A novel rhesus macaque model of Huntington’s disease recapitulates key neuropathological changes along with motor and cognitive decline
Figures
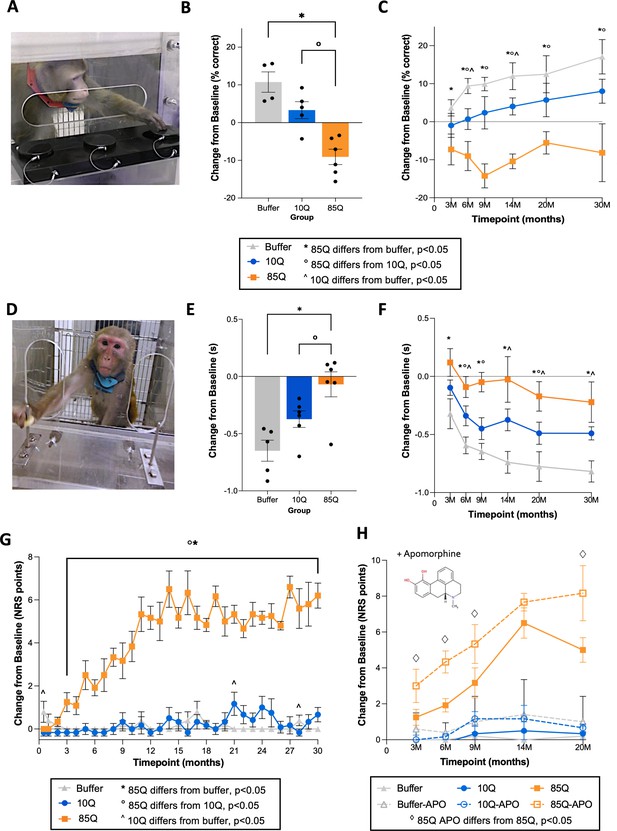
Working memory and motor deficits in 85Q-treated animals.
(A) Example of an animal performing the 3-Choice Spatial Delayed Response (SDR) task. (B) Change in SDR performance (% correct) on the SDR task, collapsed across timepoints. (C) Change in SDR performance (% correct), expanded across timepoints. All SDR data are expressed as mean ± standard error of the mean (SEM) (85Q – n = 6, 10Q – n = 5, Buffer – n = 4). The key details significant group differences at each timepoint (*, ^, and ° symbols), demonstrating that working memory changes emerge in this model beginning at 3–6 months post-85Q administration. (D) Example of an animal performing the Lifesaver Retrieval Task. (E) Performance collapsed across timepoint, plotted as the change in Retrieval Latency (seconds) from baseline. (F) Performance expanded across timepoints, plotted as the change in Retrieval Latency (seconds) from baseline. All Lifesaver Retrieval data are expressed as mean ± SEM (85Q – n = 6, 10Q – n = 6, Buffer – n = 5). The key details significant group differences at each timepoint and shows that fine motor skill learning impairment emerges in 85Q animals as early as 3 months post-85Q administration. (G) Mean changes in motor phenotypes plotted for each group. The key shows significant group differences at each timepoint, indicating that motor phenotypes first emerge around 2–3 months post-surgery. (H) Neurological rating scale (NRS) scores (difference from baseline) pre- and post-apomorphine administration plotted for each group, with pre-apomorphine NRS scores depicted by solid lines and post-apomorphine NRS scores depicted by dashed lines. The • symbol indicates significant paired comparisons at each timepoint, showing that 85Q-treated animals displayed NRS scores that were modulated by apomorphine at 3, 6, 9, and 20 months post-surgery. All NRS data are expressed as mean ± SEM (85Q – n = 6, 10Q – n = 6, Buffer – n = 5). *p < 0.05, 85Q versus Buffer, op < 0.05, 85Q versus 10Q, ^p < 0.05, Buffer versus 10Q by three-way analysis of variance (ANOVA).
-
Figure 1—source data 1
Spatial Delayed Response (SDR), Lifesaver, and neurological rating scale (NRS) scores.
Table 1 reports scores from all the behavioral tasks at each of the post-surgical timepoints, as well as overall averages. All scores are reported as change from baseline measures.
- https://cdn.elifesciences.org/articles/77568/elife-77568-fig1-data1-v1.xlsx
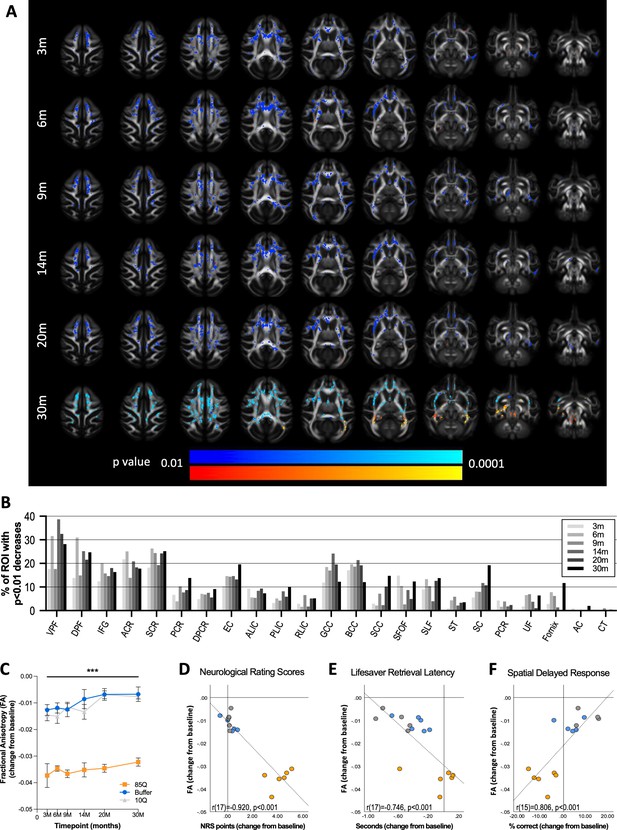
85Q-mediated microstructural changes in cerebral white matter.
