Hepatic AMPK signaling dynamic activation in response to REDOX balance are sentinel biomarkers of exercise and antioxidant intervention to improve blood glucose control
Figures
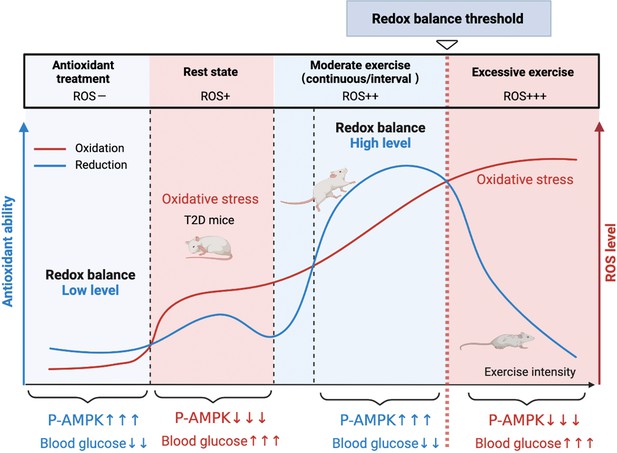
A model of the different mechanisms of exercise and antioxidant intervention in diabetes.
A graphical abstract of this study. Moderate exercise upregulated compensatory antioxidant capability and reached a high-level redox balance, whereas antioxidant intervention achieved a low-level redox balance by inhibiting oxidative stress for treating diabetes. ROS: reactive oxygen species; AMPK: AMP-activated protein kinase.
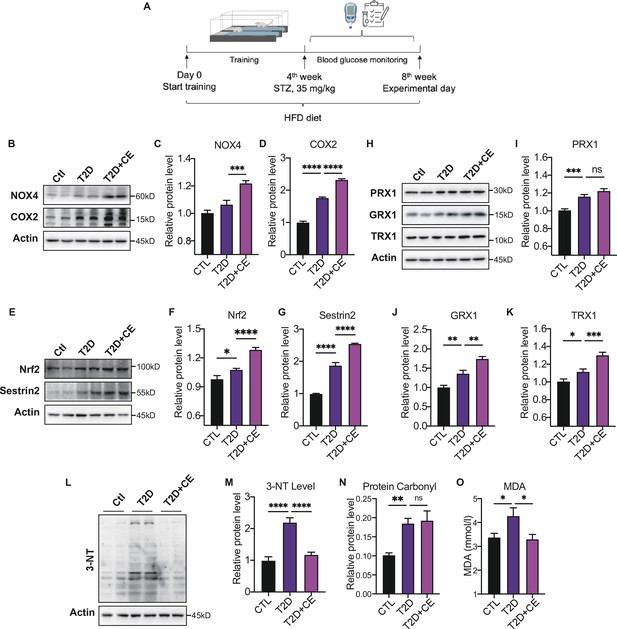
Moderate exercise induced reactive oxygen species (ROS) production in exercise group and increased the antioxidant status.
(A) Experimental design. Type 2 diabetic model (T2DM) rats model was fed by high-fat diet plus a low dose of streptozotocin (STZ) injection (35 mg/kg). The high-fat diet (HFD, 60% calories from fat) was started from the 1st week to the 8th week. The exercise intervention was started from 1st week to 4th week. (B–D). Representative protein level and quantitative analysis of NADPH oxidase 4 (NOX4) (67 kDa), cyclooxygenase 2 (COX2) (17 kDa) and Actin (45 kDa) in the rats in the control (Ctl), T2D, and T2D+continuous exercise (CE) groups.(E–G). Representative protein level and quantitative analysis of Nrf2(97 kDa), Sestrin2 (56 kDa) and Actin (45 kDa) in the rats in the Ctl, T2D, and T2D+CE groups.(H–K). Representative protein level and quantitative analysis of PRX1 (27 kDa), Grx1 (17 kDa), Trx1 (12 kDa), and Actin (45 kDa) in the rats in the Ctl, T2D and T2D+CE groups. The rat livers were homogenized by 1% SDS and analyzed by Western blots with the appropriate antibodies. (L–M). Representative protein level and quantitative analysis of 3-NT and Actin (45 kDa) in the rat in the Ctl, T2D and T2D+CE groups. (N–O). Liver protein carbonylation (N) and MDA content (O) level was detected in the rats of Ctl, T2D, T2D+CE groups. (ns: not significant; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 compared with all groups by one-way ANOVA and Tukey’s post hoc test; data are expressed as the mean ± SEM; n=4–8 per group).
