Therapeutic inhibition of keratinocyte TRPV3 sensory channel by local anesthetic dyclonine
Figures
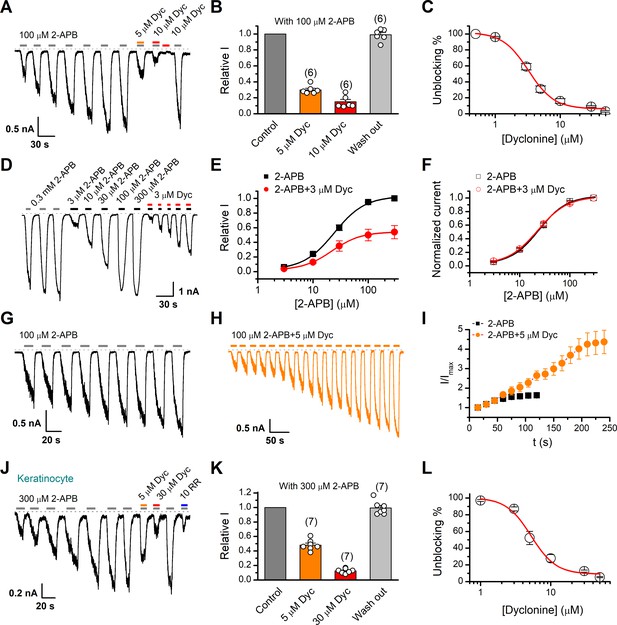
Inhibition of transient receptor potential vanilloid-3 (TRPV3) currents by dyclonine (Dyc).
(A) Inhibition of 2-aminoethoxydiphenylborate (2-APB)-evoked currents by Dyc in a representative HEK 293T cell expressing mouse TRPV3. After sensitization by repeated application of 100 μM 2-APB, the cell was exposed to 5 or 10 μM Dyc with 100 μM 2-APB or 10 μM Dyc only as indicated by the bars. Membrane currents were recorded in whole-cell configuration, and the holding potential was –60 mV. (B) Summary of relative currents elicited by 100 μM 2-APB in the presence of 0, 5, or 10 μM Dyc. Numbers of cells are indicated in parentheses. (C) The dose-response curve for Dyc inhibition of TRPV3 currents was fitted by Hill equation (IC50 = 3.2 ± 0.24 μM and nH = 2.2 ± 0.32, n = 6). (D) Representative whole-cell current traces showing the responses to varying concentrations of 2-APB without or with 3 μM Dyc after full sensitization of TRPV3. (E) Concentration-response curves of 2-APB without or with Dyc. Data are shown as relative values to the current evoked by 300 μM 2-APB. Solid lines are fits to Hill equation, yielding EC50 = 22.93 ± 0.02 μM and nH = 1.6 ± 0.1 without Dyc (n = 6); and EC50 = 22.03 ± 0.86 μM and nH = 1.7 ± 0.1 with 3 μM Dyc (n = 6). (F) Dose-response curves normalized to its own maximum of each condition. (G, H) Representative whole-cell recordings for the sensitization of TRPV3 currents elicited by repeated applications of 100 μM 2-APB in the absence (G) and presence (H) of 5 μM Dyc. (I) Time courses toward the peak currents elicited by repeated application of 100 μM 2-APB with or without Dyc (n = 9). Currents were normalized to each initial values. (J) The 2-APB-evoked inward currents were reversibly inhibited by Dyc in primary mouse epidermal keratinocytes. Representative inward currents were firstly activated by repeated application of 300 μM 2-APB at the holding potential of –60 mV, and then inhibited by 5 or 30 μM Dyc or 10 μM ruthenium red (RR) as indicated. (K) Summary of relative currents elicited by 300 μM 2-APB with or without Dyc. (L) Dose dependence of Dyc effects on TRPV3 currents in cultured keratinocytes. The solid line corresponds to a fit by Hill equation with IC50 = 5.2 ± 0.71 μM and nH = 2.4 ± 0.75 (n = 6). The dotted line indicates zero current level.
