The role of anterior insular cortex inputs to dorsolateral striatum in binge alcohol drinking
Figures
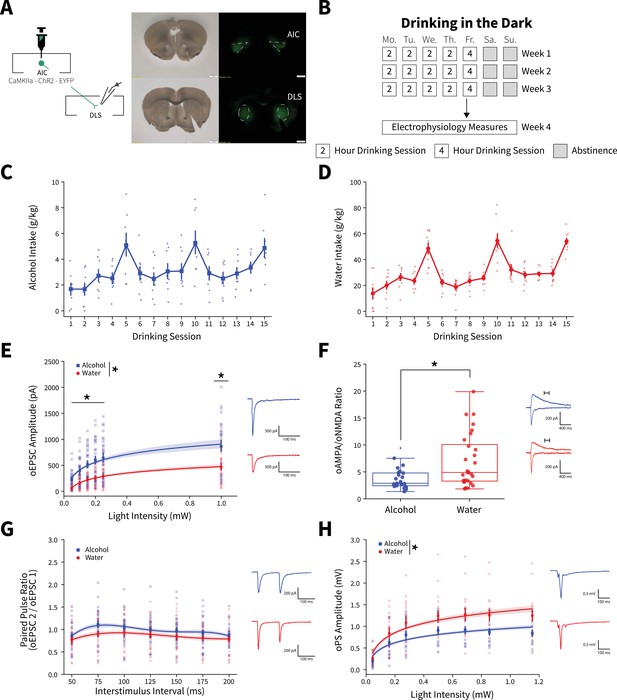
Binge alcohol consumption alters synaptic plasticity of anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS).
(A) Representation of viral strategy and expression to record AIC input responses within the DLS. (B) Schematic of Drinking in the Dark (DID) protocol for electrophysiology experiments. (C) Group and individual animals’ alcohol consumption (n=8 animals). (D) Group and individual animals’ water consumption (n=10 animals). (E) Binge alcohol consumption increased optically evoked postsynaptic current (oEPSC) amplitude of AIC inputs within the DLS (two-way mixed analysis of variance (ANOVA), Fluid F(1,50) = 15.6084, p=0.0002; Fluid × Light Intensity F(5,250) = 6.0674, p=0.000025; alcohol: 26 recordings, n=5 animals; water: 26 recordings, n=6 animals). (F) Binge alcohol consumption reduced optically evoked AMPA (oAMPA) to NMDA (oNMDA) glutamate receptor current ratios (Mann-Whitney, U=189, p=0.0072; alcohol: 24 recordings, n=5 animals; water: 28 recordings, n=6 animals). (G) There was no main effect of binge alcohol consumption on paired-pulse ratios of oEPSCs (oPPR) compared to water consumption (two-way mixed ANOVA, Fluid F(1,55) = 2.5544, p=0.1157; alcohol: 30 recordings, n=6 animals; water: 27 recordings, n=5 animals). (H) Binge drinking alcohol decreased optically evoked population spike (oPS) amplitudes produced by photoexciting AIC inputs within the DLS (two-way mixed ANOVA, Fluid F(1,28) = 4.3484, p=0.0463; Fluid × Light Intensity F(6,168) = 2.9574, p=0.0090; alcohol: 14 recordings, n=4 animals; water: 16 recordings, n=6 animals). Error bars and shading indicate ± SEM. Box plot whiskers represent interquartile range.
-
Figure 1—source data 1
Binge alcohol consumption alters synaptic plasticity of anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS).
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig1-data1-v1.zip
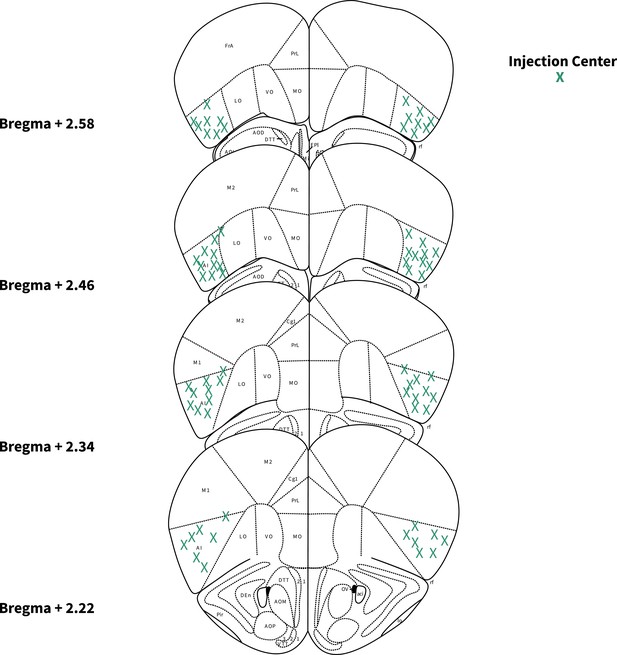
Locations of anterior insular cortex (AIC) injections for dorsolateral striatum (DLS) electrophysiology.
