Isolated catatonia-like executive dysfunction in mice with forebrain-specific loss of myelin integrity
Figures

Targeting proteolipid protein (PLP) expression in the forebrain of mice.
(A) Scheme illustrating the generation of mice with forebrain-specific loss of PLP (PLP conditional knockout [cKO]). Forebrain specificity is mediated by the Emx1IREScre driver line. Note that these mice are myelinated despite the absence of PLP from most cortical oligodendrocytes. (B) Immunofluorescence of PLP stained in the forebrain of PLP cKO mice and controls. (B’) Quantification of the PLP-immunopositive areas in the cortex (CTX), corpus callosum (CC), and striatum (STR). For statistical analysis, a two-sided Student’s t-test was performed. Datapoints represent individual mice (n). n=4 for controls and n=4 for PLP cKO. Age of animals 11 months. (C) Light-sheet microscopy to detect PLP in forebrain PLP cKO mice. Whole-mount brain sections (1 mm) were subjected to immunostaining and clearing. Boxed areas are enlarged in the lower panels. White arrows indicate sparse PLP-immunolabeling in forebrain cortical structures and the hippocampus of PLP cKO in comparison to control mice. Autofl: autofluorescence. Age of the animals is 3 months. (D) Immunoblot detection of PLP and its smaller isoform DM20, myelin basic protein (MBP), and actin in lysates of the prefrontal cortex and cerebellum of forebrain PLP cKO mice. Abundance of the protein of interest (POI) was normalized to actin. Respective quantifications are shown in the lower panel. For statistical analysis, a two-sided Student’s t-test was performed. Datapoints represent individual mice (n). n=3 for Ctrl and n=4 for PLP cKO. Age of the animals was 4 months. All data are shown as means ± SEM. Levels of significance were defined as p>0.05 (*), p>0.01 (**) and p>0.001 (***).
-
Figure 1—source data 1
Uncropped/full western blots for western blot bands shown in Figure 1D.
- https://cdn.elifesciences.org/articles/70792/elife-70792-fig1-data1-v1.zip

Loss of myelin integrity in recombined areas in forebrain proteolipid protein (PLP) conditional knockout (cKO) mice.
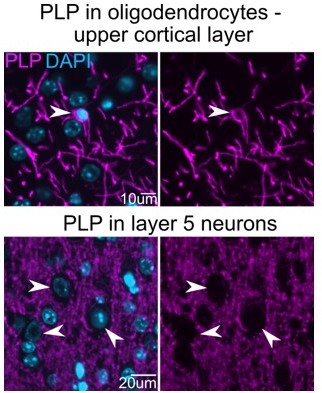
(A) Immunofluorescence staining of myelin (myelin basic protein [MBP] and PLP colabeling) in coronal brain slices of PLP cKO and control mice. Age of animals was 11 months. (B) Closeup images of boxed areas in the medial prefrontal cortex (mPFC) shown in A. Note the reduced number of MBP and PLP-immunostained fibers in PLP cKO mice indicative of dysmyelination. (C) Quantification of images shown in A in the region of interests (ROIs) indicated in the scheme on the left. UL-mPFC: upper layer medial prefrontal cortex. Bars represent means ± SEM, datapoints represent individual mice (n=3 for each group). For statistical analyses two-sided Student’s t-tests were performed. Levels of significance were defined as p>0.05 (*), p>0.01 (**) and p>0.001 (***).
