Assessing the effects of stress on feeding behaviors in laboratory mice
Figures
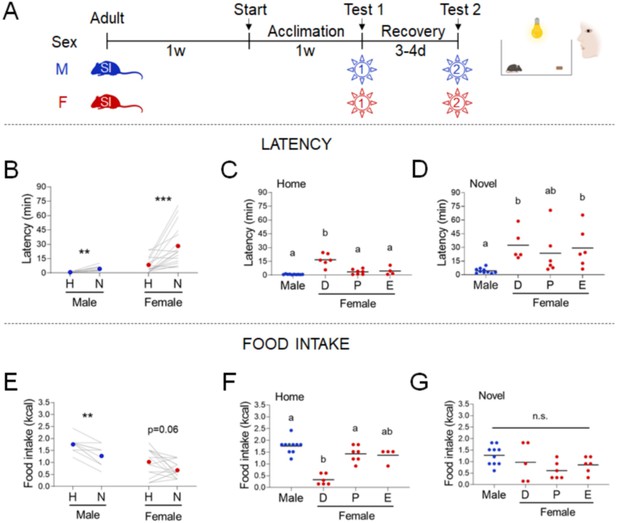
Effects of sex and estrous cyclicity in the standard novelty-suppressed feeding (NSF) assay.
(A) Experimental paradigm for the standard NSF assay performed on the bench, in the morning following an overnight fast. (B) Latency to eat in home (H) and novel (N) tests in males (blue, n = 10, paired t-test: t = 4.094; df = 9; p = 0.027) and females (red, n = 17, Wilcoxon test: W = 141; p = 0.0002). (C) Latency to eat in the home test in males (blue, n = 10) and females (red, n = 17) categorized by estrous cycle stage (D: diestrus; P: proestrus; E: estrus) (one-way analysis of variance [ANOVA]: F(3, 23) = 22.52; p < 0.0001). (D) Latency to eat in the novel test in males (blue, n = 10) and females (red, n = 17) categorized by estrous cycle stage (Kruskal–Wallis test: H(3) = 16.80; p = 0.0008). (E) Food intake in home (H) and novel (N) tests in males (blue, n = 10, Wilcoxon test: W = −36; p = 0.0078) and in females (red, n = 17, Wilcoxon test: W = −67; p = 0.0562). (F) Food intake in males (blue, n = 10) and females (red, n = 17) categorized by estrous cycle stage in the home test (Kruskal–Wallis test: H(3) = 15.86; p = 0.0012). (G) Food intake in males (blue, n = 10) and females (red, n = 17) categorized by estrous cycle stage in the novel test (one-way ANOVA: F(3, 23) = 2,370; p = 0.0968). Significant differences denoted by different letters. **p < 0.01 and ***p < 0.001 between home and novel tests. SI, social isolation; n.s., not significant. See Figure 1—source data 1.
-
Figure 1—source data 1
Standard novelty-suppressed feeding (NSF) assay in both sexes and across estrous cycle.
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig1-data1-v1.xlsx
-
Figure 1—source data 2
Sex hormones across estrous cycles.
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig1-data2-v1.xlsx
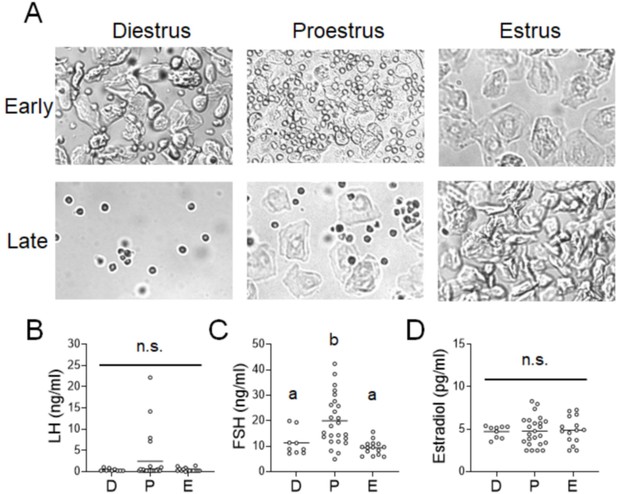
Evaluation of the estrous phase with cytology and hormone level measurements.
