Functional abnormalities in the cerebello-thalamic pathways in a mouse model of DYT25 dystonia
Figures
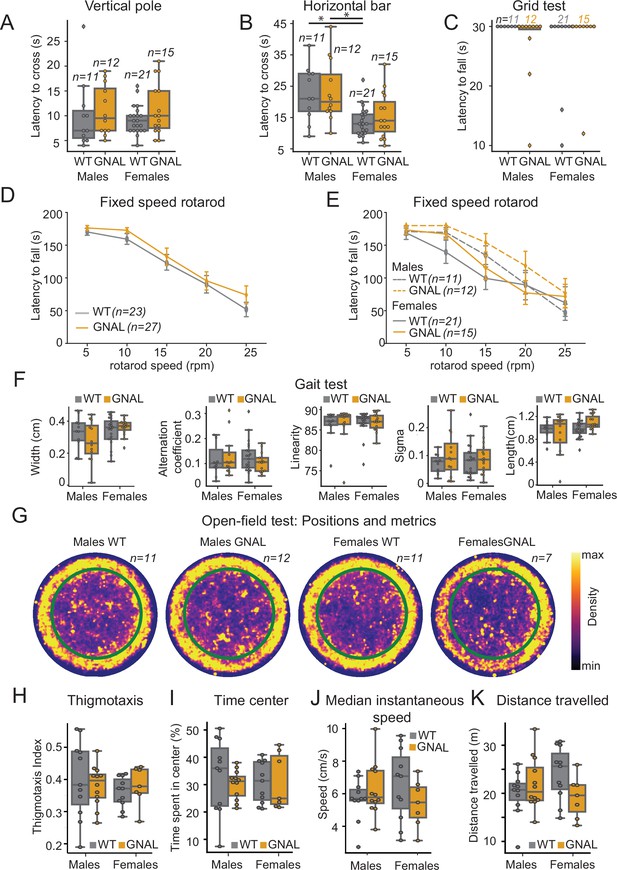
Young male and female Gnal+/- mice do not display motor coordination impairments.
(A) Latency to climb down the vertical pole in male and female Gnal+/- (GNAL) and wildtype (WT) mice. (B) Latency to cross the horizontal bar. (C) Latency to fall during the grid test (30 s cutoff). (D) Latency to fall during the fixed-speed rotarod test separated by genotype, and (E) by gender and genotype. (F) Gait width, alternation coefficient, movement linearity, sigma, and stride length during the gait test. (G) Two-dimensional histograms showing the density of position of mice during open-field sessions, separated by gender and genotype. Thigmotaxis index (H), percentage of time spent in center (I), median instantaneous speed (J), and total distance traveled (K) during the open-field test. Reported statistics are the comparison of groups separated by gender and genotype using a Kruskal–Wallis test, followed by a Dunn’s post-hoc test corrected using Holm–Sidak method. *p<0.05.
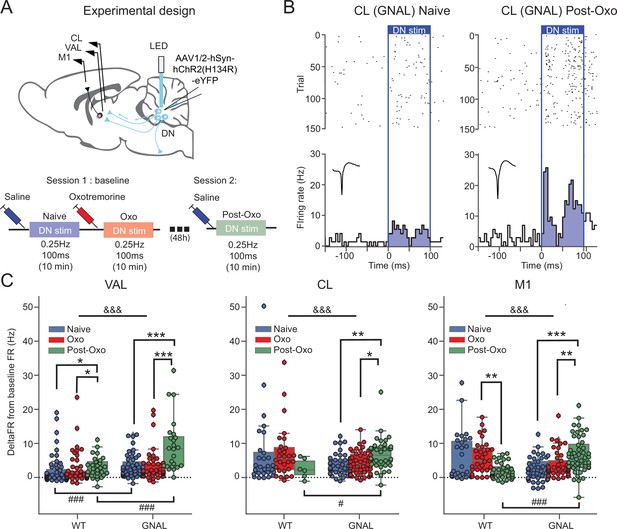
Exposure to oxotremorine causes a long-lasting increase in thalamic and cortical response to dentate nucleus (DN) stimulation in Gnal+/- mice.