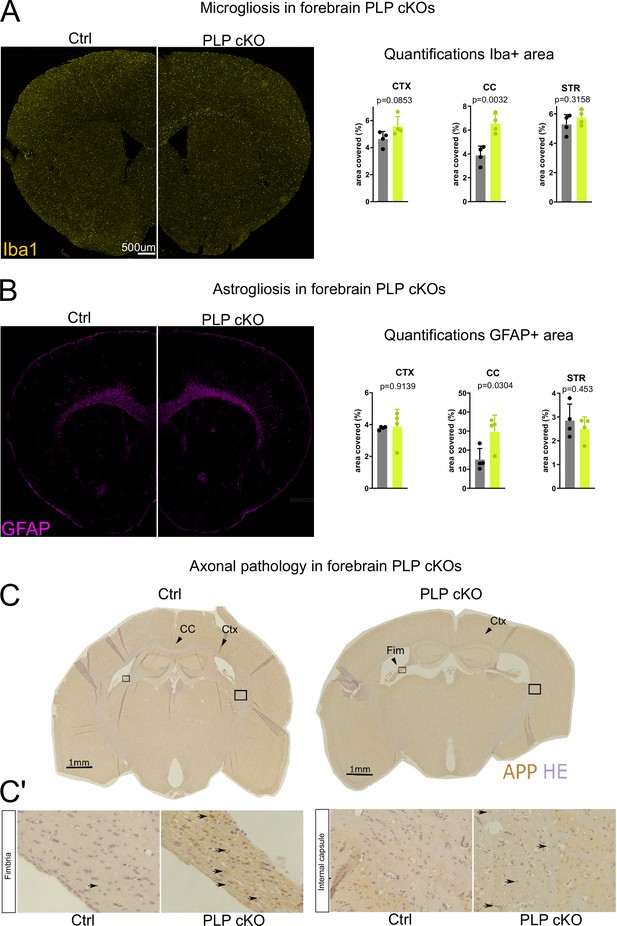
Neuropathological changes in the subcortical white matter in forebrain proteolipid protein (PLP) conditional knockout (cKO) mice.
(A) Immunofluorescent characterization of microgliosis (IBA1 immunolabeling) in forebrain PLP cKO mice (overview coronal brain section). Quantifications for IBA1+area for region of interests (ROIs) (CTX: cortex, CC: corpus callosum, STR: striatum) are shown on the right. Bars represent means ± SEM and datapoints represent individual mice (n=4 per group). For statistical analyses two-sided Student’s t-tests were performed. Age of the animals was 22 months. (B) Immunofluorescent characterization of astrogliosis (GFAP immunolabeling) in forebrain PLP cKO mice (overview coronal brain section). Quantifications for GFAP+area for ROIs (CTX: cortex, CC: corpus callosum, STR: striatum) are shown on the right. Bars represent means ± SEM and datapoints represent individual mice (n=4 per group). For statistical analyses two-sided Student’s t-tests were performed. Age of the animals was 22 months. (C) Immunohistochemical characterization of axonal swellings (amyloid precursor protein [APP] immunolabeling) in forebrain PLP cKO mice (overview coronal brain section). Note the apparent fimbria atrophy and ventricular enlargement in forebrain PLP cKO mice. Age of analysis 22 months. (C’) Closeup images of boxed areas in C. Black arrows indicate axonal spheroids in the fimbria and internal capsule of cKO mice, which are essentially absent in Ctrl mice. Levels of significance were defined as p>0.05 (*), p>0.01 (**) and p>0.001 (***).
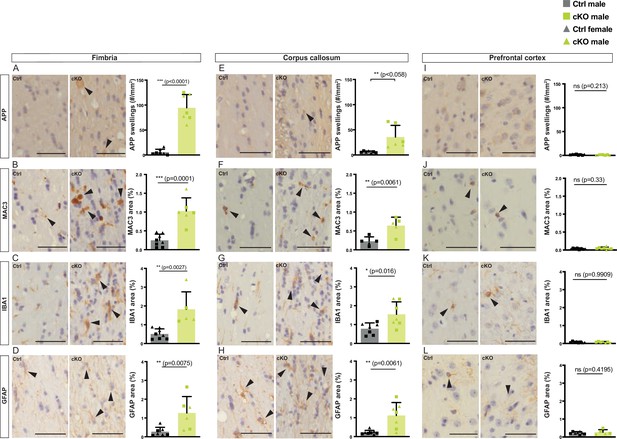
Neuropathological changes in the subcortical white matter in forebrain proteolipid protein (PLP) conditional knockout (cKO) mice.