Bilateral injection centers of AAV-ChR2-EYFP in the AIC as verified by histology (male: alcohol: n=13 animals; water: n=15 animals; female: alcohol: n=4 animals; water: n=4 animals).
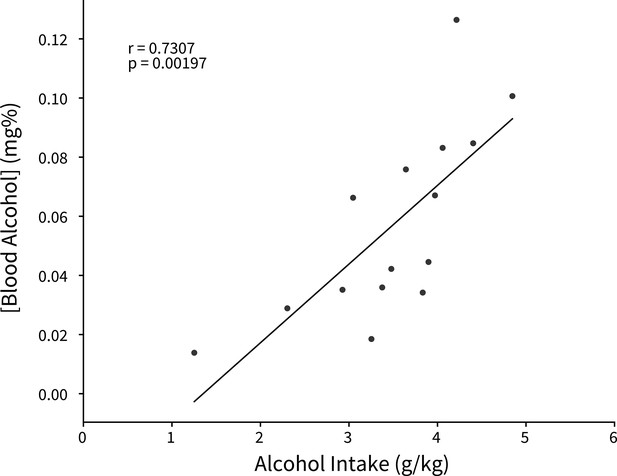
Binge alcohol consumption is positively correlated with blood alcohol concentration.
Alcohol intake was measured from a 2 hr Drinking in the Dark (DID) session and retro-orbital blood samples were collected within 30 min of the completion of the session to calculate blood alcohol concentration (Pearson’s correlation, r=0.7307, p=0.00197).
-
Figure 1—figure supplement 2—source data 1
Binge alcohol consumption is positively correlated with blood alcohol concentration.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig1-figsupp2-data1-v1.zip
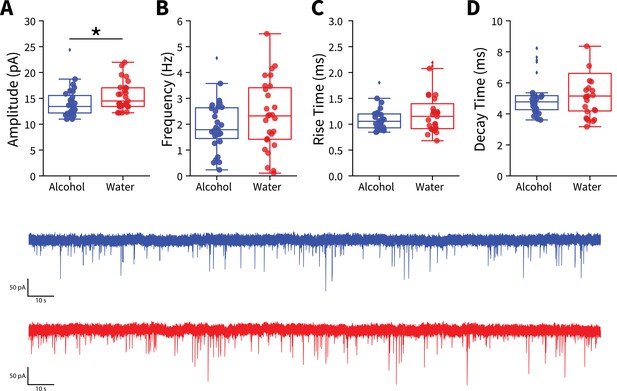
Binge alcohol consumption alters amplitudes of spontaneous excitatory synaptic activity in the dorsolateral striatum (DLS).
Spontaneous excitatory postsynaptic currents (sEPSCs) were measured in DLS medium spiny neurons from mice after 3 weeks of binge alcohol or water consumption. (A) Binge alcohol consumption decreased sEPSC amplitudes compared to water drinkers (Mann-Whitney, U=247.5, p=0.0441; alcohol: 27 recordings, n=6 animals; water: 27 recordings, n=5 animals). sEPSC (B) frequency (t test, t(52) = 1.2846, p=0.2046), (C) rise time (Mann-Whitney, U=318, p=0.4292), or (D) decay time (Mann-Whitney, U=310.5, p=0.3621) between alcohol and water exposed animals (alcohol: 27 recordings, n=6 animals; water: 27 recordings, n=5 animals).
-
Figure 1—figure supplement 3—source data 1
Binge alcohol consumption alters amplitudes of spontaneous excitatory synaptic activity in the dorsolateral striatum (DLS).