(A) Representative pictures of vaginal cytology used to characterize phases of the estrous cycle. (B) Circulating levels of luteinizing hormone (LH) across the estrous cycle (Kruskal–Wallis test: H(2) = 0.1165; p = 9434). (C) Circulating levels of follicle-stimulating hormone (FSH) across the estrous cycle (Kruskal–Wallis test: H(2) = 17.20; p = 0.0002). (D) Circulating levels of estradiol across the estrous cycle (one-way analysis of variance [ANOVA]: F(2, 45) = 0.02807). Significant differences denoted by different letters. n.s., not significant. See Figure 1—source data 2.
Effects of minimal environmental stress on the novelty-suppressed feeding (NSF) assay in females.
(A) Experimental paradigm for the automated NSF assay performed in the automated recording system, in the morning following an overnight fast, in adult female mice socially isolated 2 weeks before the tests (n = 21). (B) Latency to eat in the home test in females categorized by estrous cycle stage (D: diestrus; P: proestrus; E: estrus) (Kruskal–Wallis test: H(2) = 1.269; p = 0.5479). (C) Latency to eat in the novel test in females categorized by estrous cycle stage (one-way analysis of variance [ANOVA]: F(2, 18) = 1.385; p = 0.2757). (D) Latency to eat in home (H) and novel (N) tests (Wilcoxon test: W = 151; p = 0.0071). (E) Food intake in the home test in females categorized by estrous cycle stage (one-way ANOVA: F(2, 18) = 0.0701; p = 0.9326). (F) Food intake in the novel test in females categorized by estrous cycle stage (one-way ANOVA: F(2, 18) = 0.2336; p = 0.7941). (G) Food intake in home (H) and novel (N) tests (paired t-test: t = 1.810; df = 20; p = 0.0854). **p < 0.01 between home and novel tests. SI, social isolation; n.s., not significant. See Figure 2—source data 1.
-
Figure 2—source data 1
Effect of minimal environmental stress on the standard novelty-suppressed feeding (NSF) assay in females.
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig2-data1-v1.xlsx
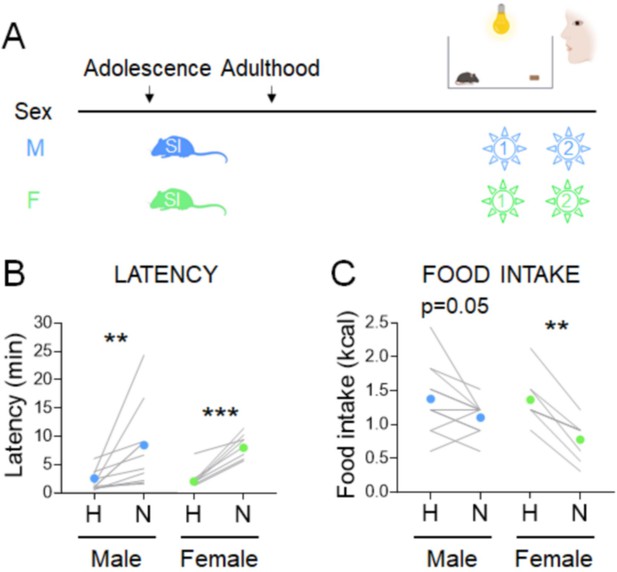
Effects of adolescent social isolation on the novelty-suppressed feeding (NSF) assay in males and females.
(A) Experimental paradigm for the NSF assay performed in the morning following an overnight fast, in adult mice socially isolated at 5 weeks of age. (B) Latency to eat in home (H) and novel (N) tests in males (blue, n = 11, Wilcoxon test: W = 66; p = 0.001) and in females (green, n = 8, Wilcoxon test: W = 36; p = 0.0078). (C) Food intake in home (H) and novel (N) tests in males (blue, n = 11, paired t-test: t = 2.193; df = 10; p = 0.0531) and in females (green, n = 8, paired t-test: t = 6.347; df = 7; p = 0.0004). **p < 0.01, ***p < 0.001 between home and novel tests. SI, social isolation. See Figure 3—source data 1.
