Rapid and precise genome engineering in a naturally short-lived vertebrate
Figures
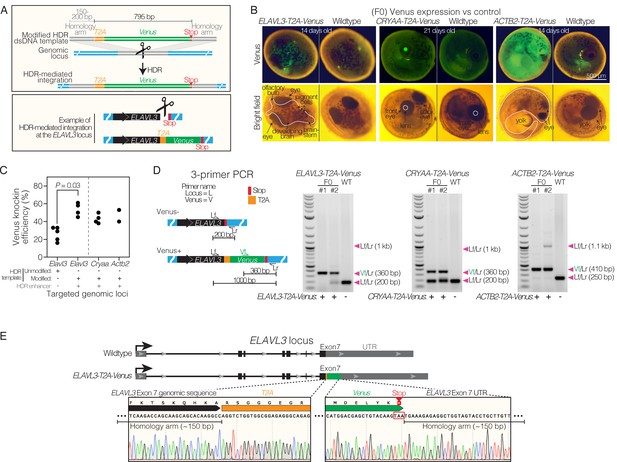
Efficient homology directed repair for precise knock-in at different genomic locations in killifish.
(A) Schematic of T2A-Venus insertion at the ELAVL3 locus. (B) Images of F0 Venus positive and wildtype 14–21-day-old embryos for each targeted locus (ELAVL3, CRYAA, and ACTB2). Twenty-one-day-old embryos were dried on coconut fiber for 7 days prior to imaging and have altered autofluorescence compared to 14-day-old embryos that were not yet put on coconut fiber. See Figure 1—figure supplement 1. (C) Efficiency of T2A-Venus knock-in at each locus (determined by visual inspection of Venus fluorescence in developed embryos) and efficiency of knock-in at ELAVL3 with a dsDNA HDR template lacking chemical modification and without the small molecule HDR enhancer; 2–5 independent injection replicates per condition; n=32–249 injected embryos per replicate. P-value calculated using a two-tailed Mann-Whitney test. See Figure 1—figure supplement 2. Raw data in Supplementary file 4. (D) Left, 3-primer PCR schematic showing locus-specific external primers forward (Lf) and reverse (Lr) and internal forward Venus primer (Vf). Right, gel images of 3-primer PCR for each locus comparing F0 with wildtype (WT) fish. Arrowheads indicate each primer pair and its expected amplification product length. Scoring Venus positive (+) or negative (-) for each fish is indicated below the gel images. Note that the relatively large ~1 kb Lf/Lr product in the transgenic F0 animals is likely to be outcompeted by the shorter Vf/Lr amplification product during the PCR reaction. Raw gel image in Figure 1—source data 1 and Figure 1—source data 2. (E) Top, comparison of ELAVL3 locus for wildtype and ELAVL3-T2A-Venus. Bottom, precise in-frame insertion of T2A-Venus in exon 7, immediately before the stop codon of ELAVL3 and followed by the ELAVL3 untranslated region (UTR).
-
Figure 1—source data 1
Original unedited gel shown in panel D.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig1-data1-v1.zip
-
Figure 1—source data 2
Original unedited gel with relevant bands labeled shown in panel D.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig1-data2-v1.zip
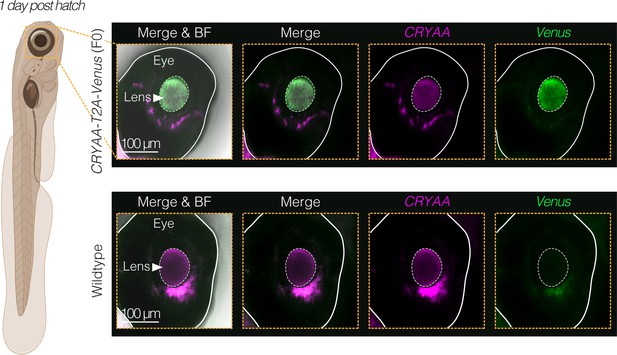
Venus expression in F0 CRYAA-T2A-Venus transgenic animals.
In situ hybridization (via HCR) of sagittal sections of the eye of larval stage (1 day post hatching) (top row) F0 CRYAA-T2A-Venus and (bottom row) wildtype killifish showing merged images of the Venus transcript (green) and the CRYAA transcript (magenta) with and without bright field illumination (BF) as well as images from separate channels. The bright signal below the lens appears in all channels, suggesting it is autofluorescence. Scale bar = 100 μm. Fry schematic created with BioRender.com.
