Cerebellar modulation of memory encoding in the periaqueductal grey and fear behaviour
Figures
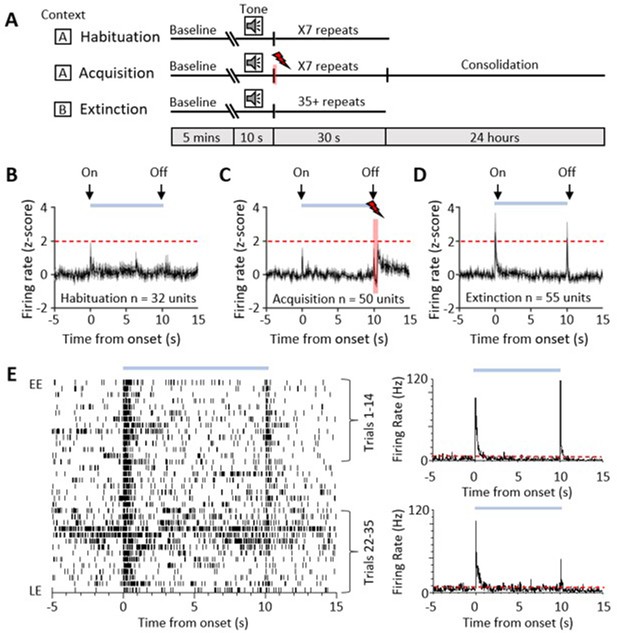
Single-unit ventrolateral periaqueductal grey (vlPAG) responses during auditory cued fear delay conditioning.
(A) Schematic representation of the fear conditioning paradigm composed of habituation, acquisition, and extinction sessions. Habituation and acquisition were carried out in context A, whilst extinction training was in context B. During acquisition, a conditioned stimulus (CS) tone was paired with an unconditioned stimulus (US) footshock (see Materials and methods for details). (B) Peri event time histogram (PETH) showing average firing rate of all available single units (n = 32) recorded during presentation of the unconditioned tone (10 s) during habituation in eight control animals. Time 0, tone onset. Light blue bar shows time the tone is on. (C) Same as (B) but all available single units (n = 50) recorded during acquisition from 10 control animals (shaded red and lightening symbol indicates time of US footshock when the stimulus artefact prevented neural recording). (D) Same as (B) but all available single units (n = 55) recorded during presentation of the unreinforced CS+ throughout extinction from 10 control animals. For (B–D), individual unit activity was z-score normalised to a 5 s baseline before tone onset. Horizontal dashed red line represents significance level (p<0.05). PETHs show mean ± SEM; 40 ms bins. (E) One example of a type 1 onset and offset single unit recorded during extinction training. Data displayed as raster plot from early extinction (EE) to late extinction (LE) with corresponding PETH for EE (trials 1–14) and LE (trials 22–35) (40 ms bins), time 0 onset of CS+. Horizontal dashed red line represents significance level (p<0.05).
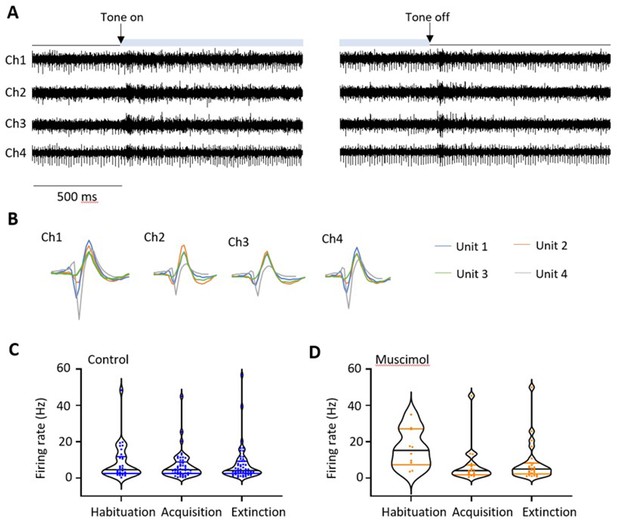
Tetrode recordings.
(A) Example electrophysiological data recorded from a ventrolateral periaqueductal grey (vlPAG) tetrode (30 kHz sampling rate, 600–3 kHz bandpass filter, Ch1-4, tetrode channels 1–4). Raw data captured during early extinction training around the time of CS+ onset (left, tone on) and CS+ offset (right, tone off). Light blue bar shows time the tone is on. (B) Spikes were sorted using all channels in MClust to identify individual units; in this case, four units (colour coded) were isolated. Individual units were classified as described in Materials and methods. (C) Violin plots showing distribution of baseline firing rates for all available single units recorded in vlPAG during three phases of fear conditioning: habituation (n = 30 units), acquisition (n = 50 control units), and extinction (n = 55 control units). Horizontal black lines show median, and blue lines IQR firing rate for each phase of conditioning. (D) Same as (C), but for single units recorded during muscimol experiments. Habituation (n = 10 units), acquisition (n = 17 units), and extinction (n = 25 units). Horizontal black lines show median, and orange lines IQR firing rate for each phase of conditioning.
-
Figure 1—figure supplement 1—source data 1
Numerical data to support graphs in Figure 1—figure supplement 1.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig1-figsupp1-data1-v2.docx
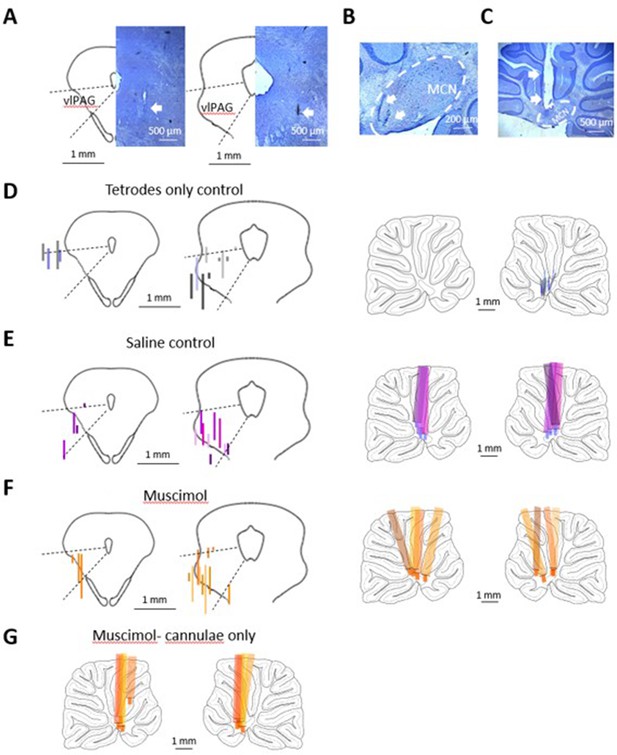
Histological verification of implants.
