Dynamic spreading of chromatin-mediated gene silencing and reactivation between neighboring genes in single cells
Figures
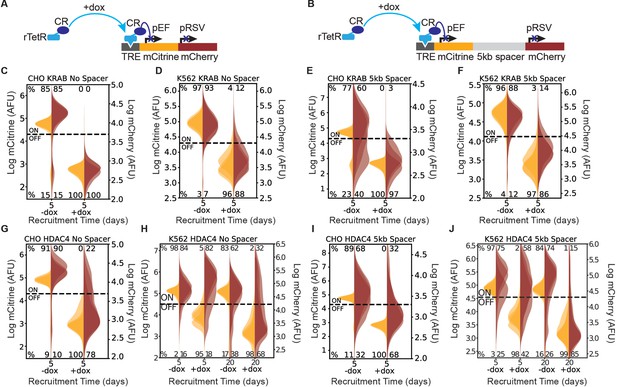
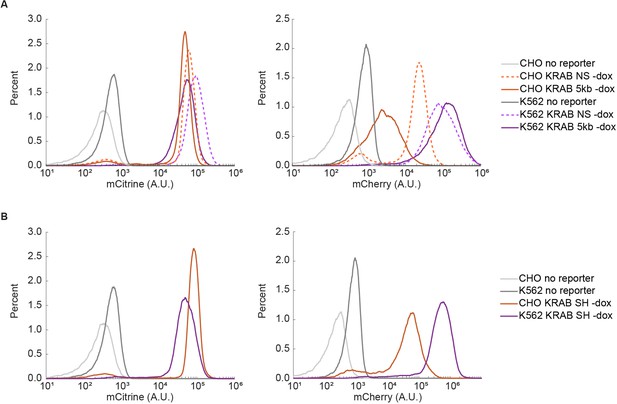
Recruitment of chromatin regulators to synthetic dual-fluorescent reporters results in transcriptional silencing of both genes.
(A, B) Recruitment of a chromatin regulator (dark blue oval) via addition of dox allows for the binding of the rTetR-CR fusion to TRE (Tet Responsive Element, dark grey), upstream of a dual-gene reporter expressing mCitrine (yellow) and mCherry (red) separated by either (A) no spacer or (B) 5 kb of lambda DNA (gray). (C–J) Fluorescence distributions of mCitrine and mCherry measured by flow cytometry either without CR recruitment (-dox), or after 5 or 20 days of recruitment ( + dox) of either KRAB or HDAC4 at the NS or 5 kb reporters in either CHO-K1 or K562 as indicated in each title. Percentages of cells ON (high-fluorescence, top) or OFF (low-fluorescence, bottom) are calculated based on a threshold (dotted line). Data from independent clonal cell lines for CHO-K1 or biological replicates of multiclonal populations for K562 are shown as overlaid semi-transparent distributions (n = 3). A Welch’s unequal variances T-test comparing the percent of cells off in -dox versus + dox for each gene, showed that the percent of cells silenced were statistically significant (p < 0.05) and reproducible for NS and 5 kb reporters in all cell types tested (Appendix 1—table 1).
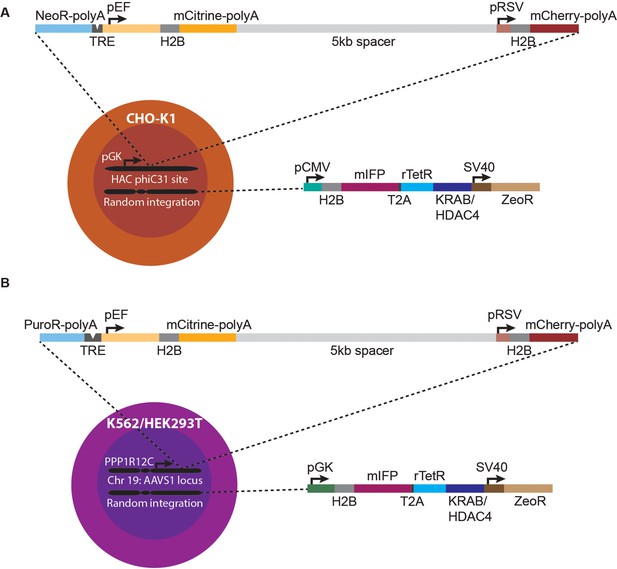
Reporter constructs used in different cell lines for analyzing spreading of transcriptional changes.
(A) Dual mCitrine-mCherry reporter (top) is integrated in CHO-K1 cells in the MI-HAC (Yamaguchi et al., 2011) at the phiC31 integrase site. Upon integration, the neomycin resistance gene (NeoR) starts being expressed from the pGK promoter in the MI-HAC. (B) Dual mCitrine-mCherry reporter is integrated in K562 or HEK293T cells by TALENS at the AAVS1 locus. Upon integration, the puromycin resistance gene (PuroR) is driven by the promoter for the PPP1R12C gene (Hockemeyer et al., 2011). Both mCitrine and mCherry genes have SV40 polyA tails which should aid in transcriptional termination of each gene. NeoR in CHO-K1and PuroR in K562/HEK293T both have a BGH polyA.The TRE in CHO-K1 has 5 TetO binding sites, while the TRE in K562/HEK293T has 9 TetO binding sites. In both cell types, each fluorescent protein is fused to H2B which stabilizes the fluorophore and localizes it to the nucleus, allowing better tracking of single cells in time-lapse microscopy. Chromatin regulators fusions of rTetR to either KRAB or HDAC4 are expressed via random integration by piggyBac transposase, the approximate expression of which can also be monitored by fluorescence of the H2B-mIFP fusion. The size of each element is drawn approximately to scale.
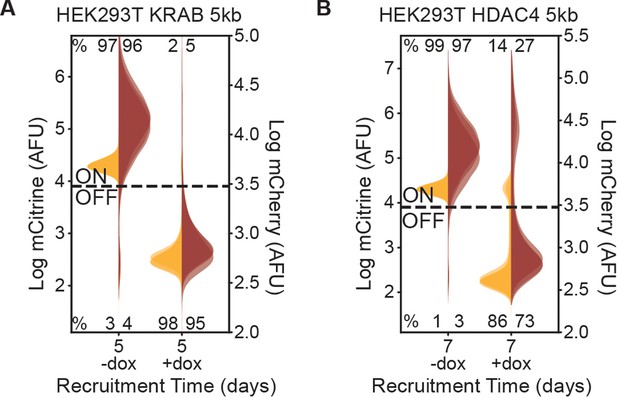
Recruitment of KRAB and HDAC4 to 5 kb reporter in HEK293T.
(A–B) Fluorescence distributions of mCitrine and mCherry measured by flow cytometry either without CR recruitment (-dox) or after 5 or 7 days of recruitment (+ dox) of either (A) KRAB or (B) HDAC4 at the 5 kb reporters in HEK293T. Biological replicates of multiclonal populations are shown as overlaid semi-transparent distributions (n = 3 ).
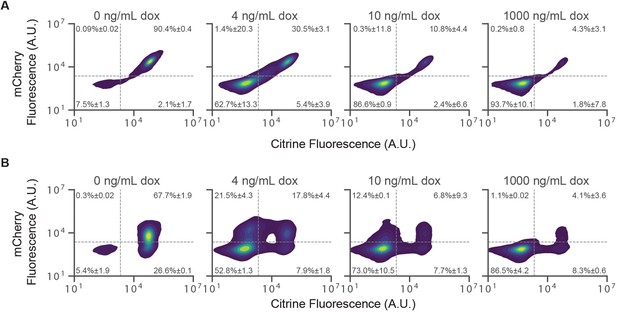
Silencing by KRAB at lower dox concentrations.
(A–B) 2D scatter plots of mCitrine and mCherry fluorescence measured by flow cytometry in CHO-K1 cells with KRAB recruitment for 5 days at 4 ng/mL , 10 ng/mL, and 1000 ng/mL in the (A) NS reporter or (B) 5 kb reporter. Percent of cells in each quadrant is shown with standard error of mean (n = 2 ).