-
Figure 2—source data 1
Full western blot images.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig2-data1-v2.pdf
-
Figure 2—source data 2
Normalized grey value of western blot data.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig2-data2-v2.xlsx
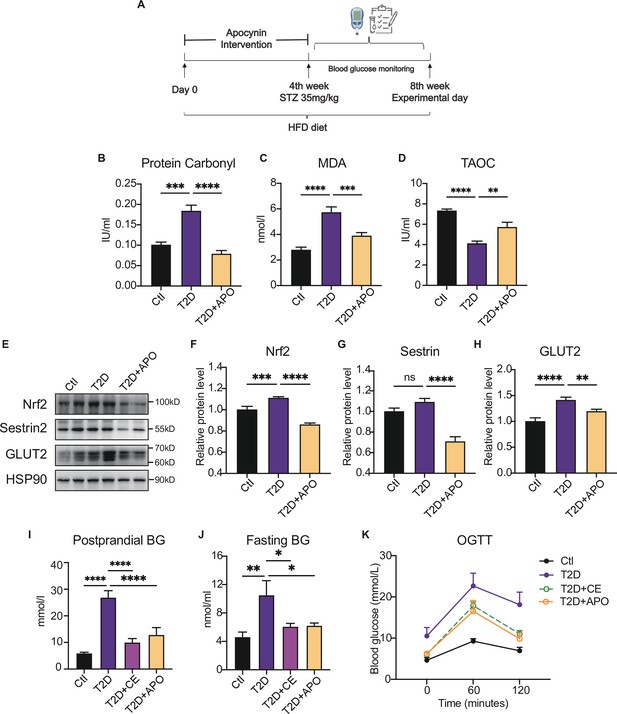
Antioxidant intervention alleviates blood glucose through promoting the upregulation of reducing levels.
(A) Experimental design. Type 2 diabetic model (T2DM) rats model was fed by high-fat diet plus a low dose of streptozotocin (STZ) injection (35 mg/kg). The apocynin intervention was started from 1st week to 4th week. (B–D) Liver protein carbonylation (B), MDA content (C) and TAOC (D) level were detected in the rats of control (Ctl), T2D and T2D+APO groups. (E–H) Representative protein level and quantitative analysis of Nrf2 (97 kDa), Sestrin2 (57 kDa), Glut2 (60–70 kDa), and HSP90 (90 kDa) in the rats in the Ctl, T2D and T2D+APO groups. (I) Postprandial blood glucose levels of Ctl, T2D, T2D+continuous exercise (CE) and T2D+APO groups at the end of 8th week. (J) Fasting blood glucose levels of Ctl, T2D, T2D+CE and T2D+APO groups at the end of 8th week. (K) Blood glucose level after oral glucose administration (0 min, 60 min, and 120 min) in Ctl, T2D, T2D+CE and T2D+APO groups at the end of 8th week (*p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 compared with all groups by one-way ANOVA and Tukey’s post hoc test; data are expressed as the mean ± SEM; n=4–8 per group).
-
Figure 3—source data 1
Full western blot images.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig3-data1-v2.pdf
-
Figure 3—source data 2
Normalized protein carbonylation, MDA content, TAOC, grey value of western blot data and blood glucose level.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig3-data2-v2.xlsx
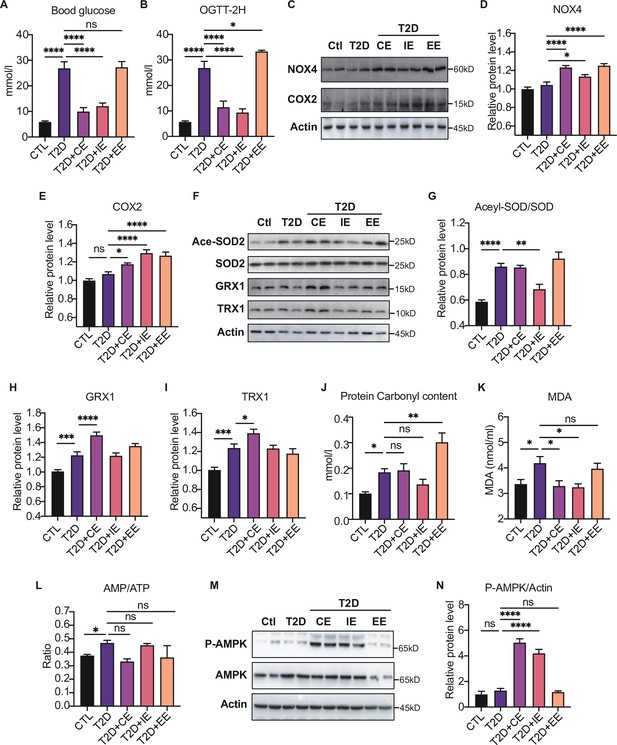
Moderate exercise-generated reactive oxygen species (ROS) promotes activation of AMP-activated protein kinase (AMPK) by phosphorylation and reduces blood glucose level, while excessive exercise- generated oxidative stress reduces AMPK expression and exacerbates diabetes.