(A) Photomicrographs of two example tetrode tracks (arrows) in periaqueductal grey (PAG). Outlines show left half of two coronal sections (anteroposterior [AP] levels –6.5 and –7.5) of the PAG. Dotted lines indicate approximate location of the ventrolateral sector (vlPAG). Note that a lack of correspondence between the physiological and histological boundaries of the lateral border of PAG has been reported previously (McMullan and Lumb, 2006). (B) Same as (A) but tracks in sagittal sections (±1 mm of midline) medial cerebellar nucleus (MCN) and (C) example track of a cerebellar cannula. (D) Pairs of figurines to the left show two coronal sections (AP levels –6.5 and –7.5) of the PAG. Pairs of figurines to the right show sagittal sections of left and right cerebellum (±1 mm of midline). Tetrodes-only controls (n = 6 rats, tetrodes implanted on one side of PAG and also in the contralateral MCN), tracks colour coded for each animal, and length of each track indicates extent of unit recording in that track; reconstruction of tetrode tracks in the vlPAG (left) and contralateral MCN (right). (E) Same as (D) but for saline controls (tetrodes implanted on one side in PAG and cannulae implanted bilaterally in MCN for saline infusion; n = 4 rats). In the cerebellar sections, the whole length of each cannula track has been reconstructed, and the blue-coloured tips indicate the extension of the internal cannula for infusions. (F) Same as (E) but cannulae used for infusion of muscimol (n = 4 rats). In the cerebellar tracks, orange-coloured tips indicate the extension of the internal cannulae for infusions. (G) Same as (F) but only cannulae implanted in left and right cerebellum for infusion of muscimol (n = 4 rats).
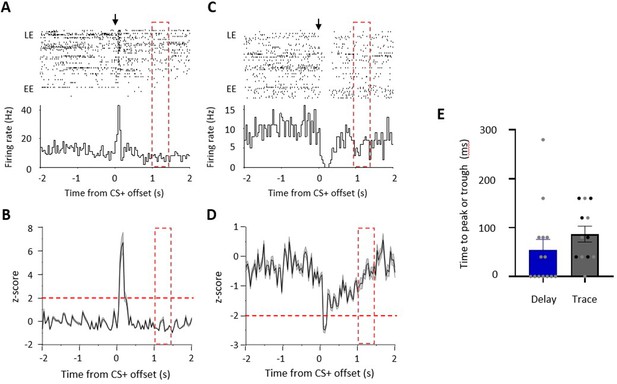
Ventrolateral periaqueductal grey (vlPAG) offset responses during auditory cued fear trace conditioning.
(A) Example type 1 offset unit in vlPAG recorded during extinction following acquisition where a 1 s trace was introduced between conditioned stimulus (CS) offset and the unconditioned stimulus (US) during acquisition. Raster plot and peri event time histogram (PETH) showing activity relative to CS+ offset (arrow in raster and time 0 in PETH). Time of the US during acquisition is shown by red dotted rectangle; 40 ms bins. (B) Same as (A) but mean PETH ± SEM for all type 1 offset units (n = 6). Horizontal dashed red line represents significance level (p<0.05). (C) Same as (A) but example type 4 offset unit. (D) Same as (B) but mean PETH ± SEM for all type 4 offset units (n = 6). (E) Bar chart plotting mean time to peak or trough in activity for all units after CS+ offset (delay, n = 19 units recorded after delay conditioning; trace, n = 12 units recorded after trace conditioning; individual units for trace experiments: light grey type 1 offset units and dark grey type 4 offset units). No significant difference in timing between trace and delay (p>0.05).
-
Figure 1—figure supplement 3—source data 1
Numerical data to support graphs in Figure 1—figure supplement 3.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig1-figsupp3-data1-v2.docx
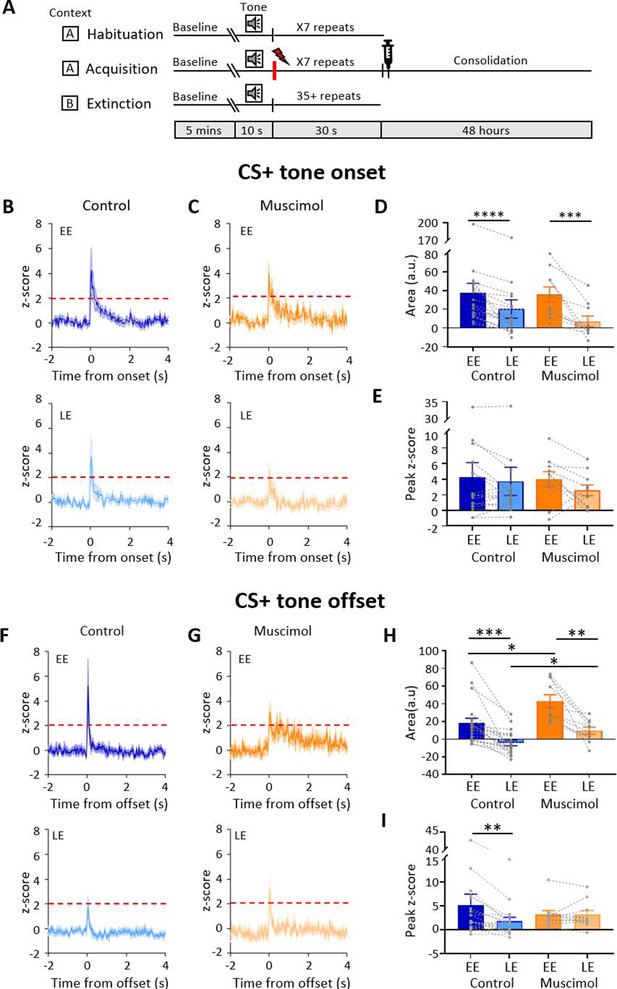
Effect of medial cerebellar nucleus (MCN) inactivation during consolidation on ventrolateral periaqueductal grey (vlPAG) type 1 onset and offset responses during extinction.