(A) ONPRC18 FA template with overlaying p-value maps shown at a range of p < 0.01 to p < 0.0001. Blue voxels indicate regions of significant FA decrease in Group 85Q, and red voxels indicate regions of significant FA increase. Although there were slight changes in FA in the Buffer- and 10Q-treated animals over time, none of the contrasts reached statistical significance (not pictured). (B) Histogram illustrating the percent volume of each ROI where significant FA changes were identified in Group 85Q corresponding to the blue voxels in panel (A). The line chart in (C) illustrates the average magnitude of change in FA under the significant voxels at each timepoint. Data are expressed as mean ± standard error of the mean (SEM). Scatterplots illustrating one-tailed Pearson correlations between FA decreases in white matter (WM) and behavior (both collapsed across timepoint) for three different behavioral measures: (D) neurological rating scale (NRS), (E) Lifesaver Retrieval Task, and (F) Spatial Delayed Response (SDR) task. ***p < 0.001; 85Q differs from Buffer and 10Q. Abbreviations: FA, fractional anisotropy; ROI, region of interest; VPF, ventral prefrontal WM; DPF, dorsal prefrontal WM; IFG, inferior frontal gyrus WM; ACR, anterior corona radiata; SCR, superior corona radiata; PCR, posterior corona radiata; DPCR, dorsal posterior corona radiata; EC, external capsule; ALIC, anterior limb of the internal capsule; PLIC, posterior limb of the internal capsule; RLIC, retrolenticular limb of the internal capsule; GCC, genu of corpus callosum; BCC, body of corpus callosum; SCC, splenium of corpus callosum; SFOF, superior fronto-occipital fasciculus; SLF, superior longitudinal fasciculus; ST, stria terminalus; SC, superior cingulum; PCR, perihippocampal cingulum; UF, uncinate fasciculus; AC, anterior commissure; CT, corticospinal tract.
-
Figure 2—source data 1
Diffusion tensor imaging (DTI).
Table 1 reports the percent volume of regions of interest (ROIs) that had p < 0.01 significant changes in fractional anisotropy (FA) identified by voxel-wise analyses in Group 85Q. Both control groups (10Q and Buffer) did not have any significant (p < 0.01) changes and are not included in this table. Table 2 reports the average change in FA (from baseline) under the p < 0.01 voxels at each timepoint, for all 17 animals on study. These data were also averaged across timepoint and correlated with average behavioral scores (also included in Table 2).
- https://cdn.elifesciences.org/articles/77568/elife-77568-fig2-data1-v1.xlsx
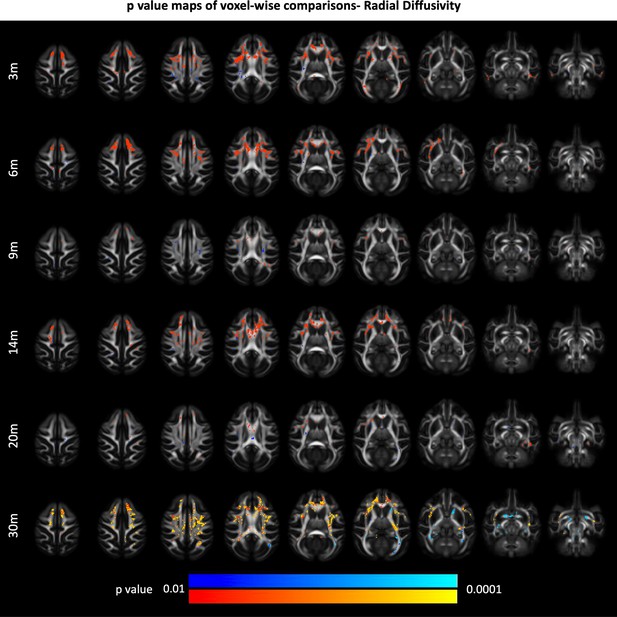
85Q-mediated changes in white matter radial diffusivity (RD).
(A) ONPRC18 FA template with overlaying p-value maps shown from the ranges of p < 0.01 to 0 < 0.0001. Blue voxels indicate areas of significant RD decrease in Group 85Q, and red voxels indicate areas of significant RD increase. Although there were slight changes in the Buffer and 10Q over time, none of the contrasts reached statistical significance (not pictured).