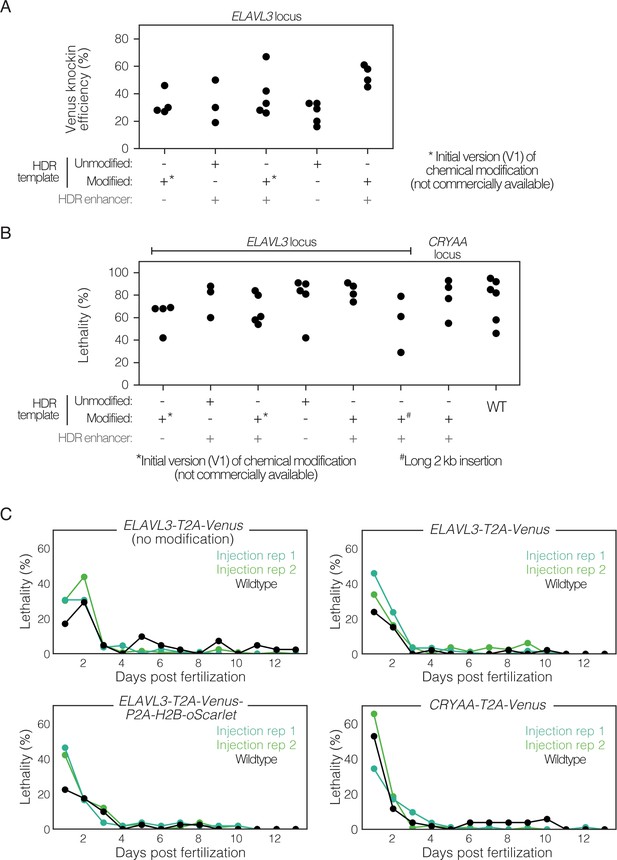
Comparison of knock-in efficiency and embryo lethality using chemically modified dsDNA HDR templates and small molecule HDR enhancer.
(A) Knock-in efficiency when using different knock-in reagents for insertion of T2A-Venus at the ELAVL3 locus – chemically modified dsDNA HDR templates versus unmodified dsDNA HDR templates, and the use of small molecule HDR enhancers (+/-HDR enhancer). Two types of HDR modifications were tested: the initial (V1) version of the chemical modification which is not commercially available (indicated by *) and the commercially available chemical modification (i.e. Alt-R HDR Donor Blocks, see Materials and methods). The commercially available modification was used for all subsequent work in this paper (see Materials and methods). Note that the condition (-) HDR enhancer (+) Alt-R HDR Donor Blocks was not tested. 3–5 independent injection replicates per condition; n=50–249 injected embryos per replicate. Each dot represents the efficiency for a replicate. Raw data in Supplementary file 4. (B) Total embryo lethality over two weeks after CRISPR/Cas9-mediated knock-in with different knock-in reagents for insertion of T2A-Venus at the ELAVL3 locus, compared to non-injected wildtype control embryos. The initial (V1) version of the chemical modification which is not commercially available is indicated by (*). For one condition, a longer 2 kb insertion (T2A-Venus-P2A-H2B-oScarlet) was used indicated by (#). 3–6 independent injection replicates per condition; n=50–249 injected embryos per replicate. Each dot represents the lethality for a replicate. No significant difference between groups (one-way ANOVA; p-value = 0.4). Raw data in Supplementary file 4. (C) Embryo daily lethality over two weeks after fertilization and injection with CRISPR/Cas9-mediated knock-in reagents (two injection replicates; green shades) and a matched (same clutch of embryos) non-injected wildtype control (black) for four different injection conditions. Each dot represents lethality for a replicate; n=40–121 embryos per replicate. Raw data in Figure 1—figure supplement 2—source data 1.
-
Figure 1—figure supplement 2—source data 1
Raw data for panel C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig1-figsupp2-data1-v1.zip
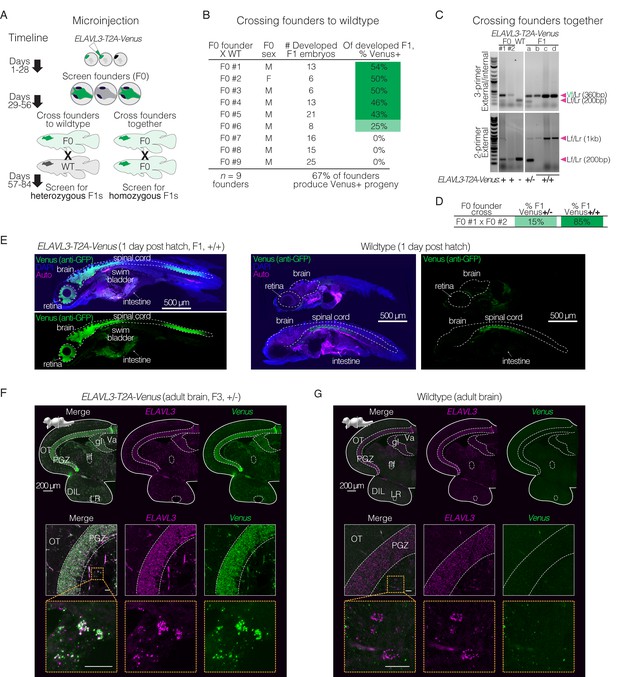
Rapid generation of stable knock-in lines in the killifish.