(A) Schematic timeline representing the fear conditioning protocol for muscimol animals. (B) Group data for control animals showing average z-scored type 1 onset responses (n = 18 single units from n = 7 animals) during early extinction (EE, upper panel) and late extinction (LE, lower panel); solid lines in each plot show mean z-score, shaded regions ± SEM; horizontal dashed red lines show significance level (p<0.05). Time 0, CS+ tone onset. (C) Same as (B) but grouped data for muscimol animals (n = 10 single units from n = 3 animals). (D) Bar charts showing average type 1 onset response area (arbitrary units) during EE and LE for single units recorded in control versus muscimol animals. Pairs of data points connected with dashed lines show change in mean response area for each single unit over extinction training. Bars show group means ± SEM. Paired t-test, ****p<0.0001, ***p<0.001. (E) Same as (D) but grouped data for peak z-score. (F) Same as (B) but control type 1 offset responses (n = 19 single units from n = 7 animals) during EE (upper panel) and LE (lower panel). Time 0, CS+ tone offset. (G) Same as (F) but grouped data for muscimol animals (n = 10 units from n = 3 animals). (H) Same as (D) but showing average type 1 offset response area (arbitrary units) during EE and LE for single units recorded in control versus muscimol animals. Paired t-test, ***p<0.001, **p<0.01; unpaired t-test, **p<0.01, *p<0.05. (H) Same as (I) but grouped data for peak z-score. Paired t-test, *p<0.05.
-
Figure 2—source data 1
Numerical data to support graphs in Figure 2.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig2-data1-v2.docx
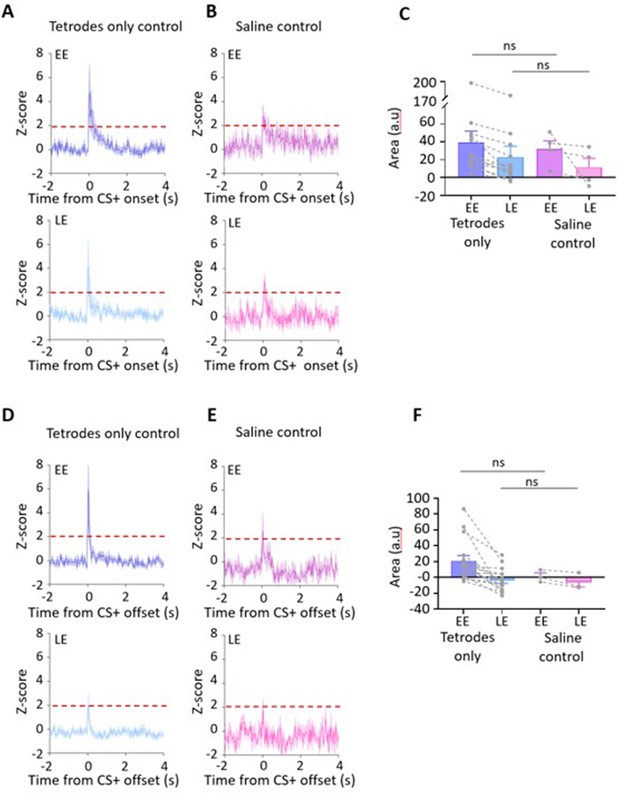
Comparison of single-unit results for tetrodes-only versus saline control animals.
(A) Group data for tetrodes-only control animals (n = 14 units) recorded on day 2, showing average z-scored type 1 onset responses during early extinction (EE, upper panel) and late extinction (LE, lower panel). Time 0, CS+ tone onset. Solid lines in each plot show mean z-score, shaded regions ± SEM. Horizontal dashed red lines show significance level (p<0.05). (B) Same as (A) but grouped data for saline control animals recorded on day 3 (n = 4 units). (C) Bar charts showing average type 1 onset response area (arbitrary units) during EE and LE for single units recorded in tetrodes-only and saline control animals. Pairs of data points connected with dashed lines show change in mean response area for each single unit over extinction training. Bars show group means ± SEM. ns, no significant difference (EE, t(16) = 0.2773, p=0.785; LE, t(16) = 0.4828, p=0.6358, unpaired t-test). (D) and (E) same as (A) and (B) but for type 1 offset responses. Time 0, CS+ tone offset. (D) Tetrodes-only control unit offset responses recorded on day 2 (n = 16 units). (E) Saline control unit offset responses recorded on day 3 (n = 3 units). (F) Same as (C). ns, no significant difference (EE, t(17) = 1.263, p=0.224 and LE, t(17) = 0.2263, p=0.824, unpaired t-test).
-
Figure 2—figure supplement 1—source data 1
Numerical data to support graphs in Figure 2—figure supplement 1.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig2-figsupp1-data1-v2.docx
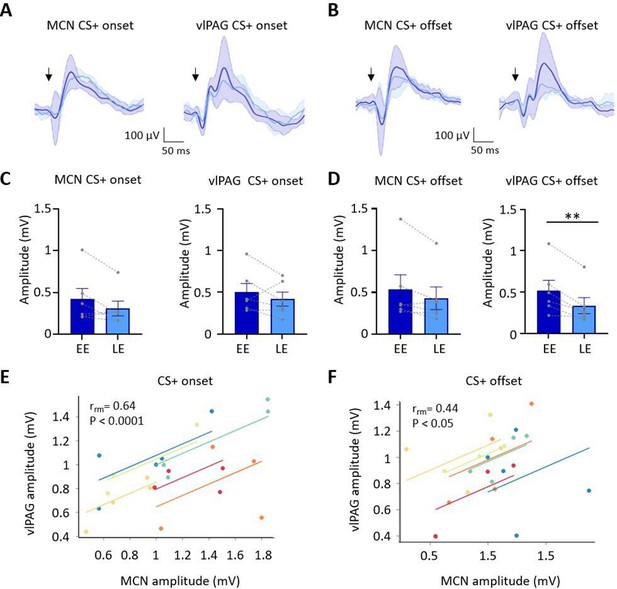
Auditory event-related field potentials (ERPs) recorded simultaneously in the medial cerebellar nucleus (MCN) and ventrolateral periaqueductal grey (vlPAG) during extinction.