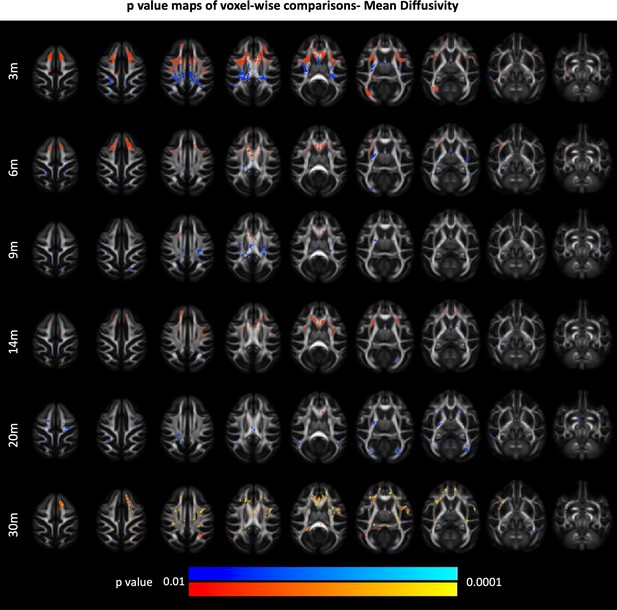
85Q-mediated changes in white matter mean diffusivity (MD).
(A) ONPRC18 FA template with overlaying p-value maps shown from the ranges of p < 0.01 to p < 0.0001. Blue voxels indicate areas of significant MD decrease in Group 85Q, and red voxels indicate areas of significant MD increase. Although there were slight changes in the Buffer and 10Q over time, none of the contrasts reached statistical significance (not pictured).
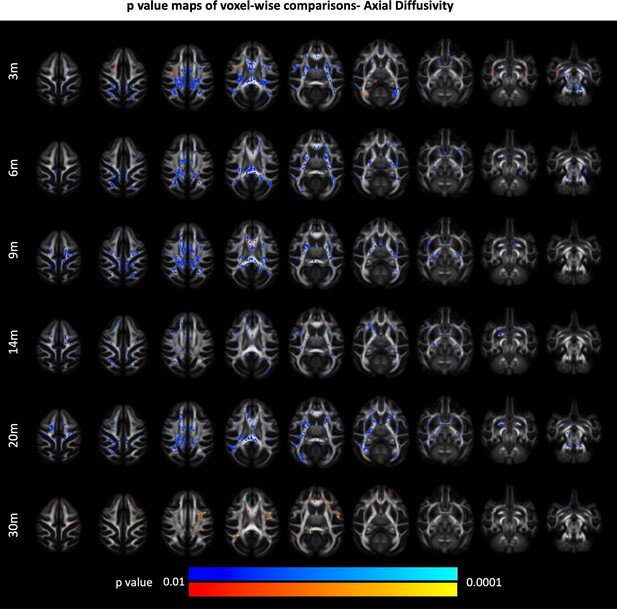
85Q-mediated changes in white matter axial diffusivity (AD).
(A) ONPRC18 FA template with overlaying p-value maps shown from the ranges of p < 0.01 to p < 0.0001. Blue voxels indicate areas of significant AD decrease in Group 85Q, and red voxels indicate areas of significant AD increase. Although there were slight changes in the Buffer and 10Q over time, none of the contrasts reached statistical significance (not pictured).
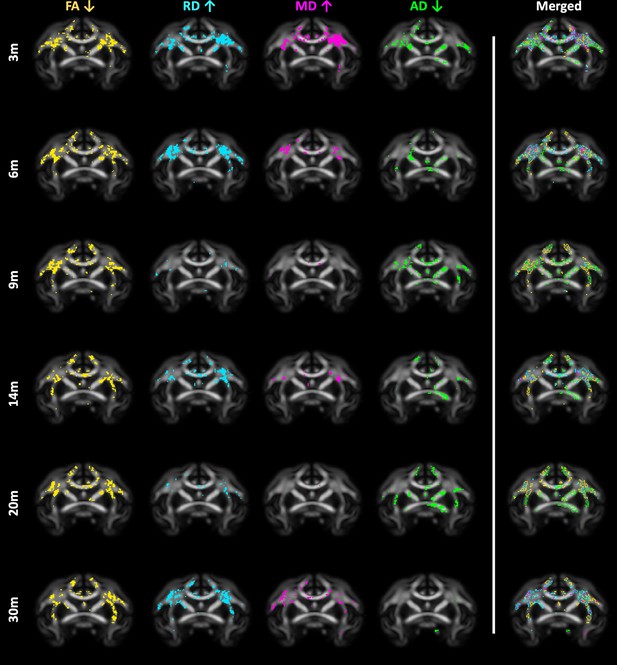
85Q-mediated changes in all diffusivity measures.
