Reverting the mode of action of the mitochondrial FOF1-ATPase by Legionella pneumophila preserves its replication niche
Figures
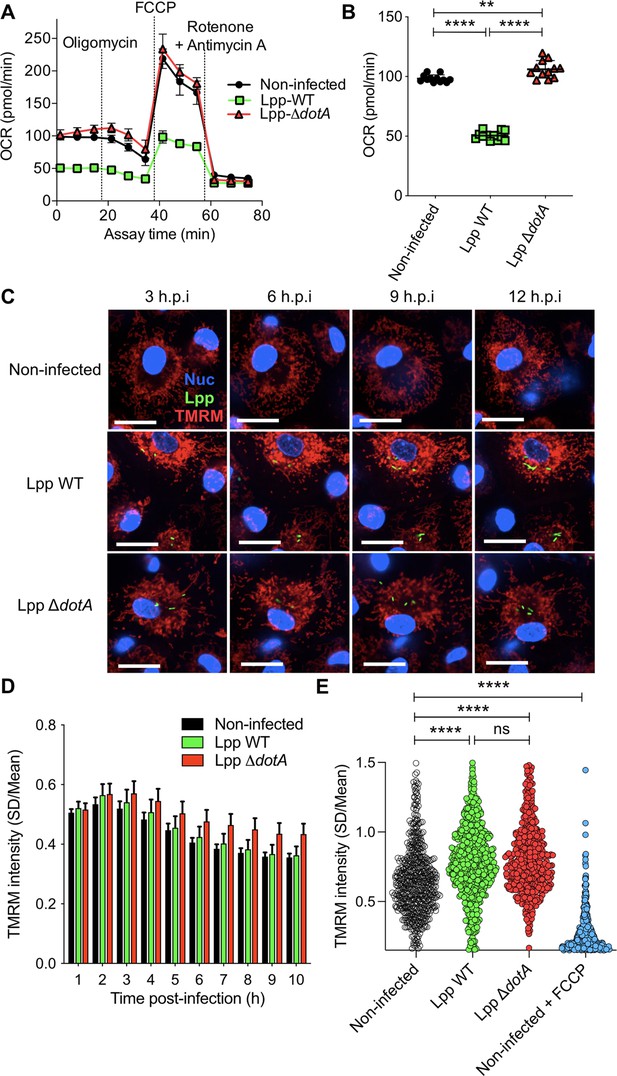
Despite a reduction of oxidative phosphorylation (OXPHOS), human monocyte-derived macrophages (hMDMs) maintain their Δψm during infection by L. pneumophila.
(A) hMDMs were infected with L. pneumophila strain Paris (Lpp) wild-type (WT), a type 4 secretion system (T4SS)-deficient ΔdotA mutant, or left uninfected (noninfected). At 6 hr post-infection (hpi), a cellular respiratory control assay was performed by measuring oxygen consumption rate (OCR) during the sequential addition of mitochondrial respiratory inhibitors (see also Figure 1—figure supplement S1A). (B) Basal respiration of hMDMs in the same conditions as in (A), at 6 hpi. (C) hMDMs were infected as in (A) with GFP-expressing bacteria (green), nuclei of host cells were stained with Hoechst (Nuc, blue), and Δψm was monitored from 1 to 12 hpi using tetramethylrhodamine methyl ester (TMRM) dye in nonquenching conditions (10 nM). Representative confocal microscope images of noninfected and infected cells at 3, 6, 9, and 12 hpi are shown. Intracellular bacterial replication can be observed in Lpp-WT infected hMDMs at 12 hpi. Bar: 20 μm. (D) Quantification of TMRM intensity at 1–10 hpi (expressed as SD/mean) in the assays described in (C). Data from four independent experiments with a total of 10 replicates. Single-cell data from the entire time course are shown in Figure 1—figure supplement S1C. (E) hMDMs were infected with GFP-expressing bacteria or left uninfected (noninfected). At 6 hpi, nuclei of host cells were stained with Hoechst and Δψm was measured using TMRM dye in nonquenching conditions (10 nM), and FCCP (10 μM) was added or not to noninfected cells as a control to monitor complete mitochondrial depolarization. Single-cell analysis from assays performed in three donors is shown in Figure 1—figure supplement S1D. **p<0.01; ****p<0.00001; ns, nonsignificant (Mann–Whitney U test).