(A) Group average ERPs recorded at CS+ onset in the MCN and vlPAG in control animals (n = 6 rats), arrows indicate time of tone onset; each waveform shows mean ± SEM; dark blue, average ERP during early extinction (EE), light blue, average ERP during late extinction (LE). (B) Same as (A) but ERPs recorded simultaneously at CS+ offset (n = 6 rats). (C) Plots showing mean peak-to-peak amplitude of ERPs recorded at CS+ onset in EE versus LE; left panel, MCN; right panel, vlPAG (n = 6 rats, means ± SEM). Pairs of data points connected with dashed lines show the change in mean amplitude over extinction training for individual animals. (D) Same as (C) but for CS+ offset (n = 6 rats, paired t-test, **p<0.01). (E) Repeated-measures correlation (rrm) for ERPs at CS+ onset (n = 6 rats) comparing the amplitude of ERPs recorded simultaneously in MCN and vlPAG. Each colour represents data and line of best fit for an individual animal. (F) Same as (E) but for CS+ offset (n = 6 rats).
-
Figure 3—source data 1
Numerical data to support graphs in Figure 3.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig3-data1-v2.docx
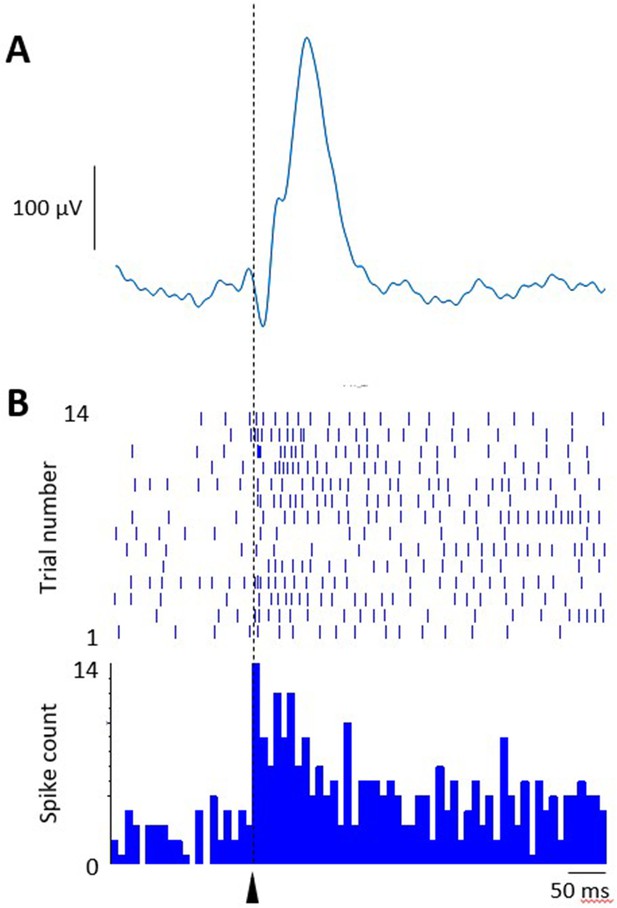
Relationship between unit activity and event-related potentials (ERPs).
(A) Example ERP recorded in the ventrolateral periaqueductal grey (vlPAG) during early extinction training at CS+ offset. Average of 14 trials. (B) Example single-unit data recorded simultaneously from the same vlPAG recording site. Vertical dashed line and arrowhead indicate CS+ offset at time 0. Raster plot 14 trials, peri event time histogram (PETH), 10 ms bin width.
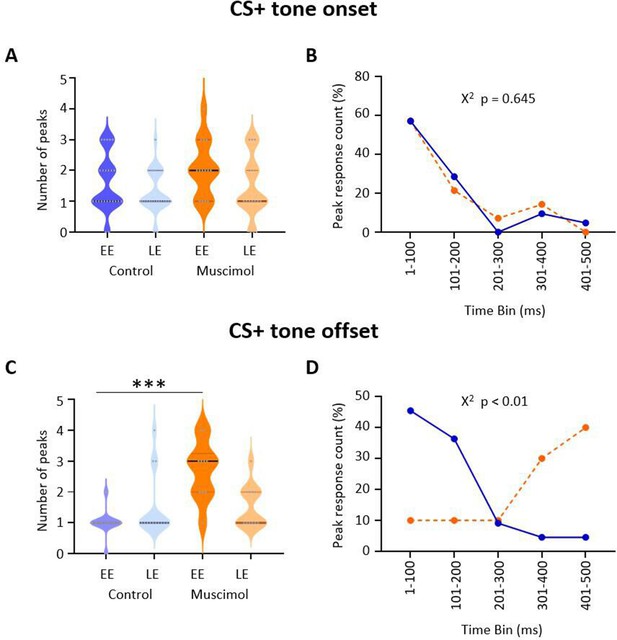
Effect of medial cerebellar nucleus (MCN) inactivation during consolidation on the timing of ventrolateral periaqueductal grey (vlPAG) type 1 onset and offset responses during extinction.
(A) Violin plots showing the number of significant peaks of activity (≥2 SD from baseline) that occur in the initial 500 ms following CS+ onset for early extinction (EE) and late extinction (LE) in control rats (blue, n = 18 units) and muscimol rats (orange, n = 10 units). (B) Proportion of peak responses as a function of time after CS+ onset. For each unit, the 100 ms time bin in which the maximum peak response occurred in the first 500 ms after CS+ onset was expressed as a percentage of total count during EE. Control (blue); muscimol (orange). Chi-squared test, p>0.05. (C) Same as (A) but for CS+ offset. ***p<0.001, unpaired t-test. Control (blue, n = 19); muscimol (orange, n = 10). (D) Same as (B) but for CS+ offset. Chi-squared test, p<0.01.