(A) ONPRC18 FA template with overlaying p-value maps shown at a threshold of p < 0.01. First column (yellow voxels) indicates areas of increased fractional anisotropy (FA). Second column (turquoise voxels) indicates areas of increased radial diffusivity (RD). Third column (magenta voxels) indicates areas of increased mean diffusivity (MD). Forth column (green voxels) indicates areas of decreased axial diffusivity (AD). Fifth column shows the merged maps, to highlight areas of overlap. In general, FA decreases were closely aligned with RD and MD increases.
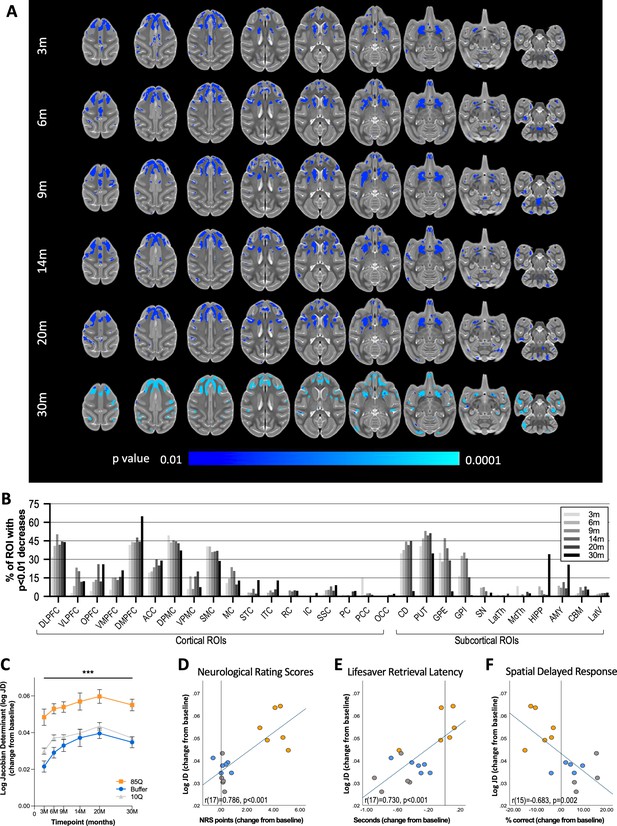
85Q-mediated tissue atrophy in cortico-basal ganglia gray matter.
(A) ONPRC18 T2w template with overlaying p-value maps shown at a threshold of p < 0.01 to p<0.0001. Blue voxels indicate regions of significant TBM contraction (increased log Jacobian Determinants) in Group 85Q. Although there were slight changes in the Buffer- and 10Q-treated animals over time, none of the contrasts reached statistical significance (not pictured). (B) Histogram illustrating the percent volume of each cortical and subcortical ROI where significant TBM contractions were identified (corresponding to the blue voxels in A). (C) A mask that merged together the thresholded p-value maps from each timepoint was created. Line charts illustrate the average log Jacobian Determinate changes under this mask for each group separately. Data are expressed as mean ± standard error of the mean (SEM) (85Q – n = 6, 10Q – n = 6, Buffer – n = 5), repeated measures analysis of variance (ANOVA). (D) Scatterplots illustrating two-tailed Pearson correlations between TBM contractions in GM and behavior (both collapsed across timepoint) for three different behavioral measures: (D) neurological rating scale (NRS), (E) Lifesaver Retrieval Task, and (F) Spatial Delayed Response (SDR) task. ***p < 0.01; 85Q differs from Buffer and 10Q. Abbreviations: TBM, tensor-based morphometry; ROI, region of interest; LogJD, log Jacobian Determinant; DLPFC, dorsolateral prefrontal cortex; VLPFC, ventrolateral prefrontal cortex; OPFC, orbitofrontal cortex; VMPFC, ventromedial prefrontal cortex; DMPFC, dorsomedial prefrontal cortex; ACC, anterior cingulate cortex; DPMC, dorsal premotor cortex; VPMC, ventral premotor cortex; SMC, supplemental motor cortex; MC, primary motor cortex; STC, superior temporal cortex; ITC, inferior temporal cortex; RC, rhinal cortex; IC, insular cortex; SSC, somatosensory cortex; PC, parietal cortex; PCC, posterior cingulate cortex; OCC, occipital cortex; CD, caudate; PUT, putamen; LatTH, lateral thalamus; MdTH, medial thalamus; HIPP, hippocampus; AMY, amygdala; SN, substantia nigra; GPI, internal globus pallidus; GPE, external globus pallidus; CBM, cerebellum; LatV, lateral ventricles.
-
Figure 3—source data 1
Tensor-based morphometry (TBM).
Table 1 reports the percent volume of regions of interest (ROIs) that had p < 0.01 significant changes in log Jacobian Determinants identified by voxel-wise analyses in Group 85Q. Both control groups (10Q and Buffer) did not have any significant (p < 0.01) changes and are not included in this table. Table 2 reports the average change in log JD (from baseline) under the p < 0.01 voxels at each timepoint, for all 17 animals on study. These data were also averaged across timepoint and correlated with average behavioral scores (also included in Table 2).