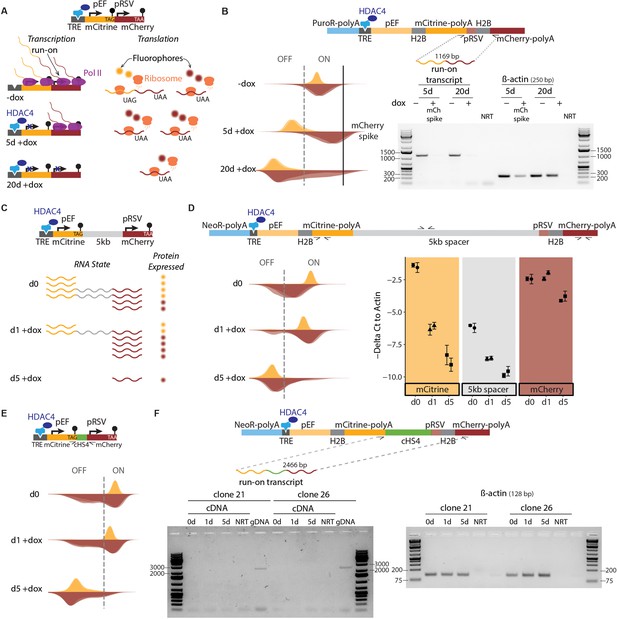
Transcriptional run-on from pEF over pRSV.
(A) Schematic representation of transcription (left) and translation (right) of NS reporter (top) in K562 during HDAC4 recruitment. Transcriptional run-on from pEF into pRSV occurs despite the presence of polyA (-dox row, left). Note that mCitrine and mCherry genes both have stop codons at their 3’ end (black stop signs) and neither gene has an IRES at the 5’ end, such that mCherry can only be translated from transcripts initiated at pRSV (-dox row, right). When pEF is silenced by HDAC4 for 5 days (5d + dox row , left), run-on no longer occurs and transcription initiation at the pRSV increases, resulting in more mCherry protein (5d + dox row , right). After 20 days of HDAC4 recruitment (20d + dox row), both genes are silenced. (B) (top) Schematic of the NS spreading reporter , showing primers for amplifying run-on transcript. (Left) Fluorescence distribution of mCitrine and mCherry across timepoints of HDAC4 recruitment at the NS reporter in K562, matching schematics in (A) for each row. Flow cytometry data from three replicates of recruitment are shown as overlaid semi-transparent distributions. Vertical dashed line shows the threshold for ON/OFF populations. After 5 days of recruitment (5d + dox) cells were sorted for mCherry expression above those in the -dox sample (mCherry spike, to the right of black line). (Right) Agarose gel showing PCR products that use as template cDNA reverse transcribed from mRNA extracted from either: cells with no recruitment (-dox), cells sorted on high mCherry (mCherry spike on the left), or cells with 20 days of recruitment (20d + dox). PCR primers amplify either a region spanning across the mCitrine and mCherry genes (labeled 1169 bp run-on transcript at the top) or beta-actin as a control for cDNA input. (C) ) Schematic representation of mRNA and fluorescent proteins levels of 5 kb reporter in CHO-K1 during HDAC4 recruitment. Transcription termination fails in mCitrine polyA causing Pol II to run-on into 5 kb lambda spacer and mCherry gene without dox (d0), reducing initiation of transcription of mCherry until pEF is silenced at day 1 of dox (d1), allowing pRSV to increase expression of mCherry before being silenced by the chromatin regulator at day 5 of dox (d5). (D) (Top) Schematic of the 5 kb spreading reporter, showing the positions of the primers used for qPCR. (Left) Fluorescence distributions of reporter genes across timepoints of HDAC4 recruitment to 5 kb reporter in CHO-K1. Flow cytometry data from independent clonal cell lines are shown as overlaid semi-transparent distributions. Vertical dashed lines show the threshold for ON/OFF populations. (Right) qPCR was performed on cDNA reverse transcribed from mRNA on regions of mCitrine, 5 kb lambda spacer, and mCherry after 0, 1, and 5 days of HDAC4 recruitment. Delta Cts with respect to Beta-actin are shown for two clones used as biological replicates. Error bars are one standard deviation of three technical replicates within each clone. Beta-actin was used as a control for normalization. (E) (Top) Schematic of HDAC4 recruitment at a construct where the reporter genes are separated by a full cHS4 insulator (SH). (Bottom) Fluorescence distributions of reporter genes across timepoints of HDAC4 recruitment to the SH construct in CHO-K1. Flow cytometry data from three replicates of recruitment are shown as overlaid semi-transparent distributions. Vertical dashed line shows the threshold for ON/OFF populations. (F) (Top) Schematic of the SH spreading reporter, showing PCR primers that detect the mRNA produced if run-on occurs. (Bottom, left) Agarose gel showing PCR products for a region spanning across the mCitrine and mCherry genes including the cHS4 insulator was performed on cDNA from CHO-K1 HDAC4 SH cells in two clones at 0, 1, and 5 days of HDAC4 recruitment. Genomic DNA (gDNA) was used as a control for primer validation. (Bottom, right) Beta-actin was amplified as a control for cDNA input.
-
Figure 1—figure supplement 4—source data 1
Original gel images from RT-PCR.
- https://cdn.elifesciences.org/articles/75115/elife-75115-fig1-figsupp4-data1-v1.zip
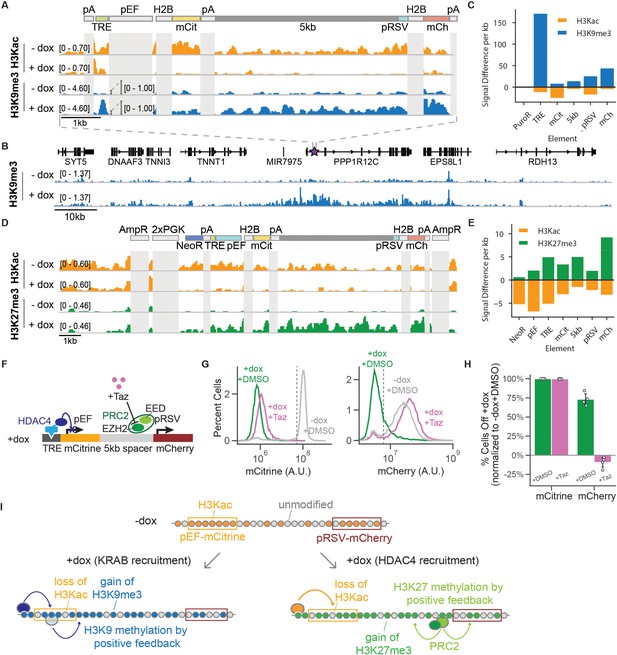
CUT& RUN data shows changes in histone modifications at silenced genes.
(A) Genome browser tracks showing counts per million (CPM)-normalized reads after CUT& RUN for histone 3 acetyl-lysine (H3Kac) and histone 3 lysine 9 trimethylation (H3K9me3) with (+ dox) and without (-dox) recruitment of rTetR-KRAB to the 5kb-spacer reporter in K562 cells. Non-unique regions resulting in ambiguous alignment, including pEF, H2B, and polyA tails (pA), are masked in light gray. (B) Genome browser tracks of H3K9me3 with (+ dox) and without (-dox) recruitment of rTetR-KRAB, looking at the surrounding locus where the 5 kb reporter is integrated in cells (purple star within first intron of PPP1R12C). This snapshot does not include an in situ representation of the reporter, which instead was appended to the end of the reference genome to preserve gene annotations. (C) Quantification of signal difference for H3K9me3 and (H3Kac) with (+ dox) and without (-dox) recruitment of KRAB for 5 days to the 5 kb reporter in K562 cells. (D) Genome browser tracks showing CPM-normalized reads after CUT& RUN for H3Kac and histone 3 lysine 27 trimethylation (H3K27me3) with (+ dox) and without (-dox) recruitment of rTetR-HDAC4 to the 5kb-spacer reporter in CHO-K1 cells. Non-unique regions resulting in ambiguous alignment, including AmpR, PGK, H2B, and polyA tails (pA), are masked in light gray. (E) Quantification of signal difference for H3Kac and H3K27me3 with (+ dox) and without (-dox) recruitment of HDAC4 for 5 days to the 5 kb reporter in K562 cells. (F) Addition of Tazemetostat (Taz) inhibits the EZH2 methyltransferase from PRC2. (G) Fluorescence distributions of mCitrine (left) and mCherry (right) measured by flow cytometry either without CR recruitment (-dox, gray) or after 7 days of HDAC4 recruitment (+ dox + DMSO, green) and with Taz(+ dox + Taz, pink) at the 5 kb reporter in CHO-K1. (H) Percentages of cells OFF normalized by the -dox + DMSO condition, based on threshold shown in panel G (dotted line)(n = 4). (I) In the absence of CR recruitment, both genes have H3Ac across the reporter (top). Upon recruitment of KRAB (bottom left), we see a loss of H3Kac and gain of H3K9me3 across the dual-gene reporter through both DNA looping from rTetR-KRAB as well as positive feedback loops for spread of methylation, resulting in a distance-dependent delay of transcriptional silencing between two genes. Upon recruitment of HDAC4 (bottom right), we see a loss of H3Kac as well as a gain of H3K27me3 across the reporter through positive feedback loops for spread of methylation by PRC2, resulting in distance-independent delay of transcriptional silencing between two genes.