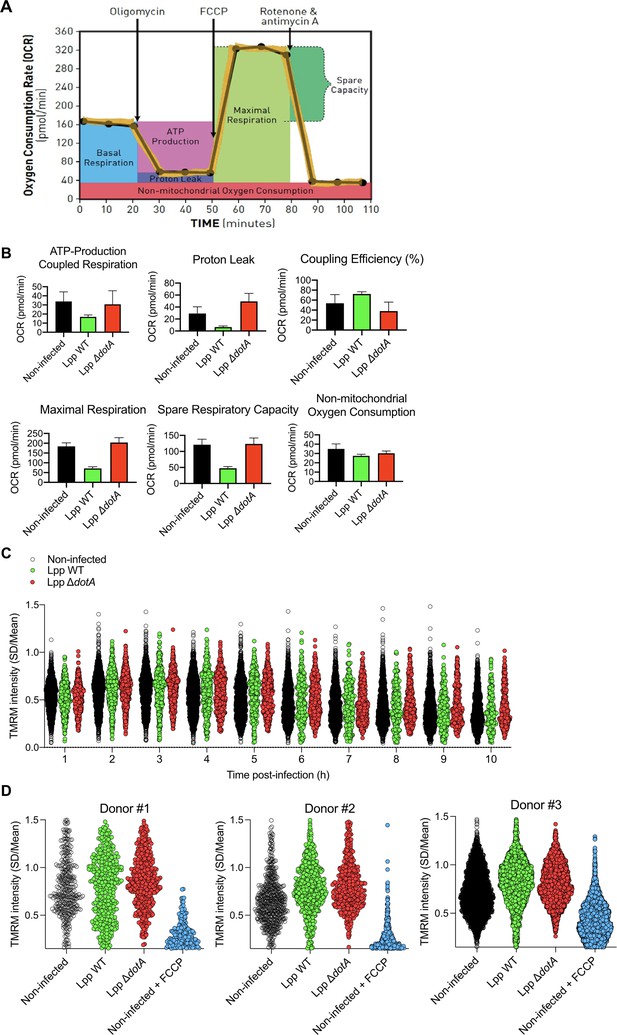
Oxidative phosphorylation (OXPHOS) and Δψm during infection by L. pneumophila.
(A) Bioenergetic profiles of the key parameters of mitochondrial respiration during a mitochondrial respiratory control assay using the Seahorse XF Mitostress kit. Sequential compound injections measure basal respiration, ATP production, proton leak, maximal respiration, spare respiratory capacity, and nonmitochondrial respiration (source: Seahorse Bioscience). (B) Human monocyte-derived macrophages (hMDMs) were infected with L. pneumophila strain Paris (Lpp) wild-type (WT), a type 4 secretion system (T4SS)-deficient ΔdotA mutant, or left uninfected (noninfected). At 6 hr post-infection (hpi), a cellular respiratory control assay (Seahorse) was performed by measuring oxygen consumption rate (OCR) during the sequential addition of mitochondrial respiratory inhibitors. Quantification of key parameters of mitochondrial functioning, derived from this assay (Figure 1A), is shown. (C) hMDMs were infected as in (B) with GFP-expressing bacteria, nuclei of host cells were stained with Hoechst, and Δψm was monitored using tetramethylrhodamine methyl ester (TMRM) dye in nonquenching conditions (10 nM). Single-cell analysis of TMRM intensity at 1–10 hpi (expressed as SD/mean) is shown. (D) hMDMs were infected with GFP-expressing bacteria or left uninfected (noninfected). At 6 hpi, nuclei of host cells were stained with Hoechst and the Δψm was measured using TMRM dye in nonquenching conditions (10 nM), and FCCP (10 μM) was added or not to noninfected cells as a control to monitor complete mitochondrial depolarization. Single-cell analysis is shown from assays performed from hMDMs purified from three different donors.
