Cholinergic and noradrenergic axonal activity contains a behavioral-state signal that is coordinated across the dorsal cortex
Figures
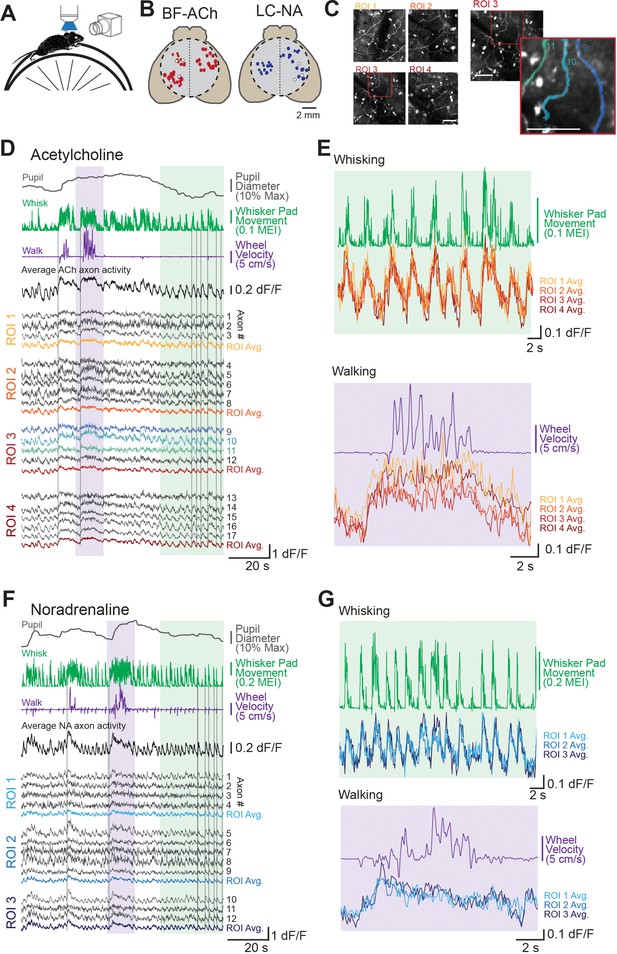
Two-photon imaging of dorsal cortical cholinergic and noradrenergic activity reveals strong state dependence.
(A) Schematic of experimental setup. Awake mice were placed atop a running wheel underneath a two-photon mesoscope objective for imaging of axonal activity through GCaMP6s. A second side camera was used to capture facial movements. (B) To allow for imaging across a large region of cortex, mice were implanted with an 8 mm diameter glass cranial window (dotted circle) on the dorsal brain surface. Recordings could be collected from regions of interest (ROIs) within a 5 × 5 mm field of view. Estimated locations of all ROIs are shown for all BF-ACh (left) and LC-NA (right) axon segment recordings. Size of each circle corresponds to the size of the corresponding square ROI, ranging from 50 × 50 to 400 × 400 µm. Typically three or four ROIs, always within the same hemisphere, were used for imaging. White circles represent ROIs shown in panels (D–G). (C) Left: mean projection image of four simultaneously recorded ROIs (corresponding data shown in panel D). Right: example of Suite2P-defined axons. Three axons are shown in the inset image, which correspond to the three blue dF/F traces in panel (D). Scale bars represent 50 µm. (D–G) Example recordings of GCaMP6s activity in 17 BF-ACh simultaneously imaged axon segments in four ROIs (D, E) and 13 LC-NA axon segments in three ROIs LC-NA (F, G). Pupil diameter, whisker pad movement (represented as motion energy index [MEI], as described in ‘Materials and methods’), and wheel movement (both rotational and vertical) are shown alongside dF/F of individual axons recorded simultaneously across ROIs shown in panel (B). Vertical gray lines in (D) and (F) are for reference and are aligned to the onset of examples of walking or whisker movements (see Animation 1 and Video 1). Average activity across all axons simultaneously monitored as well as within a ROI are shown (D, F). Colored regions in (D) and (F) are expanded for illustration in (E) and (G). Axon GCaMP6s data in (E) and (G) are represented as ROI averages. Histological investigation revealed that GCaMP6s expression was specific for cholinergic and noradrenergic neurons and axons (see Figure 1—figure supplement 1).