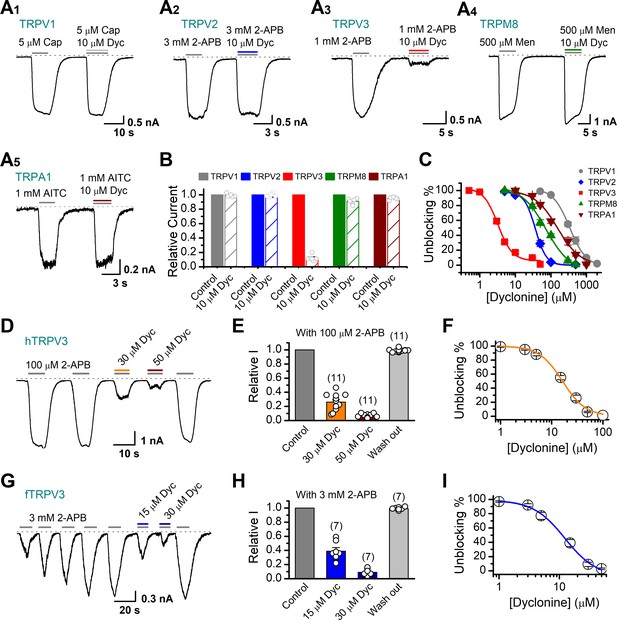
Dyclonine (Dyc) is a potent inhibitor of transient receptor potential vanilloid-3 (TRPV3) channel.
(A) Representative inward current traces from whole-cell voltage-clamp recordings show the inhibitory effects of 10 μM Dyc on TRPV1 (A1), TRPV2 (A2), TRPV3 (A3), TRPM8 (A4), or TRPA1 (A5) channels (Cap: capsaicin; Men: menthol). Bars represent duration of drug application. (B) Summary of relative currents before and after Dyc (10 μM) treatment. Numbers of cells are indicated in parentheses. (C) Dose-response curves of Dyc for inhibition of indicated ion channel currents. Solid lines represent fits by Hill equation, with IC50 = 337.4 ± 19.4 μM and nH = 2.0 ± 0.31 for TPRV1 (n = 7), IC50 = 31.1 ± 2.7 μM and nH = 2.9 ± 0.50 for TPRV2 (n = 8), IC50 = 81.8 ± 12.7 μM and nH = 1.2 ± 0.20 for TRPM8 (n = 6), and IC50 = 154.7 ± 15.6 μM and nH = 1.3 ± 0.15 for TRPA1 (n = 6). For comparison, the dose-response curve of TRPV3 channel from Figure 1C is displayed in red with IC50 = 3.2 ± 0.24 μM and nH = 2.2 ± 0.32 (n = 6). (D) Suppression of 2-aminoethoxydiphenylborate (2-APB)-evoked currents by Dyc in a human TRPV3 (hTRPV3)-expressing HEK 293T cell. Representative inward current trace shows the reversible block effect of Dyc (30 and 50 μM) at the holding potential of –60 mV. (E) Summary of inhibition of hTRPV3 by Dyc. Membrane currents were normalized to the responses elicited by the saturated concentration of 2-APB (100 μM) alone. (F) Dose-response curve for Dyc on blocking of hTRPV3. Solid line represents a fit to a Hill equation, yielding IC50 = 16.2 ± 0.72 μM and nH = 1.91 ± 0.14 (n = 11). (G) Inhibition of frog TRPV3 (fTRPV3) currents by Dyc. Representative whole-cell currents at –60 mV in a fTRPV3-expressing HEK 293T cell. After sensitization by repeated application of 3 mM 2-APB, the cell was exposed sequentially to 15 and 30 μM Dyc with 3 mM 2-APB. (H) Summary of inhibition of relative currents elicited by 3 mM 2-APB, 3 mM 2-APB with Dyc 15 or 30 μM. (I) Concentration-response curve of Dyc on the inhibition of fTRPV3 currents. Solid line represents a fit by a Hill equation, with IC50 = 12.31 ± 1.6 μM and nH = 1.6 ± 0.34 (n = 7). The dotted line indicates zero current level.