-
Figure 4—source data 1
Numerical data to support graphs in Figure 4.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig4-data1-v2.docx
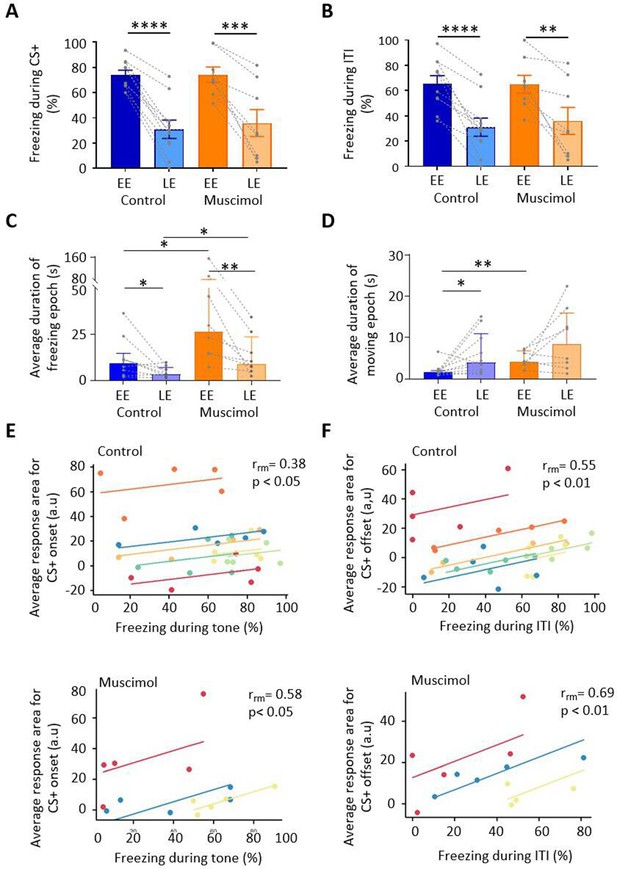
Freezing activity during extinction for control and muscimol animals.
(A) The percentage of total time the conditioned tone (CS+) was presented during early extinction (EE) and late extinction (LE) that freezing epochs occurred in control (n = 10 rats) versus muscimol (n = 8 rats). Pairs of data points connected with dashed lines show the change in mean percentage freezing per animal over extinction training. Bars show group median ± IQR. ****p<0.0001; ***p<0.001. (B) Same as (A) but for inter-tone interval (ITI). (C) Same as (A) but for the average duration of time that animals displayed freezing behaviour after the CS+. Bars show group means ± SEM. Paired t-test. **p<0.01, *p<0.05. (D) Same as (C) but for average duration of moving epochs. (E) Repeated-measures correlation (rrm) for control (upper panel, n = 7 rats) and muscimol (lower panel, n = 3 rats), comparing the integrated response area of units (arbitrary units, a.u.) at CS+ onset as a function of percentage of time freezing during presentation of the CS+. Each colour represents data and line of best fit for an individual animal. (F) Same as (E) but unit activity at CS+ offset in relation to time freezing during ITI.
-
Figure 5—source data 1
Numerical data to support graphs in Figure 5.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig5-data1-v2.docx
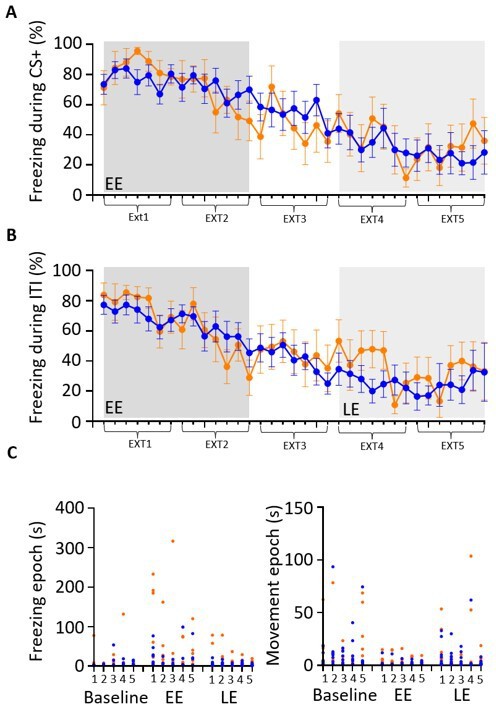
Trial-by-trial comparison of conditioned behaviour in muscimol and control animals.
(A) Grouped data showing the average percentage of time spent freezing during presentation of each CS+ tone as a function of extinction training. Five blocks of extinction (Ext1–5) with early extinction (EE, ext1,2) shaded dark grey and late extinction (LE, ext4,5) shaded light grey. Control data (blue, n = 10 rats) and muscimol data (orange, n = 8 rats). Plots show mean ± SEM. (B) Same as (A) but during the inter-tone interval (ITI). (C) Scatterplots for baseline, EE, and LE showing the duration of the first five freezing epochs (left panel) and first five movement epochs (right panel) for each block of extinction training (1–5, see Materials and methods for further details). Each data point represents the freezing or movement epoch for an individual animal. Control,blue; muscimol, orange.
-
Figure 5—figure supplement 1—source data 1
Numerical data to support graphs in Figure 5—figure supplement 1.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig5-figsupp1-data1-v2.docx
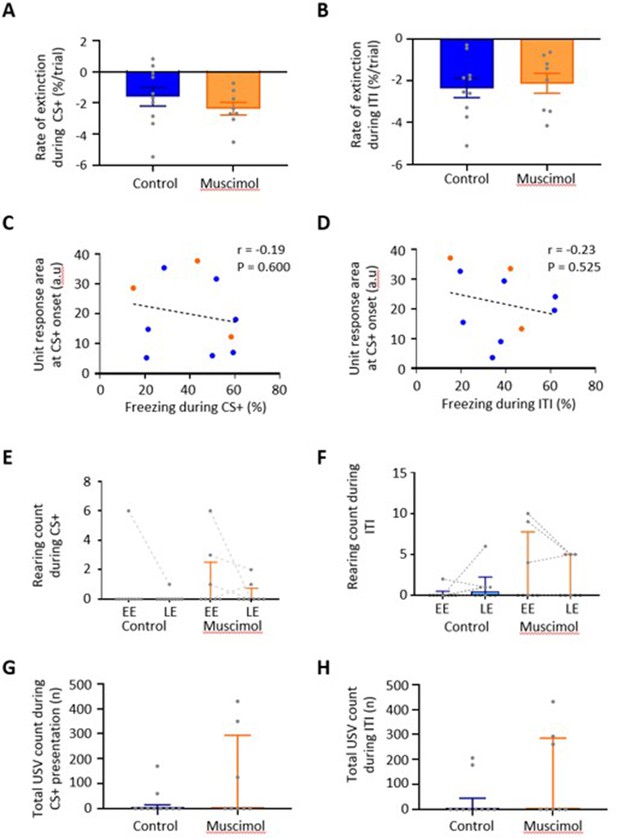
Conditioned fear-related behaviours.