(A) Schematic of generating a stable knock-in line by either crossing F0 X WT (left) or F0 X F0 (right), with timelines to verified heterozygous or homozygous animals. (B) Germline transmission of T2A-Venus at the ELAVL3 locus was verified by crossing F0 X WT (n=9 breeding pairs), with 6–25 developed embryos per pair screened visually or by PCR. (C) Crossing F0 animals positive for T2A-Venus at the ELAVL3 locus (F0 X F0). Gels showing F0 parents (left) and F1 progeny (a, b, c, and d; right) with 3-primer PCR (top) and external PCR (bottom) using Venus and locus-specific primers shown in Figure 1D. The external PCR shows both heterozygous (a) and homozygous (b, c, and d) F1 progeny for ELAVL3-T2A-Venus. Arrowheads indicate each primer pair and its expected amplification product length. Scoring for each lane of the gel is indicated below the gel images. F0 animals are likely mosaic so only ‘+’ or ‘-’ was assigned based on the 3-primer PCR result. See Figure 2—figure supplement 1 and Figure 2—figure supplement 2. Raw gel images in Figure 2—source data 1; Figure 2—source data 2 and Figure 2—source data 3. (D) Percent of fully developed and Venus-positive F1 progeny from the F0 X F0 cross that are heterozygous (+/-) or homozygous (+/+) for insertion of Venus in ELAVL3-T2A-Venus animals. (E) Immunofluorescence of sagittal sections of larval (1 day post hatch) F1 homozygous ELAVL3-T2A-Venus (left) compared with wildtype (right) killifish showing merged images of Venus (stained with anti-GFP antibody; green), DAPI (nuclei; blue), and autofluorescence (‘Auto’; magenta) as well as separate images from different channels for Venus (stained with anti-GFP antibody; green). In both ELAVL3-T2A-Venus and wildtype individuals, we observe background green signal for example in the intestine and ventral to the spinal cord. This background signal is not identical between ELAVL3-T2A-Venus and wildtype samples, likely due to slight differences in depth of the sagittal slice. Scale bar = 500 μm. (F) Top row, in situ hybridization (via HCR) of coronal brain section of an adult (3 months old) F3 heterozygous ELAVL3-T2A-Venus male showing merged images of Venus transcript (green) and the ELAVL3 transcript (magenta) as well as images from separate channels. Scale bar = 200 μm. Upper left corner, sagittal view of the killifish brain indicating the plane of the coronal section. Middle row, zoom in on the periglomerular gray zone (PGZ) of the optic tectum (OT). Bottom row, zoom in on individual ELAVL3-expressing cells. While ELAVL3 (magenta) and Venus (green) signal largely colocalize, there is some non-colocalizing background signal. This background is similar to signal observed in wildtype animals (see G). Scale bar = 20 μm. See Figure 2—figure supplement 3. (G) Top row, in situ hybridization (via HCR) of coronal brain section of an adult (3 months old) wildtype male showing merged images of Venus transcript (green) and the ELAVL3 transcript (magenta) as well as images from separate channels. Scale bar = 200 μm. Upper left corner, sagittal view of the killifish brain indicating the plane of the coronal section. Middle row, zoom in on the periglomerular gray zone (PGZ) of the optic tectum (OT). Bottom row, zoom in on individual ELAVL3-expressing cells. We observe some background green signal in wildtype animals; however, overall green signal is much less than what is observed in the ELAVL3-T2A-Venus animals (F). Scale bar = 20 μm.
-
Figure 2—source data 1
Original unedited gel shown in panel C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig2-data1-v1.zip
-
Figure 2—source data 2
Original unedited gel shown in panel C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig2-data2-v1.zip
-
Figure 2—source data 3
Original unedited gel with relevant bands labeled shown in panel C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig2-data3-v1.zip
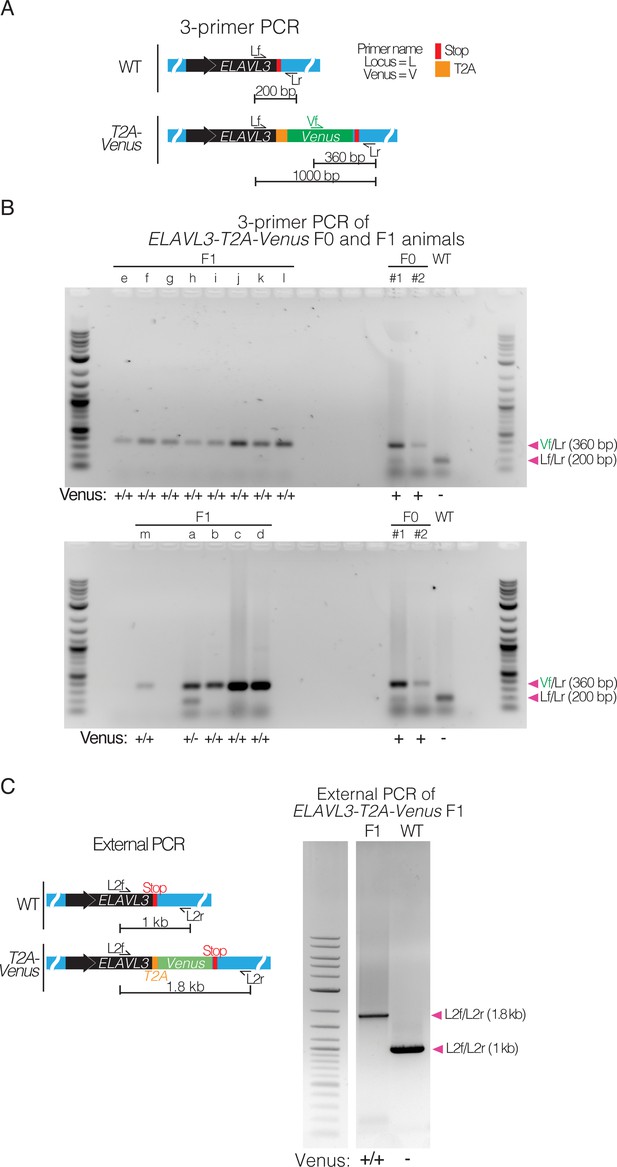
PCR amplification of F0 parents and F1 progeny confirms T2A-Venus integration and germline transmission at the ELAVL3 locus.