(A) Schematics describing the experimental design of thalamo-cortical recordings coupled with DN stimulation (top) and experimental timeline (bottom). (B) Examples of peristimulus time histogram (PSTH) and corresponding raster plot, centered on the onset of the cerebellar stimulation, of centrolateral thalamus (CL) neurons from the same recording site in a Gnal+/- mouse under saline condition, before being exposed to oxotremorine (‘naive,’ left) and 48–72 hr after being exposed to oxotremorine (‘Post-Oxo,’ right). Inset represents the average waveforms of the recorded neuron. (C) Distributions of responses to DN stimulations in naive condition (blue), under the acute effect of oxotremorine (red) and in saline post-oxo condition (green). Two-way ANOVA, with state and genotype as factors, followed by a Dunn’s post-hoc test corrected using Holm–Sidak method to compare states within genotype. *p<0.05, **p<0.01, ***p<0.001; &&&p<0.001 for ANOVA interaction term between states and genotype.
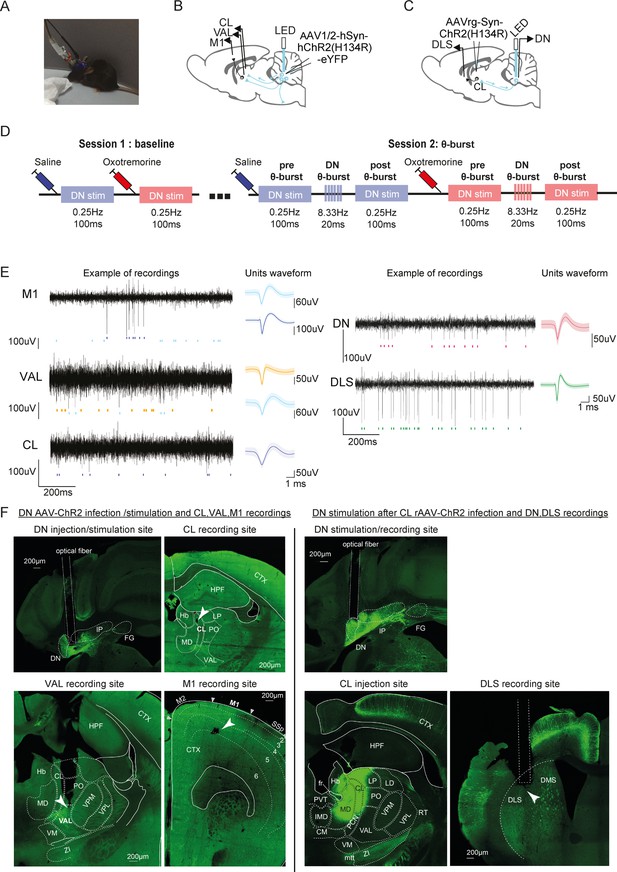
Electrophysiology recordings in thalamo-cortical and cerebello-striatal networks.
(A) Picture of a freely moving mouse during a recording coupled with optogenetic DN stimulation. (B) Schematics describing the experimental design of thalamo-cortical recordings coupled with DN stimulation. Guide cannulas with bundles of electrodes were inserted into left motor (M1), CL, and VAL. (C) Schematics describing the experimental design of cerebello-striatal recordings coupled with dentate-CL (DN-CL) stimulation. Guide cannulas with bundles of electrodes were inserted into the DN and DLS; an optic fiber was inserted in the right DN. (D) Experimental timeline describing the protocol of two recording sessions: baseline (left) and θ-burst (right). (E) Example high-pass-filtered traces recorded in the M1, VAL, CL, DN, and DLS coupled with the corresponding spike-sorted units (left) and their average waveform (mean ± SD, right). (F) Example of cannula placement, optic fiber placement, and electrode position revealed by electrolytic lesions in M1, VAL, CL, DN, and DLS. The guide cannula for the electrodes is visible only when the angle of the brain slices was close enough to the angle of penetration. The localization of the targeted area and neighboring nuclei is outlined based on the Mouse Brain Allen Atlas (2011, version 2) (http://help.brain-map.org/download/attachments/2818169/AllenReferenceAtlas_v2_2011.pdf?version=1&modificationDate=1319667383440&api=v2) and adjusted manually to correspond to the landmarks. CL: centrolateral thalamus; CM: centromedial thalamus; CTX: cortex; DLS: dorsolateral striatum; DMS: dorsomedial striatum; DN: dentate nucleus; IMD: intermediodorsal thalamus; IP: interposed nucleus; FG: fastigial nucleus; Hb: habenula; HPF: hippocampal formation; LD: laterodorsal thalamus; LP: lateral posterior thalamus; MD: mediodorsal thalamus; M1: primary motor cortex; M2: secondary motor cortex; PO: posterior thalamus; PVT: paraventricular thalamus; RT: reticularis thalamus; VAL: ventral anterior lateral thalamus; VM: ventromedial thalamus; VPM/VPL: ventral posterior medial/lateral thalamus; ZI: zona incerta; fr: fasciculus retroflexus; mmt: mammillothalamic tract.