(A) Postprandial blood glucose levels of control (Ctl), type 2 diabetic (T2D), T2D+continuous exercise (CE), T2D+intermittent exercise (IE) and T2D+excessive exercise (EE) groups at the end of 8th week. (B) Blood glucose level after oral glucose administration in Ctl, T2D, T2D+CE, T2D+IE and T2D+EE groups at the end of 8th week. (C–E) Representative protein level and quantitative analysis of NADPH oxidase 4 (NOX4) (67 kDa), cyclooxygenase 2 (COX2) (17 kDa) and HSP90 (90 kDa) in the rats in the Ctl, T2D, T2D+CE, T2D+IE, and T2D+EE groups. (F–I) Representative protein level and quantitative analysis of Ace-SOD2 (27 kDa), SOD2 (17 kDa), Grx1 (17 kDa), Trx1 (12 kDa) and Actin (45 kDa) in the rats in the Ctl, T2D, T2D+CE, T2D+IE and T2D+EE groups. (J–L) Liver protein carbonylation content (J), liver MDA content (K) and AMP/ATP ratio (L) were detected in the rats of Ctl, T2D, T2D+CE, T2D+IE and T2D+EE groups. (M–N). Representative protein level and quantitative analysis of P-AMPK (67 kDa), AMPK (67 kDa) and Actin (45 kDa) in the rats in the Ctl, T2D, T2D+CE, T2D+IE and T2D+EE groups. (ns: not significant; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 compared with all groups by one-way ANOVA and Tukey’s post hoc test; data are expressed as the mean ± SEM; n=4–8 per group).
-
Figure 4—source data 1
Full western blot images.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig4-data1-v2.pdf
-
Figure 4—source data 2
Normalized grey value of western blot data.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig4-data2-v2.xlsx
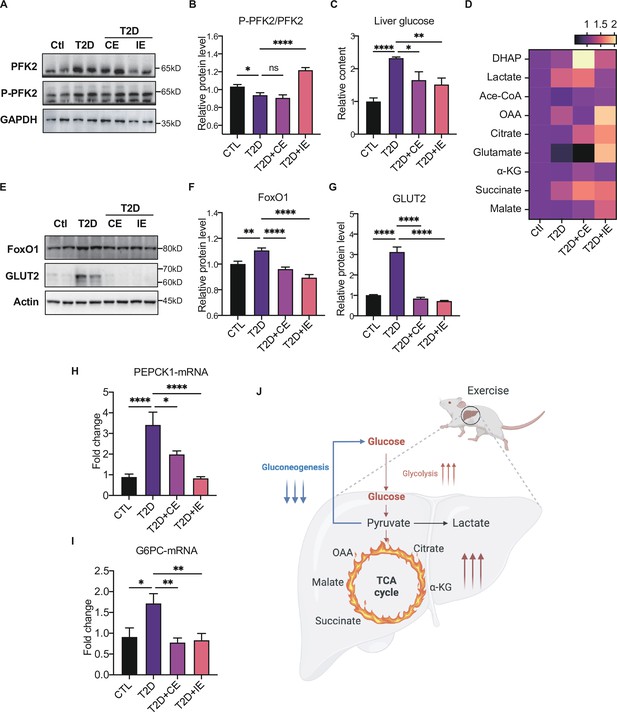
Moderate exercise promoted glycolysis and mitochondrial tricarboxylic acid cycle and inhibited the gluconeogenesis in the liver of diabetic rats.
