Ecology, more than antibiotics consumption, is the major predictor for the global distribution of aminoglycoside-modifying enzymes
Figures
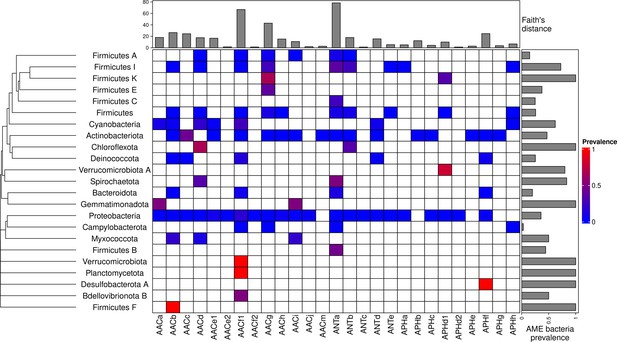
Prevalence of AME-gene-carrying bacteria across the phylogeny of Eubacteria.
The phylogenetic tree corresponds to an aggregation of bac120 phylogeny (Parks et al., 2018) to the phylum level. In the heatmap, blank boxes correspond to the observed absence of a CHG in a phylum. For the other colors, blue to red boxes stand for CHG frequencies from near-zero to one. Gray bars in the top part correspond to the Faith’s distance (the sum of the lengths of all the branches on the bac120 tree) for the species in which each CHG was found. Gray bars on the right correspond to the prevalence of AME bacteria for each phylum, i.e. the proportion of genomes in which at least one AME gene was found.
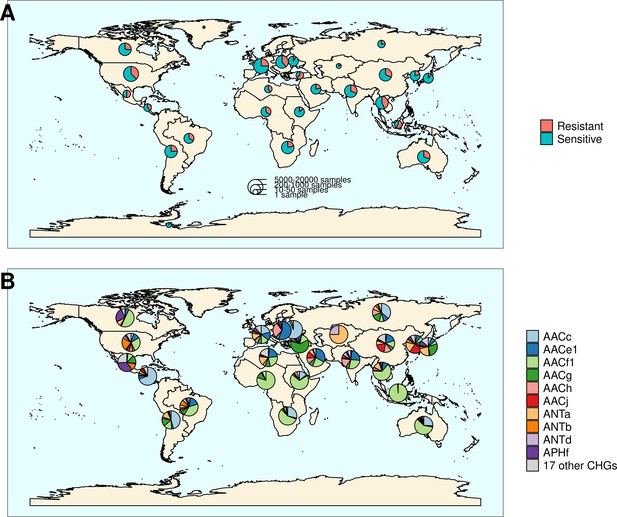
Distribution of aminoglycoside-resistant bacteria and AME-encoding genes over the world.
(A) Distribution of sampled aminoglycoside-resistant bacteria in the world. The frequencies of resistant and sensitive bacteria are displayed in red and blue, and the size of pies represents the number of genomes sampled in a given region. (B) Distribution of sampled AME genes in the world. Pie size is irrelevant to the number of samples.
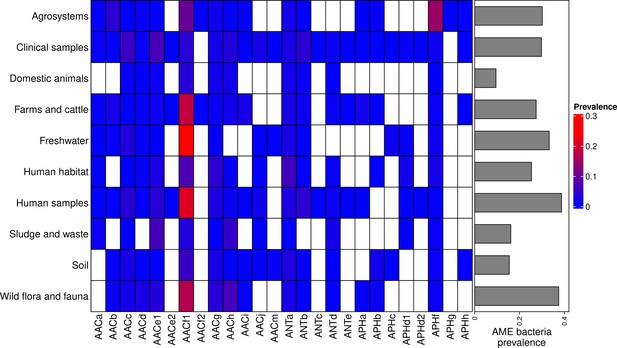
Prevalence of AME-gene-carrying bacteria across land biomes.
In the heatmap, blank boxes correspond to the observed absence of a CHG. For the other colors, blue to red boxes stand for CHG frequencies from near-zero to 0.3. Gray bars on the right correspond to the prevalence of AME bacteria for each biome, i.e. the prevalence of genomes in which at least one AME gene was found. APHe is excluded from the heatmap because it was sampled in seawater only.