(A,E,I) Representative light microscopic images of cross-sectioned fimbria, corpus callosum, and prefrontal cortex immunolabeled for amyloid precursor protein (APP) in forebrain PLP cKO and control mice at age 22 months. (B,F,J) Representative light microscopic images of cross-sectioned fimbria, corpus callosum, and prefrontal cortex immunolabeled for MAC3 in forebrain PLP cKO and control mice at age 22 months. (C,G,K) Representative light microscopic images of cross-sectioned fimbria, corpus callosum, and prefrontal cortex immunolabeled for IBA1 in forebrain PLP cKO and control mice at age 22 months. (D,H,L) Representative light microscopic images of cross-sectioned fimbria, corpus callosum, and prefrontal cortex immunolabeled for GFAP in forebrain PLP cKO and control mice. Analyses were performed at 22 months. Genotype-dependent quantifications are shown on the right. Bars represent means ± SEM; datapoints represent individual animals; male and female mice are indicated by squares and triangular datapoints, respectively. Scale bars 50 µm. For statistical analyses two-sided Student’s t-tests were performed for the Ctrl versus cKO comparison. p-Values are given in the bar graphs. All data are shown as means ± SEM. Levels of significance were defined as p>0.05 (*), p>0.01 (**) and p>0.001 (***).
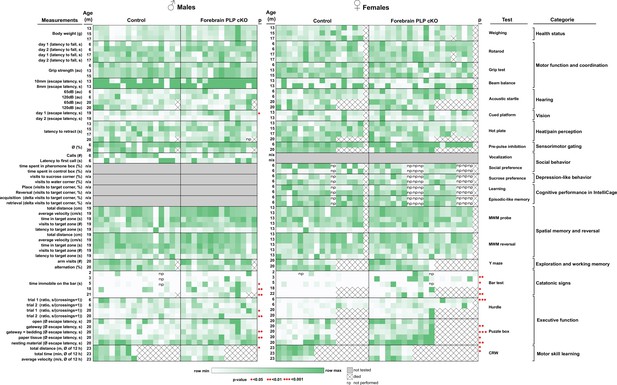
Behavioral deep-phenotyping of male and female forebrain proteolipid protein (PLP) conditional knockout (cKO) mice.
Heatmap representation of behavioral data in the categories of overall health status, motor function and coordination, hearing, vision, heat/pain perception, sensorimotor gating, social behavior, depression-like behavior, cognitive performance in the IntelliCage, spatial memory and reversal in the Morris water maze (MWM), exploration and working memory, catatonic signs and executive function. Each heatmap column represents individual animals (n-numbers). Statistical significance is indicated by red dots. Raw values, n-numbers of mice tested per experiment, mean, SEM, and p-values are specified in Table 1.
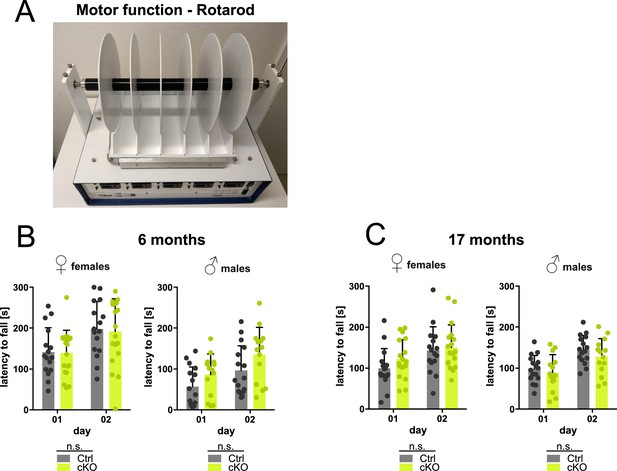
Forebrain proteolipid protein (PLP) conditional knockout (cKO) mice display normal latency to fall from the rotarod.
(A) Representative image of a mouse subjected to the rotarod test assessing motor performance. (B) Groups of female and male PLP cKO (n=19 or 14, respectively) and Ctrl (n=16 per sex) mice were assessed in the rotarod at ages 6 months on 2 consecutive days (day 1 and day 2). (C) Groups of female and male PLP cKO (n=18 or 14, respectively) and Ctrl (n=16 per sex) mice were assessed in the rotarod at ages 17 months on 2 consecutive days (day 1 and day 2). Note that cKO mice performed normally on the rotarod. Bars represent means ± SEM. Datapoints represent individual mice; not significant (n.s.) by repeated measures two-way analysis of variance (ANOVA). Levels of significance were defined as p>0.05 (*), p>0.01 (**) and p>0.001 (***).

Forebrain proteolipid protein (PLP) conditional knockout (cKO) mice display catatonia-like signs along with impaired executive function.