(A) 3-primer PCR schematic showing locus-specific external primers forward (Lf) and reverse (Lr) and internal Venus forward primer (Vf) at the ELAVL3 locus. (B) Crossing F0 animals positive for T2A-Venus at the ELAVL3 locus (F0 X F0). Gels showing F0 parents (right) and F1 progeny (a-m; left) with 3-primer PCR using Venus and locus-specific primers shown in (A). Arrowheads indicate each primer pair and its expected amplification product length. Scoring for each lane of the gel is indicated below the gel images. Full gel images of 3-primer PCR on F0 and F1 animals for gels shown in Figure 2C. Raw gel image in Figure 2—figure supplement 1—source data 1 and Figure 2—figure supplement 1—source data 2. (C) PCR amplification at the ELAVL3 locus of F1 homozygous ELAVL3-T2A-Venus killifish compared to WT with forward and reverse primers external to the homology arms (i.e. external PCR). PCR amplification on sample from F1 homozygous ELAVL3-T2A-Venus killifish produces a band at the expected length (1.8 kb). Arrowheads indicate each primer pair and its expected amplification product length. Scoring for each well of the gel is indicated below the gel images. Raw gel image in Figure 2—figure supplement 1—source data 3 and Figure 2—figure supplement 1—source data 4.
-
Figure 2—figure supplement 1—source data 1
Original unedited gel shown in panel B.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig2-figsupp1-data1-v1.zip
-
Figure 2—figure supplement 1—source data 2
Original unedited gel with relevant bands labeled shown in panel B.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig2-figsupp1-data2-v1.zip
-
Figure 2—figure supplement 1—source data 3
Original unedited gel shown in panel C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig2-figsupp1-data3-v1.zip
-
Figure 2—figure supplement 1—source data 4
Original unedited gel with relevant bands labeled shown in panel C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig2-figsupp1-data4-v1.pdf
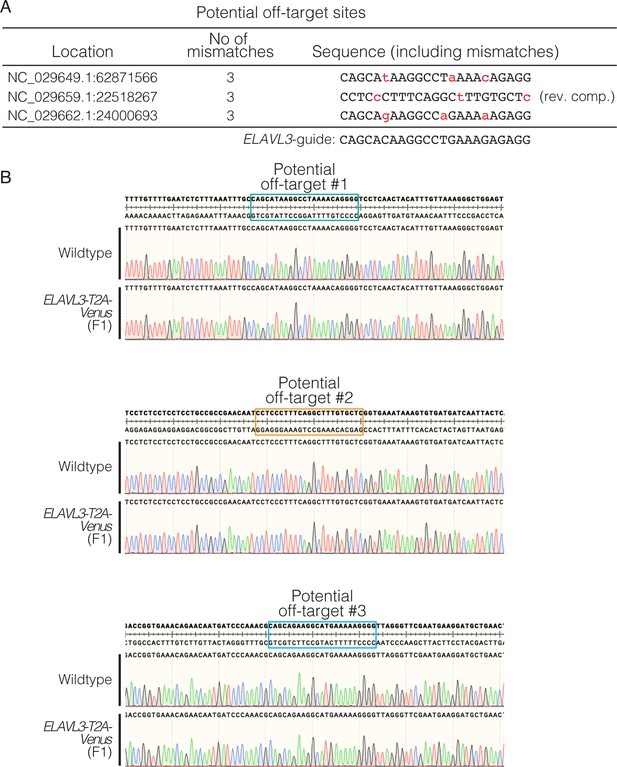
Evaluation of potential off-target effects in homozygous F1 CRISPR/Cas9 knock-in fish.
(A) The predicted most likely off-target sites for the ELAVL3 gRNA (predicted using CHOPCHOP). These three loci each have three mismatches from the ELAVL3 gRNA (indicated by lowercase, red text). (B) Sequencing results of ELAVL3-T2A-Venus homozygous F1 (generated by crossing F0s) at the three predicted most likely off-target sites. No off-target editing was observed at any of the predicted off-target sites.