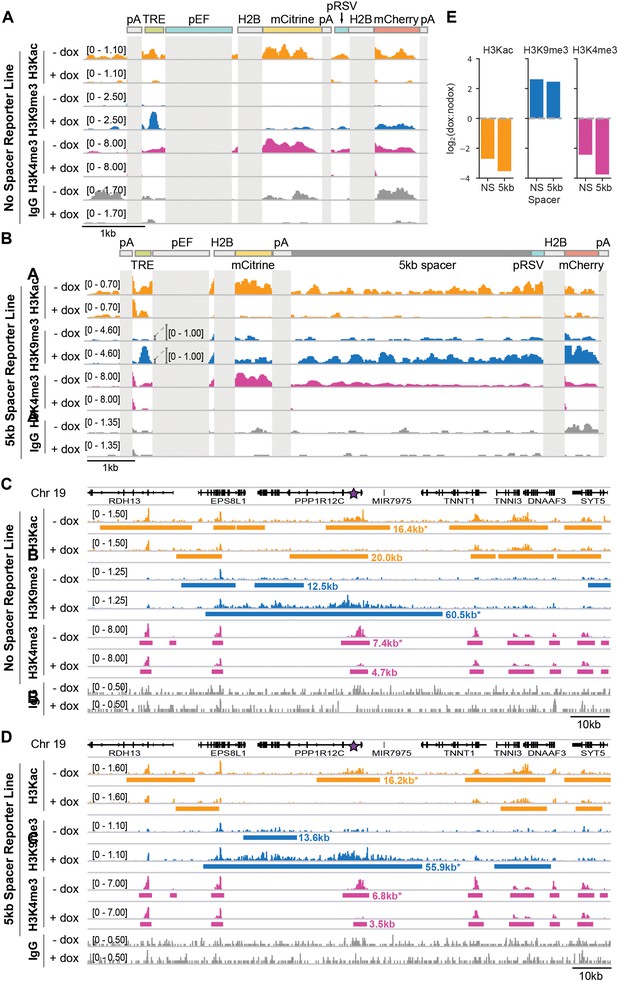
Changes in chromatin modifications at the two-gene reporter and surrounding AAVS1 locus after recruitment of KRAB for 5 days in K562 cells.
(A-B) Genome browser tracks of histone 3 acetyl-lysine (H3Kac), histone 3 lysine 9 trimethylation (H3K9me3), histone 3 lysine 4 trimethylation (H3K4me3), and IgG, as measured by CUT& RUN in K562 cells with and without dox-mediated recruitment of rTetR-KRAB to the (A) no-spacer reporter and (B) 5 kb reporter. Numbers in square brackets indicate y-axis (CPM) scaling. Reporter elements that also appear in the human genome or that are duplicated within the reporter (i.e. pEF, H2B, and polyA [pA]) are masked in light gray. (C-D) Genome browser tracks of H3Kac, H3K9me3, H3K4me3, and IgG ChromaBlocks-identified domains with and without recruitment of rTetR-KRAB, looking at the surrounding locus where the (C) NS reporter or (D) 5 kb reporter is integrated in cells (purple star within first intron of PPP1R12C, which is oriented in the reverse direction). Note that this snapshot does not include an in situ representation of the reporter, which instead has been appended to the reference genome as a separate chromosome in order to preserve gene annotations. Domains for H3K9me3, H3Kac, and H3K4me3 are depicted as horizontal bars and were called with ChromaBlocks to estimate spreading distance with KRAB recruitment. Domains including or proximal to the integration site are annotated with their domain lengths (excludes the reporter length). (E) Quantification of modification level changes with KRAB recruitment. Coordinates of the ChromaBlocks-identified domains with asterisks in (C) and (D) were used to define a region for signal integration for the corresponding dox-treated (+ dox) or untreated (-dox) sample. Log2 ratios of the total dox signal to total nodox signal within these regions are shown for each histone modification for each reporter cell line.
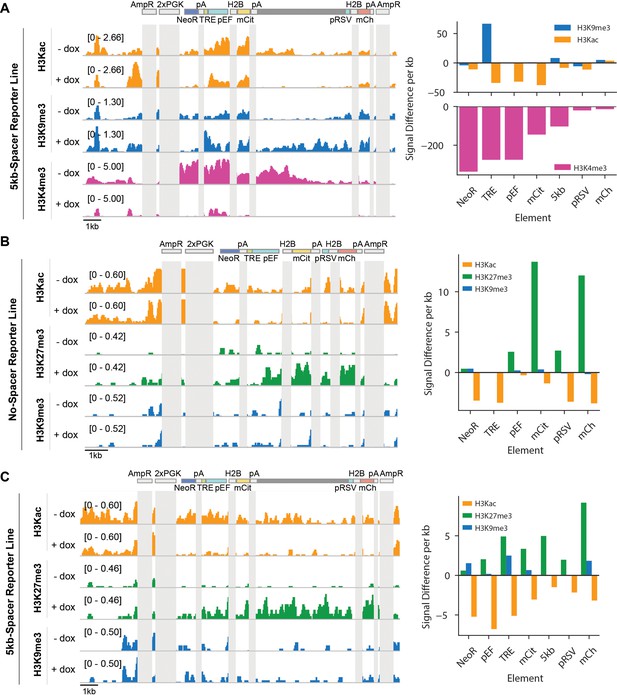
Recruitment of KRAB or HDAC4 for 5 days in CHO-K1 cells induces changes in chromatin modifications at the two-gene reporters.
Genome browser tracks of histone 3 acetyl-lysine (H3Kac), histone 3 lysine 9 trimethylation (H3K9me3), histone 3 lysine 27 trimethylation (H3K27me3), and histone 3 lysine 4 trimethylation (H3K4me3) with (+ dox) and without (-dox) recruitment of (A) rTetR-KRAB to the 5kb-spacer reporter, (B) rTetR-HDAC4 to the NS reporter and (C) rTetR-HDAC4 to the 5 kb reporter. Reporter and MI-HAC elements that appear multiple times or share significant sequence identity (i.e. H2B, polyA [pA], PGK promoters, ampicillin resistance [AmpR]) are masked in light gray).
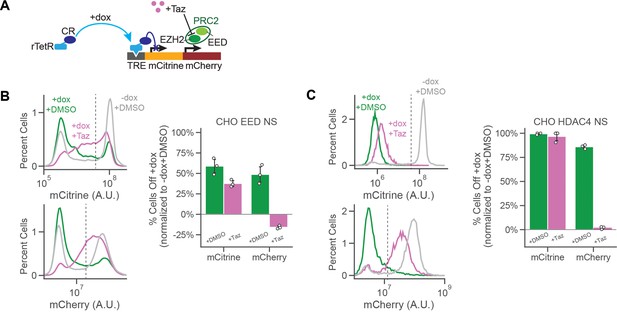
EZH2 inhibitor Tazemetostat affects silencing after recruitment of EED and HDAC4.
(A) Addition of Tazemetostat (Taz) inhibits the EZH2 methyltransferase from PRC2. (B–C) Fluorescence distributions of mCitrine (top left) and mCherry (bottom left) measured by flow cytometry either without CR recruitment (-dox, gray) or after 7 days of (B) EED or (C) HDAC4 recruitment ( + dox + DMSO , green) and with Taz( + dox + Taz , pink) at the NS reporter in CHO-K1. Percentages of cells OFF normalized by the -dox + DMSO condition (right), based on threshold (dotted line) (n = 3 ).