(A) Representative image of a mouse subjected to the bar test. The time a mouse remains immobile upon placement of its forelimbs on a horizontal bar is measured. (B) Bar test of female and male groups of forebrain PLP cKO (n=12-20 or 13-14, respectively) and Ctrl (n=10-17 or 15-16, respectively) mice were tested at the indicated ages. Bars represent means ± SEM; datapoints represent individual mice. Mann-Whitney U-tests wereperformed for statistical analyses. (C) Puzzle box setup with closed and open lid. The time to escape from a well-lit open space into a dark shelter with increasingly difficult configurations (open entry, addition of a gateway, gateway filled with bedding, entry filled with paper tissue, entry filled with nesting material) is taken as a readout. (D) Female and male groups of forebrain PLP cKO (n=12 or 14, respectively) and Ctrl (n=11 or 16, respectively) mice were assessed at age 20 or 18 months, respectively. Bars represent means ± SEM; datapoints represent individual mice. Repeated measures two-way analysis of variance ( ANOVA); significance level for the factor ‘genotype’ is given on the left; Sidak multiple comparison test was conducted as post hoc test and p-values are given in the graph. (E) Modified hurdle test assessing the escape latency of mice by crossing 5 cm hurdles toward the periphery (arrow) upon placement into the center (marked by a star) of a circular open field arena. (F) Female and male groups of forebrain PLP cKO (n=11-12 or 14, respectively) and Ctrl (n=17-19 or 16, respectively) mice were assessed at the age of 6 and 20 months. trial 1 or 2, T1 or T2. Bars represent mean ± SEM; datapoints represent individual mice. Mann-Whitney U-test was performed for statistical analysis. Levels of significance were defined as p>0.05 (*), p>0.01 (**) and p>0.001 (***). The precise number of mice tested per experiment, mean, SEM, and p-values are specified in Table 1.
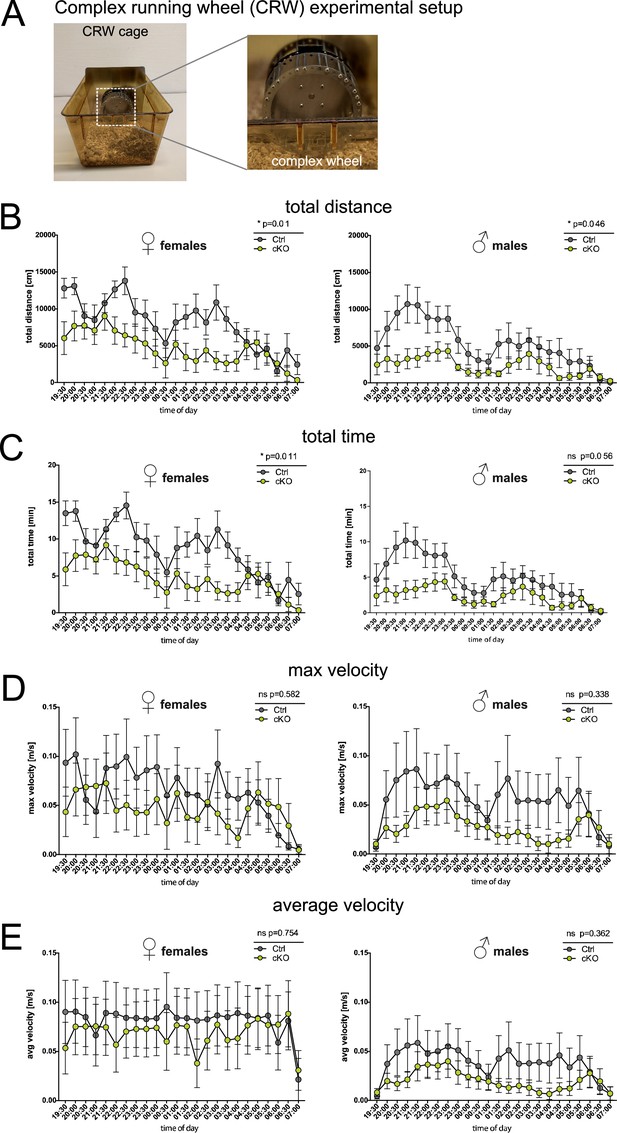
Forebrain proteolipid protein (PLP) conditional knockout (cKO) mice display reduced running distance but normal velocity in the complex running wheel (CRW) paradigm.