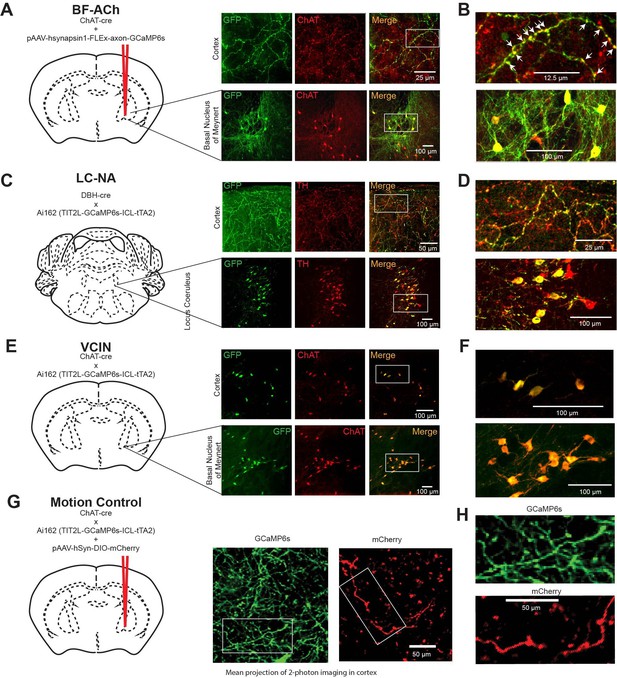
Histological verification of GCaMP6s labeling specificity for cholinergic and noradrenergic neurons and axons.
Relates to Figures 1—5. (A) Cholinergic axons were labeled with GCaMP6s through local injection of pAAV-hsynapsin1-FLEx-axon-GCaMP6s into the region of the basal forebrain, as illustrated. Immunohistochemical staining for GFP (see ‘Materials and methods’) revealed multiple axons in the neocortex to be immunopositive. Immunostaining for ChAT revealed numerous ChAT+ puncta throughout the cortex. Overlap of the GFP+ axons and ChAT+ puncta revealed that the presumed presynaptic terminals of the GCaMP6s axons were positive for ChAT (merged image). (B) Expansion of the example axons (white box in A) reveals the ChAT+ puncta along GFP+ axons (e.g. arrowheads). Examination of the basal nucleus of Meynert revealed many neurons that were both GFP+ and ChAT+. Cell counts revealed that the vast majority of GFP+ neurons were also ChAT+ (n = 516/528 neurons; 98%; N = 2 mice). (C) Histological verification of DBH cre X Ai162 mice demonstrating that locus coeruleus neurons and axons in the cerebral cortex that are labeled with GFP (GCaMP6s) are also immunopositive for tyrosine hydroxylase (TH+) and therefore are noradrenergic. The vast majority of cells immunopositive for GFP in the locus coeruleus are also immunopositive for TH (n = 559/584 neurons; 96%; N = 2 mice). (D) Expansion of axons and cells in (C) for clarity. (E) Cholinergic interneurons in the cerebral cortex are positive for both GFP and ChAT+ in ChAT-cre X Ai162 mice. Examples of local interneurons in layers 1–3 of the neocortex (and cells in the basal forebrain) that are immunopositive for both GFP and ChAT and therefore are presumed cholinergic. The large majority of GFP+ neurons in the cortex (n = 298/302; 99%) and basal forebrain (n = 387/399; 97%) were also ChAT+. (F) Close-up of cells from (E) for clarity. (G) Example axons as observed in the two-photon mesoscope that were observed in the green (GCaMP6s) and red (mCherry) channels following viral expression of pAAV-hSyn-DIO-mCherry in ChAT-cre X Ai162 mice. The cortical axons in both the green and red channels were of similar size. Images here are artificially colored green and red in (G) and (H) for clarity and consistency with parts (A–F). (H) Close up of axons for clarity.