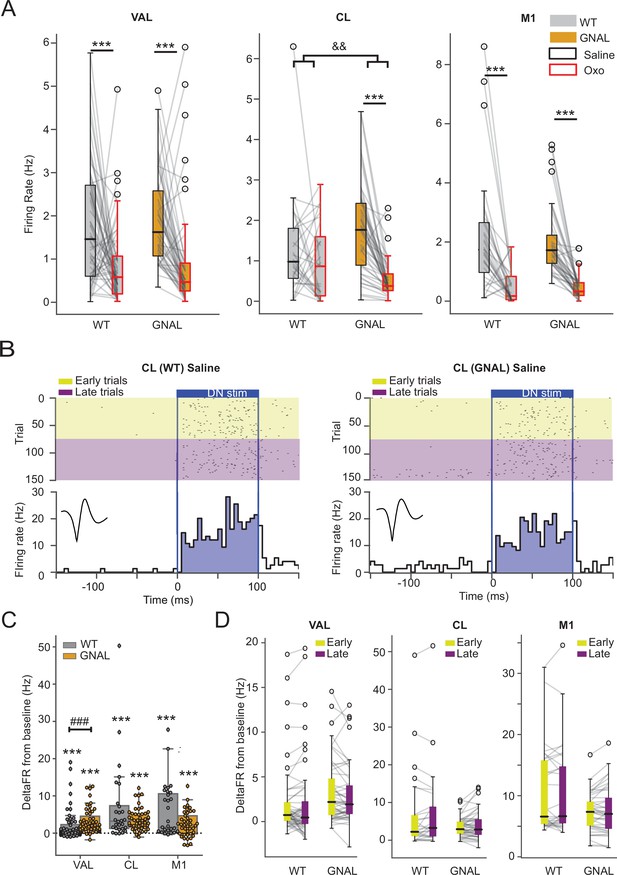
Effects of initial exposure to oxotremorine and dentate nucleus (DN) stimulation on the thalamo-cortical network in wildtype (WT) and Gnal+/- mice.
(A) Comparison of mean firing rate under saline and oxotremorine conditions in ventrolateral thalamus (VAL), centrolateral thalamus (CL), and primary motor cortex (M1). *p<0.05, **p<0.01, ***p<0.001 paired difference between treatment for the cells, &&p<0.01 interaction between treatment and genotypes. (B) Example of peristimulus time histogram (PSTH) and corresponding raster plot, centered on the onset of the cerebellar stimulation, of a CL neuron under saline condition, from a WT mouse (left) and Gnal+/- mouse (right). Inset represents the average waveform of the neuron. The first half of the stimulations is categorized as early (yellow) and the second half as late (purple). (C) Distributions of responses to DN stimulation under saline condition in VAL, CL, M1 (same as Figure 2C), here with comparisons between genotypes. Wilcoxon test for paired samples *p<0.05, **p<0.01, ***p<0.001 and Mann–Whitney test for independent samples ###p<0.001. (D) Comparison of the responses between early and late DN stimulation under saline condition, showing no evolution of responses due to low-frequency repetitive DN stimulation. No significant difference according to Wilcoxon test.
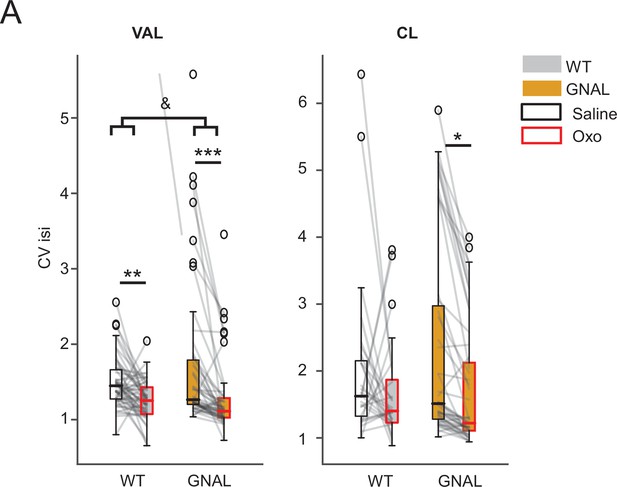
Effects of initial exposure to oxotremorine on the regularity of thalamic firing patterns in wildtype (WT) and Gnal+/- mice.