(A) Experimental setup to study voluntary running behavior and motor skill learning in the CRW. (B) Total running distance in the CRW of female and male groups of PLP cKO and Ctrl mice at age 23 months. (C) Total running time of female and male groups of PLP cKO and Ctrl mice at age 23 months in the CRW. (D) Maximal velocity reached of female and male groups of PLP cKO and Ctrl mice at age 23 months in the CRW. (E) Average velocity of female and male groups of PLP cKO and Ctrl mice at age 23 months in the CRW. Both female and male cKO mice displayed reduced running distance but normal average running velocity. A trend toward reduced running time reached significance in female but not in male cKO mice. Note that the result is interpreted as reflecting that cKO mice show normal motor capability but impaired executive function. A total of 7-8 mice per sex and genotype were used. Superimposed plot with connecting lines; repeated measures two-way analysis of variance (ANOVA); not significant, n.s.; *, p<0.05.
Tables
Behavioral deep-phenotyping of male and female forebrain proteolipid protein (PLP) conditional knockout (cKO) mice.
Precise n-numbers, mean values, SEM, and p-values for all behavioral experiments are given.
| Males | Females | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Plpflox/Y | Plpflox/Y *EmxCre | Plpflox/flox | Plpflox/flox *EmxCre | |||||||||
| Behavioral paradigms | Age (months) | (N) | Mean ± SEM | (N) | Mean ± SEM | p-Value | Age (months) | (N) | Mean ± SEM | (N) | Mean ± SEM | p-Value |
| Health status | ||||||||||||
| Body weight (g) | 13 | 16 | 36.39±0.77 | 14 | 39.23±1.29 | 0.06 | 13 | 16 | 31.16±1.17 | 19 | 29.64±0.94 | 0.25 |
| 15 | 16 | 37.69±0.68 | 14 | 40.51±1.31 | 0.06 | 15 | 16 | 31.52±1.24 | 19 | 30.50±1.01 | 0.52 | |
| 17 | 16 | 36.86±0.85 | 14 | 39.13±1.56 | 0.2 | 17 | 16 | 32.11±1.38 | 18 | 31.08±1.17 | 0.68 | |
| Motor function and coordination | ||||||||||||
| Rotarod day 1 (latency to fall, s) | 6 | 16 | 56.94±11.97 | 14 | 86.14±13.59 | p=0.12 | 6 | 16 | 141.40±14.97 | 19 | 139.00±12.84 | 0.9 |
| Rotarod day 2 (latency to fall, s) | 6 | 16 | 96.28±15.12 | 14 | 135.40±17.97 | p=0.10 | 6 | 16 | 198.20±16.75 | 19 | 192.80±16.87 | 0.84 |
| Rotarod day 1 (latency to fall, s) | 17 | 16 | 97.69±8.25 | 14 | 88.79±11.71 | p=0.53 | 17 | 16 | 99.38±12.03 | 18 | 122.00±11.14 | 0.18 |
| Rotarod day 2 (latency to fall, s) | 17 | 16 | 145.3±8.56 | 14 | 127.7±11.85 | p=0.23 | 17 | 16 | 143.20±14.43 | 18 | 150.40±12.92 | 0.71 |
| Grip strength (a.u.) | 6 | 16 | 72.42±4.41 | 14 | 74.43±5.00 | 0.76 | 6 | 16 | 71.85±3.55 | 19 | 75.61±3.75 | 0.48 |
| 13 | 16 | 88.69±3.35 | 14 | 91.12±5.22 | 0.69 | 13 | 16 | 93.98±3.77 | 19 | 84.44±3.96 | 0.09 | |