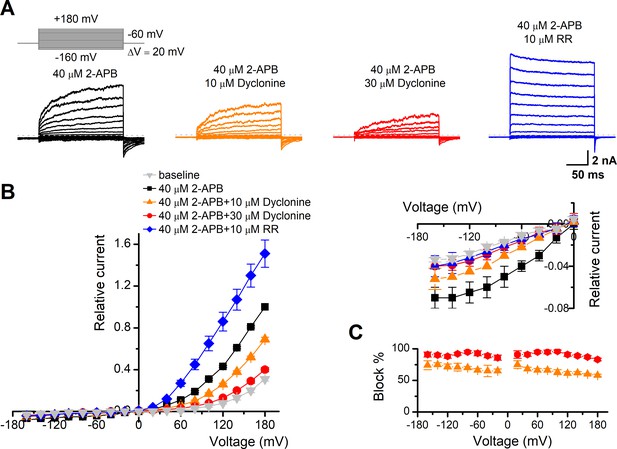
The inhibitory effect of dyclonine on transient receptor potential vanilloid-3 (TRPV3) channel is voltage-independent.
(A) Representative whole-cell currents evoked by voltage steps (inset) together with 40 μM 2-aminoethoxydiphenylborate (2-APB) in the absence and presence of 10, 30 μM dyclonine or 10 μM ruthenium red (RR) in HEK 293T cells expressing mouse TRPV3. Currents were elicited with 200 ms test pulses ranging from –160 mV to +180 mV in 20 mV increments within the same cells, and the holding potential was –60 mV. Calcium-free standard bath solution and a CsCl-filled recording electrode were used. The dotted line indicates zero current level. (B) Current-voltage relations for data in (A). Current amplitudes were normalized to the maximum responses at +180 mV in the presence of 40 μM 2-APB. Each point represents mean values (± SEM) from eight independent cells. (Inset) The inhibition effects of dyclonine and RR on TRPV3 currents at negative holding potentials are magnified and displayed on the right. Note that dyclonine had an inhibitory effect on TRPV3 currents at both positive and negative potentials, but RR only inhibited TRPV3 channel currents at negative potentials while enhanced TRPV3 currents at positive potentials (blue trace). (C) Percentage block of TRPV3 currents by dyclonine (10 and 30 μM) as a function of membrane potential. Error bars represent SEM.
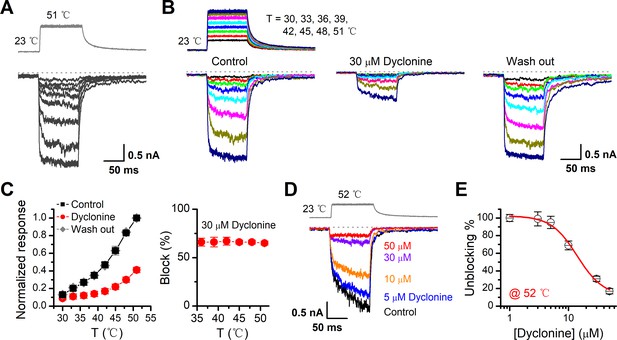
Inhibition of heat-activated transientreceptor potential vanilloid-3 (TRPV3) currents by dyclonine.