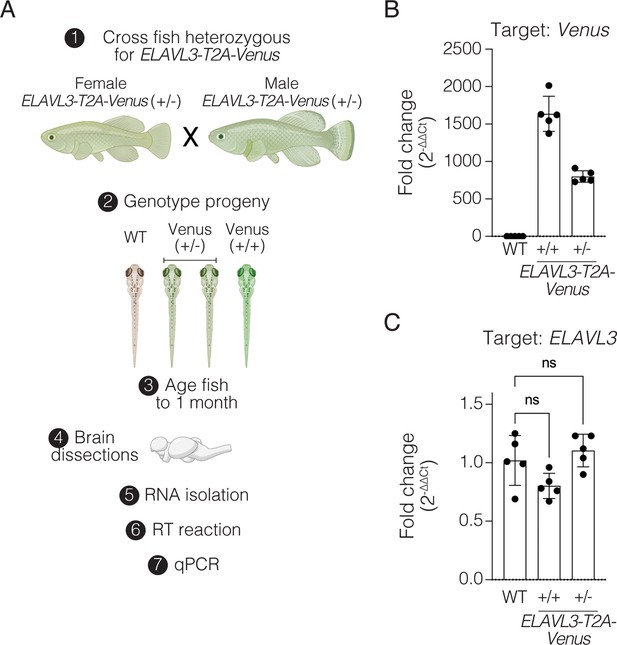
Assessment of Venus and ELAVL3 transcript levels in ELAVL3-T2A-Venus knock-in and wildtype control.
(A) Schematic of experiment: crossing F2 heterozygous ELAVL3-T2A-Venus killifish to generate F3 wildtype, heterozygous, and homozygous siblings for the ELAVL3-T2A-Venus allele followed by genotyping, brain dissections, RNA isolation, reverse transcription (RT), and quantitative PCR (qPCR). Primer pairs for ELAVL3 and housekeeping genes were designed such that they are separated by at least one intron. Primer sets used show no amplification with no RT control. Created with BioRender.com. (B) Fold change of the Venus transcript level in F3 heterozygous and homozygous ELAVL3-T2A-Venus animals relative to wildtype. n=5 animals per genotype. Bar is mean +/- SEM. Each dot represents fold change in transcript level in one fish. Raw data in Figure 2—figure supplement 3—source data 1. (C) Fold change of the ELAVL3 transcript level in F3 heterozygous and homozygous ELAVL3-T2A-Venus animals relative to wildtype. n=5 animals per genotype. Bar is mean +/- SEM. Each dot represents fold change in transcript level in one fish. No significant difference (ns) between groups (one-way ANOVA with Dunnett’s multiple comparison test for statistical comparisons). Raw data in Figure 2—figure supplement 3—source data 1.
-
Figure 2—figure supplement 3—source data 1
Raw data for panels B and C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig2-figsupp3-data1-v1.zip
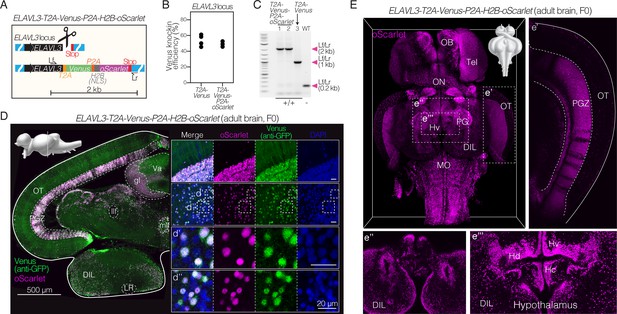
Efficient and stable knock-in of a large 2 kb insertion in killifish.
(A) Schematic of design of a T2A-Venus-P2A-H2B-oScarlet sequence for targeted knock-in at the ELAVL3 locus and locus-specific external primers forward (Lf) and reverse (Lr). (B) Knock-in efficiency comparing 2 kb insertion (ELAVL3-T2A-Venus-P2A-H2B-oScarlet) to the 0.8 kb insertion (ELAVL3-T2A-Venus) determined by visual inspection of developed F0 embryos for Venus fluorescence; 3–4 independent injection replicates per condition; n=80–157 embryos injected per replicate. No significant difference between groups (two-tailed Mann-Whitney test; p-value = 0.6). Data in Supplementary file 4. (C) PCR amplification at the ELAVL3 locus using locus-specific external primers forward (Lf) and reverse (Lr) shown in (A) comparing amplicon length from two F1 ELAVL3-T2A-Venus-P2A-H2B-oScarlet animals (lane 1 and 2), one F1 ELAVL3-T2A-Venus animal (lane 3), and one wildtype animal (lane WT), showing a single band at the expected length in each case. Scoring for each lane of the gel is indicated below the gel image. See Figure 3—figure supplement 1. Raw gel image in Figure 3—source data 1 and Figure 3—source data 2. (D) Left, immunofluorescence of coronal brain section of adult (3 months old) F0 ELAVL3-T2A-Venus-P2A-H2B-oScarlet male, showing expression of Venus and oScarlet. Scale bar = 500 μm. Upper left corner, sagittal view of the killifish brain indicating the plane of the coronal section. Right, select regions showing separate channels for oScarlet (magenta), Venus (stained with anti-GFP antibody; green), DAPI (nuclei; blue) as well as merged channels. (d’) and (d’’): zoomed in individual cells. Scale bar = 20 μm. oScarlet expression is confined to nuclei while Venus expression is observed throughout cell bodies and projections. The DAPI positive, oScarlet/Venus negative cells are likely non-neuronal brain cell types (e.g., astrocytes, oligodendrocytes, etc.). (E) Brain-wide expression of nuclear-localized oScarlet (magenta) in adult (1 month old) F0 ELAVL3-T2A-Venus-P2A-H2B-oScarlet male. Select regions are highlighted: (e’) the optic tectum (OT), (e’’) the most ventral view of the hypothalamus, and (e’’’) the periventricular hypothalamus. Strips in the PGZ that appear to lack oScarlet expression could be due to mosaicism (i.e. lack of knock-in in some cells) in the F0 individual.