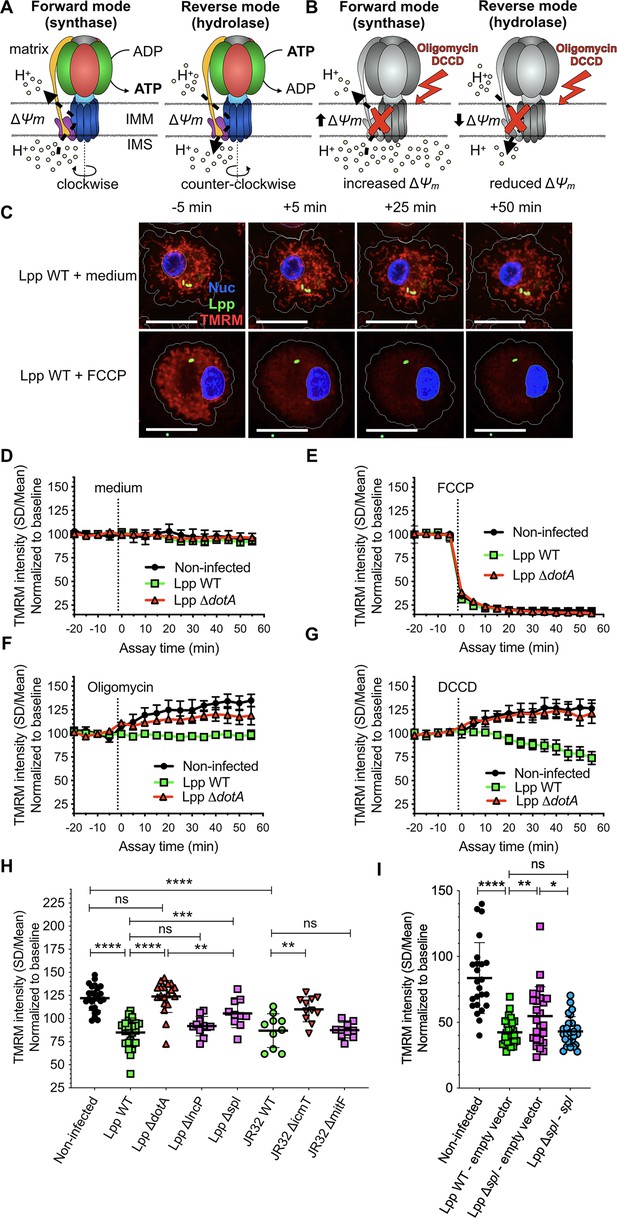
The mitochondrial FOF1-ATPase works in the ‘reverse mode’ during infection of human monocyte-derived macrophages (hMDMs) by L. pneumophila.
(A) In the ‘forward mode’ of the mitochondrial ATPase, the Δψm generated by the electron transport chain is used by the FOF1-ATPase to synthesize ATP. The ‘reverse mode’ of the FOF1-ATPase leads to ATP hydrolysis to pump H+ to the intermembrane space (IMS). IMM, inner mitochondrial membrane. (B) When the FOF1-ATPase is inhibited by oligomycin or dicyclohexylcarbodiimide (DCCD), an increase in Δψm indicates that the ATPase was working in the ‘forward mode’ (H+ accumulate in the IMS), while a decrease in Δψm indicates functioning in the ‘reverse mode’ (H+ cannot be translocated to the IMS by the FOF1-ATPase to sustain the Δψm). (C) hMDMs were infected with GFP-expressing bacteria (green) or left uninfected (noninfected). At 5.5 hr post-infection (hpi), cells were labeled with Hoechst to identify the cell nucleus (Nuc, blue) and tetramethylrhodamine methyl ester (TMRM) (red) to quantify Δψm. At 6 hpi, addition of medium (no changes) or FCCP (complete depolarization) was used as controls. Representative confocal images of Lpp-WT-infected hMDMs (6 hpi) at 5 min before the addition of medium (top) or FCCP (bottom), and at 5, 25, and 50 min after addition of medium or FCCP. Bar: 20 μm. (D) Quantification of (C) before (baseline) and after the addition of medium. Each dot represents mean ± SD of three independent experiments with a total of eight replicates. (E) Same as (D) but FCCP was added. (F) Same as (D) but oligomycin was added. (G) Same as (D) but DCCD was added. (H) Same as (C) but infection was performed with Lpp-WT, Lpp-ΔdotA, Lpp-ΔlncP, Lpp-Δspl, L. pneumophila strain Philadelphia JR32 (JR32)-WT, JR32-ΔicmT or JR32-ΔmitF. TMRM values (SD/mean) at 50 min after DCCD addition are shown. Data from a minimum of three experiments per strain with 10 or more replicates per strain. (I) hMDMs were infected with GFP-expressing bacteria or left uninfected (noninfected). At 5.5 hpi, cells were labeled with Hoechst to identify the cell nucleus and TMRM to quantify Δψm. At 6 hpi, addition of DCCD revealed whether the FOF1-ATPase works in the ‘reverse mode.’ Infection was performed with Lpp-WT expressing empty pBCKS vector (empty vector), Lpp-Δspl-expressing empty vector and Lpp-Δspl-expressing pBCKS-spl vector, which express LpSpl (complemented strain, Lpp-Δspl:: spl). Data from three donors are shown. Each dot represents a replicate. *p<0.1; **p<0.01; ***p<0.001; ****p<0.00001; ns, nonsignificant (Mann–Whitney U test).
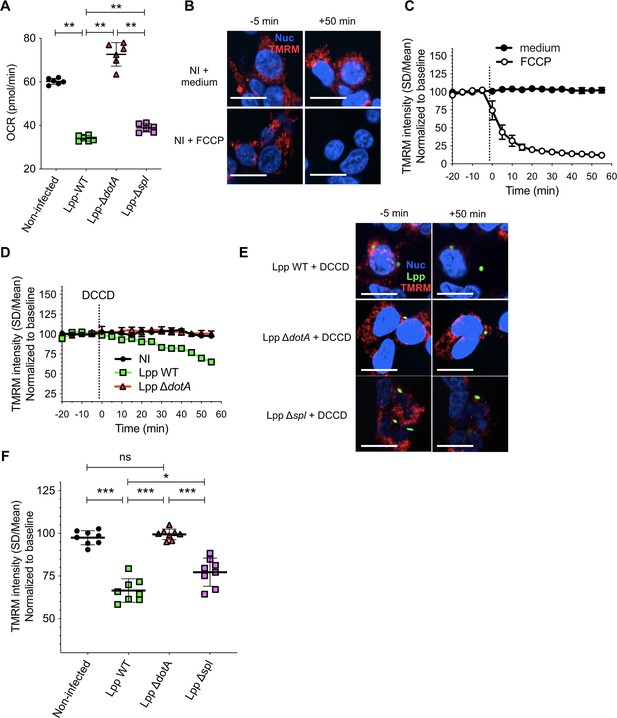
Basal respiration of human monocyte-derived macrophages (hMDMs) infected with L. pneumophila Δspl mutant and validation of the model in HEK-293 cells.