(A) Sensitization of TRPV3 by heat. Heat-evoked TRPV3 currents in response to repeated temperature jumps. Temperature pulse generated by infrared laser diode irradiation was stepped from room temperature to 51°C in 1.5 ms and then clamped for 100 ms. (B) Effects of dyclonine on heat-activated TRPV3 currents. Heat-evoked current traces were recorded in whole-cell configuration, which were stabilized by sensitization of repeated fast temperature jumps as shown in (A). Temperature jumps shown on the top had a duration of 100 ms and a rise time of 1.5 ms. Bath solution with 0 or 30 μM dyclonine was applied by brief perfusion to the patch just before temperature stimulation on the same cells. (C) The average plot compares the temperature responses in the absence and presence of 30 μM dyclonine (left, n = 6). Currents were normalized by their maximum responses under control condition, respectively. Note that data from control and washout are superimposed. Percentage block of heat-evoked TRPV3 currents by 30 μM dyclonine as a function of temperature is shown on the right. (D) Representative inward currents evoked by a series of identical temperature jumps inhibited by dyclonine in a concentration-dependent manner. The temperature pulse (52°C) is shown in gray. Holding potential was –60 mV. (E) Dose dependence of dyclonine effects on heat-activated TRPV3 currents. The solid line represents a fit to Hill equation with IC50 = 14.1 ± 2.5 μM and nH = 1.9 ± 0.54 (n = 10). All whole-cell recordings were got from TRPV3-expressing HEK 293T cells held at –60 mV.
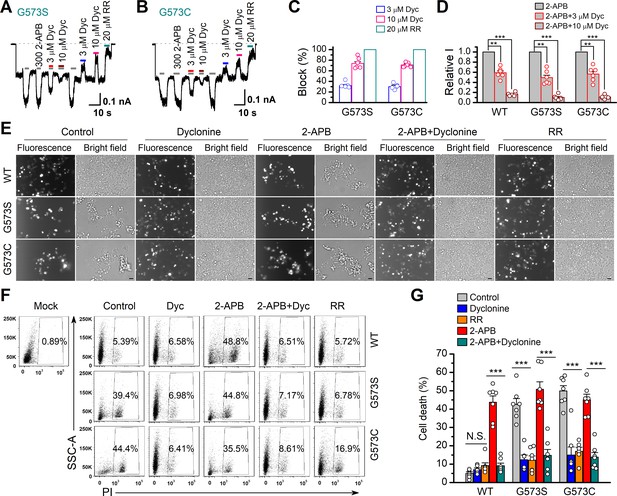
Dyclonine (Dyc) rescued cell death caused by expression of overactive transient receptor potential vanilloid-3 (TRPV3) mutant.
(A, B) Effects of Dyc on whole-cell currents recorded from TRPV3 (G573S) and TRPV3 (G573C) expressed in T-Rex 293 cells, showing that Dyc (3 and 10 μM) reversibly inhibited the response to 300 μM 2-aminoethoxydiphenylborate (2-APB) and the spontaneous activities at –60 mV. 20 μM ruthenium red (RR) was applied for subtracting leak currents. Bars represent duration of drug application. (C) Averaged inhibition of spontaneous activities of G573 mutants by Dyc and RR. (D) Summary of relative whole-cell currents of TRPV3 (wild-type [WT]), G573S, and G573S with or without Dyc treatment. Error bars represent SEM. (E) Bright-field and fluorescence images showing the cell survival. The GFP-tagged TRPV3 (WT) and two mutants (G573C and G573S) in pcDNA4/TO vector were respectively transfected into T-Rex 293 cells, and then treated with doxycycline (20 ng/ml) for 16 hr post-transfection to induce gene expression in the presence of drugs as indicated. Images of cells were taken at 12 hr after induction. Scale bar, 50 μm. (F) Flow cytometry analysis of the percentage of dead cells. Cells transfected with the desired plasmids are as indicated. After the gene expression induced by doxycycline, the cells were treated with Dyc (50 μM), 2-APB (30 μM), RR (10 μM), or the combination of 2-APB and Dyc, and then stained with propidium iodide, followed by flow cytometry to analyze cell survival. (G) Summary plots of cell death rates under different treatments. Data were averaged from seven independent experiments. *** p<0.0001.