-
Figure 3—source data 1
Effects of long social isolation starting at adolescence on the standard novelty-suppressed feeding (NSF) assay.
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig3-data1-v1.xlsx
-
Figure 3—source data 2
Parsing the effects of length vs. timing of social isolation on the standard novelty-suppressed feeding (NSF) assay in females.
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig3-data2-v1.xlsx
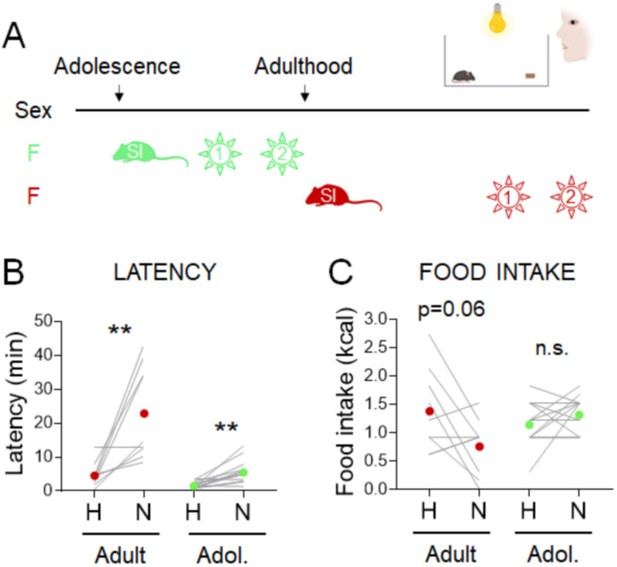
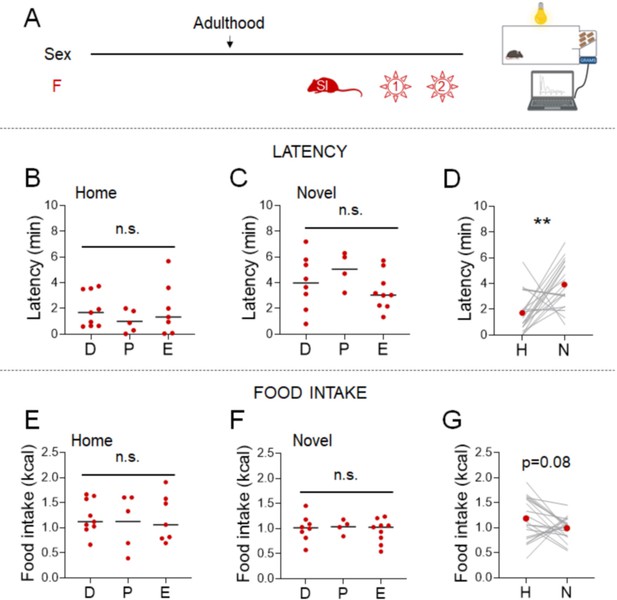
Parsing the effects of length vs. timing of social isolation on the standard novelty suppressed feeding (NSF) assay in females.
(A) Experimental paradigm for the manual novelty-suppressed feeding (NSF) assay in adolescent and adult females. (B) Latency to eat in home (H) and novel (N) tests in adult females socially isolated from 8 weeks of age for 7 weeks (red, n = 9, Wilcoxon test: W = 43; p = 0.0078) and in 7-week-old female mice socially isolated at 5 weeks of age (green, n = 13, Wilcoxon test: W = 85; p = 0.0012). (C) Food intake in home (H) and novel (N) tests in adult females socially isolated at 8 weeks of age for 7 weeks (red, n = 9, paired t-test: t = 2.211; df = 8; p = 0.058) and in 7-week-old female mice socially isolated at 5 weeks of age (green, n = 13, Wilcoxon test: W = 18; p = 0.2656). **p < 0.01. SI, social isolation; n.s., not significant. See Figure 3—source data 2.