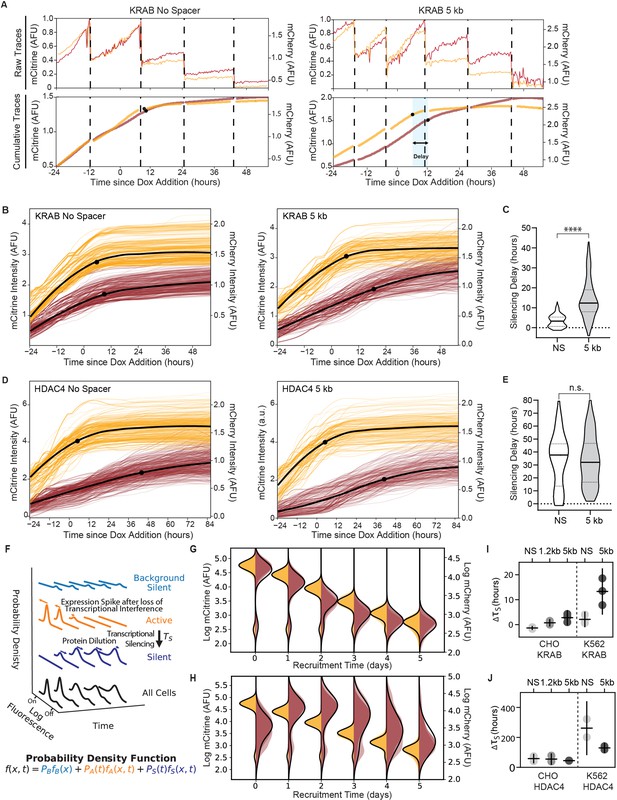
Single-cell data measures delay in transcriptional silencing of the two genes.
(A) (Top) Example raw traces measured by time-lapse microscopy showing total fluorescence of mCitrine (yellow) and mCherry expression (red) as a function of time in an individual cell lineage in CHO-K1 with the NS reporter (left) and 5 kb reporter (right). Dotted lines denote cell divisions. Recruitment of KRAB starts after 24 hours by dox addition. (Bottom) Cumulative single-cell traces stitched across cell divisions showing estimated silencing time points (black dots, Materials and methods) and silencing delay (blue shading). (B) Cumulative single-cell traces of mCitrine (yellow) and mCherry (red) in CHO-K1 cells with NS reporter (left, n = 296) and 5 kb reporter (right, n = 218) with the mean trace (black curves) and median silencing times (dots) as a function of time since recruitment of KRAB by dox addition at time 0. (C) Distribution of delay times between silencing of mCitrine and mCherry in individual cells as shown in (A) after recruitment of KRAB in (NS reporter median silencing delay = 3.3 hr; 5 kb reporter median silencing delay = 12.3 hr; statistically significant difference by Welch’s unequal variances T-test). (D) Cumulative single-cell traces as a function of time relative to HDAC4 recruitment (NS, n = 211; 5 kb, n = 291), as in (B). (E) Distribution of delay times after recruitment of HDAC4 (NS reporter median silencing delay = 38 hours; 5 kb reporter median silencing delay = 32 hours; no statistically significant difference by Welch’s unequal variances T-test). (F) A probabilistic model consisting of three states: background silent (light blue), active (orange), silent (dark blue). Each state has its own weight and distribution, and all states are summed to a final probability density function (black) that describes the probability of finding a cell with fluorescence at time. This model is used to fit daily flow cytometry data to extract transcriptional silencing times () upon CR recruitment, while taking into account: stochastic transitions of cells from the active to the silent state upon CR-mediated silencing, spike in expression after loss of transcriptional interference, and mRNA and protein degradation and dilution (Materials and methods). (G, H) Overlaid daily distributions of mCitrine (transparent yellow) and mCherry (transparent red) fluorescence from flow cytometry during recruitment of (G) KRAB and (H) HDAC4 with average model fit (black line) (n = 3). (I) Silencing delays between mCitrine and mCherry after KRAB recruitment extracted from daily flow cytometry time-courses using the model in (F) for different spreading reporters: NS, 1.2 kb, 5 kb. Each dot represents a clone for CHO-K1 (left) and a biological replicate for K562 (right); horizontal bar is mean delay, vertical bar is 90% confidence interval from the fit estimated using the t-distribution (n = 3). (J) Silencing delays between mCitrine and mCherry after HDAC4 recruitment (CHO-K1, n = 3 clones; K562 n = 2 biological replicates); same notation as (I).
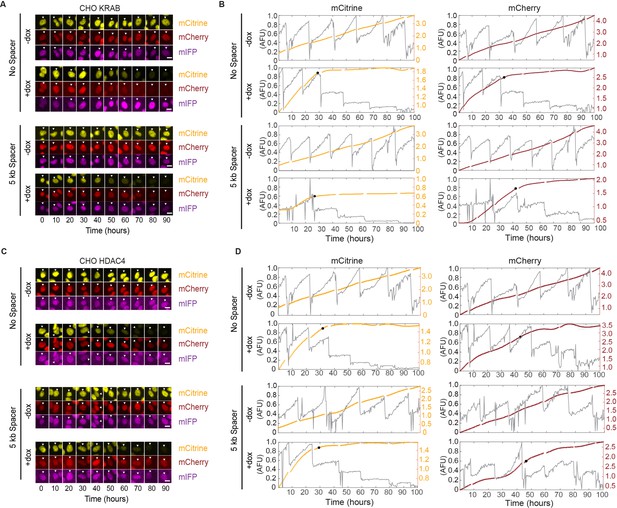
Example images and single-cell analysis of silencing dynamics from time-lapse microscopy of CHO-K1 cells.
(A, C) Filmstrips at 10 hr intervals of representative tracked single-cells upon dox induced (A) KRAB and (C) HDAC4 silencing from time-lapse movies in all channels. Scale bar represents 10 µm. (B, D) Plot of nuclear intensity of the single-cell tracked in the filmstrip (A,C) in the mCitrine and mCherry channels. Gray traces represent the integrated nuclear intensity prior to stitching. The computationally stitched trace is shown in yellow (mCitrine), or red (mCherry), respectively. The black dot represents the time at which the cell was considered silenced.
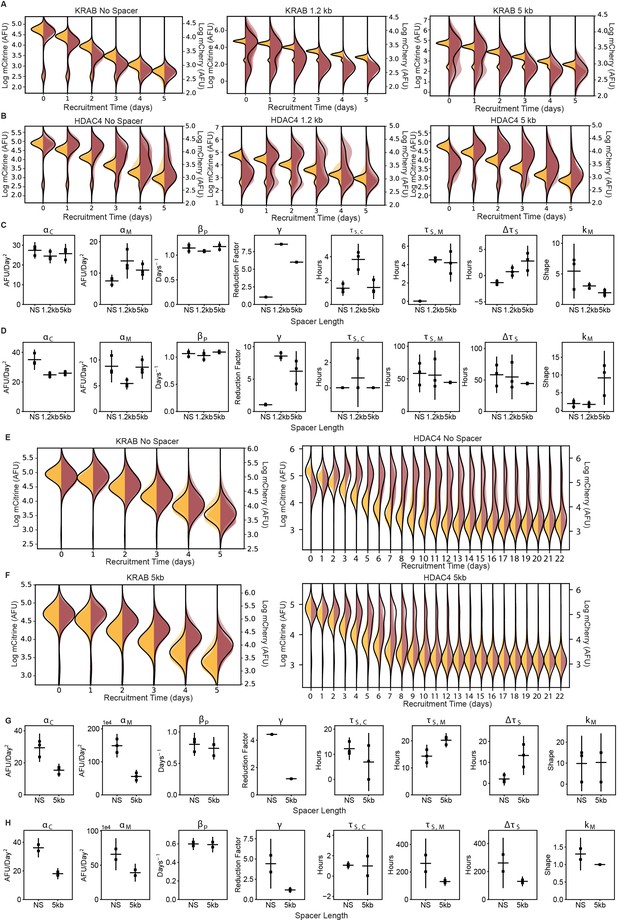
Dynamics of silencing measured by flow cytometry and fit by gene expression model.