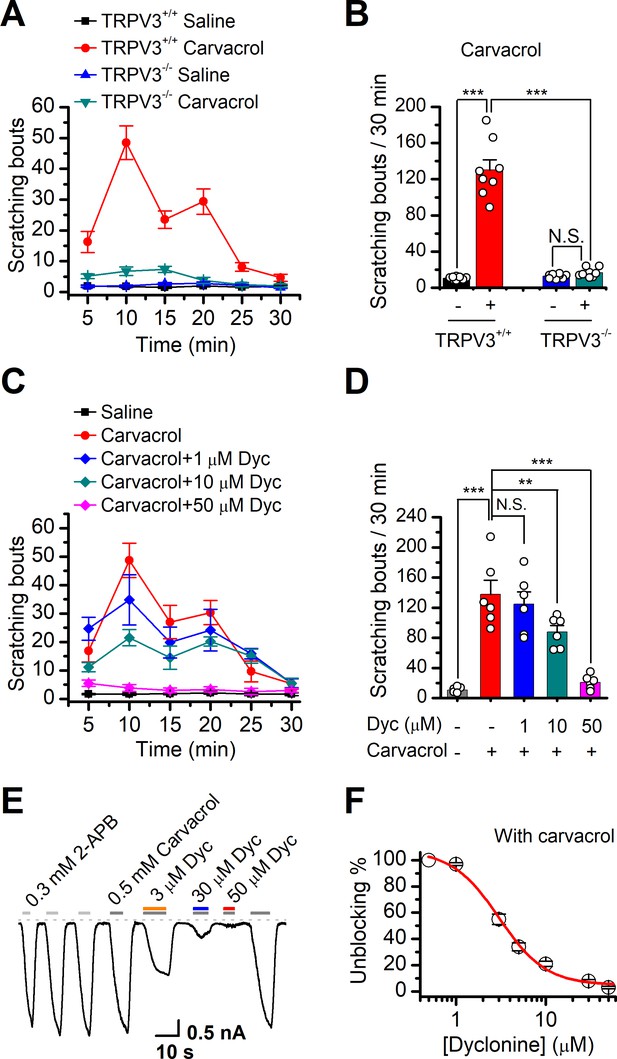
Dyclonine (Dyc) suppresses scratching behavior induced by carvacrol.
(A) Summary of the time courses of neck-scratching behaviors in wild-type transientreceptor potential vanilloid-3 (TRPV3) and TRPV3 knock out (C57BL/6) mice after intradermal injection of 50 μl carvacrol (0.1%) or normal saline (0.9% NaCl) containing 0.1% ethanol into the mouse neck. Time for scratching bouts was plotted for each 5 min interval over the 30 min observation period. (B) Quantification of the cumulative scratching bouts over 30 min under different treatments, showing that intradermal injection of carvacrol elicited a remarkable increase in the number of scratching bouts in TRPV3+/+ but not TRPV3-/- mice (n = 6; N.S.: no significance; *p<0.05; **p<0.01; ***p<0.001, by one-way ANOVA). (C) Time courses of neck-scratching behaviors in response to intradermal injection of 50 μl carvacrol (0.1%), with pretreatment of normal saline (0.9% NaCl), or different concentrations (1, 10, and 50 μM) of Dyc in the same site. (D) Summary plots of the number of scratching bouts over 30 min under different treatments as indicated, showing that Dyc dose-dependently alleviated carvacrol-evoked acute itch (n = 6; N.S.: no significance; *p<0.05; **p<0.01; ***p<0.001, by one-way ANOVA). (E) Inhibition of carvacrol-evoked currents by Dyc in a representative HEK 293T cell expressing TRPV3. After sensitization by repeated application of 300 μM 2-aminoethoxydiphenylborate (2-APB), the cell was exposed to 3, 30, or 50 μM Dyc with 500 μM carvacrol as indicated by the bars. Membrane currents were recorded in a whole-cell configuration, and the holding potential was –60 mV. (F) The dose-response curve for Dyc inhibition of carvacrol-evoked TRPV3 currents was fitted by Hill equation (IC50 = 3.5 ± 0.34 μM and nH = 2.1 ± 0.41, n = 8).