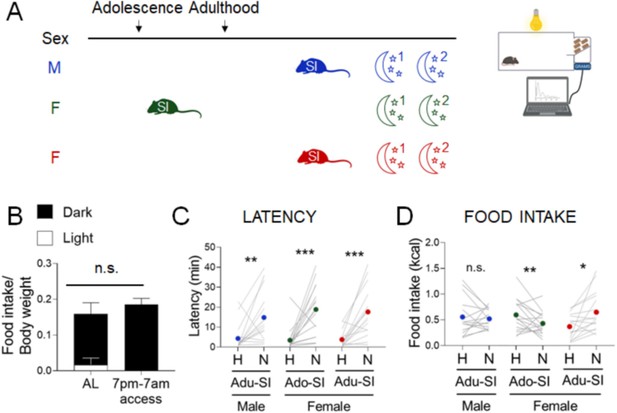
Effects of diurnal factors on the novelty-suppressed feeding (NSF) assay in males and females.
(A) Experimental paradigm for the automated NSF assays performed at the onset of the dark phase in adult mice. (B) Food intake per g body weight across the light cycle (white bar) and dark cycle (black bar) of female mice fed ad libitum (AL) or on a 7 pm to 7 am schedule (n = 7, paired t-test: t = 1.777; df = 6; p = 1259). (C) Latency to eat in home (H) and novel (N) tests in males socially isolated 2 weeks before the tests (blue, n = 15, Wilcoxon test: W = 92; p = 0.0067), females socially isolated during adolescence (green, n = 16, Wilcoxon test: W = 120; p = 0.0008), and females socially isolated 2 weeks before the tests (red, n = 12, Wilcoxon test: W = 126; p = 0.0003). (D) Food intake in home (H) and novel (N) tests in males socially isolated 2 weeks before the tests (blue, n = 15, Wilcoxon test: W = −4.0; p = 0.9229), females socially isolated during adolescence (green, n = 16, paired t-test: t = 2.166; df = 15; p = 0.0468), and females socially isolated 2 weeks before the test (red, n = 12, Wilcoxon test: W = 106; p = 0.0038). *p < 0.05, **p < 0.01, ***p < 0.001 between home and novel tests. SI, social isolation; n.s., not significant. See Figure 4—source data 1.
-
Figure 4—source data 1
Stress assay performed in the dark phase of the light cycle.
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig4-data1-v1.xlsx
-
Figure 4—source data 2
Parsing the effects of the time of day vs. prandial state.
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig4-data2-v1.xlsx
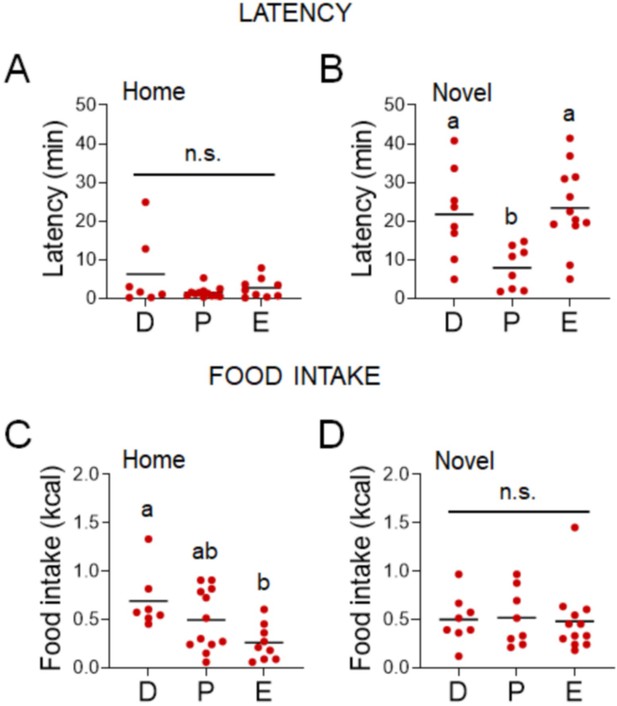
Influences of the estrous cycle when the stress assay is performed in the dark phase of the diurnal cycle.