(A) Rate of extinction as measured by the slope of the line of best fit for change in percentage of time spent freezing during presentation of the CS+ over the first 21 presentations of the CS+ during extinction training in control (n = 10 rats) versus muscimol (n = 8 rats) experiments. Individual data points show mean rate of change per animal. Bars show group means ± SEM. (B) Same as (A) but the change in percentage of time spent freezing during the inter-tone interval (ITI) for the first 21 CS+ presentations during extinction training in control (n = 10 rats) versus muscimol (n = 8 rats) experiments. (C) Scatterplot showing the mean unit response area (arbitrary units) at CS+ onset as a function of percentage time spent freezing activity in early extinction (EE) and late extinction (LE) during CS+ presentation. Individual data points represent individual animals. Blue, control; orange, muscimol. Pearson correlation r = –0.19, p>0.05. (D) Same as (C) but for freezing during the ITI and unit response area at CS+ offset. Pearson correlation r = –0.23, p>0.05. (E) The total number of rears during CS+ presentation during extinction training in control (n = 10 rats) versus muscimol (n = 8 rats) experiments. Individual data points connected with dashed lines show change in mean number of rears per animal over extinction training. Bars show group median and IQR (EE, U = 30, p=0.275; LE, U = 33.5, p=0.444, Mann–Whitney test). (F) Same as (E) but rearing count for ITI in control (n = 10 rats) versus muscimol (n = 8 rats) experiments (EE, U = 33, p=0.490; LE, U = 36.5, p=0.7629, Mann–Whitney test). (G) Total number of ultrasonic vocalisations (USVs) during presentation of the CS+ in all extinction training trials for control (n = 10 rats) versus muscimol (n = 8 rats) experiments. Individual data points show mean count per animal. Bars show group median and IQR (U = 31, p=0.3725, Mann–Whitney test). (H) Same as (G) but USV count in the ITI for control (n = 10 rats) versus muscimol (n = 8 rats) experiments (U = 30, p=0.3039, Mann–Whitney test).
-
Figure 5—figure supplement 2—source data 1
Numerical data to support graphs in Figure 5—figure supplement 2.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig5-figsupp2-data1-v2.docx
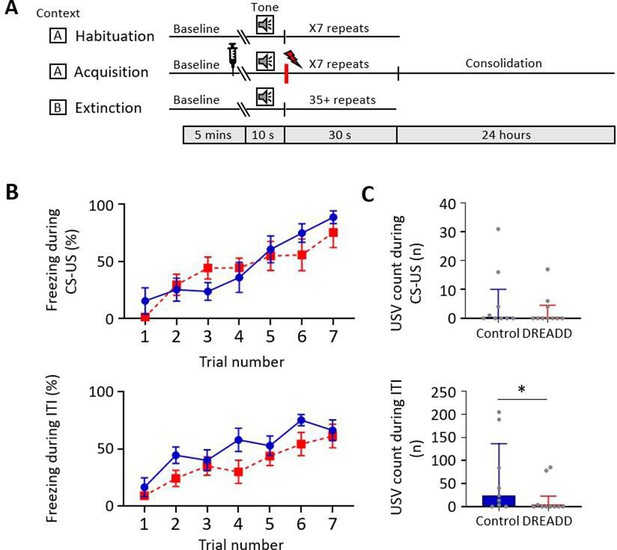
Effect of medial cerebellar nucleus-periaqueductal grey (MCN-PAG) pathway modulation on behaviour during acquisition.
(A) Schematic timeline representing the fear conditioning protocol for the DREADDs experiment. (B) The effect of clozapine N-oxide (CNO) delivery into the ventrolateral PAG (vlPAG) during acquisition on freezing behaviour measured during presentation of the paired conditioned stimulus-unconditioned stimulus (CS-US) (upper plot), and the inter-tone interval (ITI) (lower plot). Blue plot, control animals (n = 9 rats); red plot, DREADD (hM4D(Gi)) animals, n = 10 rats; data points mean ± SEM. (C) The number of ultrasonic vocalisations (USVs) recorded in control versus DREADD animals during CS-US presentation (upper panel) and during the ITI (lower panel); bars show median ± IQR. Mann–Whitney, one-tailed test, *p<0.05.
-
Figure 6—source data 1
Numerical data to support graphs in Figure 6.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig6-data1-v2.docx
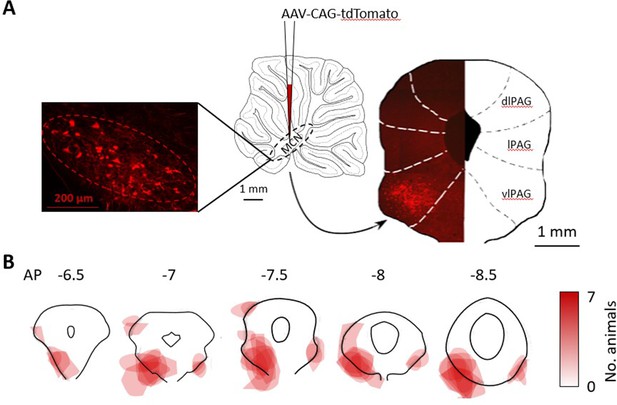
Anatomical mapping of the medial cerebellar nucleus-periaqueductal grey (MCN-PAG) pathway.
(A) Schematic showing a sagittal section of the cerebellum injection into the MCN of an anterograde tracer (AAV-CAG-tdTomato viral vector). To the left, high-power photomicrograph showing transfected neurons in MCN. To the right, coronal section shows representative pattern of terminal labelling in the contralateral PAG in one animal (dlPAG, lPAG, vlPAG; dorsolateral, lateral, and ventrolateral PAG). (B) Coronal sections of the PAG at different anteroposterior (AP) levels showing the pattern of terminal labelling in all available animals (n = 7 rats). The darker the red shading, the larger the number of animals with terminal labelling in that area.