(A-B) Overlaid daily distributions of mCitrine (transparent yellow) and mCherry (transparent red) fluorescence from flow cytometry during recruitment of (A) KRAB and (B) HDAC4 in CHO-K1 for each spreading reporter indicated in the titles with average fit to the model in Figure 3F (black line) (n = 3 clones). (C-D) Parameters from probabilistic model fit (Figure 3F) of flow cytometry data after (C) KRAB and (D) HDAC4 recruitment in CHO-K1. Each dot represents a clone of the given reporter, horizontal bar is mean delay, vertical bar is 90% confidence interval estimated using the t-distribution. (E-F) Overlaid replicates of daily distributions of mCitrine (transparent yellow) and mCherry (transparent red) fluorescence from flow cytometry, with average model fit (black line), during recruitment of KRAB (left, n = 3) and HDAC4 (right, n = 2) to (E) NS and (F) 5 kb reporter in K562. (G-H) Parameters from probabilistic model fit of flow cytometry data after (G) KRAB and (H) HDAC4 recruitment in K562. Each dot represents a replicate of the given reporter line, horizontal bar is mean delay, vertical bar is 90% confidence interval estimated using the t-distribution.
Steady-state expression in the absence of dox in CHO-K1 versus K562 cells.
Fluorescence distributions of pEF-mCitrine (left) and pRSV-mCherry (right) from flow cytometry in the absence of dox-mediated recruitment in CHO-K1 (orange) versus K562 (purple) in different reporter constructs: (A) NS and 5 kb , and (B) SH insulator. Note that pRSV-mCherry is always expressed at higher levels in K562 compared to CHO-K1.
Spreading of silencing movie for KRAB NS.
Figure 3—videos 1–4 are zoomed-in views of spreading of silencing for KRAB and HDAC4 in the NS and 5 kb reporters in CHO-K1. Each movie is digitally cropped to follow a subset of cells captured in the full frame. mCitrine fluorescence is pseudo-colored as yellow, and mCherry fluorescence is pseudo-colored in red. Timestamps are in HH:MM relative to dox addition at 0 h.
Spreading of silencing movie for KRAB 5 kb.
Figure 3—videos 1–4 are zoomed-in views of spreading of silencing for KRAB and HDAC4 in the NS and 5 kb reporters in CHO-K1. Each movie is digitally cropped to follow a subset of cells captured in the full frame. mCitrine fluorescence is pseudo-colored as yellow, and mCherry fluorescence is pseudo-colored in red. Timestamps are in HH:MM relative to dox addition at 0 h.
Spreading of silencing movie for HDAC4 NS.
Figure 3—videos 1–4 are zoomed-in views of spreading of silencing for KRAB and HDAC4 in the NS and 5 kb reporters in CHO-K1. Each movie is digitally cropped to follow a subset of cells captured in the full frame. mCitrine fluorescence is pseudo-colored as yellow, and mCherry fluorescence is pseudo-colored in red. Timestamps are in HH:MM relative to dox addition at 0 h.
Spreading of silencing movie for HDAC4 5 kb.
Figure 3—videos 1–4 are zoomed-in views of spreading of silencing for KRAB and HDAC4 in the NS and 5 kb reporters in CHO-K1. Each movie is digitally cropped to follow a subset of cells captured in the full frame. mCitrine fluorescence is pseudo-colored as yellow, and mCherry fluorescence is pseudo-colored in red. Timestamps are in HH:MM relative to dox addition at 0 hr.
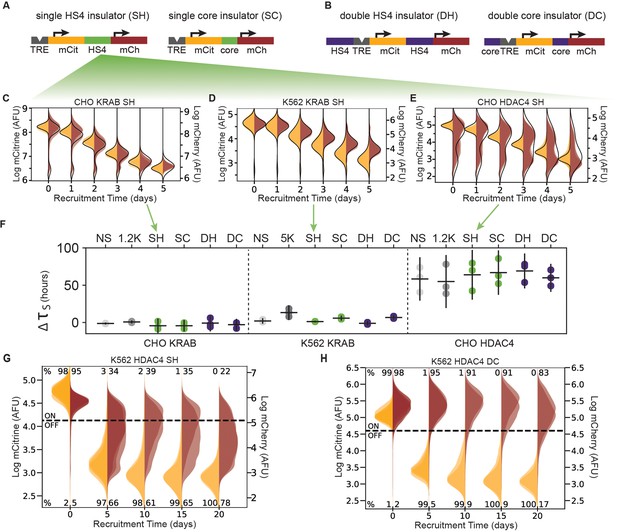
The role of the cHS4 insulators in spreading of transcriptional silencing across genes.
(A) Single insulator geometries between mCitrine (mCit) and mCherry (mCh) fluorescent genes, with full 1.2 kb HS4 insulator (SH, left) and 250 bp core insulator (SC, right). (B) Double insulator geometries with full-length 1.2 kb HS4 insulator (DH, left) and 250 bp core insulator (DC, right) flanking the TRE-pEF-mCitrine region. (C–E) Overlaid replicates of daily distributions of mCitrine (yellow) and mCherry (red) fluorescence from flow cytometry during recruitment of (C) KRAB in CHO-K1, (D) ) KRAB in K562, or (E) HDAC4 in CHO-K1, to SH reporters with average model fit (black line) (n = 3). (F) Silencing delay times between mCitrine and mCherry in different insulator reporters after chromatin regulator recruitment for 5 days (each dot is a replicate, horizontal bar is mean delay, vertical bar is 90 % confidence interval estimated using the t-distribution). (G–H) Overlaid replicates of daily distributions of mCitrine (yellow) and mCherry (red) fluorescence from flow cytometry during extended recruitment of HDAC4 to (G) SH reporter or (H) DC reporter in K562 (n = 3 ).
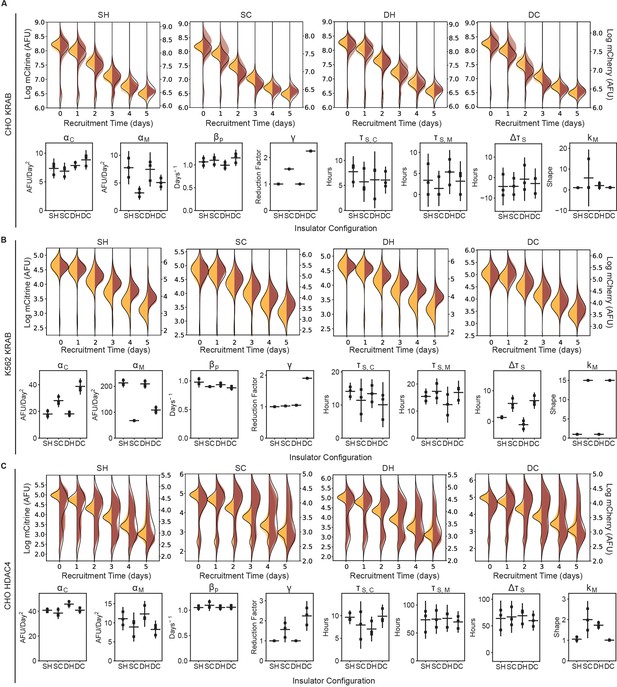
The effect of all insulator configurations on spreading dynamics of transcriptional silencing in CHO-K1 and K562.
(Top panels of A-C) Overlaid replicates of daily distributions of mCitrine (transparent yellow) and mCherry (transparent red) fluorescence from flow cytometry and average model fits (black lines) during recruitment of : (A) KRAB in CHO-K1, (B) K562 with KRAB, and (C) CHO-K1 with HDAC4. (Bottom panels of A-C) Parameters from probabilistic model fit of flow cytometry data of insulator reporters with (A) KRAB recruitment in CHO-K1, (B ) K562 with KRAB, and (C) CHO-K1 with HDAC4. Each dot represents a replicate of the given insulator reporter line, horizontal bar is mean delay, vertical bar is 90 % confidence interval estimated using the t-distribution.
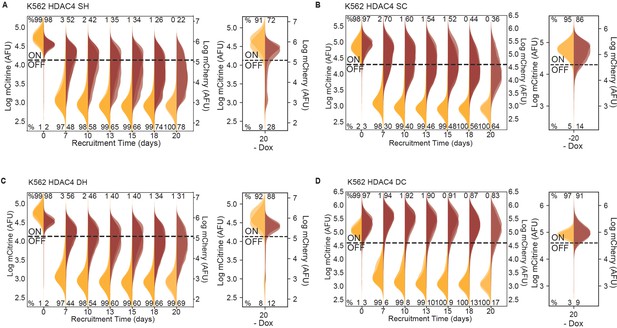
Insulators attenuate spreading of silencing via HDAC4 in K562.