(A) Home test latency across the estrous cycle (D: diestrus; P: proestrus; E: estrus) when the assay is performed at the onset of the dark phase (n = 28, Kruskal–Wallis test: H(2)= 0.5602; p = 0.7557). (B) Novel test latency across the estrous cycle when the assay is performed at the onset of the dark phase (n = 28, one-way analysis of variance [ANOVA]: F(2, 25) = 1.197; p = 0.0053). (C) Home test latency across the estrous cycle when the assay is performed at the onset of the dark phase (n = 28, Kruskal–Wallis test: H(2) = 7.649; p = 0.0218). (D) Novel test latency across the estrous cycle when the assay is performed at the onset of the dark phase (n = 28, Kruskal–Wallis test: H(2) = 0.4612; p = 0.7941). Significant differences denoted by different letters. SI, social isolation; n.s., not significant. See Figure 4—source data 1.
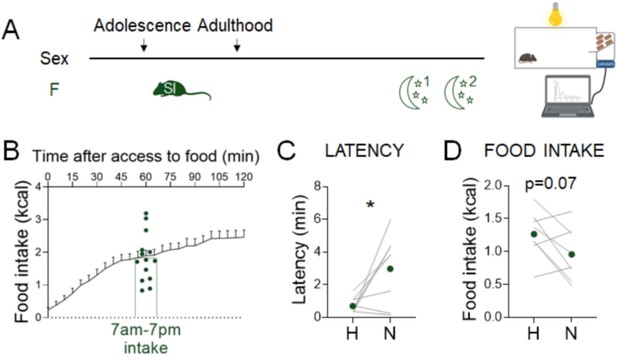
Parsing the effects of the time of day vs. prandial state on the effects of novel environment stress on feeding behavior.
(A) Experimental paradigm for the automated novelty-suppressed feeding (NSF) assays performed at the onset of the dark phase following 90% calorie restriction in adult females (n = 8). (B) Representation of how 7 am to 7 pm average intake preceding an overnight fast was matched on a 7 pm to 7 am food access schedule. (C) Latency to eat in home (H) and novel (N) tests (paired t-test: t = 2.932, df = 7; p = 0.022). (D) Food intake in home (H) and novel (N) tests (paired t-test: t = 2.104, df = 7; p = 0.0734). *p < 0.05. SI, social isolation; n.s., not significant. See Figure 4—source data 2.
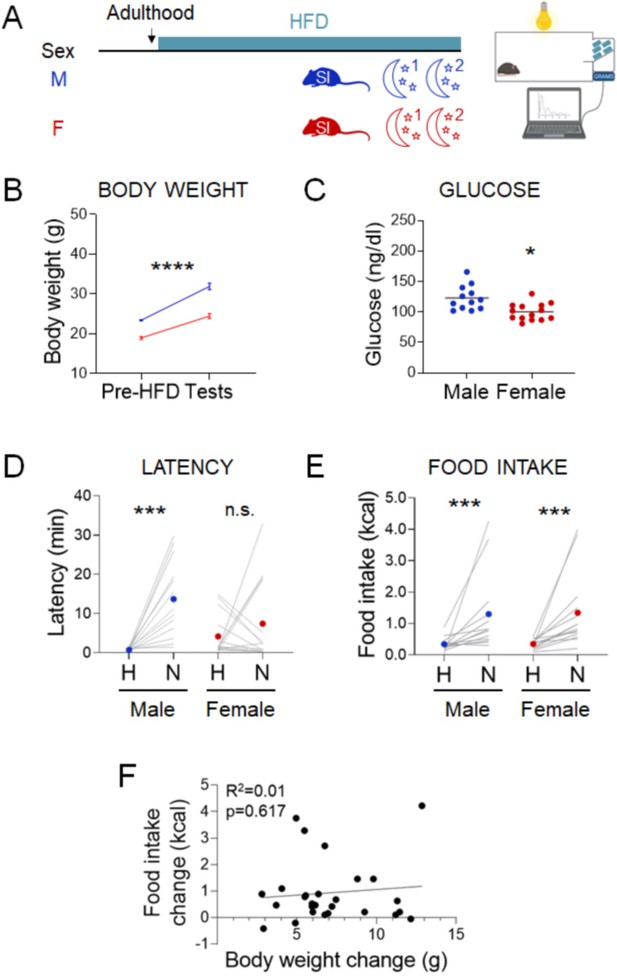
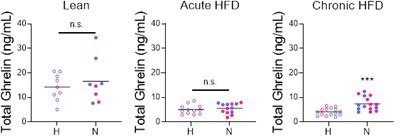
Effects of chronic exposure to high-fat diet (HFD) on the novelty-suppressed feeding (NSF) assay in males and females.