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig1-figsupp3-data1-v1.zip
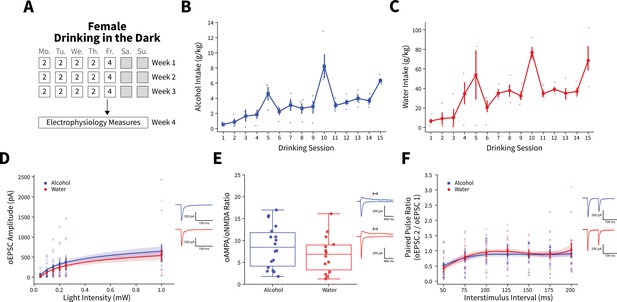
Female binge alcohol consumption does not alter synaptic plasticity of anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS).
(A) Schematic of Drinking in the Dark (DID) protocol for electrophysiology experiments. (B) Group and individual animals’ alcohol consumption (n=4 animals). (C) Group and individual animals’ water consumption (n=4 animals). (D) Female binge alcohol consumption did not alter optically evoked postsynaptic current (oEPSC) amplitude of AIC inputs within the DLS (two-way mixed analysis of variance [ANOVA], Fluid F(1,25) = 0.5136, p=0.4802; alcohol: 16 recordings, n=4 animals; water: 11 recordings, n=4 animals). (E) Female binge alcohol consumption did not alter optically evoked AMPA (oAMPA)/optically evoked NMDA (oNMDA) glutamate current ratios (t test, t(29.425) = 1.089, p=0.285; alcohol: 18 recordings, n=4 animals; water: 14 recordings, n=4 animals). (F) There was no effect of binge alcohol consumption on paired-pulse ratios of oEPSCs (oPPR) (two-way mixed ANOVA, Fluid F(1,27) = 0.0870, p=0.7703; alcohol: 17 recordings, n=4 animals; water: 12 recordings, n=4 animals). Error bars and shading indicate ± SEM. Box plot whiskers represent interquartile range.
-
Figure 1—figure supplement 4—source data 1
Female binge alcohol consumption does not alter synaptic plasticity of anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS).
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig1-figsupp4-data1-v1.zip
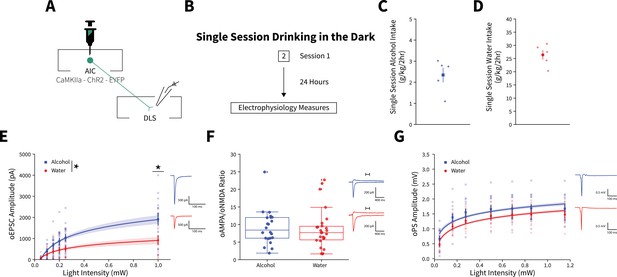
A single binge alcohol session alters optically evoked glutamate currents, but does not change the AMPA/NMDA current ratio or alter dorsolateral striatum (DLS) network effects.
(A) Representation of viral strategy and expression to record anterior insular cortex (AIC) input responses within the DLS. (B) Schematic of Drinking in the Dark (DID) protocol for electrophysiology experiments. (C) Group and individual animals’ alcohol consumption (n=5 animals). (D) Group and individual animals’ water consumption (n=5 animals). (E) A single alcohol DID session increased optically evoked postsynaptic current (oEPSC) amplitude of AIC inputs within the DLS compared to water controls (two-way mixed analysis of variance [ANOVA], Fluid F(1,27) = 5.6876, p=0.0244; Fluid × Light Intensity F(5,135) = 8.2821, p=0.000001; alcohol: 15 recordings, n=5 animals; water: 14 recordings, n=5 animals). A single DID alcohol session did not alter (F) optically evoked NMDA (oNMDA)/optically evoked AMPA (oAMPA) ratios (Mann-Whitney, U=324.0, p=0.4413; alcohol: 22 recordings, n=5 animals; water: 26 recordings, n=6 animals) or (G) optically evoked population spike (oPS) amplitudes (two-way mixed ANOVA, Fluid F(1,24) = 2.7840, p=0.1082; alcohol: 14 recordings, n=5 animals; water: 12 recordings, n=5 animals). Error bars and shading indicate± SEM. Box plot whiskers represent interquartile range.