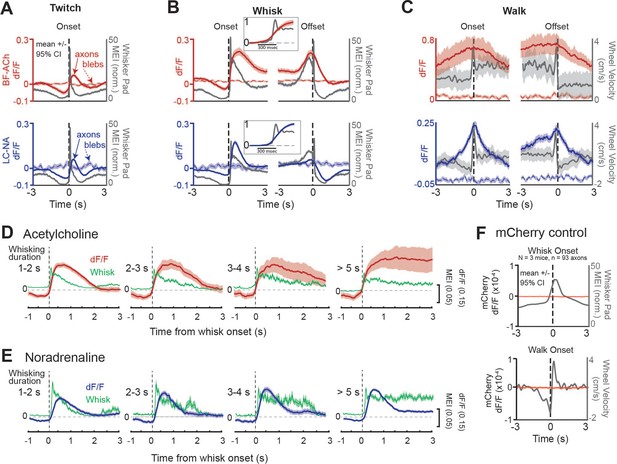
Basal forebrain-derived cholinergic (BF-ACh) and locus coeruleus-derived noradrenergic (LC-NA) activity track behavioral state transitions.
(A–C) Mean GCaMP6s fluorescence in BF-ACh (top) and LC-NA (bottom) axons aligned to the onset and offset of whisker twitching (A), a bout of full whisking (B), and walking (C). Gray traces represent averaged whisker pad motion energy (A, B) or walking speed (C). Shaded region represents a 95% confidence interval. Fluorescence of nearby blebs (dotted lines) was not affected by whisking or walking. (B), insets. Expanded time base, with data normalized from 0 to 1, for comparison of onset kinetics of average whisker movements and cholinergic/noradrenergic axon activity. (D, E) Activation of cholinergic (D) and noradrenergic (E) axons during whisking bouts of varying durations, from 1 to 2, 2–3, 3–4, and >5 s. Note that the cholinergic (red traces) axonal responses maintain activity during prolonged whisking (green traces), while the noradrenergic axonal responses (blue traces) are more transient. Shaded region represents a 95% confidence interval. For all panels, ACh data obtained from N = 5 mice, n = 397 axon segments over 1108 twitches, 330 whisking onsets, 201 whisking offsets, and 101 walking bouts. NA data obtained from N = 6 mice, n = 283 axon segments over 644 twitches, 400 whisking onsets, 353 whisking offsets, and 34 walking bouts. (F) Mean non-activity-dependent mCherry fluorescence (red traces) in axons aligned to the onset of whisking (top) and walking (bottom). Gray traces represent whisker pad motion energy (top) and walking speed (bottom). Note that the dF/F of mCherry axons is three orders of magnitude smaller than that of GcaMP6s axons and does not fluctuate at the onset of walking or whisking. For mCherry control experiments, 93 axon segments were imaged in three mice during 147 whisker twitches, 564 whisking onsets, 529 whisking offsets, and 105 walking bouts.