(A) Comparison of CVisi under saline and oxotremorine conditions in ventrolateral thalamus (VAL) and centrolateral thalamus (CL). *p<0.05, **p<0.01, ***p<0.001 paired difference between treatment for the cells, &p<0.05 interaction between treatment and genotypes.
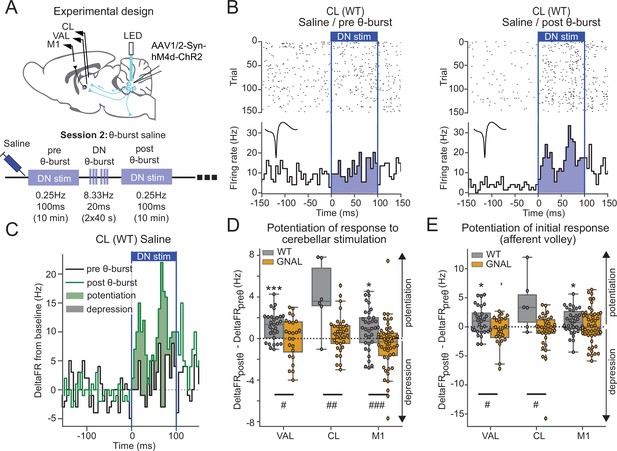
Asymptomatic Gnal+/- mice display an abnormal cerebello-thalamic plasticity induced by dentate nucleus (DN) θ-bursts.
(A) Schematics describing the experimental design of thalamo-cortical recordings coupled with dentate nucleus-centrolateral thalamus (DN-CL) stimulation (top) and experimental timeline (bottom). All the mice received oxotremorine (0.1 mg/kg) in session 1, which took place 2–3 days before. (B) Example of peristimulus time histogram (PSTH) and corresponding raster plot, centered on the onset of the cerebellar stimulation, of a CL neuron from a wildtype (WT) mouse under saline condition, before θ-burst (left) and after θ-burst (right). Inset represents the average waveform of the neuron. (C) Overlay of PSTHs for the neuron shown in panel (B); the difference between the histograms is filled to highlight the potentiation or depression of the responses. (D) Impact of θ-burst stimulations administered in saline condition on the response to 100 ms DN stimulations. (E) Impact of θ-burst stimulations administered in saline condition on the afferent volley in response to 100 ms DN stimulation. Wilcoxon test for paired samples *p<0.05, **p<0.01, ***p<0.001. Mann–Whitney test for independent samples #p<0.05, ##p<0.01, ###p<0.001 difference between genotypes.
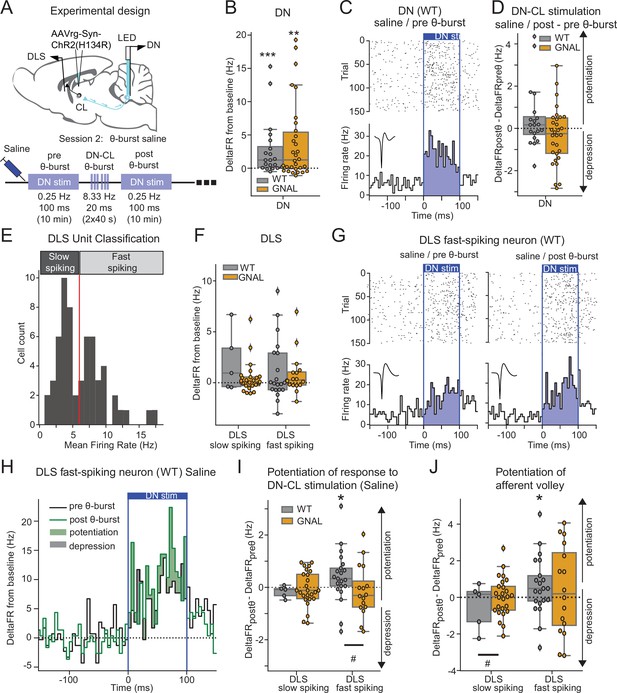
Dentate nucleus-centrolateral thalamus (DN-CL) θ-bursts potentiate the response of fast-spiking dorsolateral striatum (DLS) neurons to DN stimulation in WT wildtype (mice), but not in Gnal+/- mice.