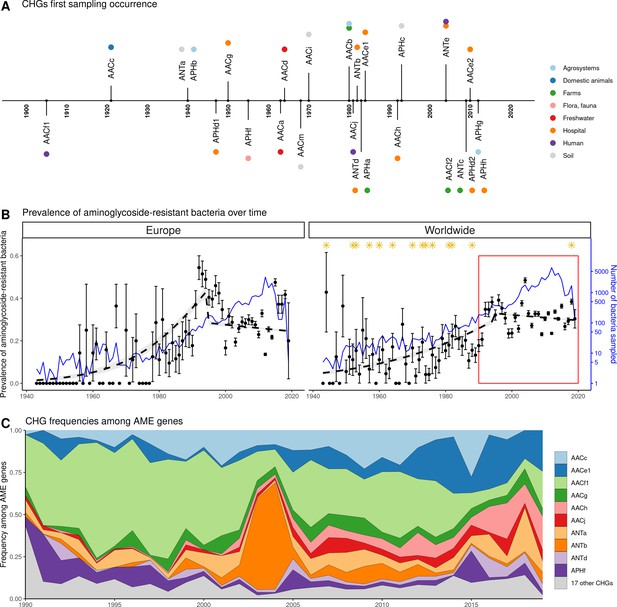
Global time trends for aminoglycoside resistance.
(A) First sampling occurrence for each CHG in the analyzed dataset. First sampling occurrence of APHe is unknown. (B) Evolution of the worldwide and European prevalences of AME bacteria between 1943 and 2019. The dots and error bars represent the measured prevalence (± standard error) of AME bacteria each year. The dotted line represents a binomial regression of the prevalence of AME bacteria over time, fitted separately before 1995 and after 1995. The blue curve represents the number of bacteria genomes sampled each year (in logarithmic scale). Orange stars represent the dates at which the following aminoglycoside drugs were released for commercial use: streptomycin, neomycin, hygromycin, kanamycin, paromomycin, capreomycin, gentamicin, ribostamycin, dibekacin, tobramycin, amikacin, sisomicin, netilmicin, micronomicin, isepamycin, plazomicin. The red rectangle displays the period of time analyzed in panel: (C) Evolution of worldwide CHG frequencies among sampled AME genes between 1990 and 2019. 17 CHGs for which less than 400 sequences were sampled are grouped and displayed in gray.
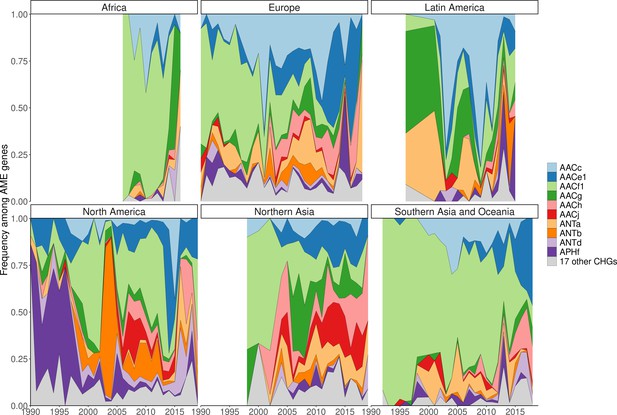
Evolution of continental CHG frequencies among sampled AME genes between 1990 and 2019.
IMAGE24 regions were gathered to higher continental ensembles in order to obtain more representative frequencies. Western and Central Europe and ‘Ukraine’ were gathered to Europe; Canada and the USA to North America; the rest of the Americas to Latin America; all African landmasses to Africa; Oceania, Southeastern Asia, ‘Indonesia’, and ‘India’ to Southern Asia and Oceania; Korea, Japan, Middle East, Turkey, ‘China’, ‘Asia-Stan’, and ‘Russia’ to Northern Asia. CHG frequencies were only computed when at least 10 AME-gene-carrying bacteria had been sampled in a continent in a year.
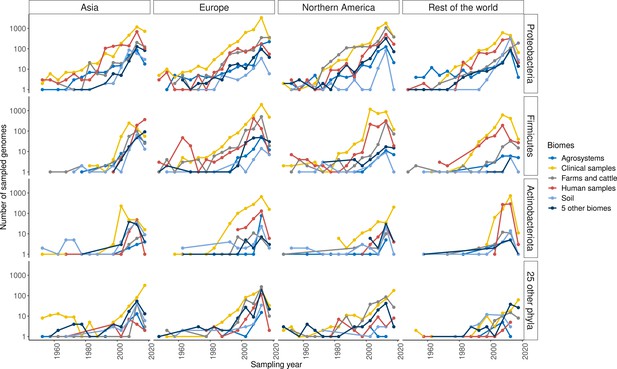
Distribution of sampling over time, space, ecology, and phylogeny.
Each point represents a 5-year period between 1945 and 2019. Rows display different phyla, columns different continents, and colors different biomes. IMAGE24 regions were gathered to higher geographical entities for simplification. Only the biomes, phyla, and geographical regions with the most sampled genomes are displayed, the others being gathered to single categories.