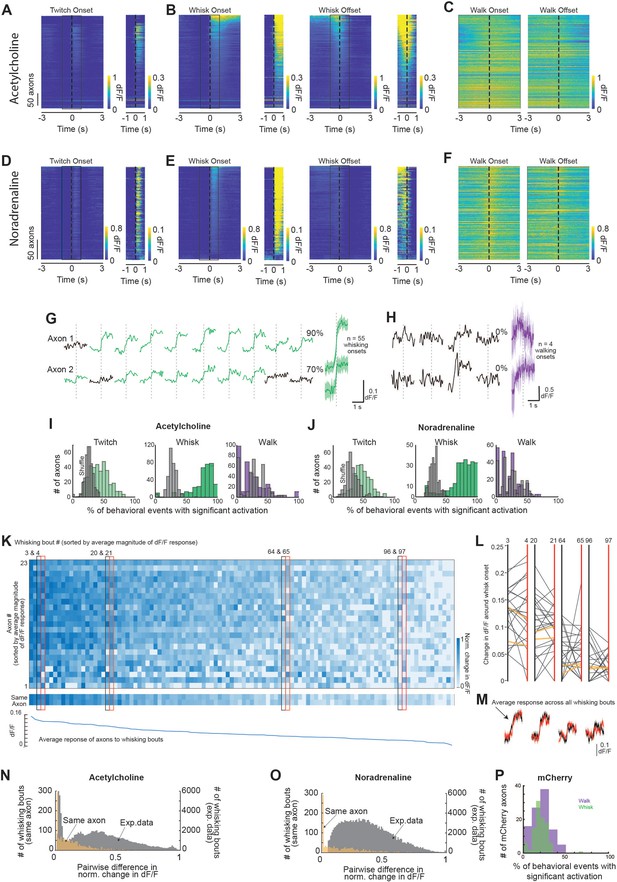
Individual axonal responses to behavioral state changes.
Relates to Figures 1 and 2. (A–F) Same data as displayed in Figure 2A–C, but separated to visualize heterogeneity of individual axon responses to behavioral state changes. Axons are sorted based on the magnitude of dF/F change at the onset of whisking. For (A, B, D, E), two scales of dF/F are shown for clarity. (G, H) Example raw traces of axon segments illustrating the probability that the axon segment showed a significant response in relation to the onset of a bout of whisking (G) or the onset of wheel movement (H). Green traces were those detected to have a significant response on that trial. Black traces did not. In this illustration, Axon 1 exhibited 90% probability of significant activation with whisking, but 0% chance that the 1 s after onset of walking was significantly different than the 1 s prior to walking onset. Axon segment 2 showed a 70% probability or significant response to onset of whisking and a 0% rate of difference for GCaMP6s average signal difference during the 1 s following, versus 1 s preceding, walk onset. (I, J) Probability that a given basal forebrain-derived cholinergic (BF-ACh) or locus coeruleus-derived noradrenergic (LC-NA) axon will increase fluorescence alongside the onset of a twitch, whisk, or walking bout (1 s after vs. 1 s before onset). Gray bars are the distribution resulting from a shuffle (see ‘Materials and methods’). Responsiveness from n = 397 axons (N = 5 mice) was measured at the onset of 1108 twitches, 330 whisking bouts, and 101 walking bouts for ACh (I). Responsiveness from n = 283 axons (N = 6 mice) was measured at the onset of 644 twitches, 400 whisking bouts, and 34 walking bouts for NA (J). (K) Representative example of axonal activity at the onset of whisking bouts. Darker colors represent a stronger activation around whisking onset (determined by subtracting mean dF/F during 1 s prior to whisking onset from mean dF/F of 1 s following whisking onset and normalizing across all whisking bouts). To visualize activity of axons around whisking bouts of similar magnitude, the plot is sorted horizontally by average magnitude of axonal responses to each whisking bout. Axons are shown from highest average responsiveness to whisking (top) to lowest (bottom). Note that some axons respond oppositely to whisking bouts of similar magnitude (pairs of examples shown in black and red boxes). Middle: same analysis for two segments of the same axon recorded during this example session. Note the relative uniformity of responses between these same-axon segments compared to the heterogeneity shown across all recorded axons. Bottom: average change in dF/F across all simultaneously recorded axons at the onset of whisking bouts. This metric was used to sort across whisking bouts. (L) Change in dF/F at the onset of whisking bouts of similar magnitude. Examples are taken from whisking bouts boxed in (K). Yellow lines represent changes in dF/F of the two same-axon segments. Segments of the same axon generally respond similarly to whisks of similar magnitude while there is a clear diversity in response across all 23 recorded axons. (M) Averaged dF/F of all recorded axons around the onset of whisking bouts. Color of the dF/F trace corresponds to the color of the correspondingboxes in (K) and the vertical lines in (L). (N, O) Comparison of pairwise differences in normalized change in dF/F between all simultaneously imaged axon segments for each whisking bout. For both BF-ACh (N) and LC-NA (O), the distribution of differences between activation amplitudes between pairs of heterogeneous axon segments was more broadly distributed than for segments of the same axons. For ACh, N = 5 mice, n = 7272 different axon pairs and n = 18 same axon pairs. For NA, N = 6 mice, n = 1699 different axon pairs and n = 30 same axon pairs. (P) Same analysis as in (I) and (J) but for non-calcium-dependent mCherry axons.