(A) Schematics describing the experimental design of cerebello-striatal recordings coupled with DN-CL stimulation (top) and experimental timeline (bottom). (B) Distributions of response amplitude in the DN to optogenetic stimulation in saline condition, before θ-bursts. (C) Example of peristimulus time histogram (PSTH) and corresponding raster plot, centered on the onset of the cerebellar stimulation, of a DN neuron from a WT mouse under saline condition, before θ-bursts. Inset represents the average waveform of the neuron. (D) Impact of θ-burst stimulations administered in saline condition on the amplitude of response to 100 ms DN-CL stimulation. (E) Illustration of the criteria used to classify DLS neurons as slow spiking and fast spiking based on their mean firing rate using a threshold of 6 Hz (red line). (F) Distributions of striatal responses to DN-CL stimulation in saline condition, before θ-burst. (G) Example of a PSTH and corresponding raster plot, centered on the onset of the DN-CL stimulation, of a DLS fast-spiking neuron from a WT mouse under saline condition, before θ-burst (left) and after θ-burst (right). Inset represents the average waveform of the neuron. (H) Overlay of the PSTHs from panel (G); the difference between the histograms is filled to highlight the potentiation or depression of responses. (I) Impact of θ-burst stimulations administered in saline condition on the response to 100 ms DN-CL stimulation. (J) Impact of θ-burst stimulations administered in saline condition on the afferent volley in response to 100 ms DN-CL stimulation. Wilcoxon test for paired samples *p<0.05, **p<0.01, ***p<0.001. Mann–Whitney test for independent samples #p<0.05, ##p<0.01, ###p<0.001 for differences between genotypes.
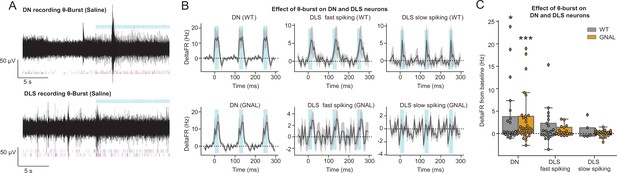
Dentate nucleus-centrolateral thalamus (DN-CL) θ-bursts elicit excitation in DN neurons and dorsolateral striatum (DLS) fast-spiking neurons.
(A) Example high-pass-filtered traces recorded in the DN and DLS during θ-burst, with the corresponding spike-sorted units, showing an increased firing rate induced by θ-burst. θ-burst stimulations are represented by the blue lines. (B) Average peristimulus time histograms (PSTHs) (mean ± SD) of DN, DLS fast-spiking and slow-spiking neurons during DN-CL θ-burst for wildtype (WT) mice (top) and Gnal+/- (bottom). (C) Distributions of responses to DN-CL stimulation under saline condition in DN, DLS fast-spiking and slow-spiking neurons. Wilcoxon test for paired samples *p<0.05, **p<0.01, ***p<0.001. Mann–Whitney test for independent samples #p<0.05, ##p<0.01, ###p<0.001 difference between genotypes.
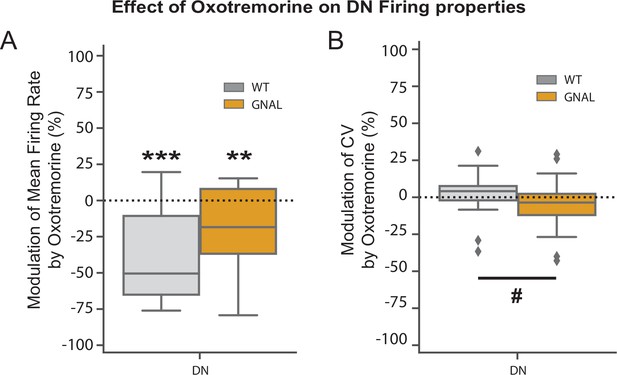
Effect of oxotremorine on the firing of the dentate nucleus (DN).