(A) hMDMs were infected with L. pneumophila strain Paris WT (Lpp-WT), a type 4 secretion system (T4SS)-deficient ΔdotA mutant, a LpSpl-deficient Δspl mutant, or left uninfected (noninfected). At 6 hr post-infection (hpi), basal oxygen consumption rate (OCR) was measured using the Seahorse technology. Each dot represents the value of one replicate from a representative experiment. (B) HEK-293 cells stably expressing the FcγRII receptor to efficiently internalize IgG-opsonized L. pneumophila were labeled with Hoechst to identify the cell nucleus (Nuc, blue) and tetramethylrhodamine methyl ester (TMRM) (red) to quantify Δψm. Addition of medium (no changes) or FCCP (complete depolarization) to noninfected (NI) HEK-293 cells was used as controls. Representative confocal images at 5 min before the addition of medium (top) or FCCP (bottom), and at 50 min after addition of medium or FCCP. Bar: 20 μm. (C) Quantification of (B) before (baseline) and after the addition of medium/FCCP (dotted line). Each dot represents mean ± SD of three independent experiments. (D) HEK-293 cells were infected with GFP-expressing bacteria (WT, ΔdotA mutant) opsonized using an anti-flagellin antibody, or left uninfected (noninfected). At 6 hpi, dicyclohexylcarbodiimide (DCCD) was added to the cells and changes Δψm were monitored. (E) Representative confocal images at 5 min before the addition of DCCD and at 50 min after addition of DCCD. Green: GFP-expressing bacteria. Bar: 20 μm. (F) Same as (D) but infection was performed with opsonized Lpp-WT, Lpp-ΔdotA, or Lpp-Δspl strains. TMRM values (SD/mean) at 50 min after DCCD addition are shown. Data from three experiments per strain with six or more replicates per strain. *p<0.1; **p<0.01; ***p<0.001; ns, nonsignificant (Mann–Whitney U test).
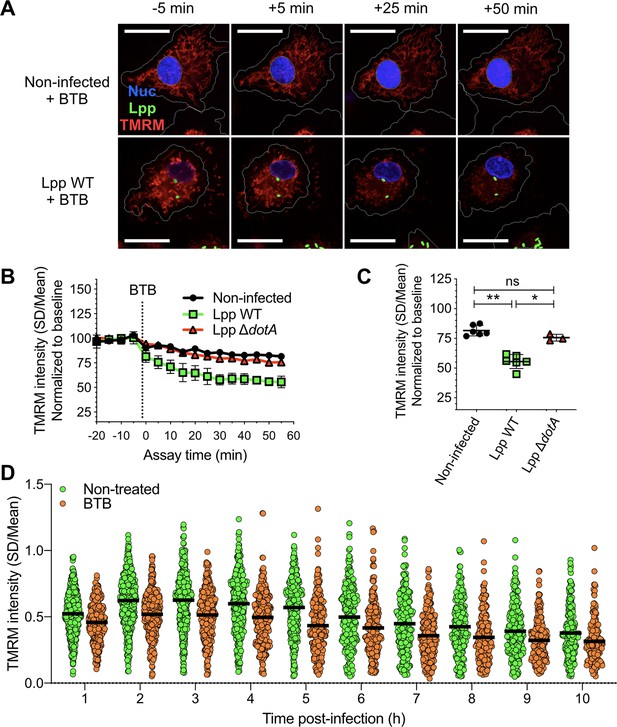
Inhibition of the ‘reverse mode’ of mitochondrial FOF1 ATPase reduces the Δψm of L. pneumophila-infected human monocyte-derived macrophages (hMDMs).
(A) hMDMs were infected with GFP-expressing bacteria (green), Lpp-WT or Lpp-ΔdotA, or left uninfected (noninfected). At 5.5 hr post-infection (hpi), cells were labeled with Hoechst to identify cell nucleus (Nuc, blue) and tetramethylrhodamine methyl ester (TMRM) (red) to quantify Δψm. At 6 hpi, BTB06584 (BTB, 50 μM), a specific inhibitor of the ‘reverse mode’ of the ATPase, was added and Δψm monitored. Representative confocal microscopy images of noninfected (top) and Lpp-WT-infected (bottom) hMDMs (6 hpi) at 5 min before the addition and at 5, 25, and 50 min after the addition of BTB. Bar: 20 μm. (B) Quantification of (C) before (baseline) and after the addition of BTB. Each dot represents the mean ± SD of three independent experiments with a total of six replicates. (C) Same infection conditions than (A) but TMRM values (SD/mean) at 50 min after BTB addition are shown. Data from three experiments with a total of six replicates (three replicates for Lpp-ΔdotA). (D) Single-cell analysis of Δψm in Lpp-WT-infected hMDMs treated with BTB (50 μM) or left untreated (nontreated). Single-cell data from one representative experiment *p<0.05; **p<0.01; ns, nonsignificant (Mann–Whitney U test).