(A–B) Representative protein level and quantitative analysis of P-PFK2 (64 kDa), PFK2 (64 kDa) and GAPDH (37 kDa) in the rats in the control (Ctl), T2D, T2D+continuous exercise (CE) and T2D+intermitten exercise (IE) groups. (C). Liver glucose level after oral glucose administration in Ctl, T2D, T2D+CE, and T2D+IE groups at the end of 8th week. (D) Relative concentrations of substrates for glycolysis (DHAP and Lactate) and the tricarboxylic acid cycle (citrate, succinate and malate) in the rats of Ctl, T2D, T2D+CE, and T2D+IE groups. The concentration of substrates was analyzed by LC-MS/MS. (E–G) Representative protein level and quantitative analysis of FoxO1 (82 kDa), GLUT2 (60–70 kDa) and Actin (45 kDa) in the rats in the Ctl, T2D, T2D+CE, and T2D+IE groups. (H–I) Expression of hepatic Pepck and G6C mRNA in the Ctl, T2D, T2D+CE, and T2D+IE groups were evaluated by real-time PCR analysis. Values represent mean ratios of Pepck and G6pase transcripts normalized to GAPDH transcript levels. (J) Schematic diagram illustrating the effect of CE and IE on glycolysis, gluconeogenesis and mitochondrial tricarboxylic acid cycle (ns: not significant; *p<0.05, **p<0.01, ****p<0.0001 compared with all groups by one-way ANOVA and Tukey’s post hoc test; data are expressed as the mean ± SEM; n=6–8 per group).
-
Figure 5—source data 1
Full western blot images.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig5-data1-v2.pdf
-
Figure 5—source data 2
Normalized grey value of western blot and mRNA data.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig5-data2-v2.xlsx
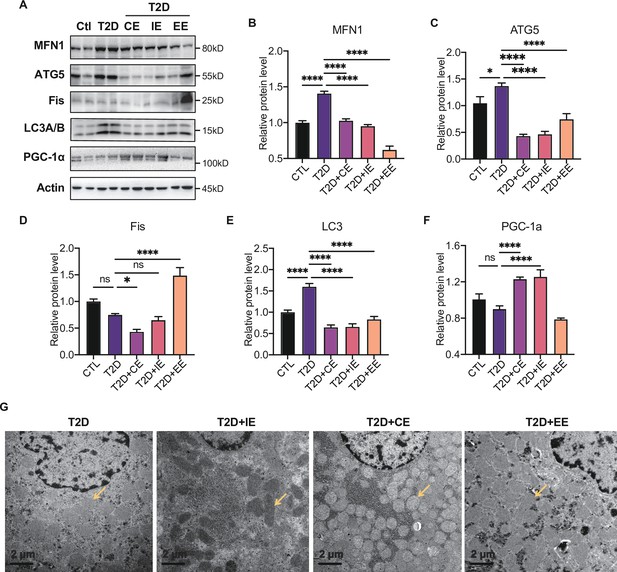
Moderate exercise inhibited hepatic mitophagy, while excessive exercise exhibited opposite effect and inhibited the mitochondrial biogenesis.
(A–F) Representative protein level and quantitative analysis of MFN1 (82 kDa), ATG5 (55 kDa), FIS (25 kDa), LC3A/B (14,16 kDa), PGC-1α (130 kDa), and Actin (45 kDa) in the rats in the control (Ctl), T2D, T2D+continuous exercise (CE), T2D+IE, and T2D+excessive exercise (EE) groups. (G) Transmission electron microscope (TEM) analysis of the ultrastructure of hepatocytes in the rats in the T2D, T2D+CE, T2D+IE, and T2D+EE groups (The yellow arrows point to mitochondria). (Scale bar = 2 μm; ns: not significant; *p<0.05, **p<0.01, ***p<0.001, ****p<0.0001 compared with all groups by one-way ANOVA and Tukey’s post hoc test; data are expressed as the mean ± SEM; n=8 per group).
-
Figure 6—source data 1
Full western blot images.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig6-data1-v2.pdf
-
Figure 6—source data 2
Normalized grey value of western blot data.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig6-data2-v2.xlsx
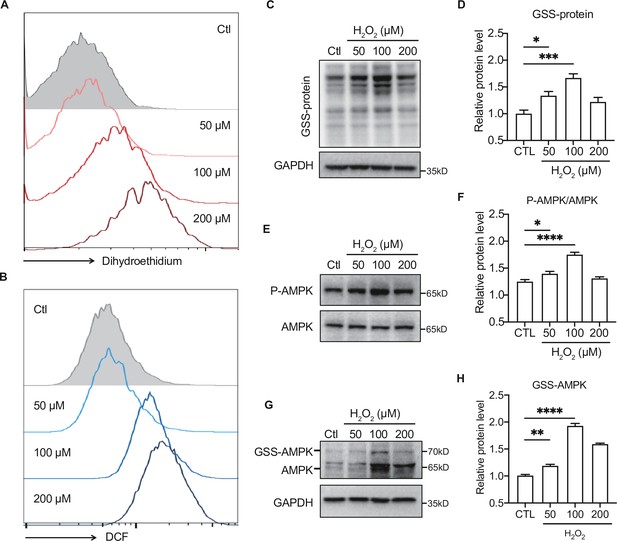
Moderate reactive oxygen species (ROS) activates AMP-activated protein kinase (AMPK) through GRX-mediated glutathionylation.