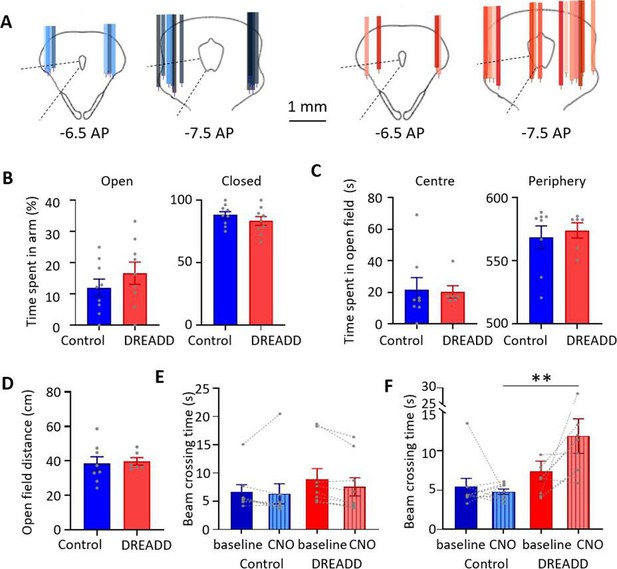
Effect of DREADDs on general motor and affective behaviour.
(A) Histological reconstruction of cannulae placement in periaqueductal grey (PAG) in control (n = 9 rats, tracks colour coded blue to black for different animals) and DREADD animals (n = 10 rats, tracks colour coded orange to red for different animals). For each track, the tips indicate the extension of the internal cannula for infusions. (B) Percentage of the total time in an elevated plus maze spent in the open arm (left) and in the closed arm (right) for control (n = 9 rats) versus DREADD (n = 9 rats) experiments. Individual data points show percentage of time per animal. Bars show mean ± SEM (open arm, t(16) = 1.036, p=0.316; closed arm, t(16) = 1.036, p=0.316; unpaired t-test). (C) Time spent in the centre versus the periphery of the open field arena for control versus DREADD animals. Individual data points show total time per animal. Bars show mean ± SEM (centre, t(12) = 0.145, p=0.887; periphery, t(12) = 0.463, p=0.651, unpaired t-test). (D) The total distance moved in the open field arena in control (n = 8 rats) versus DREADD (n = 6 rats) experiments. Individual data points show total distance per animal. Bars show mean ± SEM (t(12) = 0.267, p=0.794, unpaired t-test). (E) Beam walking traversal times in control (n = 9 rats) and DREADD (n = 9 rats) experiments before (baseline) and after infusion of clozapine N-oxide (CNO) into ventrolateral PAG (vlPAG) for the two groups of animals. Pairs of data points connected by dotted lines show change in mean crossing time per animal between baseline and CNO. Bars show mean ± SEM (time to traverse, t(16) = 0.508, p=0.618, unpaired t-test). (F) Same as (E) but beam traversal times in control (n = 9 rats) and DREADD (n = 9 rats) experiments before (baseline) and intraperitoneal injection of CNO. Unpaired t-test **p<0.001.
-
Figure 6—figure supplement 2—source data 1
Numerical data to support graphs in Figure 6—figure supplement 2.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig6-figsupp2-data1-v2.docx
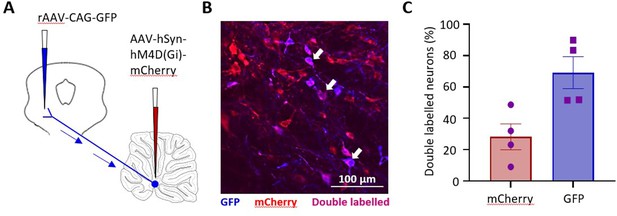
Anatomical evaluation of DREADD transfection of medial cerebellar nucleus-periaqueductal grey (MCN-PAG) pathway.
(A) Schematic showing experimental arrangement for anatomical assessment of DREADD transfection. In each animal, a green fluorescent retrograde tracer (rAAV-CAG-GFP) was injected into the ventrolateral sector of PAG (shown as false colour blue) and in the same animals MCN was transfected with inhibitory DREADD virus that expressed red fluorescence (AAV-hSyn-hM4D(Gi)-mCherry). (B) Photomicrograph showing examples of single- (red or blue) and double-labelled neurons (purple, arrows) in MCN. (C) Graph plotting double-labelled neurons in MCN as a percentage of the two single-labelled populations (n = 4 rats). Individual data points per animal. Mean ± SEM.
-
Figure 6—figure supplement 3—source data 1
Numerical data to support graphs in Figure 6—figure supplement 3.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig6-figsupp3-data1-v2.docx
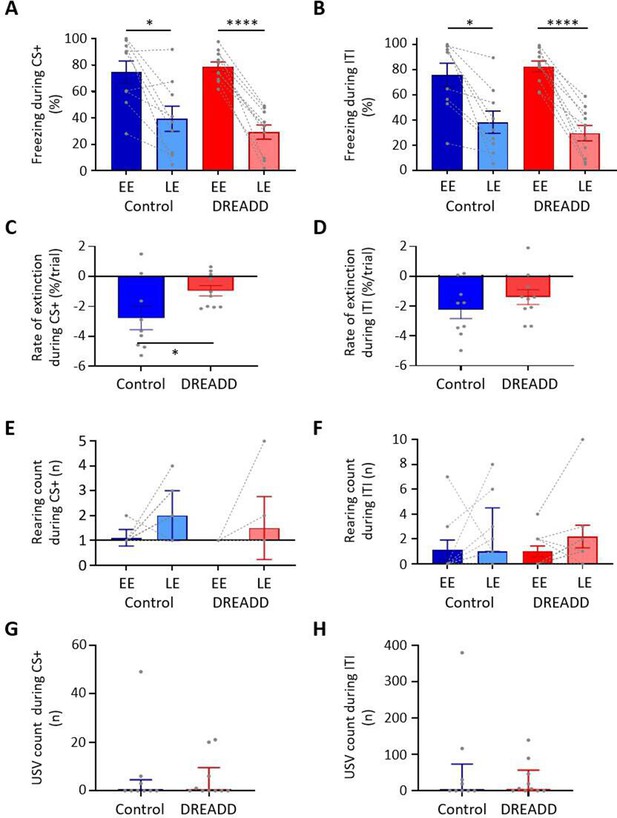
Effect of medial cerebellar nucleus-periaqueductal grey (MCN-PAG) pathway modulation on behaviour during extinction.