-
Figure 3—source data 1
Original unedited gel shown in panel C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig3-data1-v1.zip
-
Figure 3—source data 2
Original unedited gel with relevant bands labeled shown in panel C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig3-data2-v1.pdf
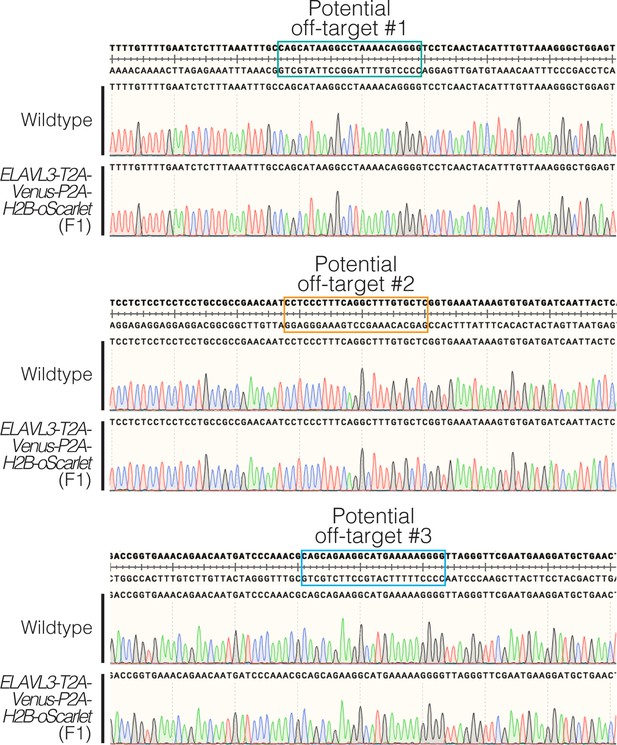
Evaluation of potential off-target effects in homozygous F1 CRISPR/Cas9 knock-in fish.
Sequencing results of ELAVL3-T2A-Venus-P2A-H2B-oScarlet homozygous F1 (generated by crossing F0s) at the three predicted most likely off-target sites. No off-target editing was observed at any of the predicted off-target sites.
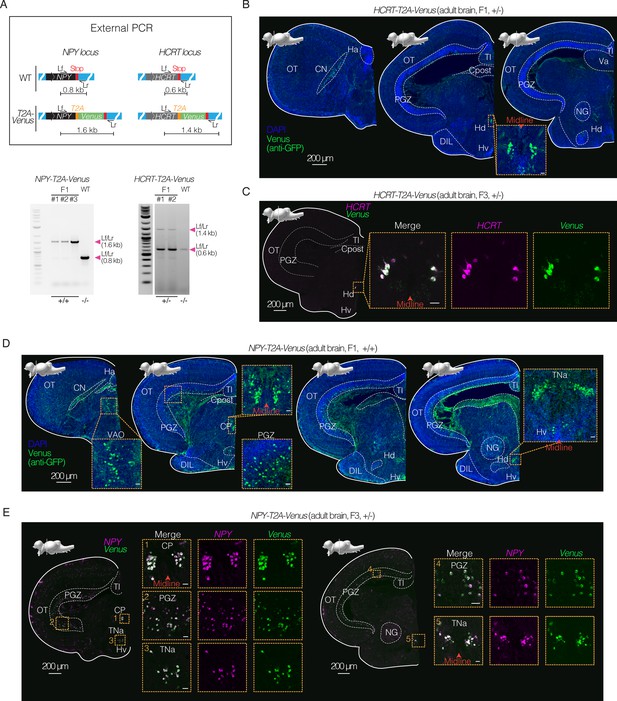
Expression in specific neuronal populations using CRISPR/Cas9 knock-in lines in killifish.