- https://cdn.elifesciences.org/articles/77568/elife-77568-fig3-data1-v1.xlsx
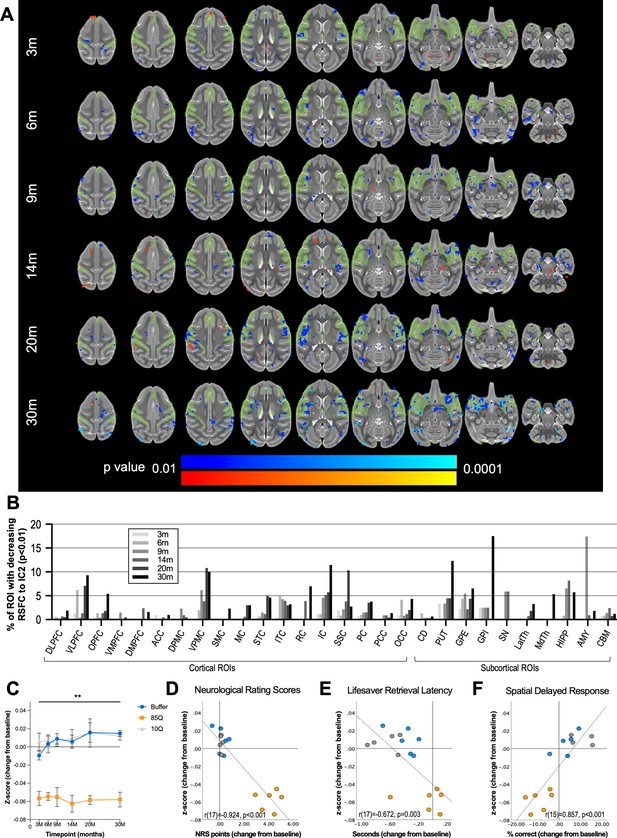
85Q-mediated alterations in patterns of brain-wide resting-state functional connectivity.
(A) ONPRC18 T2w template with overlaying map of voxels showing high temporal correlation with independent component 2 (IC2), depicted in green. Additional overlaying p-value maps are shown at a threshold of p < 0.01 to p < 0.0001. Blue voxels indicate regions of significantly reduced RSFC (decreased z-score) with IC2 in Group 85Q, and red voxels indicate regions of significant increased RSFC (increased z-score) with IC2. Although there were slight changes in the Buffer- and 10Q-treated animals over time, none of the contrasts reached statistical significance (not pictured). (B) Histogram illustrating the percent volume of each cortical and subcortical region of interest (ROI) where significantly decreased z-scores were identified at each timepoint (corresponding to the blue voxels in A). (C) A mask that merged together the thresholded p-value maps from each timepoint was created for regions of RSFC decreases with IC2. Line charts illustrate the average magnitude of RSFC changes (from baseline) under this mask for each group separately, ±1 standard error of the mean (SEM). (D) Scatterplots illustrating two-tailed Pearson correlations between RFSC changes in GM and behavior (both collapsed across timepoint) for three different behavioral measures: (D) neurological rating scale (NRS), (E) Lifesaver Retrieval Task, and (F) Spatial Delayed Response (SDR) task. RSFC, resting-state functional connectivity. **p < 0.001; 85Q differs from Buffer and 10Q. Abbreviations: DLPFC, dorsolateral prefrontal cortex; VLPFC, ventrolateral prefrontal cortex; OPFC, orbitofrontal cortex; VMPFC, ventromedial prefrontal cortex; DMPFC, dorsomedial prefrontal cortex; ACC, anterior cingulate cortex; DPMC, dorsal premotor cortex; VPMC, ventral premotor cortex; SMC, supplemental motor cortex; MC, primary motor cortex; STC, superior temporal cortex; ITC, inferior temporal cortex; RC, rhinal cortex; IC, insular cortex; SSC, somatosensory cortex; PC, parietal cortex; PCC, posterior cingulate cortex; OCC, occipital cortex; CD, caudate; PUT, putamen; GPE, external globus pallidus; GPI, internal globus pallidus; SN, substantia nigra; LatTH, lateral thalamus; MdTH, medial thalamus; HIPP, hippocampus; AMY, amygdala; CBM, cerebellum.
-
Figure 4—source data 1
IC2 RSFC.
Table 1 reports the percent volume of regions of interest (ROIs) that had p < 0.01 significant changes in IC2 z-scores identified by voxel-wise analyses in Group 85Q. Both control groups (10Q and Buffer) did not have any significant (p < 0.01) changes and are not included in this table. Table 2 reports the average change in z-score (from baseline) under the p < 0.01 voxels at each timepoint, for all 17 animals on study. These data were also averaged across timepoint and correlated with average behavioral scores (also included in Table 2).