-
Figure 1—figure supplement 5—source data 1
A single binge alcohol session alters optically evoked glutamate currents, but does not change the AMPA/NMDA current ratio or alter dorsolateral striatum (DLS) network effects.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig1-figsupp5-data1-v1.zip
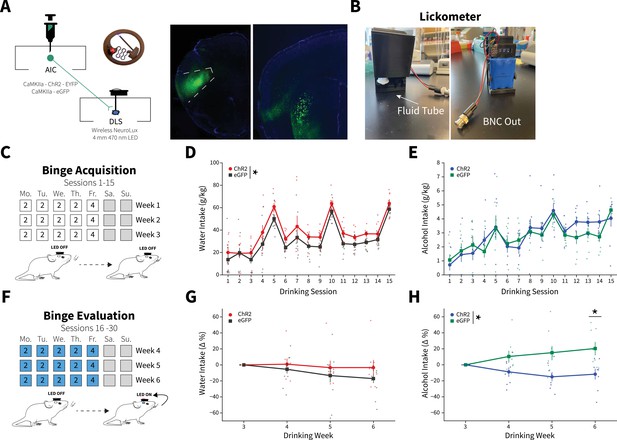
Anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS) modulate binge alcohol intake, but not water intake.
(A) Representation of viral strategy and expression to modulate AIC inputs within the DLS. (B) Photographs and description of lickometer. (C) Schematic of Drinking in the Dark (DID) protocol for binge alcohol or water acquisition. (D) Group and individual animals’ water consumption differed by viral expression during binge acquisition (two-way mixed analysis of variance [ANOVA], Virus F(1,16) = 8.1951, p=0.0113; ChR2: n=8 animals, eGFP: n=11 animals). (E) Group and individual animals’ alcohol consumption did not differ by viral expression grouping during binge acquisition (two-way mixed ANOVA, Virus F(1,14) = 0.1829, p=0.6754; ChR2: n=8 animals, eGFP: n=8 animals). (F) Schematic of DID protocol for binge alcohol or water evaluation. (G) Photoexciting AIC inputs within the DLS during DID did not alter water intakes (two-way mixed ANOVA, Virus F(1,17) = 1.0087, p=0.3293; ChR2: n=8 animals, eGFP: n=11 animals), but (H) decreased alcohol intake (two-way mixed ANOVA, Virus F(1,14) = 8.5743, p=0.0110; Virus × Drinking Week F(3,42) = 4.7132, p=0.0063; Drinking Week 6, p=0.0281; ChR2: n=8 animals, eGFP: n=8 animals). Error bars indicate± SEM. All post hoc comparisons are Sidak corrected.
-
Figure 2—source data 1
Anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS) modulate binge alcohol intake, but not water intake.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig2-data1-v1.zip
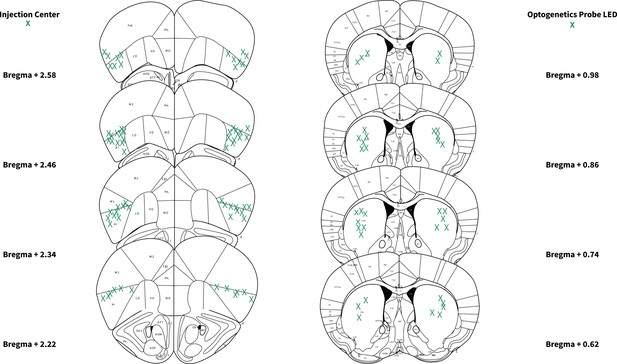
Locations of anterior insular cortex (AIC) injections and dorsolateral striatum (DLS) wireless optogenetic probe placements.
Bilateral injection centers of AAV-ChR2-EYFP or AAV-eGFP in the AIC and unilateral (left: n=18 probes; right: n=17 probes) wireless optogenetic probe LED locations as verified by histology (alcohol: n=16 animals; water n=19 animals).
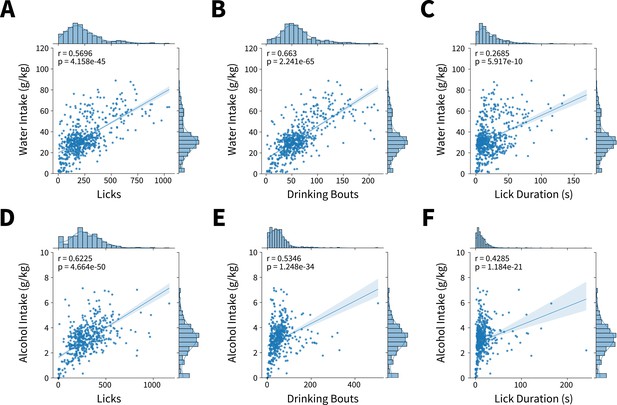
Microstructure features measured from lickometers correlate with alcohol and water intakes.