Overlaid replicates of distributions of mCitrine (transparent yellow) and mCherry (transparent red) fluorescence from flow cytometry data during extended recruitment of HDAC4 (left, n = 3) and 20 days without recruitment (right, n = 2) in K562 with insulator geometries defined in Figure 4A&B: (A) SH, (B) SC, (C) DC, and (D) DH.
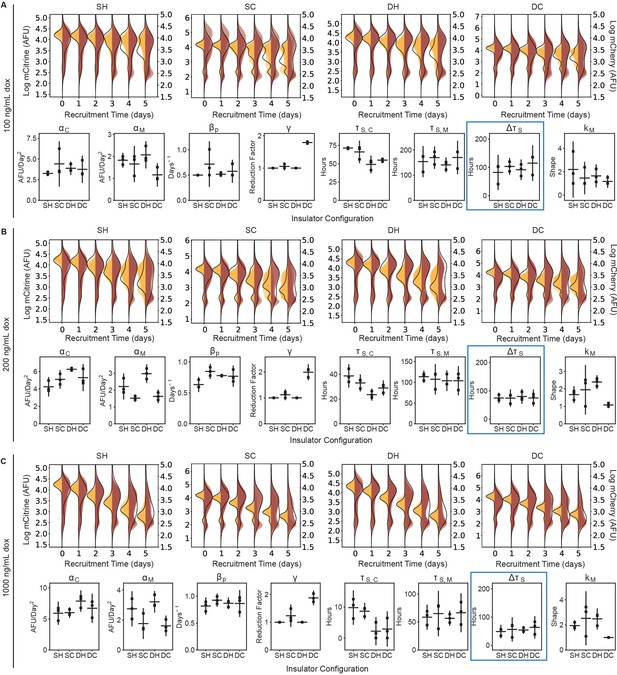
Dynamics of spreading upon weaker gene targeting insulator reporters with HDAC4 at lower dox concentrations in CHO-K1.
(Top panels of A-C) Overlaid replicates of daily distributions of mCitrine (transparent yellow) and mCherry (transparent red) fluorescence from flow cytometry and average model fits (black lines) during recruitment of HDAC4 in CHO-K1 at (A) 100 ng/mL, (B) 200 ng/mL, and (C) 1000 ng/mL (saturating dox). (Bottom panels of A-C) Parameters from probabilistic model fit of flow cytometry data for insulator reporters with HDAC4 recruitment in CHO-K1 at: (A) 100 ng/mL, (B) 200 ng/mL, and (C) and 1000 ng/mL. Each dot represents a replicate of the given insulator reporter line, horizontal bar is mean delay, vertical bar is 90 % confidence interval estimated using the t-distribution (n = 3). Silencing delay times () highlighted in blue.
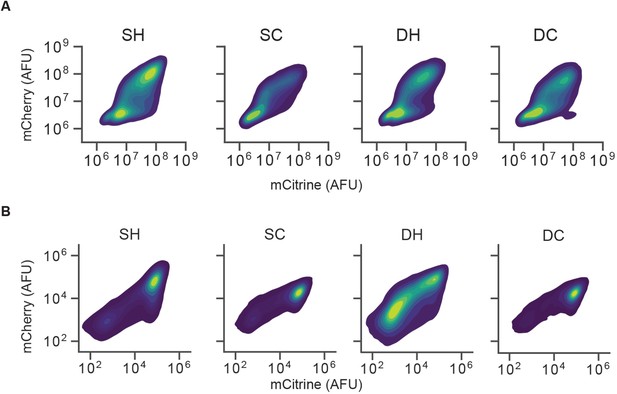
Insulators do not block spreading of silencing with weaker gene targeting at lower dox concentrations.
2D density plots of mCitrine and mCherry fluorescence measured by flow cytometry in CHO-K1 cells with insulator constructs indicated in the title of each graph after 5 days of (A) 200 ng/mL dox recruitment of HDAC4, or (B) 4 ng/mL dox recruitment of KRAB.
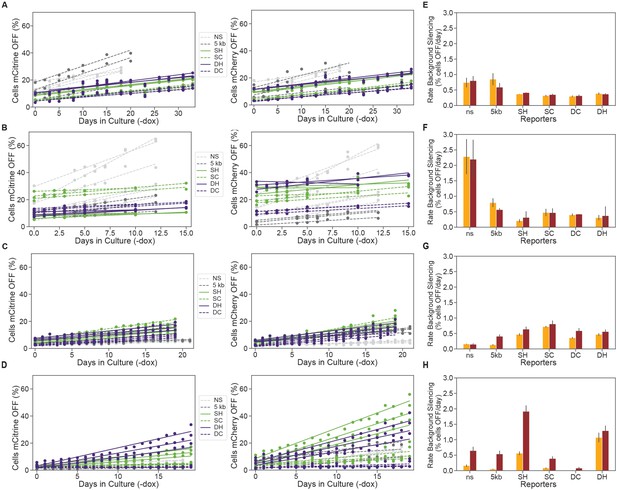
Insulators prevent background silencing of reporter genes.
(A-D) Background levels of mCitrine (left) and mCherry silencing (right) in reporters with lambda spacers or insulators in the absence of dox for: (A) CHO-K1 KRAB, (B) CHO-K1 HDAC4, (C) K562 KRAB, and (D) K562 HDAC4 cell lines. (E-H) Rates of background silencing of mCitrine and mCherry in the absence of dox in: (E) CHO-K1 KRAB, (F) CHO-K1 HDAC4, (G) K562 KRAB, and (H) K562 HDAC4 cell lines. Error bars represent standard error of mean for three clones or replicates.
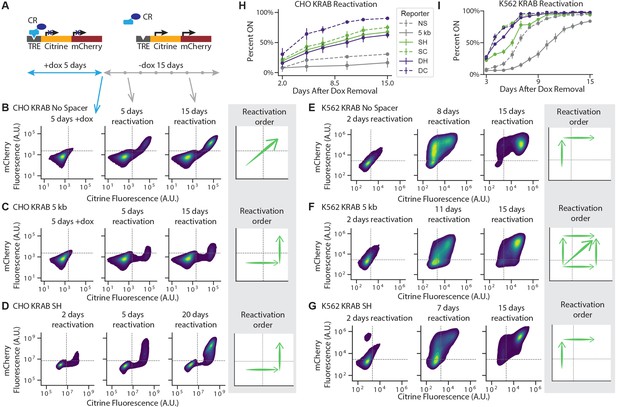
Reactivation of gene expression spreads between the two genes.
(A) Schematic of experimental setup: each CR is recruited upstream of the mCitrine gene for 5 days. Recruitment is then stopped by removing dox and reactivation is monitored every few days by flow cytometry. (B-G) 2D density plots of mCitrine and mCherry fluorescence from flow cytometry show pattern of gene reactivation in: (B) CHO-K1 KRAB NS (n = 3 clones), (C) CHO-K1 KRAB 5 kb (n = 4 clones), (D) CHO-K1 KRAB SH (n = 3), (E) K562 KRAB NS (n = 3), (F) K562 KRAB 5 kb (n = 3), and (G) K562 KRAB SH (n = 3). (H-I) Percent of cells in which at least one reporter gene reactivated over time after dox removal after KRAB release in (H) CHO-K1 and (I) K562. Replicates are from either independent clonal cell lines, where indicated, or from biological replicates of multiclonal populations.
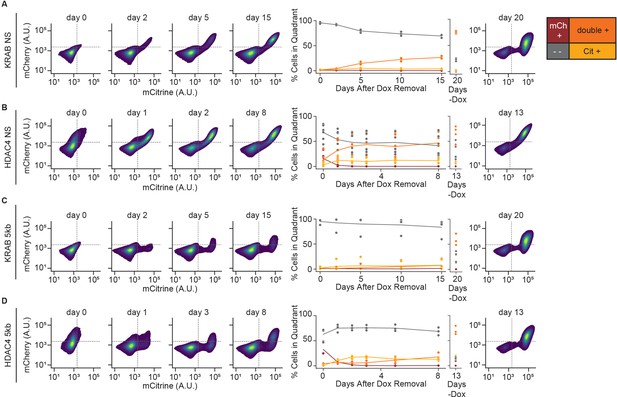
Reactivation of gene expression in CHO-K1 NS and 5 kb reporter lines.