| 15 | 16 | 88.35±3.83 | 14 | 84.64±5.97 | 0.6 | 15 | 16 | 93.50±4.67 | 19 | 89.53±6.38 | 0.63 | |
| Beam balance 10 mm (escape latency, s) | 13 | 16 | 100.00±12.23 | 14 | 113.00±10.08 | 0.36 | 13 | 16 | 100.00±25.86 | 19 | 75.60±14.62 | 0.75 |
| Beam balance 8 mm (escape latency, s) | 13 | 16 | 100.00±8.40 | 14 | 98.25±9.54 | 0.92 | 13 | 16 | 100.00±20.63 | 19 | 90.99±17.93 | 0.86 |
| Hearing | ||||||||||||
| Acoustic startle at 65 dB (a.u.) | 6 | 16 | 0.87±0.09 | 14 | 0.95±0.15 | 0.6 | 6 | 16 | 0.82±014 | 19 | 0.74±0.10 | 0.73 |
| Acoustic startle at 120 dB (a.u.) | 6 | 16 | 2.88±0.49 | 14 | 3.59±0.56 | 0.34 | 6 | 16 | 2.82±0.32 | 19 | 3.53±0.46 | 0.23 |
| Acoustic startle at 65 dB (a.u.) | 18 | 15 | 0.93±0.07 | 13 | 0.85±0.11 | 0.55 | 20 | 11 | 0.87±0.12 | 12 | 1.00±0.12 | 0.42 |
| Acoustic startle at 120 dB (a.u.) | 18 | 15 | 2.25±0.31 | 13 | 1.87±0.30 | 0.39 | 20 | 11 | 2.33±0.31 | 12 | 2.17±0.25 | 0.7 |
| Vision | ||||||||||||
| Cued platform training day 1 (escape latency, s) | 19 | 16 | 25.30±3.35 | 14 | 40.59±6.14 | 0.03 | 13 | 16 | 24.46±3.98 | 19 | 28.23±3.93 | 0.61 |
| Cued platform training day 2 (escape latency, s) | 19 | 16 | 6.13±0.76 | 14 | 10.77±2.92 | 0.07 | 13 | 16 | 11.90±2.98 | 19 | 12.08±2.42 | 0.42 |
| Heat/pain perception | ||||||||||||
| Hot plate (latency to retract, s) | 13 | 16 | 21.40±1.12 | 14 | 22.95±1.46 | 0.69 | 13 | 16 | 17.25±1.00 | 19 | 15.44±0.95 | 0.22 |
| 15 | 16 | 16.96±1.24 | 14 | 15.81±1.32 | 0.42 | 15 | 16 | 15.89±0.83 | 19 | 15.34±0.60 | 0.59 | |
| 17 | 16 | 13.12±1.26 | 14 | 12.13±1.02 | 0.71 | 17 | 16 | 16.04±1.00 | 18 | 15.37±1.04 | 0.65 | |
| 20 | 15 | 13.95±0.96 | 12 | 13.71±0.81 | 0.85 | 20 | 11 | 14.26±0.83 | 12 | 12.59±1.30 | 0.3 | |
| Sensorimotor gating | ||||||||||||
| Pre-pulse inhibition (Ø, %) | 6 | 16 | 43.36±7.85 | 14 | 49.84±5.20 | 0.79 | 6 | 16 | 33.53±6.65 | 19 | 40.76±9.97 | 0.13 |
| 20 | 15 | 16.14±6.88 | 13 | 10.82±5.24 | 0.55 | 20 | 11 | 8.24±3.56 | 12 | 5.62±8.94 | 0.8 | |
| Social behavior | ||||||||||||
| Vocalization (calls, #) | 6 | 16 | 333.3±71.49 | 14 | 231.1±60.22 | 0.29 | n/a | n/a | n/a | n/a | n/a | n/a |
| Vocalization (latency to first call, s) | 6 | 16 | 62.23±18.53 | 14 | 67.14±16.86 | 0.15 | n/a | n/a | n/a | n/a | n/a | n/a |
| Social preference (time spent in pheromone box, %) | n/a | n/a | n/a | n/a | n/a | n/a | 6 | 15 | 64.80±9.63 | 13 | 48.38±6.98 | 0.12 |
| Social preference (time spent in control box, %) | n/a | n/a | n/a | n/a | n/a | n/a | 6 | 15 | 29.80±9.01 | 13 | 36.31±6.02 | 0.21 |
| Depression-like behavior | ||||||||||||
| Sucrose preference (visits to sucrose corner, %) | n/a | n/a | n/a | n/a | n/a | n/a | 6 | 15 | 44.20±2.48 | 13 | 43.45±3.66 | 0.86 |