(A) Water licks (Shepherd’s pi correlation, r=0.5696, p=4.1581e-45), (B) bouts (Shepherd’s pi correlation, r=0.6679, p=1.5517e-66), and (C) drinking duration (Shepherd’s pi correlation, r=0.2685, p=5.9165e-10) per Drinking in the Dark (DID) session correlate with water intakes. (D) Alcohol licks (Shepherd’s pi correlation, r=0.6225, p=4.6644e-50), (E) bouts (Shepherd’s pi correlation, r=0.5343, p=1.2482e-34), and (F) drinking duration (Shepherd’s pi correlation, r=0.4285, p=1.1841e-21) per DID session correlate with alcohol intakes. Individual datapoints represent a microstructure feature and intake from single DID session. Data were fit to a linear model represented by a line of best fit with shading indicating ± SEM. Axes histograms display distribution of microstructure features and intakes across all DID sessions.
-
Figure 2—figure supplement 2—source data 1
Microstructure features measured from lickometers correlate with alcohol and water intakes.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig2-figsupp2-data1-v1.zip
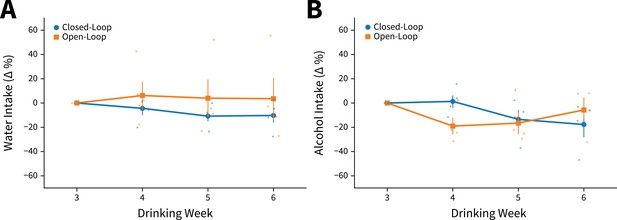
Channelrhodopsin-2 (ChR2) expressing yoked animals do not differ from closed-loop stimulated animals in alcohol and water intakes.
Intakes during photoexcitation in ChR2 expressing animals do not differ by stimulation delivery paradigm for (A) water (two-way mixed analysis of variance [ANOVA], Stimulation Paradigm F(1,6) = 0.6079, p=0.4652; closed-loop: n=4 animals, open-loop: n=4 animals) or (B) alcohol (two-way mixed ANOVA, Stimulation Paradigm F(1,6) = 0.7340, p=0.1268; closed-loop: n=4 animals, open-loop: n=4 animals). Error bars indicate± SEM.
-
Figure 2—figure supplement 3—source data 1
Channelrhodopsin-2 (ChR2) expressing yoked animals do not differ from closed-loop stimulated animals in alcohol and water intakes.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig2-figsupp3-data1-v1.zip
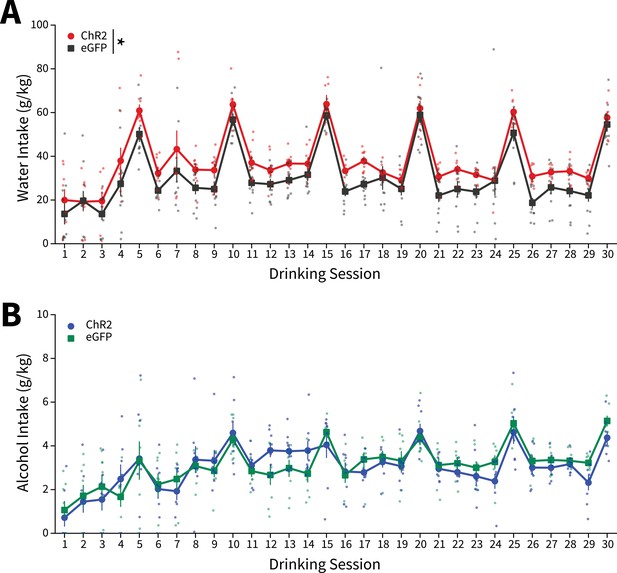
Water and alcohol intakes for all Drinking in the Dark (DID) sessions.