(A) Top, schematics of design of T2A-Venus sequence for targeted knock-in at the NPY and HCRT loci including locus-specific external primers forward (Lf) and reverse (Lr). Bottom, PCR amplification at the NPY or HCRT locus comparing amplicon length from NPY-T2A-Venus (F1 animals) versus wildtype (WT) and comparing amplicon length from HCRT-T2A-Venus (F1 animals) versus wildtype (WT). See Figure 4—figure supplement 1. Raw gel images in Figure 4—source data 1; Figure 4—source data 2 and Figure 4—source data 3. (B) Immunofluorescence of coronal brain sections of adult (4 months old) F1 heterozygous HCRT-T2A-Venus female, showing Venus expression (stained with anti-GFP antibody; green) and DAPI (nuclei; blue). Scale bar = 200 μm. Distinct nuclei indicated and labeled with abbreviated names. Above each slice is the sagittal view of the killifish brain indicating the plane of the coronal section. Inset shows zoom in on Venus positive population of cells in the dorsal hypothalamus close to the midline. Scale bar = 20 μm. See Figure 4—figure supplement 2. (C) In situ hybridization (via HCR) of coronal brain section of adult (4 months old) F3 heterozygous HCRT-T2A-Venus male, showing merged images of Venus transcript (green) and HCRT transcript (magenta). Scale bar = 200 μm. Inset shows zoom in on Venus positive and HCRT-positive population of cells in the dorsal hypothalamus close to the midline with merge and separated channels. Scale bar = 20 μm. See Figure 4—figure supplement 2. (D) Immunofluorescence of coronal brain sections of adult (3.5 months old) F1 homozygous NPY-T2A-Venus male, showing Venus expression (stained with anti-GFP antibody; green) and DAPI (nuclei; blue). Scale bar = 200 μm. Above each slice is the sagittal view of the killifish brain indicating the plane of the coronal section. Insets show zoom in on the Venus-positive populations. Scale bar = 20 μm. See Figure 4—figure supplement 2. (E) In situ hybridization (via HCR) of coronal brain sections of adult (3 months old) F3 heterozygous NPY-T2A-Venus male, showing merged image of Venus transcript (green) and the NPY transcript (magenta) in situ hybridization. Scale bar = 200 μm. Insets show zoom in on Venus-positive and NPY-positive populations. Scale bar = 20 μm. See Figure 4—figure supplement 2 and Figure 4—figure supplement 3.
-
Figure 4—source data 1
Original unedited gel shown in panel A.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig4-data1-v1.zip
-
Figure 4—source data 2
Original unedited gel shown in panel A.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig4-data2-v1.zip
-
Figure 4—source data 3
Original unedited gel with relevant bands labeled shown in panel A.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig4-data3-v1.zip
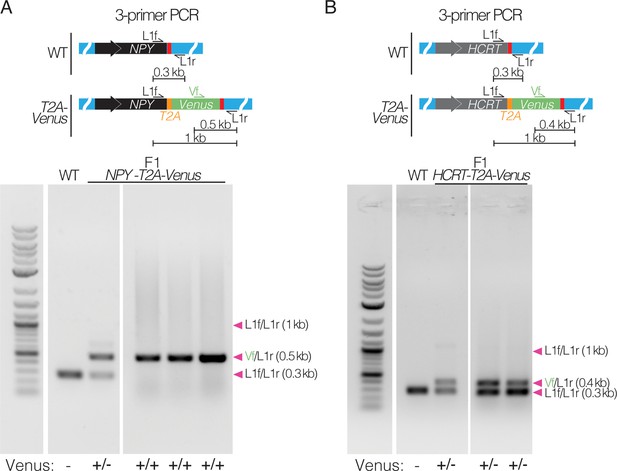
Confirmation of knock-in by PCR amplification and sequencing.
(A) 3-primer PCR amplification at the NPY locus of F1 heterozygous and homozygous NPY-T2A-Venus killifish compared to WT. Arrowheads indicate each primer pair and its expected amplification product length. Scoring Venus negative (-), heterozygous (+/-), or homozygous (+/+) for each well of the gel is indicated below the gel image. Raw gel image in Figure 4—figure supplement 1—source data 1 and Figure 4—figure supplement 1—source data 2 . The PCR product was sequenced and confirmed. (B) 3-primer PCR amplification (schematic and gel images) at the HCRT locus of F1 heterozygous HCRT-T2A-Venus killifish compared to WT. Arrowheads indicate each primer pair and its expected amplification product length. Scoring Venus negative (-), heterozygous (+/-), or homozygous (+/+) for each well of the gel is indicated below the gel image. Raw gel image in Figure 4—figure supplement 1—source data 1 and Figure 4—figure supplement 1—source data 2. The PCR product was sequenced and confirmed.
-
Figure 4—figure supplement 1—source data 1
Original gel.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig4-figsupp1-data1-v1.zip
-
Figure 4—figure supplement 1—source data 2
Original unedited gel with relevant bands labeled.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig4-figsupp1-data2-v1.zip
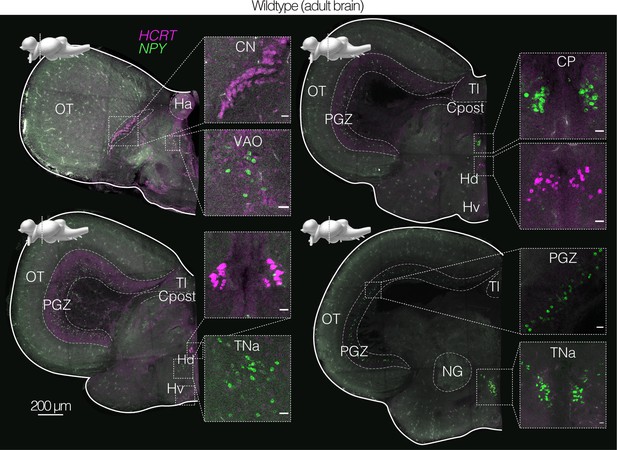
Staining of endogenous NPY and HCRT transcripts in the adult brain of wildtype killifish.