(A) Experimental paradigm for the automated NSF assay performed at the onset of the dark phase in adult chronically exposed to HFD mice socially isolated 2 weeks before the tests. (B) Body weights before exposure to HFD and at the time of the tests, in males (blue, n = 13, paired t-test: t = 11.25; df = 12; p < 0.0001) and females (red, n = 14, paired t-test: t = 13.53; df = 13; p < 0.0001). (C) Blood glucose levels in males (blue, n = 13) and females (red, n = 14, unpaired t-test: t = 3.460; df = 24; p = 0.002). (D) Latency to eat in home (H) and novel (N) tests in adult males (blue, n = 13, paired t-test: t = 4.751; df = 12; p = 0.0005) and females (red, n = 14, Wilcoxon test: W = 5; p = 0.9032). (E) Food intake in home (H) and novel (N) tests in adult males (blue, n = 13, Wilcoxon test: W = 89; p = 0.0005) and in adult females (red, n = 14, Wilcoxon test: W = 105; p = 0.0001). (F) Correlation between change in food intake in N vs. H and change in body weight before and after chronic exposure to HFD. *p < 0.05, ***p < 0.001, ****p < 0.0001. SI, social isolation; n.s., not significant. See Figure 5—source data 1.
-
Figure 5—source data 1
Stress assay performed in the dark phase of the light cycle in overweight mice chronically exposed to high-fat diet (HFD).
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig5-data1-v1.xlsx
-
Figure 5—source data 2
Stress assay performed in the dark phase of the light cycle in lean mice acutely exposed to high-fat diet (HFD).
- https://cdn.elifesciences.org/articles/70271/elife-70271-fig5-data2-v1.xlsx
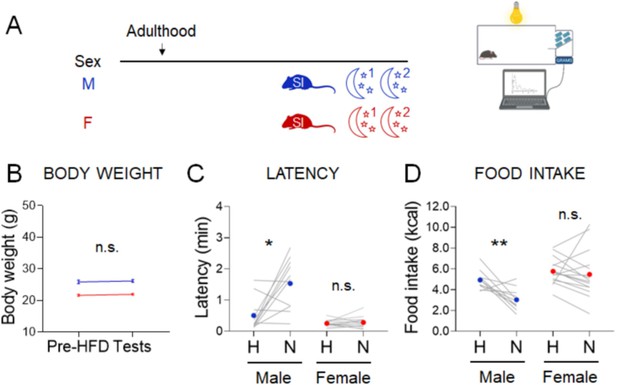
Effect of novel envrionment stress assessed in the dark phase of the light cycle in lean mice acutely exposed to high-fat diet (HFD).
(A) Experimental paradigm for the automated novelty-suppressed feeding (NSF) assay performed at the onset of the dark phase in lean adult socially isolated 2 weeks before the tests, when high-fat diet (HFD) was presented during the test. (B) Body weights before acclimation to HFD and at the time of the tests, in males (blue, n = 11, paired t-test: t = 1.266; df = 10; p = 0.2343) and females (red, n = 15, paired t-test: t = 1.389; df = 15; p = 0.1865). (C) Latency to eat in home (H) and novel (N) tests in males (blue, n = 11, Wilcoxon test: W = 46; p = 0.04) and females (red, n = 15, paired t-test: t = 0.6366; df = 14; p = 0.5347). (D) Food intake in home (H) and novel (N) tests in adult males (blue, n = 11, paired t-test: t = 3.956; df = 10; p = 0.0027) and in adult females (red, n = 15, paired t-test: t = 0.5385; df = 14; p = 0.5987). *p < 0.05; **p < 0.01. SI, social isolation; n.s., not significant. See Figure 5—source data 2.