(A) Water intakes for all sessions (two-way mixed analysis of variance (ANOVA), Virus F(1,16) = 7.2567, p=0.0160; ChR2: n=8 animals, eGFP: n=11 animals). (B) Alcohol intakes for all sessions (two-way mixed ANOVA, Virus F(1,14) = 1.1205, p=0.3075; ChR2: n=8 animals, eGFP: n=8 animals). Error bars indicate± SEM.
-
Figure 2—figure supplement 4—source data 1
Water and alcohol intakes for all Drinking in the Dark (DID) sessions.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig2-figsupp4-data1-v1.zip
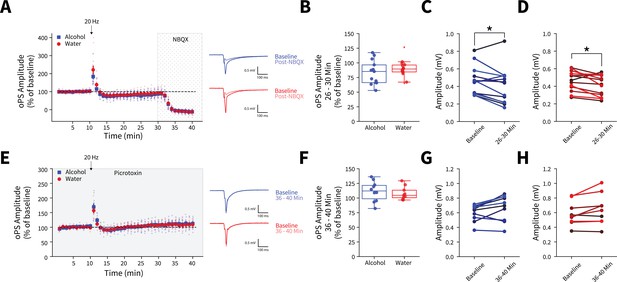
Effect of using in vivo optogenetic stimulation parameters in brain slices.
(A) The 20 Hz stimulation used for in vivo stimulation does not produce differential responses in AMPA glutamate receptor antagonist (NBQX)-sensitive optically evoked population spike (oPS) amplitudes between water and alcohol exposed animals (two-way mixed analysis of variance [ANOVA], Virus F(1,24) = 1.2962, p=0.2661; alcohol: 13 recordings, n=4 animals, water: 14 recordings, n=5 animals). (B) Summary of average 26–30 min oPS amplitudes following 20 Hz stimulation show no group differences between alcohol and water exposed animals (t test, t(25) = 0.9475, p=0.3525). 20 Hz stimulation decreased both (C) alcohol (paired t test, t(12) = 2.4345, p=0.0315) and (D) water (paired t test, t(13) = 2.6115, p=0.0215) oPS amplitudes at 26–30 min compared to baseline (0–10 min). (E) 20 Hz stimulation does not produce differential responses in oPS amplitudes between water and alcohol exposed animals in the presence of picrotoxin (two-way mixed ANOVA, Virus F(1,15) = 0.0894, p=0 0.7691; alcohol: 10 recordings, n=3 animals, water: 9 recordings, n=3 animals). (F) Summary of average 36–40 min oPS amplitudes following 20 Hz stimulation in the presence of the GABAA receptor antagonist, picrotoxin, show no group differences between alcohol and water exposed animals (t test, t(17) = 0.4556, p=0.6545). In the presence of picrotoxin, there are no differences in baseline (0–10 min) and oPS amplitudes at 36–40 min for (G) alcohol (paired t test, t(9) = 2.2445, p=0.0515) or (H) water exposed animals (paired t test, t(8) = 2.2467, p=0.0549). These data indicate that 20 Hz stimulation does not induce long-lasting glutamate receptor-mediated synaptic plasticity in either water or alcohol drinking animals. Error bars indicate ± SEM. Box plot whiskers represent interquartile range.
-
Figure 2—figure supplement 5—source data 1
Effect of using in vivo optogenetic stimulation parameters in brain slices.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig2-figsupp5-data1-v1.zip
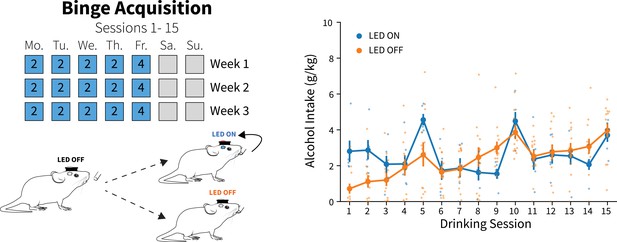
Photoexciting anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS) did not alter alcohol intakes during binge acquisition.
Photoexciting AIC inputs in the DLS did not alter alcohol intakes compared to non-stimulated alcohol drinkers during binge acquisition (two-way mixed analysis of variance [ANOVA], LED state F(1,18) = 0.2484, p=0.6243; LED ON: n=6 animals, LED OFF: n=14 animals). Error bars indicate ± SEM.