(A–B) Analysis of superoxide (A) and ROS (B) generation using hydroethidine (A) and H2DCFDA (B) probes in primary hepatocytes under H2O2 stress (50–200 μmol/L, 30 min). The fluorescence intensity was detected by flow cytometry. (C–D) Representative protein level and quantitative analysis of GSS-adduct protein and GAPDH in primary hepatocytes under H2O2 stress (50–200 μmol/L). Hepatocytes loaded with EE-GSH-biotin were incubated with/without H2O2 for 30 min, and the amounts of GSS-protein adduct formation were determined using non-reducing SDS-PAGE and Western blot analysis with streptavidin-HRP. (E–F) Representative protein level and quantitative analysis of P-AMPK (67 kDa) and AMPK (67 kDa) in primary hepatocytes under H2O2 stress (50–200 μmol/L, 30 min). (G–H). AMPK cysteine gel shift immunoblot. Cysteine dependent shifts by incubation of AMPK protein with glutathione reductase and PEG-Mal. PEG2-mal labelled glutathionylation modification shifts AMPK by ~10 kDa above the native molecular weight. Representative protein level and quantitative analysis of GSS-AMPK (72 kDa), AMPK (67 kDa) and GAPDH in primary hepatocytes under H2O2 stress.
-
Figure 7—source data 1
Full western blot images.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig7-data1-v2.pdf
-
Figure 7—source data 2
Normalized grey value of western blot data.
- https://cdn.elifesciences.org/articles/79939/elife-79939-fig7-data2-v2.xlsx
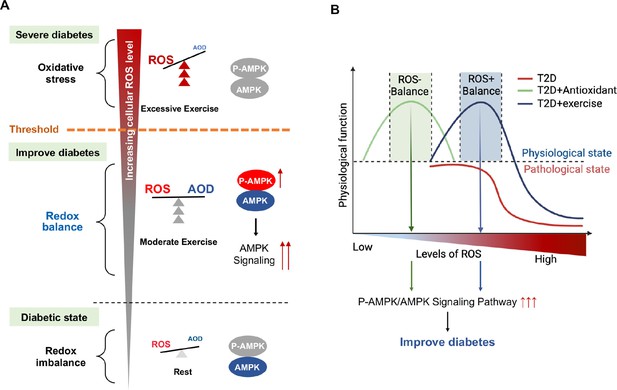
Schematic diagram of redox balance threshold.
(A) The increased reactive oxygen species (ROS) and/or decreased antioxidative capacity (AOD) cause an imbalanced redox state and declined AMP-activated protein kinase (AMPK) activity in diabetic individuals. Moderate exercise promotes the activity of antioxidant enzymes by generating benign ROS to reach redox balance, and directly promotes AMPK signaling, thus reducing glucose levels in the blood and liver. Excessive exercise causes excess ROS and exceeds the redox balance threshold, inhibiting AMPK activity and expression, thus leading to exacerbation of diabetes. (AMPK and P-AMPK in gray circles indicate decrease, red circles indicate increase, blue circles indicate no significant changes). (B) Dose-response curve of diabetic individuals with exercise and antioxidant intervention.The state of diabetic individuals is applicable to the description of a S-shaped curve, due to the high level of oxidative stress and decreased reduction level in diabetic individuals. With the increase of ROS, the physiological function of diabetic individuals gradually decreases. Moderate exercise shifts the S-shaped curve upward and to the right, forming a bell-shaped dose-response curve, thus reducing the sensitivity to oxidative stress in diabetic individuals and restoring redox homeostasis. However, with excessive exercise, ROS production increases beyond the threshold range of redox balance, resulting in decreased physiological function. The ROS at the peak of the bell-shaped curve for antioxidant interventions (optimal physiological activity) is lower than the ROS at the peak for moderate exercise. The intervals on either side of the peak correspond to the range of redox balance thresholds, where antioxidant interventions are at low levels of redox balance and exercise is at high levels of redox balance.
Additional files
-
MDAR checklist
- https://cdn.elifesciences.org/articles/79939/elife-79939-mdarchecklist1-v2.pdf
-
Supplementary file 1
Antibody information (a) and chow and HFD diet nutrition composition (b).
- https://cdn.elifesciences.org/articles/79939/elife-79939-supp1-v2.docx