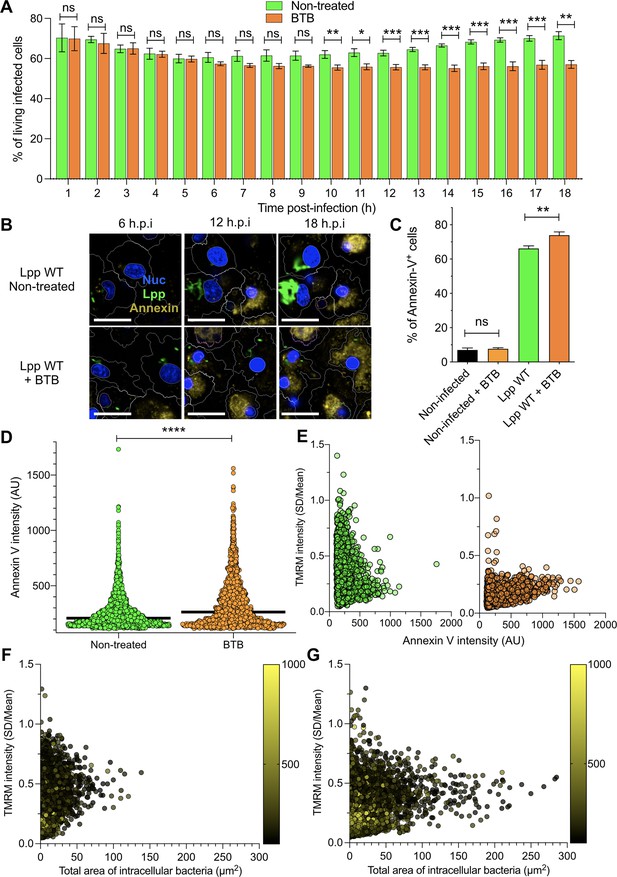
Inhibition of FO-F1 ATPase ‘reverse mode’ increases cell death in L. pneumophila-infected human monocyte-derived macrophages (hMDMs).
(A) hMDMs were infected with Lpp-WT-GFP and were nontreated or treated with 50 μM BTB. The presence of GFP-expressing bacteria in each cell was monitored, and the number of living infected cells in the whole population was graphed as a percentage of living infected cells. Data from three independent experiments with a total of seven replicates per condition and time point. (B) hMDMs were infected with Lpp-WT-GFP (green), the nuclei of host cells were stained with Hoechst (Nuc, blue), and Annexin-V Alexa Fluor 647 was added to the cell culture to monitor early cell death (Annexin, yellow) from 1 to 18 hr post-infection (hpi) in nontreated or BTB-treated hMDMs. Representative confocal images of nontreated and Lpp-WT-GFP-infected cells at 6, 12, and 18 hpi are shown. Intracellular bacterial replication can be observed in nontreated Lpp-WT-infected hMDMs at 12 and 18 hpi. Bar: 20 μm. (C) hMDMs stained as in (B) were infected with Lpp-WT-GFP or left uninfected (noninfected), and then were treated or not with BTB (50 μM). Percentage of Annexin-V+ cells at 24 hpi is shown. Data from three independent experiments with a total of seven replicates per condition (D) Single-cell analysis (12 hpi) of Annexin-V intensity of the assays described in (B). Single-cell data from one representative experiment (18 hpi shown in Figure 2—figure supplement 1A). (E) hMDMs were infected with Lpp-WT-GFP, nuclei of host cells were stained with Hoechst, and tetramethylrhodamine methyl ester (TMRM) and Annexin-V Alexa Fluor 647 were added to the cells to simultaneously monitor (1–18 hpi) Δψm and early cell death, respectively, in nontreated or BTB-treated hMDMs (representative multifield confocal images in Figure 2—figure supplement 1C). Single-cell analyses (12 hpi) of Δψm (TMRM SD/mean) and cell death (Annexin-V intensity) in more than 1600 cells per condition are shown. Single-cell data from one representative experiment. Green dots, nontreated Lpp-WT-infected single cells; orange dots, BTB-treated Lpp-WT-infected single cells. (F) Same infection conditions as in (E) but using nontreated cells. Bacterial replication was monitored in each single cell infected with Lpp-WT. Single-cell analyses (12 hpi) of Δψm (TMRM SD/mean), area of intracellular GFP-expressing bacteria (μm2), a proxy for intracellular replication, and cell death (Annexin-V intensity) in more than 3800 cells are shown. Single-cell data from one representative experiment; color scale (yellow) represents Annexin-V intensity per cell (AU). (G) Same as in (F) at 18 hpi. *p<0.05; **p<0.01; ***p<0.001; ****p<0.00001; ns, nonsignificant (Mann–Whitney U test).