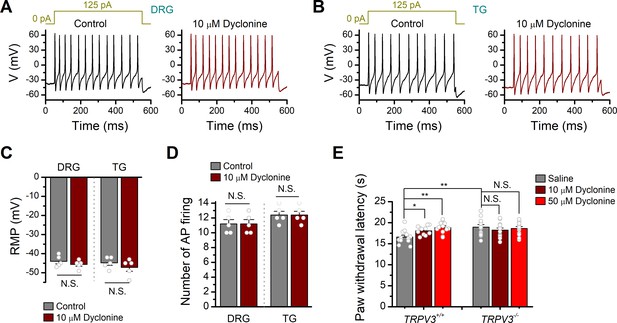
Effects of dyclonine on the excitability of mouse dorsal root ganglia (DRG) and trigeminal ganglia (TG) neurons as well as the transient receptor potential vanilloid-3 (TRPV3)-mediated nociceptive behavior.
(A, B) Current-clamp responses of DRG and TG neurons to 500 ms current pulse injection, respectively. Protocol of injected current is shown on the top. (C) Statistics plot showing no significant changes for resting membrane potential (RMP) from DRG or TG neurons in the presence of dyclonine (10 μM) compared with control. RMPs were –44 ± 1.3 mV (n = 5) to –45 ± 0.9 mV (n = 5) treated with dyclonine in DRG neurons, and –44.8 ± 1.3 mV (n = 5) to –47.2 ± 1.9 mV (n = 5) treated with dyclonine in TG neurons. (D) Comparison of averaged frequency of action potential (AP) firing (numbers of action potential firing) evoked by current injection of 125 pA lasting for 500 ms in DRG and TG neurons. (E) Paw withdrawal or licking latencies to noxious thermal stimuli at 55°C evaluating the effect of dyclonine on thermal pain sensing. The paw withdrawal latencies (PWL) were 16.57 ± 0.46 s (n = 12), 18.08 ± 0.03 s (n = 12), and 18.82 ± 0.42 s (n = 12) for hind paws with intraplantar injection with saline, 10and 50 μM dyclonine in TRPV3+/+ mice, respectively. The PWL were 18.93 ± 0.61 s (n = 12), 18.23 ± 0.52 s (n = 12), and 18.61 ± 0.46 s (n = 12) for hind paws with intraplantar injection with saline, 10and 50 μM dyclonine in TRPV3-/- mice. N.S.: no significance; *p<0.05; **p<0.01; ***p<0.001.
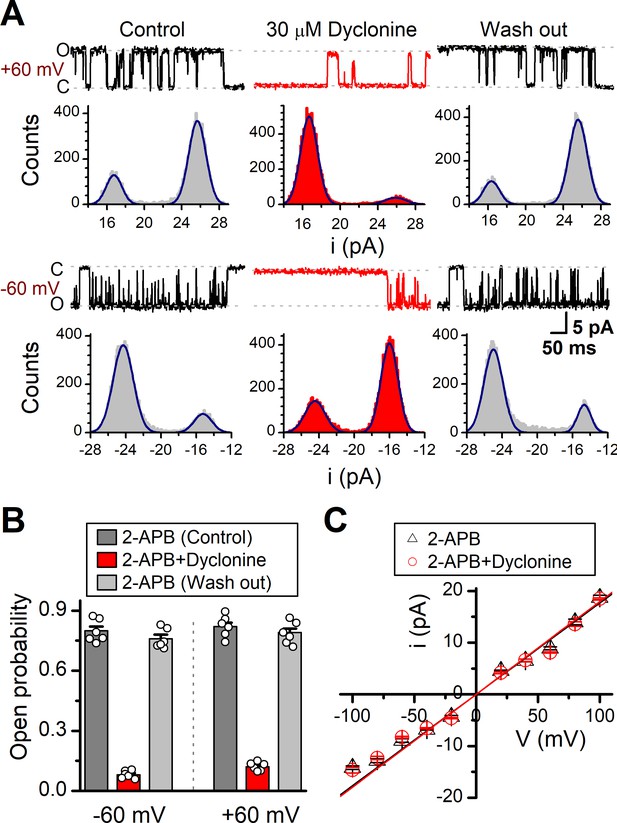
Effects of dyclonine on single-channel properties of transient receptor potential vanilloid-3 (TRPV3).