(A) Modulation of mean firing rate and (B) CV in the DN caused by oxotremorine administration. Wilcoxon test for paired samples *p<0.05, **p<0.01, ***p<0.001. Mann–Whitney test for independent samples #p<0.05, ##p<0.01, ###p<0.001 difference between genotypes.
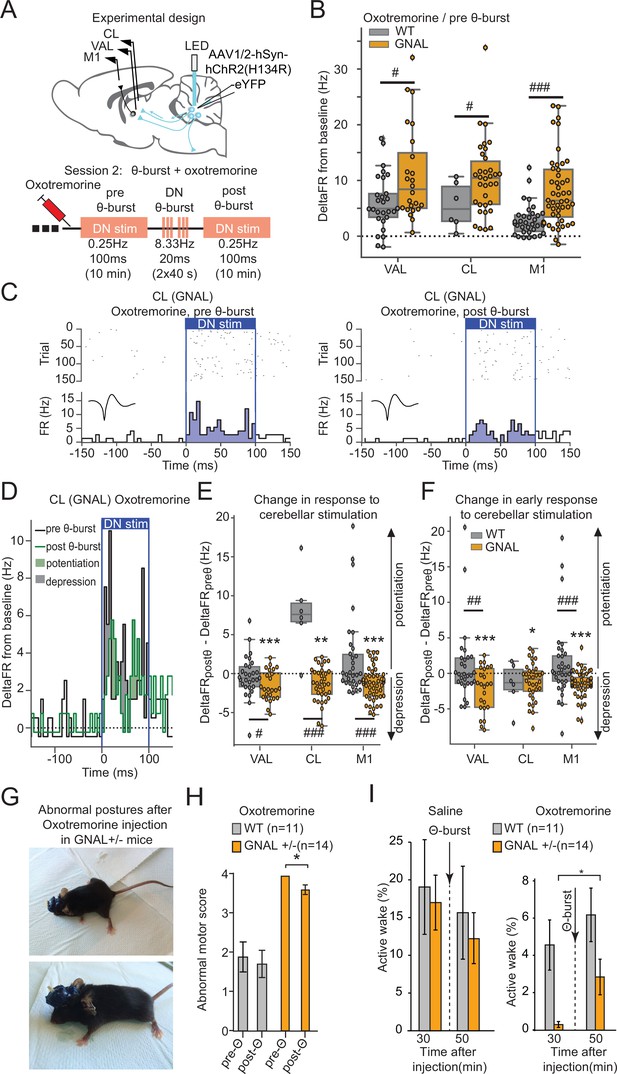
Dentate nucleus (DN) θ-bursts administered to symptomatic Gnal+/- mice induce a decreased response to DN stimulation and decrease motor symptoms of dystonia.
(A) Schematics describing the experimental design of thalamo-cortical recordings coupled with DN stimulation (top) and experimental timeline (bottom). (B) Distributions of responses to DN stimulation in oxotremorine condition, before θ-burst. (C) Example peristimulus time histogram (PSTH) and corresponding raster plot, centered on the onset of the cerebellar stimulation of a centrolateral thalamus (CL) neuron from a Gnal+/- mouse under oxotremorine condition, before θ-burst (left) and after θ-burst (right). Inset represents the average waveform of the neuron. (D) Overlay of the PSTHs from panel (C); the difference between the histograms is filled to highlight the potentiation or depression of the responses. (E) Impact of θ-burst stimulations administered in oxotremorine condition on the response to +/-ms DN stimulation. (F) Impact of θ-burst stimulations administered in oxotremorine condition on the afferent volley in response to 100 ms DN stimulation. Wilcoxon test for paired samples *p<0.05, **p<0.01, ***p<0.001; Mann–Whitney test for independent samples #p<0.05, ##p<0.01, ###p<0.001 difference between genotypes. (G) Examples of dystonic postures in Gnal+/- mice following oxotremorine administration. (H) Average dystonia scores in Gnal+/- and wildtype (WT) mice following oxotremorine administration before and after DN θ-burst stimulations. (I) Change of average active wake percentage after one session of DN θ-burst stimulations in Gnal+/- and WT mice. Wilcoxon test *p<0.05 difference between pre- and post-θ-burst stimulations.
Additional files
-
Supplementary file 1
Tables of statistical results for all comparisons reported in the text.
- https://cdn.elifesciences.org/articles/79135/elife-79135-supp1-v1.xlsx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/79135/elife-79135-mdarchecklist1-v1.docx