| Sucrose preference (visits to water corner, %) | n/a | n/a | n/a | n/a | n/a | n/a | 6 | 15 | 18.19±2.34 | 13 | 20.85±2.34 | 0.36 |
| Cognitive performance in IntelliCage | ||||||||||||
| Place learning (visits to target corner, %) | n/a | n/a | n/a | n/a | n/a | n/a | 6 | 15 | 35.78±2.33 | 13 | 34.39±2.05 | 0.66 |
| Reversal learning (visits to target corner, %) | n/a | n/a | n/a | n/a | n/a | n/a | 6 | 15 | 37.82±2.79 | 13 | 38.29±2.61 | 0.91 |
| Episodic-like memory acquisition (delta visits to target corner, %) | n/a | n/a | n/a | n/a | n/a | n/a | 6 | 15 | 3.73±2.79 | 13 | 2.31±2.07 | 0.69 |
| Episodic-like memory retrieval (delta visits to target corner, %) | n/a | n/a | n/a | n/a | n/a | n/a | 6 | 15 | 7.29±4.13 | 13 | 3.04±4.40 | 0.49 |
| Spatial memory and reversal (Morris water maze) | ||||||||||||
| Probe: total distance (cm) | 19 | 16 | 944.5±81.60 | 14 | 1137±57.98 | 0.07 | 13 | 16 | 1258±38.34 | 19 | 1328±31.19 | 0.16 |
| Probe: average velocity (cm/s) | 19 | 16 | 10.50±0.91 | 14 | 12.63±0.64 | 0.07 | 13 | 16 | 13.98±0.43 | 19 | 14.76±0.35 | 0.16 |
| Probe: time in target zone (s) | 19 | 16 | 32.61±3.80 | 14 | 36.39±3.03 | 0.45 | 13 | 16 | 27.03±1.78 | 19 | 26.14±1.38 | 0.69 |
| Probe: visits to target zone (#) | 19 | 16 | 9.44±0.95 | 14 | 11.36±0.58 | 0.11 | 13 | 16 | 14.63±0.84 | 19 | 15.32±0.72 | 0.53 |
| Probe: latency to target zone (s) | 19 | 16 | 8.16±1.71 | 14 | 5.81±1.00 | 0.44 | 13 | 16 | 6.00±1.97 | 19 | 3.74±0.55 | 0.67 |
| Reversal: total distance (cm) | 19 | 16 | 1091±71.27 | 14 | 1236±80.26 | 0.19 | 13 | 16 | 1256±36.02 | 19 | 1442±30.55 | 0.77 |
| Reversal: average velocity (cm/s) | 19 | 16 | 12.12±0.79 | 14 | 13.73±0.89 | 0.19 | 13 | 16 | 13.96±0.40 | 19 | 13.80±0.34 | 0.77 |
| Reversal: time in target zone (s) | 19 | 16 | 28.60±1.81 | 14 | 25.60±2.27 | 0.3 | 13 | 16 | 24.65±1.44 | 19 | 25.39±1.24 | 0.7 |
| Reversal: visits to target zone (#) | 19 | 16 | 8.63±0.62 | 14 | 10.21±0.78 | 0.12 | 13 | 16 | 15.56±0.94 | 19 | 14.42±0.68 | 0.32 |
| Reversal: latency to target zone (s) | 19 | 16 | 7.72±1.41 | 14 | 8.40±1.39 | 0.63 | 13 | 16 | 4.42±0.66 | 19 | 4.39±0.58 | 0.96 |
| Exploration and working memory | ||||||||||||
| Y maze (arm visits, #) | 20 | 15 | 18.33±1.54 | 13 | 19.77±1.85 | 0.55 | 20 | 11 | 16.09±2.04 | 12 | 19.17±2.32 | 0.33 |
| Y maze (alternation, %) | 20 | 15 | 60.34±3.16 | 13 | 56.85±3.41 | 0.46 | 20 | 11 | 66.53±3.69 | 12 | 60.48±4.52 | 0.32 |
| Catatonic signs | ||||||||||||
| Bar test (time immobile on the bar, s) | 2 | 15 | 0.35±0.16 | 14 | 0.51±0.28 | p=0.51 | 2 | 16 | 0.23±0.09 | 19 | 0.45±0.21 | 0.74 |