(A) Single-channel currents of TRPV3 were recorded from inside-out membrane patches of HEK 293T cells at two membrane potentials (± 60 mV) in symmetrical 150 mM NaCl and were low-pass filtered at 2 kHz. Currents were evoked by 10 μM 2-aminoethoxydiphenylborate (2-APB) in the absence and presence of dyclonine (30 μM) after sensitization induced by repetitive 300 µM 2-APB. All-point amplitude histograms of single-channel currents were shown below the current traces. The histograms were fit to sums of two Gaussian functions to determine the average amplitudes of currents and the open probabilities. Dotted lines indicate the opened channel state (O) and the closed channel state (C), respectively. (B) Summary of effects of dyclonine on TRPV3 single-channel open probability. Dyclonine (30 μM) decreased TRPV3 open probability from 0.8 ± 0.02 to 0.08 ± 0.01 at –60 mV (n = 6), and from 0.82 ± 0.02 to 0.12 ± 0.01 at +60 mV (n = 6), respectively. (C) I-V relationships of TRPV3 single-channel current evoked by 10 μM 2-APB without (black triangles) and with 30 μM dyclonine (red circles). Unitary conductance measured by fitting a linear function were 163.6 ± 6.4 pS and 179.2 ± 5.5 pS for before and after treatment by dyclonine, respectively.
Molecular residues involved in dyclonine-transient receptor potential vanilloid-3 (dyclonine-TRPV3) interaction.
(A) Overall view of the mTRPV3-dyclonine complex. Three putative binding modes (BMs) for dyclonine in the pore cavity of mTRPV3 channel (PDB ID code: 6DVZ) are denoted as BMA, BMB, and BMC (please find the details in the text), with the expanded view of BMA shown on the right. Four subunits of the tetramer are distinguished by different colors, and dyclonine in a schematic structure is shown in red. (B) (Left) Potential docking poses of dyclonine and TRPV3 channel. (Right) Cluster analysis showing all BMs distributed into three clusters, BMA, BMB, and BMC. (C) Representative whole-cell recordings show reversible blocking of 2-aminoethoxydiphenylborate (2-APB) (1 mM)-evoked responses by dyclonine (3, 10, or 30 μM) in HEK 293T cells expressing mutant TRPV3 channels as indicated, respectively. The combination of 3, 10, or 30 μM dyclonine and 2-APB was applied following the control currents evoked by a saturated concentration of 2-APB (1 mM, initial gray bar). Holding potential was –60 mV. Bars represent duration of stimuli. (D) Concentration-response curves of dyclonine on inhibition of the TRPV3 mutants. Solid lines represent fits by a Hill equation, with the half-maximal inhibitory concentration (IC50) shown in (E). For comparison, the dose-response curve of wile-type channel is displayed in gray. Four point mutations (L630W, N643A, I644W, and L655A) reduced the inhibitory efficiency of dyclonine, while the other two point mutations (L642A and I659A) enhanced the inhibitory effects of dyclonine on TRPV3 currents. (F) Average current responses of mutant channels compared with wild-type TRPV3 channels. Each substitution of putative residues except L639A retained their normal responses to 2-APB. Numbers of cells are indicated in parentheses. (G) Modulation of thiol-oxidizing and disulfide-reducing agents on the inhibitory effects of dyclonine. Whole-cell recordings from the wild-type TRPV3 and the mutants expressed in HEK 293 T cells, showing the effects of (2-(trimethylammonium) ethyl methanethiosulfonate, bromide) (MTSET) and dithiothreitol (DTT) on the responses to 2-APB with or without dyclonine after sensitization induced by 300 μM 2-APB. MTSET (1 mM) and DTT (10 mM) were locally applied for ~3 min to probe the accessibility, respectively. The responsiveness to 2-APB or 2-APB plus dyclonine was subsequently examined before and after treatments. Holding potential was –60 mV. (H) Summary of inhibition of relative currents elicited by 300 μM 2-APB, 300 μM 2-APB with dyclonine 10 or 1 μM. (I) Summary of inhibitory effects of dyclonine before and after treatments with MTSET and DTT. The dotted line indicates zero current level in all cases. Error bar represents SEM. N.S.: no significance; *p<0.05; **p<0.01; ***p<0.001.