(Left) 2D density plots of mCitrine and mCherry fluorescence from flow cytometry at different timepoints of reactivation (day 0 represents the end of 5 days of CR recruitment), (middle) percentages of cells in each quadrant as a function of release time, and (right) 2D density plots for no dox controls in: (A) NS reporter with KRAB (n = 3 clones), (B) NS reporter with HDAC4 (n = 6 clones), (C) 5 kb reporter with KRAB (n = 4 clones), and (D) 5 kb reporter with HDAC4 (n = 3 clones).
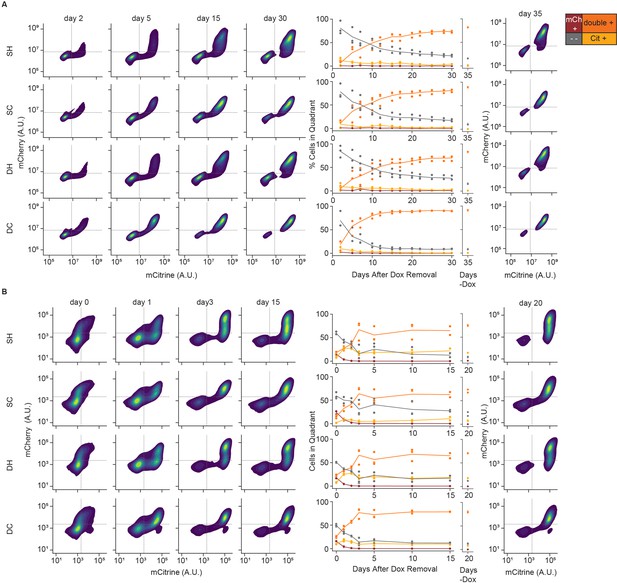
Reactivation of gene expression in CHO-K1 insulator reporter lines.
(Left) 2D density plots of mCitrine and mCherry fluorescence from flow cytometry at different timepoints of reactivation (day 0 represents the end of 5 days of CR recruitment), (middle) percentages of cells in each quadrant as a function of release time, and (right) 2D density plots for no dox controls in: (A) insulators with KRAB and (B) insulators with HDAC4. Replicates are from biological replicates of multiclonal populations (n = 3 ).
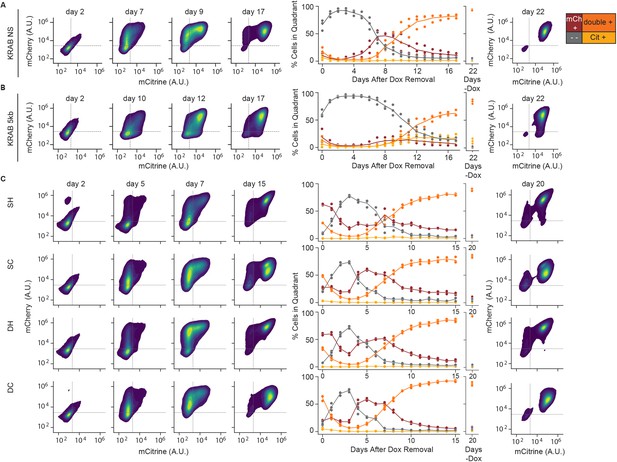
Reactivation of gene expression in K562 cell reporter lines.
(Left) 2D density plots of mCitrine and mCherry fluorescence from flow cytometry at different timepoints of reactivation (day 0 represents the end of 5 days of CR recruitment), (middle) percentages of cells in each quadrant as a function of release time, and (right) 2D density plots for no dox controls in: (A) NS, (B) 5 kb, and (C) insulator reporters after release of KRAB. Replicates are from biological replicates of multiclonal populations (n = 3 ).
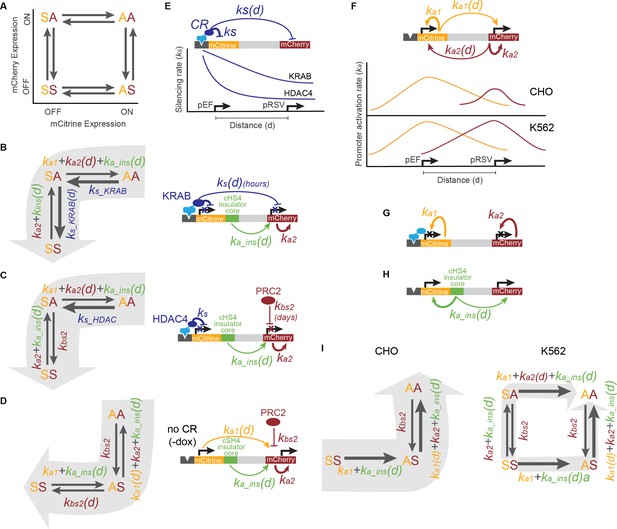
Model of multi-gene control coordinated by the action of CRs, promoters, and insulators.
(A) Four states for a two-gene reporter, where the first letter (yellow) represents the mCitrine state and the second letter (red) represents mCherry state, as either active (A) or silent (S). Arrows represent the rates at which each gene is turned on or off. (B) During KRAB recruitment, cells transition from both genes active (AA) to both gene silent (SS), with mCitrine silencing first (SA intermediate state). The silencing rates of KRAB at the nearby pEF-mCitrine (ks_KRAB ) and pRSV-mCherry (ks_KRAB(d), where d is the distance between the pEF and pRSV promoters) dominate over the activation rates of promoters (ka1 , ka2 ) and insulators (ka_ins(d),where here d is the distance between the core insulator and a nearby promoter). (C) During HDAC4 recruitment, silencing of mCitrine (AA to SA) is driven by the silencing rate of HDAC4 (ks_HDAC4 ), while silencing of mCherry (SA to SS) is driven by background silencing rate (kbs2 ) due to PRC2. Both the pRSV promoter (ka2 ) and insulator reactivation rates (ka_ins) can compete with pRSV-mCherry silencing. (D) Before CR recruitment (no dox), pEF as well as insulators can act from a distance on pRSV (ka1(d), ka_ins(d)) preventing background silencing of mCherry (kbs2 ). (E) KRAB can act on genes over a long distance (ks(d)), silencing both mCitrine and mCherry, while the range of HDAC4 silencing is much smaller (ks ), only directly affecting mCitrine. (F) In the absence of CR recruitment, promoters can activate themselves (ka1 , ka2 ), and maintain activity of genes at a distance (ka1(d), ka2 (d)). In CHO-K1, pRSV is weaker than in K562. (G) When genes are silenced, their promoters cannot act on neighboring genes, instead only reactivate themselves (ka1 , ka2 ). (H) The core element of the cHS4 insulator can maintain activity of the nearby genes and drive reactivation after CR-mediated silencing (ka_ins(d)). (I) Reactivation after silencing is driven by gene activation rates (ka1 , ka2 ) and core cHS4 insulators (ka_ins(d)) if present. In CHO-K1 cells where pEF is stronger than pRSV, mCitrine reactivates first followed by mCherry (left). However, in K562 where both promoters are equally strong, either gene can reactivate first (right).