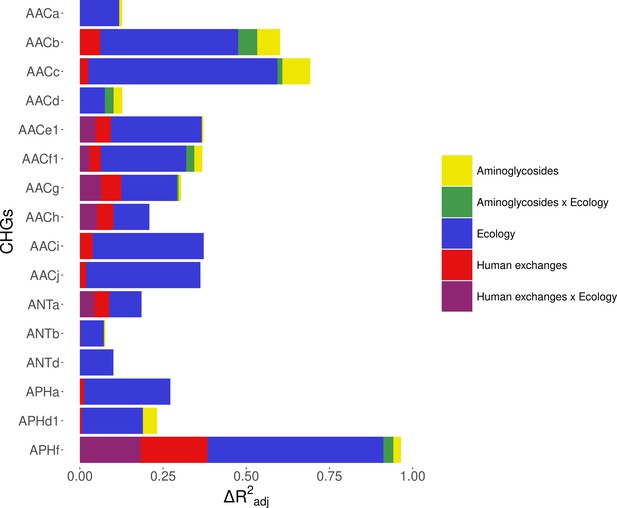
Relative importance of several factors to explain the distribution of aminoglycoside-resistant bacteria in Europe between 1997 and 2018.
Logistic regressions with a spatial Matérn correlation structure were computed to explain the frequency of 16 CHGs. This figure represents the contribution of each variable in each selected model, as the fraction of adjusted McFadden’s pseudo-R2 explained by adding this variable.
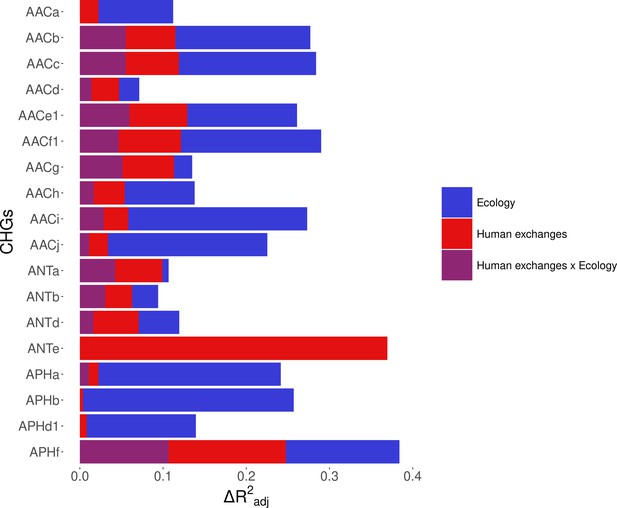
Relative importance of several factors to explain the distribution of aminoglycoside-resistant bacteria in the world between 1997 and 2018.
Logistic regression with a spatial Matérn correlation structure were computed to explain the frequency of 18 CHGs in samples. This figure represents the contribution of each variable in each selected model, as the fraction of adjusted McFadden’s pseudo-R2 explained by adding this variable.
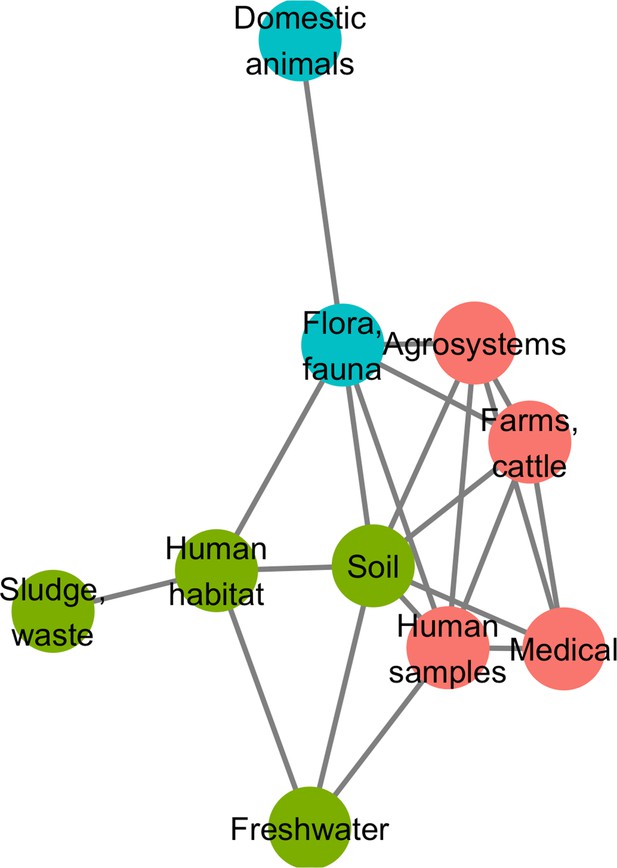
Minimum spanning network based on shared antibiotic resistance genes between biomes.