- https://cdn.elifesciences.org/articles/77568/elife-77568-fig4-data1-v1.xlsx
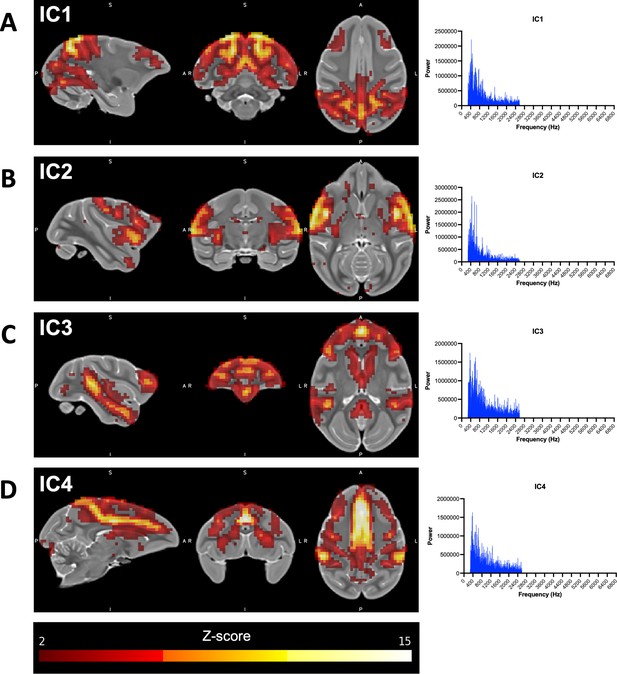
Resting-state networks of interest identified using independent component (IC) analysis.
(A–D) Illustrations showing the ONPRC18 T2w template with overlaying z-score maps from each IC of interest (shown at a threshold of z > 2), along with histograms demonstrating the low-frequency spectral power associated with each (Griffanti et al., 2017).
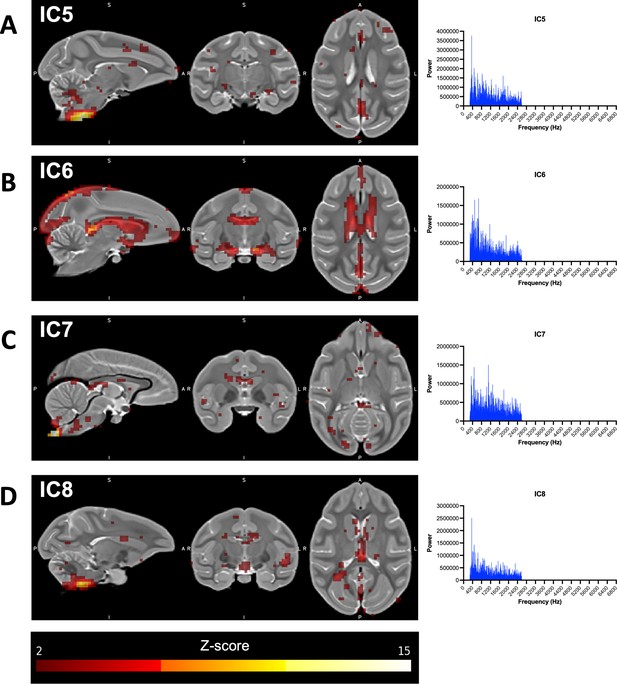
Networks identified using independent component (IC) analysis associated with noise.
(A–D) Illustrations showing the ONPRC18 T2w template with overlaying z-score maps from each omitted IC (shown at a threshold of z > 2), along with histograms illustrating the relatively even distribution of spectral frequencies in each.
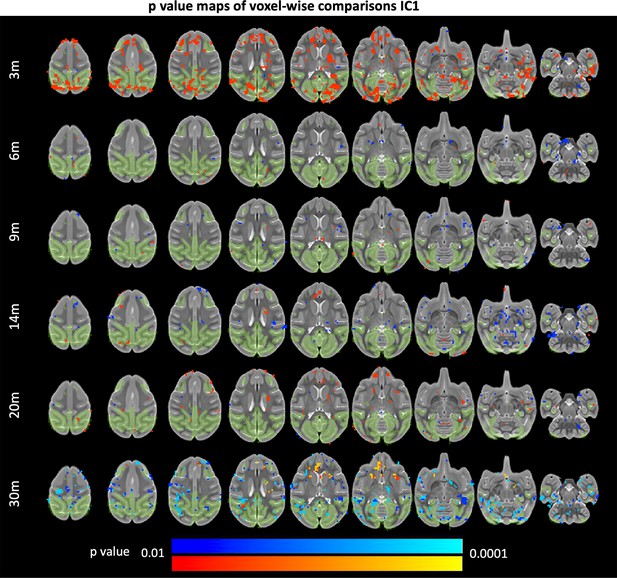
85Q-mediated changes resting-state functional connectivity: IC1.
ONPRC18 T2w template with overlaying p-value maps shown from the ranges of p < 0.01 to p < 0.0001. Blue voxels indicate regions of significant RSFC decrease in Group 85Q, and red voxels indicate areas of significant RSFC increase. Green voxels indicate regions of IC1. Although there were slight changes in the Buffer and 10Q over time, none of the contrasts reached significance (not pictured).
