a multiplicity of infection (MOI) of 25:1 with trypomastigotes WT (black), Cas9 (red), TcMscS-KD (blue), or TcMscS-KO (green). TcMscS mutants show a significant decrease in intracellular amastigotes. Values are mean ± SEM of five independent experiments in triplicate. (D) Quantification of extracellular trypomastigotes released from infected HEK-293 cells after 4 days of infection. Cells were infected at an MOI of 25:1 with WT (black), Cas9 (red), or TcMscS-KO (green) trypomastigotes and extracellular parasites were collected daily for counting. Values are mean ± SEM of four independent experiments. (E) Representative images of HEK-293 cells infected with Cas9 (Cas9), TcMscS-KD, or TcMscS-KO parasites. TcMscS was detected with specific antibodies (red); Cas9 was labeled with anti-GFP; nuclei and kinetoplasts were DAPI stained. Bar size = 10 µm. (**p<0.01, ****p< 0.0001).
-
Figure 8—source data 1
Quantification of infectivity data.
- https://cdn.elifesciences.org/articles/67449/elife-67449-fig8-data1-v2.xlsx
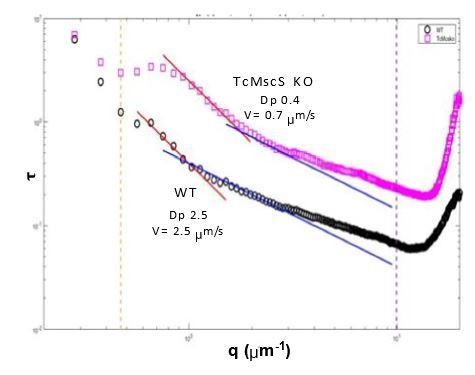
Motility analysis of WT and TcMscS KO by differential dynamic microscopy (DDM).
Dp corresponds to diffusion coefficient and V the linear velocity. There is a significant difference in the speed of KO (0.7 μm/s) vs WT parasites (2.5 μm/s) as well as the diffusional movement (0.4 vs 2.5, respectively).
Videos
Opening transition in the modeled TcMscS.
Motility of Cas9 epimastigotes.
Motility of TcMscS-KO epimastigotes.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (E. coli) | TOP10 | Invitrogen | C404010 | Cloning bacteria |
| Strain, strain background (E. coli) | BL21 (DE3) pLysS | Invitrogen | C606010 | Expression bacteria |
| Strain, strain background (E. coli) | MJF641 (Δ7) | Provided by Dr. Ian Booth (University of Aberdeen, UK) | Spheroplast strain | |
| Cell line (T. cruzi) | Y strain | ATCC | 50832 | Parasites used in this study |
| Cell line (Homo sapiens) | HFF | ATCC | SCRC-1041 CVCL_3285 | Cells used to produce infective forms |
| Cell line (H. sapiens) | HEK293 | BEI resources | NR-9313 CVCL_0045 | Cells used to produce infective forms |
| Transfected construct | Human T Cell Nucleofector Solution | LONZA | VPA-1002 | Transfection reagent for epimastigotes |
| Recombinant DNA reagent | pQE80L | QIAGEN | 32943 | Bacterial expression vector |
| Recombinant DNA reagent | Cas9/pTREXn | Addgene | RRID:Addgene_68708 | Vector for CRISPR-Cas9 -mediated KO |
| Recombinant DNA reagent | tdTomato/ pTREXb | Addgene | RRID:Addgene_68709 | Vector for KO donor cassettes |
| Recombinant DNA reagent | pUC_sgRNA | Addgene | RRID:Addgene_68710 | sgRNA backbone containing vector |
| Recombinant DNA reagent | pTREXn-eGFP | Addgene | RRID:Addgene_62544 | Expression vector for T. cruzi |
| Antibody | Anti-TcMscS | Cocalico Biologicals Inc | Generated for this work | Polyclonal antibody produced in guinea pig used for western blot and IFA analysis (1:1000 and 1:100, respectively). |
| Antibody | Anti-myc (mouse monoclonal) | SIGMA | RRID:AB_439694 (M4439) | Antibody used for western blot analysis and IFA (1: 5000 and 1:250, respectively) |
| Antibody | Anti-actin (mouse monoclonal) | SIGMA | RRID:AB_262137 (A3853) | Antibody used for IFA (1:250) |
| Antibody | Anti-Tubulin (mouse monoclonal) | SIGMA | RRID:AB_477579 (T5168) | Antibody used for IFA (1:500) and western blot (1:5000) |
| Antibody | Anti-trypanosome SSP1 (mouse monoclonal) | BEI Resources | NR-50891 | Used as trypomastigote marker for IFA (1:250) |
| Antibody | Anti-trypanosome SSP4 (mouse monoclonal) | BEI Resources | NR- 50892 | Used as amastigote marker for IFA (1:250) |
| Antibody | Anti- GFP (rabbit polyclonal) | Thermo Fisher | RRID:AB_221570 (A6455) | Antibody used for IFA (1:3000) |
| Chemical compound, drug | Fura2-AM | Thermo Fisher | F1221 | Reagent used for calcium measurements |
| Software, algorithm | pCLAMP11 | Molecular Devices | RRID:SCR_011323 | Electrophysiology acquisition and analysis software |
| Software, algorithm | Graph pad PRISM9 | http://www.graphpad.com | RRID:SCR_002798 | Statistical analysis and graph software |
Additional files
-
Supplementary file 1
Sequences of primers used.
Bold letters indicate restriction sites. Protospacer sequences are underlined, and ultramer sequences that correspond to the flanking regions of the gene are in italics.
- https://cdn.elifesciences.org/articles/67449/elife-67449-supp1-v2.docx
-
Supplementary file 2
Changes in cell volume upon hypoosmotic stress.
Peak and recovery analysis of epimastigotes’ cell volume changes under hypoosmotic stress. For all the conditions, values are the mean ± SE of n = 6 independent experiments. p-values were calculated based on one-way analysis of variance with Bonferroni post-test. Differences were considered significant when p<0.01(*).
- https://cdn.elifesciences.org/articles/67449/elife-67449-supp2-v2.docx
-
Supplementary file 3
Changes in cell volume upon hyperosmotic stress.
Peak and recovery analysis of epimastigotes’ cell volume changes under hyperosmotic stress. For all the conditions, values are the mean ± SE of n = 6 independent experiments. p-values were calculated based on one-way analysis of variance with Bonferroni post-test. Differences were considered significant when p<0.01(*).
- https://cdn.elifesciences.org/articles/67449/elife-67449-supp3-v2.docx
-
Supplementary file 4
Quantification of intracellular amastigotes.
The number of intracellular amastigotes was counted at 6 and 48 hr post-infection. For all the conditions, values are the mean ± SE of n = 5 independent experiments. p-values were calculated based on one-way analysis of variance with Bonferroni post-test. Differences were considered significant when p<0.05(*).
- https://cdn.elifesciences.org/articles/67449/elife-67449-supp4-v2.docx
-
Supplementary file 5
Quantification of extracellular trypomastigotes.
Extracellular trypomastigotes collected from the supernatant of infected cells were counted at days 4, 5, and 6 post-infection. For all the conditions, values are the mean ± SE of n = 4 independent experiments. p-values were calculated based on one-way analysis of variance with Bonferroni post-test. Differences were considered significant when p<0.05(*).
- https://cdn.elifesciences.org/articles/67449/elife-67449-supp5-v2.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/67449/elife-67449-transrepform-v2.pdf