Resistomes are represented here as vertices. Edges correspond to particularly high resemblance between two resistomes. Three modules in the network were established by the Louvain clustering algorithm: vertices are colored based on the module to which they belong.
Tables
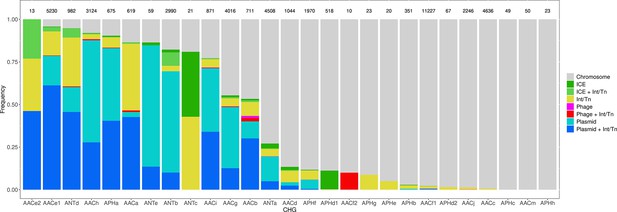
List of CHGs by biochemical function.
| Cluster | Biochemical function | Number of genes identified |
|---|---|---|
| AACa | N-acetyltransferases | 619 |
| AACb | N-acetyltransferases | 711 |
| AACc | N-acetyltransferases | 4636 |
| AACd | N-acetyltransferases | 1044 |
| AACe1 | N-acetyltransferases | 5230 |
| AACe2 | N-acetyltransferases | 13 |
| AACf1 | N-acetyltransferases | 11227 |
| AACf3 | N-acetyltransferases | 10 |
| AACg | N-acetyltransferases | 4016 |
| AACh | N-acetyltransferases | 3124 |
| AACi | N-acetyltransferases | 871 |
| AACj | N-acetyltransferases | 2246 |
| AACm | N-acetyltransferases | 50 |
| ANTa | nucleotidyltransferases | 4508 |
| ANTb | nucleotidyltransferases | 2990 |
| ANTc | nucleotidyltransferases | 21 |
| ANTd | nucleotidyltransferases | 982 |
| ANTe | nucleotidyltransferases | 59 |
| APHa | phosphotransferases | 675 |
| APHb | phosphotransferases | 351 |
| APHc | phosphotransferases | 49 |
| APHd1 | phosphotransferases | 518 |
| APHd2 | phosphotransferases | 67 |
| APHe | phosphotransferases | 20 |
| APHf | phosphotransferases | 1970 |
| APHg | phosphotransferases | 23 |
| APHh | phosphotransferases | 23 |
Additional files
-
Supplementary file 1
Description of the models selected to explain the distribution of aminoglycoside resistant bacteria in Europe between 1997 and 2018.
- https://cdn.elifesciences.org/articles/77015/elife-77015-supp1-v1.doc
-
Supplementary file 2
Description of the models selected to explain the distribution of aminoglycoside resistant bacteria in the world between 1997 and 2018.
- https://cdn.elifesciences.org/articles/77015/elife-77015-supp2-v1.doc
-
Supplementary file 3
Description of the models selected to explain the distribution of aminoglycoside resistant bacteria in Europe between 1997 and 2018 under low or high sampling.
- https://cdn.elifesciences.org/articles/77015/elife-77015-supp3-v1.doc
-
Supplementary file 4
Description of the models selected to explain the distribution of aminoglycoside resistant bacteria in Europe between 1997 and 2018, in biomes where antibiotics are prescribed.
- https://cdn.elifesciences.org/articles/77015/elife-77015-supp4-v1.doc
-
Supplementary file 5
Human exchanges variables influencing the prevalence of aminoglycoside resistant bacteria.
- https://cdn.elifesciences.org/articles/77015/elife-77015-supp5-v1.doc
-
Supplementary file 6
Classification of biomes according to the keywords of the NCBI BioSamples database.
- https://cdn.elifesciences.org/articles/77015/elife-77015-supp6-v1.doc
-
Supplementary file 7
Impact of the association of AME genes with mobile genetic elements with their duplication risk.
- https://cdn.elifesciences.org/articles/77015/elife-77015-supp7-v1.doc
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/77015/elife-77015-transrepform1-v1.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/77015/elife-77015-mdarchecklist1-v1.pdf
-
Source data 1
List of genomes that were screened for aminoglycoside resistance.
- https://cdn.elifesciences.org/articles/77015/elife-77015-data1-v1.csv
-
Source data 2
List of AME genes that were found by screening.
- https://cdn.elifesciences.org/articles/77015/elife-77015-data2-v1.csv
-
Source data 3
Distribution of screened AME genes over time, geography, and ecology.
- https://cdn.elifesciences.org/articles/77015/elife-77015-data3-v1.csv
-
Source data 4
AME sequences used to define clusters of homologous genes.
- https://cdn.elifesciences.org/articles/77015/elife-77015-data4-v1.csv