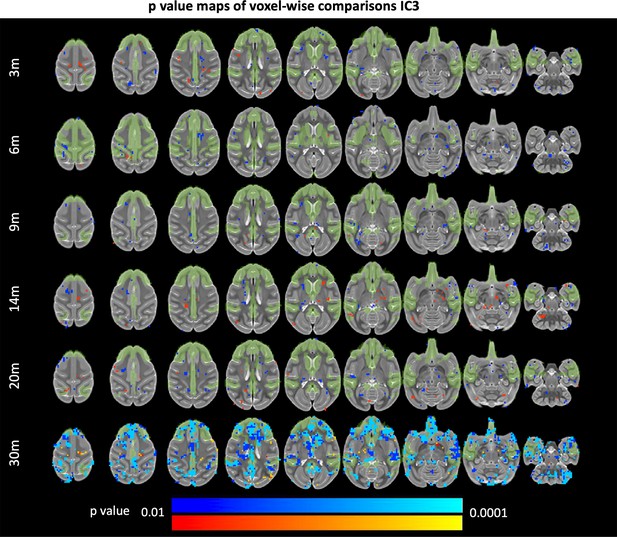
85Q-mediated changes resting-state functional connectivity: IC3.
ONPRC18 T2w template with overlaying p-value maps shown from the ranges of p < 0.01 to p < 0.0001. Blue voxels indicate regions of significant RSFC decrease in Group 85Q, and red voxels indicate regions of significant RSFC increase. Green voxels indicate regions of IC3. Although there were slight changes in the Buffer and 10Q over time, none of the contrasts reached significance (not pictured).
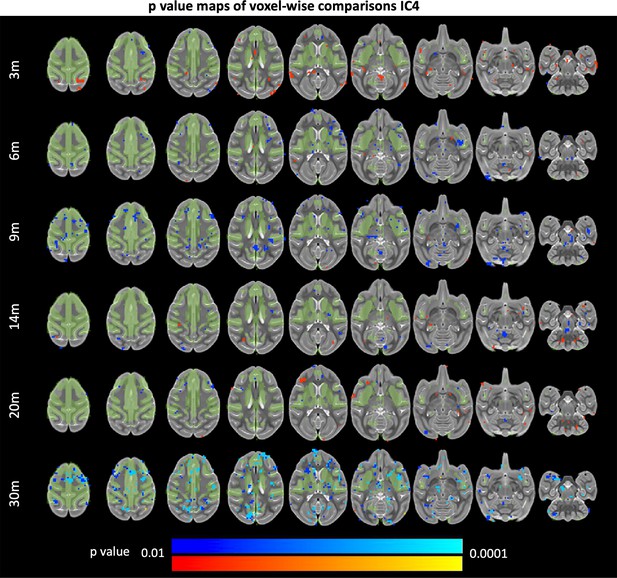
85Q-mediated changes resting-state functional connectivity: IC4.
ONPRC18 T2w template with overlaying p-value maps shown from the ranges of p < 0.01 to p < 0.0001. Blue voxels indicate regions of significant RSFC decrease in Group 85Q, and red voxels indicate regions of significant RSFC increase. Green voxels indicate regions of IC4. Although there were slight changes in the Buffer and 10Q over time, none of the contrasts reached significance (not pictured).
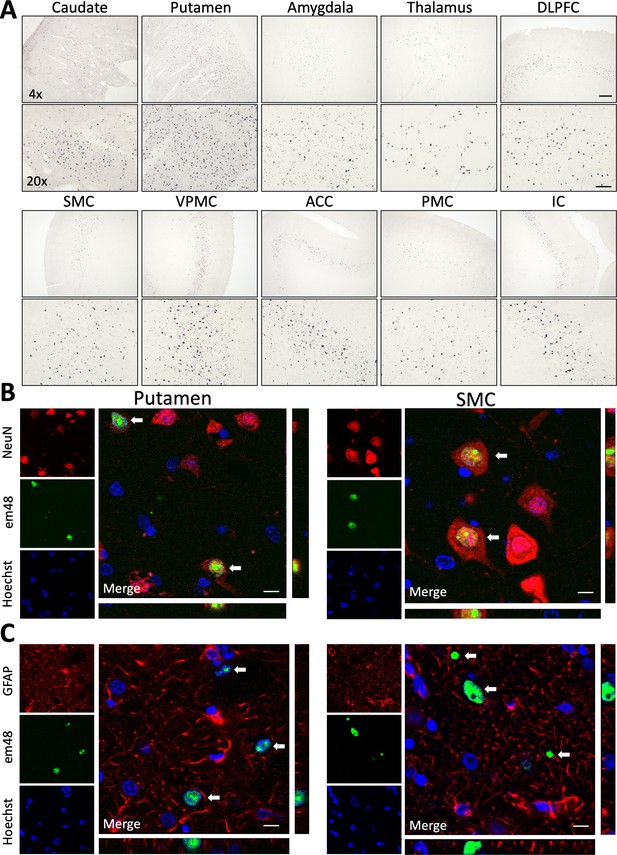
85Q-treated animals show evidence of neuronal em48+ mHTT protein inclusion body formation throughout cortical and subcortical brain regions.