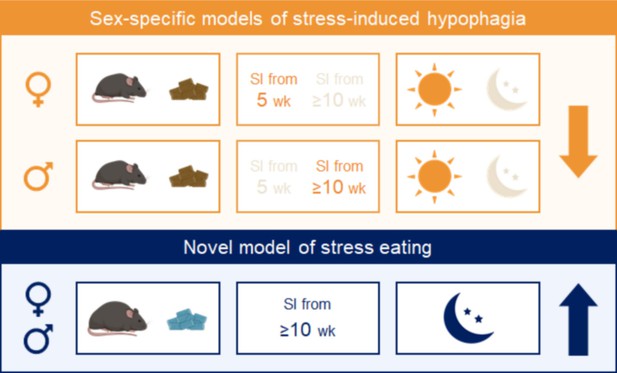
Summary.
Combining experimental variables that influence stress-induced food intake in the same direction yields consistent and reproducible effects in males and females. Sex-specific models of stress-induced hypophagia (top panel). The lean state and performing the assay in the light phase promote hypophagic responses. A short period of social isolation in adult (≥10 weeks) males elicits hypophagic responses, but a prolonged (~6 weeks) period of social isolation starting in adolescence (5 weeks) is needed to produce the same effect in females. Sex-independent model of stress-induced hyperphagia (bottom panel). Chronic exposure to high fat diet and performing the assay in the dark phase promote hyperphagic responses in both sexes.
Additional files
-
Supplementary file 1
Results of multivariable statistical analyses across groups.
(A) Summary of experimental groups. Overview of the number of cohorts and animals included in the analysis of each experimental type, as well as the number of animals excluded because they failed to train or were outliers. (B) Test type. Strength of association between two main outcomes, latency (i) and food intake (ii), and type of the assay (manual vs. automated) in the morning assay in females on chow, socially isolated in adulthood. Strength of association between the two outcomes, latency (iii) and food intake (iv), by test type (novel vs. home), and type of the assay, in the morning assay in females on chow, socially isolated in adulthood. The models were adjusted for body weight, age, and test order (home or novel test performed first). (C) Social isolation length. Strength of association between two main outcomes, latency (i) and food intake (ii), and length of social isolation (2 vs. 6–7 weeks) in both morning and dark phase assays in females on chow. Strength of association between the two outcomes, latency (iii) and food intake (iv), by test type (novel vs. home), and length of social isolation, in both morning and dark phase assays in females on chow. Strength of association between two main outcomes, latency (v) and food intake (vi), and length of social isolation (2 vs. 6–7 weeks) in the morning msuppleemanual assay in males on chow. Strength of association between the two outcomes, latency (vii) and food intake (viii), by test type (novel vs. home), and length of social isolation, in the morning manual assay in males on chow. Strength of association between two main outcomes, latency (ix) and food intake (x), sex, and length of social isolation (2 vs. 6–7 weeks) in the morning manual assay in adult males and females on chow socially isolated at 5 weeks for 7 weeks or during adulthood for 2 weeks. Strength of association between the two outcomes, latency (xi) and food intake (xii), by test type (novel vs. home), sex, and length of social isolation, in the morning manual assay in adult males and females on chow socially isolated at 5 weeks for 7 weeks or during adulthood for 2 weeks. Strength of association between two main outcomes, latency (xiii) and food intake (xiv), estrous cycle, and length of social isolation (2 vs. 6–7 weeks) in adult females on chow socially isolated at 5 weeks for 7 weeks or during adulthood for 2 weeks. The models were adjusted for body weight, age, and test order (home or novel test performed first). (D) Social isolation timing. Strength of association between two main outcomes, latency (i) and food intake (ii), and timing of social isolation (5-week-old vs. adulthood) in both morning and dark phase assays in females on chow. Strength of association between the two outcomes, latency (iii) and food intake (iv), by test type (novel vs. home), and timing of social isolation, in both morning and dark phase assays in females on chow. The models were adjusted for body weight, age, and test order (home or novel test performed first). (E) Time of day. Strength of association between two main outcomes, latency (i) and food intake (ii), and time of day (10 am vs. 7 pm) in adult females on chow (mice socially isolated at 5 weeks or for 7 weeks in the morning assay excluded). Strength of association between the two outcomes, latency (iii) and food intake (iv), by test type (novel vs. home), and time of day (10 am vs. 7 pm) in adult females on chow (mice socially isolated at 5 weeks or for 7 weeks in the morning assay excluded). The models were adjusted for body weight, age, and test order (home or novel test performed first). (F) Chow vs. chronic HFD. Strength of association between two main outcomes, latency (i) and food intake (ii), and type of diet (chow vs. chronic HFD) in the dark phase assay in adult males and females socially isolated in adulthood. Strength of association between the two outcomes, latency (iii) and food intake (iv), by test type (novel vs. home), and type of diet (chow vs. chronic HFD) in the dark phase assay in adult males and females socially isolated in adulthood. The models were adjusted for body weight, age, and test order (home or novel test performed first). (G) Acute HFD. Strength of association between two main outcomes, latency (i) and food intake (ii), and type of diet (chow vs. acute HFD) in the dark phase assay in adult males and females socially isolated in adulthood. Strength of association between the two outcomes, latency (iii) and food intake (iv), by test type (novel vs. home), and type of diet (chow vs. acute HFD) in the dark phase assay in adult males and females socially isolated in adulthood. Strength of association between two main outcomes, latency (v) and food intake (vi), sex, and type of diet (chow vs. acute HFD) in the dark phase assay in adult males and females socially isolated in adulthood. Strength of association between the two outcomes, latency (vii) and food intake (viii), by test type (novel vs. home), sex, and type of diet (chow vs. acute HFD) in the dark phase assay in adult males and females socially isolated in adulthood. Strength of association between two main outcomes, latency (ix) and food intake (x), and length of HFD (acute vs. chronic) in the dark phase assay in adult males and females socially isolated in adulthood. Strength of association between the two outcomes, latency (xi) and food intake (xii), by test type (novel vs. home), and length of HFD in the dark phase assay in adult males and females socially isolated in adulthood. Strength of association between two main outcomes, latency (xiii) and food intake (xiv), sex, and length of HFD in the dark phase assay in adult males and females socially isolated in adulthood. Strength of association between the two outcomes, latency (xv) and food intake (xvi), by test type (novel vs. home), sex, and length of HFD in the dark phase assay in adult males and females socially isolated in adulthood. The models were adjusted for body weight, age, and test order (home or novel test performed first). (H) Sex. Strength of association between two main outcomes, latency (i) and food intake (ii), and sex in mice on chow (females in the automated morning assay, adolescent females and adult females socially isolated for 6–7 weeks excluded). Strength of association between the two outcomes, latency (iii) and food intake (iv), and sex in mice on chow (females in the automated morning assay, adolescent females and adult females socially isolated for 6–7 weeks excluded). Strength of association between two main outcomes, latency (v) and food intake (vi), and sex in all mice on HFD. Strength of association between the two outcomes, latency (iii) and food intake (iv), and sex in all mice on HFD. The models were adjusted for body weight, age, and test order (home or novel test performed first). (I) Estrous cycle. Strength of association between two main outcomes, latency (i) and food intake (ii), and phase of the estrous cycle (Diestrus, Proestrus, and Estrus) in all females on chow and socially isolated in adulthood. Strength of association between two main outcomes, latency (iii) and food intake (iv), and phase of the estrous cycle (Diestrus, Proestrus, and Estrus) in all females on acute or chronic HFD. The models were adjusted for body weight, age, and test order (home or novel test performed first).
- https://cdn.elifesciences.org/articles/70271/elife-70271-supp1-v1.xlsx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/70271/elife-70271-transrepform1-v1.docx