| 3 | 15 | 0.14±0.03 | 14 | 0.88±0.34 | p=0.19 | 3 | 17 | 0.11±0.01 | 20 | 0.52±0.13 | <0.01 | |
| 5 | 15 | 0.39±0.14 | 14 | 2.63±0.82 | p<0.05 | 5 | 17 | 0.11±0.01 | 19 | 0.27±0.07 | <0.05 | |
| 18 | 16 | 0.54±0.21 | 14 | 1.19±0.29 | p<0.01 | 18 | 10 | 0.10±0.00 | 12 | 0.25±0.09 | <0.05 | |
| 21 | 15 | 0.27±0.11 | 13 | 2.06±0.70 | p<0.01 | 22 | 11 | 0.10±0.00 | 12 | 0.28±0.08 | <0.01 | |
| Executive function and motor skill learning | ||||||||||||
| Hurdle test trial 1 (ratio, s/(crossings +1)) | 6 | 16 | 10.30±1.72 | 14 | 8.34±0.98 | p=0.89 | 6 | 17 | 4.99±0.48 | 19 | 10.80±1.82 | <0.001 |
| Hurdle test trial 2 (ratio, s/(crossings +1)) | 6 | 16 | 1.75±0.17 | 14 | 1.81±0.33 | p=0.52 | 6 | 17 | 1.24±0.17 | 19 | 1.46±0.22 | 0.44 |
| Hurdle test trial 1 (ratio, s/(crossings +1)) | 20 | 16 | 6.76±1.16 | 14 | 11.28±2.04 | p<0.05 | 20 | 11 | 4.88±0.85 | 12 | 9.57±3.14 | 0.08 |
| Hurdle test trial 2 (ratio, s/(crossings +1)) | 20 | 16 | 5.49±1.02 | 14 | 8.07±1.34 | p<0.01 | 20 | 11 | 3.26±0.41 | 12 | 6.99±2.51 | 0.17 |
| Puzzle box (Ø escape latency, s): open | 20 | 16 | 89.48±19.73 | 14 | 118.10±19.82 | p=0.22 | 20 | 11 | 96.23±24.28 | 12 | 140.6±20.26 | 0.17 |
| Puzzle box (Ø escape latency, s): gateway | 20 | 16 | 48.38±8.61 | 14 | 69.95±15.01 | p=0.12 | 20 | 11 | 25.15±6.67 | 12 | 48.18±8.34 | <0.01 |
| Puzzle box (Ø escape latency, s): gateway+bedding | 20 | 16 | 41.96±8.11 | 14 | 71.88±11.72 | p<0.01 | 20 | 11 | 18.21±3.10 | 12 | 53.01±8.19 | <0.0001 |
| Puzzle box (Ø escape latency, s): paper tissue | 20 | 16 | 85.02±10.21 | 14 | 148.30±16.58 | p<0.01 | 20 | 11 | 104.5±32.19 | 12 | 224.3±32.40 | <0.05 |
| Puzzle box (Ø escape latency, s): nesting material | 20 | 16 | 245.50±21.02 | 14 | 259.70±19.55 | p=0.63 | 20 | 11 | 200.6±35.48 | 12 | 322.3±15.65 | <0.01 |
| Complex running wheel (total distance, m, Ø of 12 hr) | 23 | 8 | 1283.14±319.78 | 8 | 550,23±104.3 | p=0.047 | 23 | 7 | 1956.16±146.65 | 8 | 1118.77±253.23 | 0.04 |
| Complex running wheel (total time, min, Ø of 12 hr) | 23 | 8 | 119.0±29.27 | 8 | 54.78±10.03 | p=0.057 | 23 | 7 | 206.5±15.40 | 8 | 113.4±26.17 | 0.019 |
| Complex running wheel (maximal velocity, m/s, Ø of 12 hr) | 23 | 8 | 0.0555±0.0266 | 8 | 0.0272±0.0103 | p=0.574 | 23 | 7 | 0.0639±0.0226 | 8 | 0.0463±0.0214 | 0.582 |
| Complex running wheel (average velocity, m/s, Ø of 12 hr) | 23 | 8 | 0.0803±0.0311 | 8 | 0.0648±0.0248 | p=0.362 | 23 | 7 | 0.0812±0.0291 | 8 | 0.0686±0.0266 | 0.939 |
Additional files
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/70792/elife-70792-transrepform1-v1.pdf
-
Source data 1
Source data on all displayed figures and graphs.
- https://cdn.elifesciences.org/articles/70792/elife-70792-data1-v1.xlsx