(A) Low (×4) and high (×20) magnification brightfield photomicrographs illustrate em48+ mHTT inclusions throughout the areas of injection (caudate and putamen), as well as in several other cortical and subcortical brain regions with known afferent projections to the striatum. Examples shown here: DLPFC, dorsolateral prefrontal cortex; SMC, supplemental motor cortex; VPMC, ventral premotor cortex; ACC, anterior cingulate cortex; PMC, primary motor cortex; IC, insular cortex. Scale bar on ×4 image indicates 200 µM, scale bar on ×20 image indicates 50 µM. em48+ inclusions were not observed in 10Q- or buffer-treated controls. (B) Triple-labeled immunofluorescent confocal images from 85Q-treated animals illustrating localization of mHTT inclusions in neuronal nuclei. Examples shown here are from an area of injection (putamen – left panel) as well as a distal cortical region (supplemental motor cortex – right panel). White arrows indicate cells that are triple labeled for neurons (NeuN+), mHTT inclusions (em48+), and nuclei (stained with Hoechst 33342). (C) Triple-labeled immunofluorescent confocal images illustrating the lack of localization of mHTT inclusions in astrocytes, examples shown from the putamen and supplemental cortex. White arrows indicate cells that are labeled for mHTT inclusions (em48+) and nuclei (stained with Hoechst 33342), but not astrocytes (GFAP+). Scale bars in B and C = 10 µM. Orthogonal views from different planes are included for reference, and depict an example of one of the em48+ cells from each brain region, and for each cellular marker.
Tables
Summary of study participants and surgical cases.
Abbreviations: 85Q, fragment of mHTT protein bearing 85 glutamine repeats; 10Q, fragment of mHTT protein bearing 10 glutamine repeats; Buffer, phosphate buffered saline; VG, vector genomes; SDR, spatial delay response.
| Animal Id | Treatment group | Sex | Age at surgery (years) | Weight at surgery (kg) | Vector dose per hemisphere (vg) | Met criteria for SDR task | Met criteria for lifesaver task |
|---|---|---|---|---|---|---|---|
| 1 | 85Q | F | 10.1 | 5.7 | 3.30E+11 | Yes | Yes |
| 2 | 85Q | F | 11.8 | 6 | 3.30E+11 | Yes | Yes |
| 3 | 10Q | F | 13.5 | 9.9 | 3.30E+11 | Yes | Yes |
| 4 | 85Q | M | 5.9 | 10.7 | 3.30E+11 | Yes | Yes |
| 5 | 10Q | F | 8.7 | 6.5 | 3.30E+11 | Yes | Yes |
| 6 | Buffer | F | 12 | 8.7 | – | Yes | Yes |
| 7 | 10Q | F | 9 | 8.6 | 3.30E+11 | Yes | Yes |
| 8 | Buffer | F | 9.9 | 7.5 | – | No | Yes |
| 9 | Buffer | M | 7.7 | 11.3 | – | Yes | Yes |
| 10 | 10Q | M | 7.5 | 8.8 | 3.30E+11 | Yes | Yes |
| 11 | 85Q | F | 13 | 6.2 | 3.30E+11 | Yes | Yes |
| 12 | 85Q | F | 12.3 | 7.5 | 3.30E+11 | Yes | Yes |
| 13 | Buffer | F | 7.9 | 6 | – | Yes | Yes |
| 14 | 85Q | M | 7.1 | 11.1 | 3.30E+11 | Yes | Yes |
| 15 | Buffer | F | 8.8 | 5.9 | – | Yes | Yes |
| 16 | 10Q | F | 11.8 | 6 | 3.30E+11 | Yes | Yes |
| 17 | 10Q | M | 10 | 9.5 | 3.30E+11 | No | Yes |
Additional files
-
Supplementary file 1
Planned group comparisons for 3-Choice Spatial Delayed Response (SDR) task using one-tailed independent sample t-tests at each timepoint.
*p < 0.05, **p < 0.01, ***p < 0.001.
- https://cdn.elifesciences.org/articles/77568/elife-77568-supp1-v1.docx
-
Supplementary file 2
Planned group comparisons for Lifesaver Retrieval Latencies using one-tailed independent sample t-tests at each timepoint.
*p < 0.05, **p < 0.01, ***p < 0.001.
- https://cdn.elifesciences.org/articles/77568/elife-77568-supp2-v1.docx
-
Supplementary file 3
Neurological rating scale (NRS) for nonhuman primates.
Behaviors were scored cage-side by trained observers, blinded to treatment condition, during 30- to 45-min focal observations. Scores were summed across categories to generate a total NRS score. Higher scores indicate more severe phenotypes.
- https://cdn.elifesciences.org/articles/77568/elife-77568-supp3-v1.docx
-
Supplementary file 4
Planned group comparisons in monthly neurological rating scale (NRS) scores using one-tailed independent sample t-tests at each timepoint.
*p < 0.05, **p < 0.01, ***p < 0.001.
- https://cdn.elifesciences.org/articles/77568/elife-77568-supp4-v1.docx
-
Supplementary file 5
Planned comparisons for pre- versus post-apomorphine neurological rating scale (NRS) scores at each timepoint using one-tailed paired-sample t-tests for each group separately.
*p < 0.05, **p <0.01.
- https://cdn.elifesciences.org/articles/77568/elife-77568-supp5-v1.docx
-
Transparent reporting form
- https://cdn.elifesciences.org/articles/77568/elife-77568-transrepform1-v1.pdf