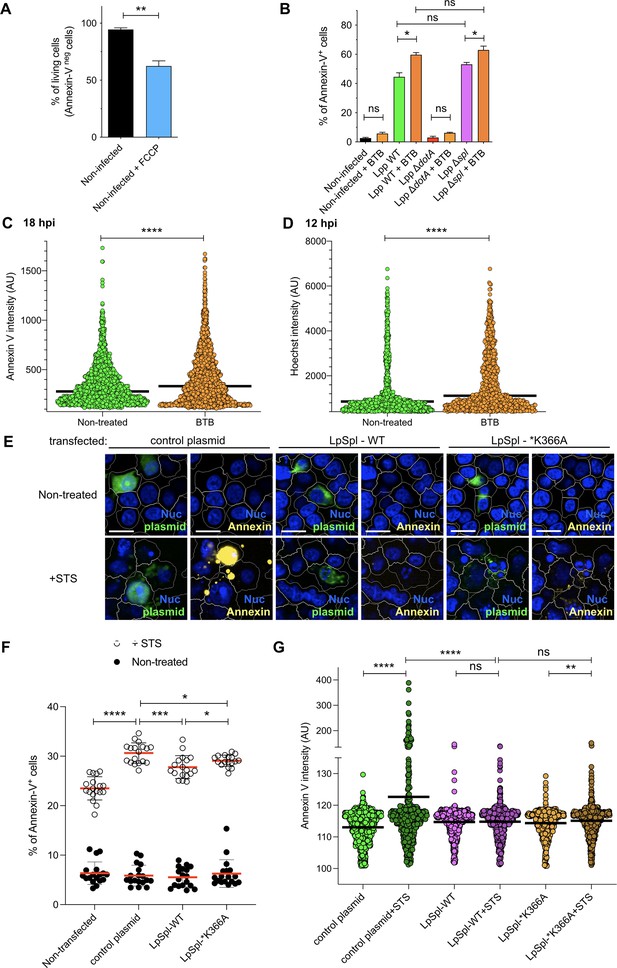
Inhibition of FO-F1 ATPase ‘reverse mode’ delays cell death in L. pneumophila-infected human monocyte-derived macrophages (hMDMs), while transfection of LpSpl into HEK-293 cells protected transfected cells from Staurosporine (STS)-induced cell death.