-
Figure 2—figure supplement 6—source data 1
Photoexciting anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS) did not alter alcohol intakes during binge acquisition.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig2-figsupp6-data1-v1.zip
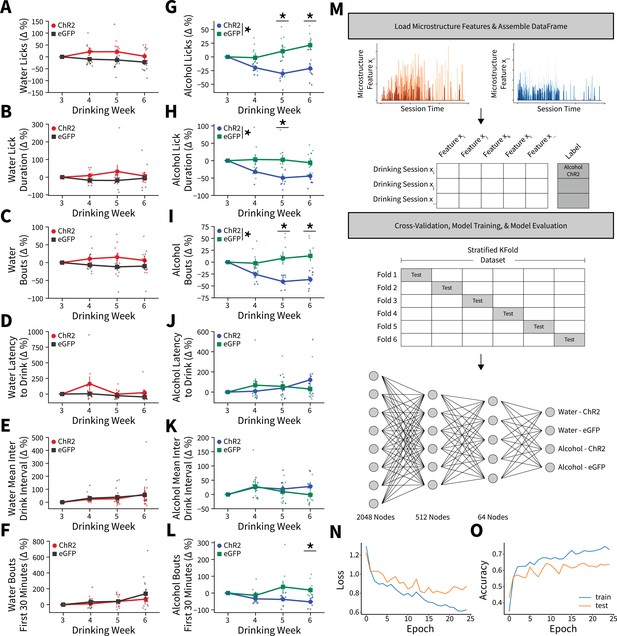
Alterations in drinking microstructure represent significant decreases in alcohol intake and are predictive of AIC→DLS alcohol-induced synaptic plasticity changes.
Photoexciting anterior insular cortex (AIC) inputs does not alter the (A) number of water licks (two-way mixed analysis of variance [ANOVA], Virus F(1,17) = 3.7529, p=0.0695; ChR2: 32 observations, n=8 animals, eGFP: 44 observations, n=11 animals), (B) water lick durations (two-way mixed ANOVA, Virus F(1,17) = 2.2136, p=0.1551; ChR2: 32 observations, n=8 animals, eGFP: 44 observations, n=11 animals), (C) water bouts (two-way mixed ANOVA, Virus F(1,17) = 3.0848, p=0.0971; ChR2: 32 observations, n=8 animals, eGFP: 44 observations, n=11 animals), (D) latency to drink water (two-way mixed ANOVA, Virus F(1,17) = 2.7012, p=0.1186; ChR2: 32 observations, n=8 animals, eGFP: 44 observations, n=11 animals), (E) mean inter-drink-interval for water drinking (two-way mixed ANOVA, Virus F(1,17) = 0.0272, p=0.8708; ChR2: 32 observations, n=8 animals, eGFP: 44 observations, n=11 animals), (F) or the number water bouts in the first 30 min of the Drinking in the Dark (DID) session (two-way mixed ANOVA, Virus F(1,17) = 0.5716, p=0.4599; ChR2: 32 observations, n=8 animals, eGFP: 44 observations, n=11 animals). Photoexciting AIC inputs (G) decreases the number of alcohol licks (two-way mixed ANOVA, Virus F(1,14) = 11.0142, p=0.0051; Virus × Drinking Week F(3,42) = 5.8888, p=0.0019; Week 5 p=0.0307, Week 6 p=0.0137; ChR2: 32 observations, n=8 animals, eGFP: 32 observations, n=8 animals), (H) decreases total alcohol lick duration (two-way mixed ANOVA, Virus F(1,14) = 6.9536, p=0.0195; Virus × Drinking Week F(3,42) = 3.8533, p=0.0160; Week 5 p=0.0458; ChR2: 32 observations, n=8 animals, eGFP: 32 observations, n=8 animals), and (I) decreases the number of alcohol bouts (two-way mixed ANOVA, Virus F(1,14) = 11.2086, p=0.0048; Virus × Drinking Week F(3,42) = 9.4893, p=0.0001; Week 5 p=0.0135, Week 6 p=0.0051; ChR2: 32 observations, n=8 animals, eGFP: 32 observations, n=8 animals). (J) Modulating AIC inputs does not impact the latency to drink alcohol (two-way mixed ANOVA, Virus F(1,14) = 0.0084, p=0.9281; ChR2: 32 observations, n=8 animals, eGFP: 32 observations, n=8 animals) or (K) the mean inter-drink-interval for alcohol drinking (two-way mixed ANOVA, Virus F(1,14) = 0.4686, p=0.5047; ChR2: 32 observations, n=8 animals, eGFP: 32 observations, n=8 animals), but (L) decreases front loading behaviors for alcohol drinking (two-way mixed ANOVA, Virus F(1,14) = 4.2003, p=0.0596; Virus × Drinking Week F(3,42) = 3.9078, p=0.0150; Week 6 p=0.0230; ChR2: 32 observations, n=8 animals, eGFP: 32 observations, n=8 animals). (M) Schematic for microstructure feature detection, dataset assembly, cross-validation, and network architecture. (N) Loss curve visualization for training and testing data. (O) Model accuracy for training and testing data. Error bars indicate ± SEM. All post hoc comparisons are Sidak corrected.