Residues in the transient receptor potential vanilloid-3 (TRPV3) channel pore for interacting with dyclonine.
(A) Receptor grid for docking generated with 35 Å × 35 Å × 35 Å dimensions. (B) Detailed binding modes (BMs) of dyclonine in BMB and BMC. The putative interaction residues are labeled, and dyclonine is displayed in sticks for emphasis. (C) Representative whole-cell recordings show reversible blocking of 2-aminoethoxydiphenylborate (2-APB) (1 mM)-evoked responses by dyclonine (10 or 30 μM) in HEK 293Tcells expressing mutant TRPV3 channels as indicated, respectively. Holding potential was –60 mV. Bars represent duration of stimuli. (D) Concentration-response curves of dyclonine on inhibition of the TRPV3 mutants. Solid lines represent fits by a Hill equation, with the half-maximal inhibitory concentration (IC50) shown in (E). For comparison, the dose-response curve of wild-type is displayed in gray. (F) Average current responses of mutant channels compared with wild-type TRPV3 channels. Only cells that expressed I637A, F666A, or I674A showed similar response as wild-type channel, while others with the substation by alanine were not functional. Numbers of cells are indicated in parentheses. (G) Structures assigned to apo/resting (left) and open states (right). (H) Cavity fostered by the pore helix and S5–S6 domains of TRPV3 channels at the resting (left) and open (right) states.

Alignment of the pore-region sequences.
Alignment of the pore sequence of mTRPV3 with other TRP channels, with the identical or similar residues shaded in colors. Meanwhile, differences in amino acids compositions present for different channels. The key residues of transient receptor potential vanilloid-3 (TPRV3) are indicated at the top, which affects the inhibitory effect of dyclonine.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Species (Mus musculus) | Trpv3-/- mice | Wang et al., 2021 | PMID:32535744 | C57BL/6J background |
| Cell line (Homo sapiens) | HEK 293T | ATCC | Cat.#:CRL-3216 | |
| Cell line (Homo sapiens) | T-Rex 293 | Thermo Fisher | Cat.#:R71007 | |
| Chemical compound | 2-APB | Sigma-Aldrich | Cat.#:D9754 | TRPV1-3 agonist |
| Chemical compound | Carvacrol | MedChemExpress | Cat.#:499752 | TRPV3 agonist |
| Chemical compound | Menthol | Sigma-Aldrich | Cat.#:M278 | TRPM8 agonist |
| Chemical compound | Capsaicin | MedChemExpress | Cat.#: HY10448 | TRPV1 agonist |
| Chemical compound | AITC | Sigma-Aldrich | Cat.#:377430 | TRPA1 agonist |
| Chemical compound | Ruthenium red | Sigma-Aldrich | Cat.#:R2751 | TRP channel inhibitor |
| Chemical compound | Poly-L-lysinehydrochloride | Sigma-Aldrich | Cat.#:2658 | |
| Chemical compound | MTEST | MedChemExpress | Cat.#:690632554 | |
| Chemical compound | DTT | Sigma-Aldrich | Cat.#:3483123 | |
| Chemical compound | Dyclonine | MedChemExpress | Cat.#:536436 | |
| Software, algorithm | Patchmaster | HEKA Electronics | ||
| Software, algorithm | OriginPro | Originlab.com | ||
| Software, algorithm | Clampfit 10 | Molecular Devices | ||
| Software, algorithm | SigmaPlot 10 | SPSS Science |