(A) Noninfected hMDMs were stained with Hoechst to identify their nuclei and challenged with FCCP (10 μM) for 18 hr, while Annexin-V Alexa Fluor 647 was added to the cell culture to monitor cell death. Percentage of living cells (Annexin-V negative) is shown. These values served as reference to interpret the results shown in Figure 4A. (B) hMDMs were infected with GFP-expressing bacteria or left uninfected (noninfected), and then were treated or not with BTB (50 μM). At 24 hr post-infection (hpi), the nuclei of host cells were stained with Hoechst and Annexin-V Alexa Fluor 647 was added to the cell culture to monitor cell death. Percentage of Annexin-V+ cells at 24 hpi is shown. *p<0.1; **p<0.01. ****p<0.0001; ns, nonsignificant (Mann–Whitney U test). (C) hMDMs were infected with Lpp-WT-GFP, the nuclei of host cells were stained with Hoechst, and Annexin-V-647 was added to the cell culture to monitor early cell death from 1 to 18 hpi in nontreated or BTB-treated hMDMs. Single-cell analysis of Annexin-V intensity at 18 hpi is shown. (D) hMDMs were infected as in (A), and Hoechst intensity in the nucleus was analyzed in single cells at 12 hpi. (E) HEK-293 cells stably expressing the FcγRII receptor were transfected with a control plasmid (pGFPmax, Lonza), with a plasmid expressing LpSpl WT (harboring an Xpress tag) or with a plasmid expressing a catalytically inactive LpSpl protein (*K366A, also harboring an Xpress tag). At 24 hr post-transfection, cells were labeled with Hoechst and Annexin-V-647 to monitor cell death, and 1 μM STS was added. At 6 hr, cells were fixed, permeabilized, blocked, and stained with primary mouse antibodies against Xpress tag and secondary anti-mouse Alexa Fluor 488 antibodies. Representative confocal images of transfected cells. Blue, nucleus (Hoechst, Nuc); yellow, Annexin-V-647; green, GFP or anti-Xpress (i.e., LpSpl). Bar: 20 μm. (F) Percentage of Annexin-V-positive cells in each condition, upon addition or not of STS during 6 hr. (G) Single-cell data of Annexin-V mean fluorescence intensity. More than 400 transfected cells were analyzed in each condition. *p<0.1; **p<0.01; ***p<0.001; ****p<0.0001; ns, nonsignificant (Mann–Whitney U test).
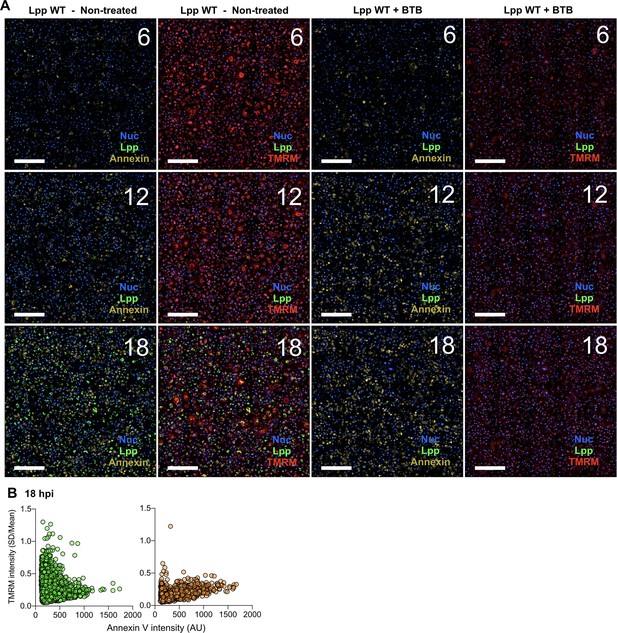
BTB treatment, Δψm, and cell death of L. pneumophila-infected human monocyte-derived macrophages (hMDMs).
(A) hMDMs were infected with Lpp-WT-GFP (green), nuclei of host cells were stained with Hoechst (Nuc, blue), and tetramethylrhodamine methyl ester (TMRM) (red) and Annexin-V Alexa Fluor 647 (yellow) were added to the cells to simultaneously monitor Δψm and early cell death, respectively, in nontreated or BTB-treated hMDMs, respectively. Confocal images of living infected cells were automatically acquired in 16 fields per well (four wells per condition) using ×60 magnification. Representative images of 16 stitched fields per condition are shown at 6, 12, and 18 hr post-infection (hpi). Bar: 200 μm. (B) hMDMs were infected with Lpp-WT-GFP, nuclei of host cells were stained with Hoechst, and TMRM and Annexin-V Alexa Fluor 647 were added to the cells to simultaneously monitor (1–18 hpi) Δψm and early cell death, respectively, in nontreated or BTB-treated hMDMs, respectively. Single-cell analyses (18 hpi) of Δψm (TMRM SD/mean) and cell death (Annexin-V intensity) in more than 1600 cells per condition are shown. Single-cell data from one representative experiment, Green dots, nontreated Lpp-WT-infected single cells; orange dots, BTB-treated Lpp-WT-infected single cells.