In situ hybridization (via HCR) of coronal brain sections of an adult (3 months old) wildtype male labeling endogenous NPY (green) and HCRT (magenta) transcripts (using gene-specific probe sets, Supplementary file 5). Scale bar = 200 μm. Distinct nuclei indicated and labeled with abbreviated names. Above each slice is the sagittal view of the killifish brain indicating the plane of the coronal section. Inset shows zoom in on NPY and HCRT positive population of cells. Scale bar = 20 μm.
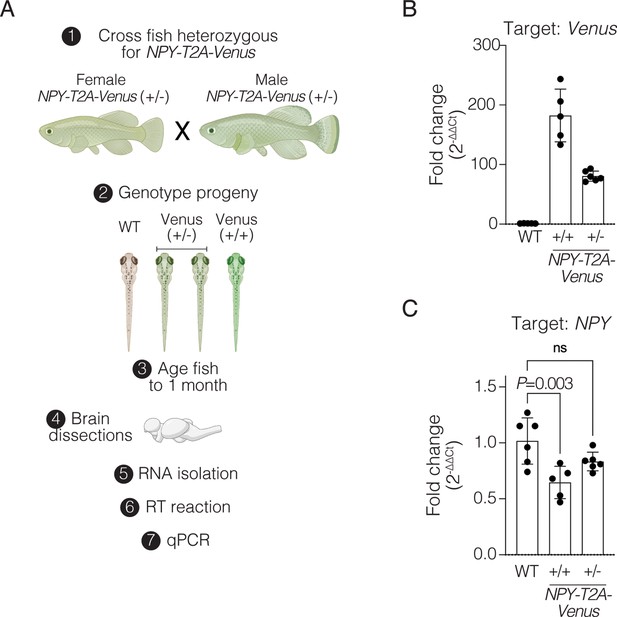
Assessment of Venus and NPY transcript levels in NPY-T2A-Venus knock-in and wildtype control.
(A) Schematic of experiment: crossing F2 heterozygous NPY-T2A-Venus killifish to generate F3 wildtype, heterozygous, and homozygous siblings for the NPY-T2A-Venus allele followed by genotyping, brain dissections, RNA isolation, reverse transcription (RT), and quantitative PCR (qPCR). Primer pairs for NPY and housekeeping genes were designed such that they are separated by at least one intron. Primer sets used show no amplification with no RT control. Created with BioRender.com. (B) Fold change of the Venus transcript level in F3 heterozygous and homozygous NPY-T2A-Venus animals relative to wildtype. n=4–6 animals per genotype. Bar is mean +/- SEM. Each dot represents fold change in transcript level in one fish. Raw data in Figure 4—figure supplement 3—source data 1. (C) Fold change of the NPY transcript level in F3 heterozygous and homozygous animals relative to wildtype. n=5–6 animals per genotype. Bar is mean +/- SEM. Each dot represents fold change in transcript level in one fish. One-way ANOVA with Dunnett’s multiple comparison test for statistical comparisons. No significant difference (ns). Raw data in Figure 4—figure supplement 3—source data 1.
-
Figure 4—figure supplement 3—source data 1
Raw data for panels B and C.
- https://cdn.elifesciences.org/articles/80639/elife-80639-fig4-figsupp3-data1-v1.xlsx
Additional files
-
MDAR checklist
- https://cdn.elifesciences.org/articles/80639/elife-80639-mdarchecklist1-v1.pdf
-
Supplementary file 1
Step-by-step protocol for CRISPR/Cas9-mediated knock-in for killifish.
- https://cdn.elifesciences.org/articles/80639/elife-80639-supp1-v1.pdf
-
Supplementary file 2
Annotated ELAVL3-T2A-Venus HDR sequence.
- https://cdn.elifesciences.org/articles/80639/elife-80639-supp2-v1.zip
-
Supplementary file 3
Sequences of primers, guide RNAs, and HDR repair templates.
- https://cdn.elifesciences.org/articles/80639/elife-80639-supp3-v1.xlsx
-
Supplementary file 4
Number of injected embryos, lethality, and knock-in efficiency for several loci and different HDR donor templates with or without HDR enhancer.
- https://cdn.elifesciences.org/articles/80639/elife-80639-supp4-v1.xlsx
-
Supplementary file 5
Probe sets for in situ hybridization chain reaction.
- https://cdn.elifesciences.org/articles/80639/elife-80639-supp5-v1.xlsx
-
Supplementary file 6
Annotated ELAVL3-T2A-Venus-P2A-H2B-oScarlet HDR sequence.
- https://cdn.elifesciences.org/articles/80639/elife-80639-supp6-v1.zip
-
Supplementary file 7
Annotated NPY-T2A-Venus HDR sequence.
- https://cdn.elifesciences.org/articles/80639/elife-80639-supp7-v1.zip
-
Supplementary file 8
Annotated HCRT-T2A-Venus HDR sequence.
- https://cdn.elifesciences.org/articles/80639/elife-80639-supp8-v1.zip