-
Figure 3—source code 1
Alterations in drinking microstructure represent significant decreases in alcohol intake and are predictive of AIC→DLS alcohol-induced synaptic plasticity changes.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig3-code1-v1.zip
-
Figure 3—source data 1
Alterations in drinking microstructure represent significant decreases in alcohol intake and are predictive of AIC→DLS alcohol-induced synaptic plasticity changes.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig3-data1-v1.zip
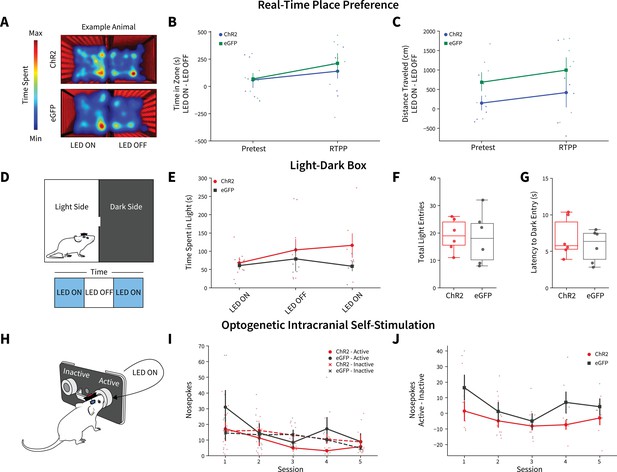
Anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS) do not modulate valence, anxiety-like, or operant responding behaviors.
(A) Example animal heatmap for real-time place preference assay. Photoexciting AIC inputs within the DLS did not alter (B) real-time place preference/avoidance behaviors (two-way mixed analysis of variance [ANOVA], Virus F(1,11) = 0.2330, p=0.6388; ChR2: n=6 animals, eGFP: n=7 animals) or (C) locomotion during real-time place preference (two-way mixed ANOVA, Virus F(1,11) = 3.8457, p=0.0757; ChR2: n=6 animals, eGFP: n=7 animals). (D) Schematic for light-dark box assay. Modulating AIC inputs did not alter (E) time spent on light side across LED epochs two-way mixed ANOVA, Virus F(1,9) = 1.6826, p=0.2268; ChR2: n=6 animals, eGFP: n=6 animals, (F) the number of total light side entries (t test, t(10) = 0.2204, p=0.83; ChR2: n=6 animals, eGFP: n=6 animals), or (G) delays to enter the dark (t test, t(10) = 0.7699, p=0.4591; ChR2: n=6 animals, eGFP: n=6 animals). (H) Schematic of operant self-stimulation testing. (I) Photoexciting AIC inputs did not alter nosepoking behaviors across session (three-way ANOVA, Virus × Poke × Session F(4,96) = 0.3108, p=0.8702; ChR2: n=6 animals, eGFP: n=6 animals). (J) Active–inactive nosepokes do not differ between viral expression when modulating AIC (two-way mixed ANOVA, Virus F(1,8) = 5.0255, p=0.0553; study sufficiently powered to detect main effect of Virus as determined by power analysis, β=0.9898; ChR2: n=6 animals, eGFP: n=6 animals). Error bars indicate ± SEM. Box plot whiskers represent interquartile range.
-
Figure 4—source data 1
Anterior insular cortex (AIC) inputs to the dorsolateral striatum (DLS) do not modulate valence, anxiety-like, or operant responding behaviors.
- https://cdn.elifesciences.org/articles/77411/elife-77411-fig4-data1-v1.zip