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Cell line (Cricetulus griseus) | CHO-K1 (with Human artificial chromosome) | Oshimura Lab, Yamaguchi et al., 2011 | ||
| Cell line (Homo sapiens) | K562 (lymphoblast, adult leukemia) | ATCC | CCL-243 | |
| Cell line (Homo sapiens) | HEK293T (epithelial, embryo kidney) | ATCC | CRL-3216 | |
| Recombinant DNA reagent | pSL007_PB_pCMV-H2B-mIFP-T2A-rTetR-ratKRAB-zeo (plasmid) | This paper | RRID: Addgene_179438 | Piggybac rTetR-CR: rat KRAB |
| Recombinant DNA reagent | pLB62_PB_pGK-H2B-mIFP-T2A-rTetR-humanKRAB-zeo (plasmid) | This paper | RRID: Addgene_179439 | Piggybac rTetR-CR: human KRAB |
| Recombinant DNA reagent | pLB37_PB_pGK-H2B-mIFP-T2A-rTetR-HDAC4-zeo (plasmid) | This paper | RRID: Addgene_179440 | Piggybac rTetR-CR: human HDAC4 |
| Recombinant DNA reagent | pSL006_phiC31_neo-5xTetO-pEF-H2B-mCitrine-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179425 | NS reporter phiC31 |
| Recombinant DNA reagent | pMH010_phiC31_neo-5xTetO-pEF-H2B-mCitrine-1.2kb,lambda-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179427 | 1.2 kb reporter phiC31 |
| Recombinant DNA reagent | pSL008_phiC31_neo-5xTetO-pEF-H2B-mCitrine-5kb,lambda-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179426 | 5 kb reporter phiC31 |
| Recombinant DNA reagent | pSL011_AAVS1-puro-9xTetO-pEF-H2B-mCitrine-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179428 | NS reporter AAVS1 |
| Recombinant DNA reagent | pSL012_AAVS1-puro-9xTetO-pEF-H2B-mCitrine-5kb,lambda-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179429 | 5 kb reporter AAVS1 |
| Recombinant DNA reagent | pMH002_phiC31_neo-5xTetO-pEF-H2B-mCitrine-HS4-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179430 | Single cHS4 (SH) reporter phiC31 |
| Recombinant DNA reagent | pMH003_phiC31_neo-5xTetO-pEF-H2B-mCitrine-coreHS4-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179431 | Single core cHS4 (SC) reporter AAVS1 phiC31 |
| Recombinant DNA reagent | pMH004_phiC31_neo-HS4-5xTetO-pEF-H2B-mCitrine-HS4-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179432 | Double cHS4 (DH) reporter phiC31 |
| Recombinant DNA reagent | pMH005_phiC31_neo-core-5xTetO-pEF-H2B-mCitrine-core-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179433 | Double core cHS4 (DC) reporter phiC31 |
| Recombinant DNA reagent | pMH011_AAVS1_puro-pA-9xTetO-pEF-H2B-mCitrine-HS4-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179434 | Single cHS4 (SH) reporter AAVS1 |
| Recombinant DNA reagent | pMH012_AAVS1_puro-pA-9xTetO-pEF-H2B-mCitrine-core-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179435 | Single core cHS4 (SC) reporter AAVS1 |
| Recombinant DNA reagent | pMH014_AAVS1_puro-pA-HS4-9xTetO-pEF-H2B-mCitrine-HS4-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179436 | Double cHS4 (DH) reporter AAVS1 |
| Recombinant DNA reagent | pMH015_AAVS1_puro-pA-core-9xTetO-pEF-H2B-mCitrine-core-pRSV-H2B-mCherry (plasmid) | This paper | RRID: Addgene_179437 | Double core cHS4 (DC) reporter AAVS1 |
| Antibody | Anti-rabbit IgG H&L chain (guinea pig, polyclonal) | Antibodies-Online | Cat #ABIN101961 | CUT&RUN (1:100) |
| Antibody | Anti-H3ac (rabbit, polyclonal) | Active Motif | Cat #39,139 | CUT&RUN (1:100) |
| Antibody | Anti-H3K4me3 (rabbit, polyclonal) | Active Motif | Cat #39,159 | CUT&RUN (1:100) |
| Antibody | Anti-H3K27me3 (rabbit, monoclonal) | Cell Signaling | Cat #9,733 S | CUT&RUN (1:50) |
| Peptide, recombinant protein | pA-MNase | Henikoff Lab | ||
| Commercial assay or kit | CUTANA ChIC/CUT&RUN Kit | EpiCypher | Cat #14 – 1048 | CUT&RUN |
| Chemical compound, drug | Doxycycline | Tocris Bioscience | 4,090 | |
| Chemical compound, drug | Tazemetostat-6438 | Selleck Chemicals | S7128 | EZH2 inhibitor |
| Software, algorithm | MACKtrack | Taylor, 2019 | https://github.com/brookstaylorjr/MACKtrack |
Statistics of reporters after silencing.
Percent of cells off with standard error of mean (SEM) for each gene in the -dox and +dox condition after the number of days indicated, with threshold drawn in Figure 1 (dotted line). Welch’s unequal variances T-test was run on the percent cells off from each replicate (n=3) and p-value is shown for comparison of -dox versus +dox for each gene.
| % mCitrine Off | % mCherry Off | |||||
|---|---|---|---|---|---|---|
| -dox | +dox | p-value | -dox | +dox | p-value | |
| CHO KRAB No Spacer (day 5) | 15±2 | 100±0.03 | 0.00055 | 15.3±2.5 | 100±0.03 | 0.00084 |
| CHO KRAB 5kb Spacer (day 5) | 23.2±6.9 | 99.9±0.1 | 0.00789 | 39.8±8.8 | 96.9±1.9 | 0.01916 |
| CHO HDAC4 No Spacer (day 5) | 9.6±2.1 | 99.6±0.3 | 0.00044 | 11±2.0 | 76.2±6.9 | 0.0071 |
| CHO HDAC4 5kb Spacer (day 5) | 11.3±2.9 | 99.9±0.03 | 0.00109 | 32.2±3.7 | 68.1±0.9 | 0.00764 |
| K562 KRAB No Spacer (day 5) | 2.6±0.2 | 95.9±2.0 | 0.00039 | 6.8±1.1 | 87.7±4.5 | 0.00198 |
| K562 KRAB 5kb Spacer (day 5) | 4.4±0.2 | 97±1.2 | 0.00012 | 12.3±1.4 | 86.1±4.3 | 0.00148 |
| K562 HDAC4 No Spacer (day 20) | 10±6.6 | 98.3±0.3 | 0.0056 | 30.5±7.2 | 67.1±4.8 | 0.01784 |
| K562 HDAC4 5kb Spacer (day 20) | 9.4±5.4 | 99.3±0.09 | 0.00352 | 24.2±5.1 | 83.8±1.1 | 0.00555 |
| HEK HDAC4 5kb Spacer (day 7) | 1.3±0.2 | 86.7±3.5 | 0.00167 | 2.5±0.5 | 73.5±3.8 | 0.00256 |
Delay times between mCitrine and mCherry silencing from model fit to flow cytometry data.
The 90% confidence interval (CI) was estimated using the t-distribution.
| Reporter | Δτs (hour) | 90% CI | ||
|---|---|---|---|---|
| CHO-K1 | KRAB | NS | -1.34 | ±0.59 |
| 1.2 kb | 0.75 | ±1.26 | ||
| 5kb | 2.78 | ±2.82 | ||
| SH | -4.36 | ±8.96 | ||
| SC | -4.29 | ±6.75 | ||
| DH | -0.78 | ±10.36 | ||
| DC | -2.92 | ±7.14 | ||
| HDAC4 | NS | 58.4 | ±28.2 | |
| 1.2 kb | 54.9 | ±34.8 | ||
| 5kb | 44.5 | ±1.1 | ||
| SH | 64 | ±32.5 | ||
| SC | 67 | ±28.9 | ||
| DH | 69.1 | ±22.6 | ||
| DC | 59.9 | ±17.9 | ||
| K562 | KRAB | NS | 2.1 | ±2.91 |
| 5kb | 13.3 | ±9.1 | ||
| SH | 1.29 | ±0.37 | ||
| SC | 5.86 | ±2.58 | ||
| DH | -1.06 | ±2.25 | ||
| DC | 6.76 | ±2.52 | ||
| HDAC4 | NS | 261 | ±177 | |
| 5kb | 130 | ±32 | ||
Primers used for investigating transcriptional interference.
| Assay | Locus | Name | Sequence (5’->3’) |
|---|---|---|---|
| PCR | Beta-actin (human genome) | ACTB-F1 | CATGTACGTTGCTATCCAGGC |
| PCR | ACTB-R1 | CTCCTTAATGTCACGCACGAT | |
| PCR | mCitrine | citrine3prime_fwd | GTAAGTGTACCCAATTCGCCCTATAGTGAG |
| PCR | mCherry | mCherry5prime_reverse | TCCTCGCCCTTGCTCACCAT |
| qPCR | mCitrine | F_cit_Set2 | CGGCGACGTAAACGGCCACAAGTTCAG |
| qPCR | R_cit_Set2 | CTTGCCGGTGGTGCAGATGAA | |
| qPCR | 5kb lambda | 5kb_lambda_Set1_F | CCACCTGTTACTGGTCGATTTA |
| qPCR | 5kb_lambda_Set1_R | GATATTCCCACCTCCGGTTAAG | |
| qPCR | mCherry | mCherry_Set1_F | AGGACGGCGAGTTCATCTA |
| qPCR | mCherry_Set1_R | CCCATGGTCTTCTTCTGCATTA | |
| qPCR | Beta-actin (CHO-K1 genome) | bActin_F | ACTGGGACGATATGGAGAAG |
| qPCR | bActin_R | GGTCATCTTTTCACGGTTGG |