(A) The percentage of total time the CS+ was presented that animals displayed freezing behaviour during early extinction (EE) (trials 1–14) or late extinction (LE) (trials 21–35) in control (n = 9 rats) versus DREADD (hM4D(Gi), n = 10 rats). Pairs of data points connected with dashed lines show the change in mean percentage freezing per animal over extinction training. Bars show group means ± SEM. ****p<0.0001, *p<0.05. (B) Same as (A) but for inter-tone interval (ITI). (C) Rate of extinction during presentation of the CS+ as measured by the change in percentage freezing over the first 21 CS+ presentations during extinction training in control (n = 9 rats) versus DREADD experiments (n = 10 rats). Individual data points show mean rate of change per animal. Bars show group means ± SEM. Unpaired t-test *p<0.05. (D) Same as (C) but for ITI. (E) The total number of rears during presentation of the CS+ during EE and LE for control (n = 9 rats) versus DREADD experiments (n = 10 rats). Pairs of data points connected with dashed lines show change in mean number of rears per animal over extinction training. Bars show group median and IQR. (F) Same as (E) but for ITI. (G) Total number of ultrasonic vocalisations (USVs) during presentation of the CS+ in extinction training in control (n = 9 rats) versus DREADD experiments (n = 10 rats). Individual data points show mean count per animal. Bars show group median and IQR. (H) Same as (G) but total number of USVs recorded during ITI.
-
Figure 7—source data 1
Numerical data to support graphs in Figure 7.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig7-data1-v2.docx
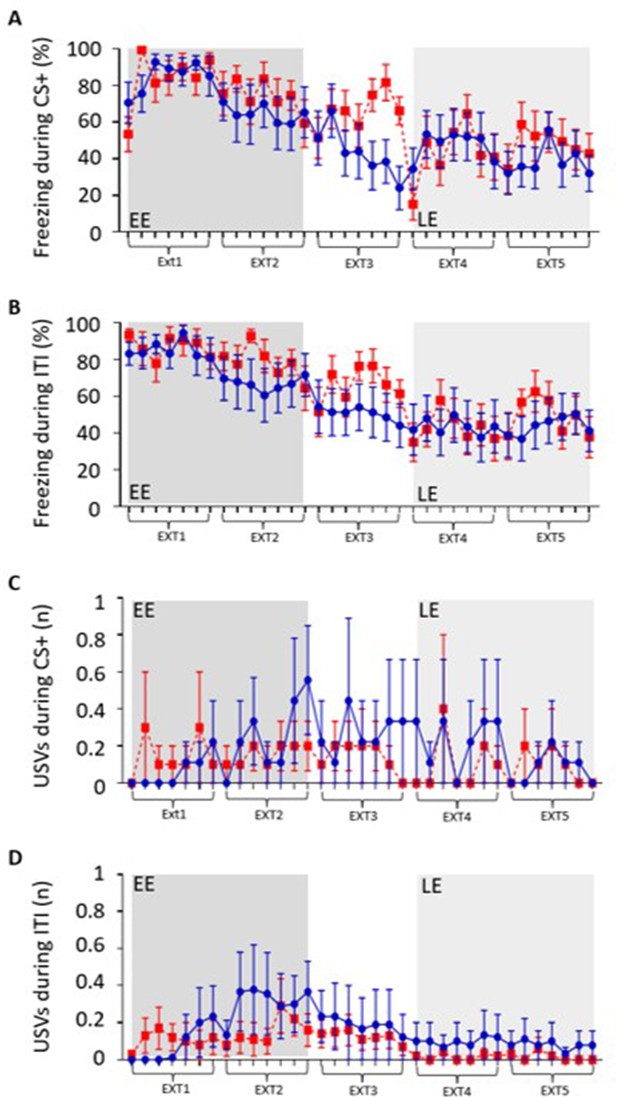
Freezing and ultrasonic vocalisation (USV) behaviour across extinction training.
(A) Grouped data showing the average percentage of time spent freezing during presentation of each CS+ tone as a function of extinction training. Each data point based on average of seven CS+ presentations per animal ± SEM. Five blocks of extinction (Ext1–5) with early extinction (EE, ext1,2) shaded dark grey and late extinction (LE, ext4,5) shaded light grey. Control data (blue, n = 9 rats) and DREADD data (red, n = 10 rats). (B) Same as (A) but during the inter-tone interval (ITI). (C) Same as (A) but grouped data showing average count of USVs emitted during each CS+ presentation as a function of extinction training. Control (blue, n = 9 rats) and DREADD (red, n = 10 rats). (D) Same as (C) but average USV count during the ITI. Mean ± SEM.
-
Figure 7—figure supplement 1—source data 1
Numerical data to support graphs in Figure 7—figure supplement 1.
- https://cdn.elifesciences.org/articles/76278/elife-76278-fig7-figsupp1-data1-v2.docx
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (species) | Sprague–Dawley (rat) male | Envigo | RRID:RGD_737903 | |
| Transfected construct | pAAV-CAG-tdTomato | Addgene, USA | RRID:Addgene_59462 | Adeno-associated viral vector (AAV1) |
| Transfected construct | rAVV-CAG-GFP | Addgene, USA | RRID:Addgene_37825 | Adeno-associated viral vector (AAVrg) |
| Transfected construct | pAAV-hSyn-hM4D(Gi)-mCherry | Addgene, USA | RRID:Addgene_50475 | Adeno-associated viral vector (AAV5) |
| Transfected construct | pAAV-hSyn-EGFP | Addgene, USA | RRID:Addgene_50465 | Adeno-associated viral vector (AAV5) |
| Chemical compound, drug | Muscimol | Sigma-Aldrich | M1523 | |
| Chemical compound, drug | Clozepine-N-oxide | Tocris Bioscience, UK | 4963 | |
| Antibody | Anti-mCherry (rabbit polyclonal) | BioVision | 5993 | (1:2000) |
| Antibody | Alexa Fluor 594 | Molecular Probes | (1:1000) | |
| Other | CerePlex µ Headstage | Blackrock Microsystems, UT | PN-9716 | |
| Software, algorithm | Blackrock Central Software Suite | Blackrock Microsystems, UT | ||
| Software, algorithm | OBS | Open Broadcaster Software; 2012–2020 | ||
| Software, algorithm | Solomon Coder | András Péter, 2019 | ||
| Software, algorithm | DeepLabCut | Wei and Kording, 2018 | ||
| Software, algorithm | MATLAB | MathWorks | RRID:SCR_001622 | |
| Software, algorithm | Spike7 | Cambridge Electronic Design Limited | ||
| Software, algorithm | NeuroExplorer | Plexon | ||
| Software, algorithm | RStudio | RStudio, USA | ||
| Software, algorithm | Prism 9 | GraphPad, USA |