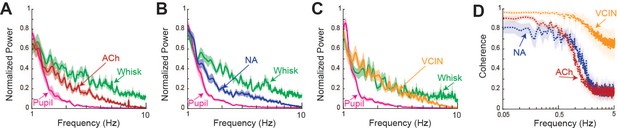
Distribution of power and coherence for pupil, cholinergic, noradrenergic, whisking, and VIP-expressing interneuron (VCIN) activity.
Related to Figures 2, 3C and D. (A) Power distribution between 1 and 10 Hz for cholinergic axon fluorescence, pupil diameter, and whisker pad motion energy. N = 5 mice, n = 397 axons. (B) Same as (A) but including noradrenergic axons. N = 6 mice, n = 283 axons. (C) Same as (A, B) but showing VCIN fluorescence. N = 7 mice, n = 98 cells. Note that whisker pad motion energy and VCIN fluorescence has more power at frequencies above approximately 1.5 Hz than cholinergic or noradrenergic axons. (D) Coherence between adjacent portions of the same acetylcholine (ACh), noradrenaline (NA) axons or segments of the same VCIN cell body. Note the large fall off of coherence for frequencies above approximately 1 Hz. The fall off for power for VCIN neurons is less marked. Plotted is the mean +/- 95% confidence intervals.
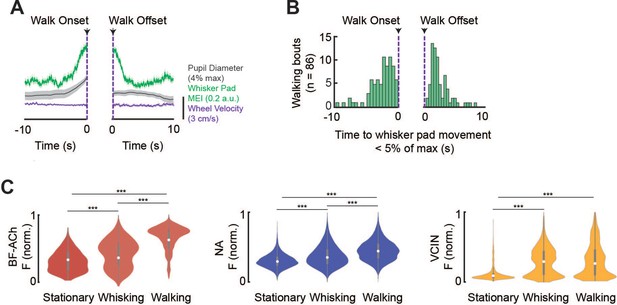
Variable timing of onset/offset of whisking and walking and fluorescence of cholinergic and noradrenergic axons and VIP-expressing interneurons (VCINs) during different states.
Related to Figure 2. (A) Average whisker movements and pupil diameter preceding and following the onset/offset of a walking bout (n = 86 walking bouts; mean +/- 95% confidence intervals plotted). Note that the average movement in the whisker pad both precedes, and follows, walk onset by several seconds. (B) Distribution of onset/offset of bouts of whisker movements to the onset/offset of a bout of walking. Note that whisker movement onset/offset is variable in comparison to walking onset/offset, and distributed over several seconds. (C) Violin plots of the normalized fluorescence of cholinergic (n = 397), noradrenergic (n = 283) axons, and VCIN (n = 98) neurons during the different behavioral states of stationary (no detected whisker movements or walking), whisking (twitching or whisking), and walking. Note the variations in the magnitude of fluorescence recorded in each population across behavioral states. ***p<0.001; One-way ANOVA with multiple comparisons post-hoc testing.
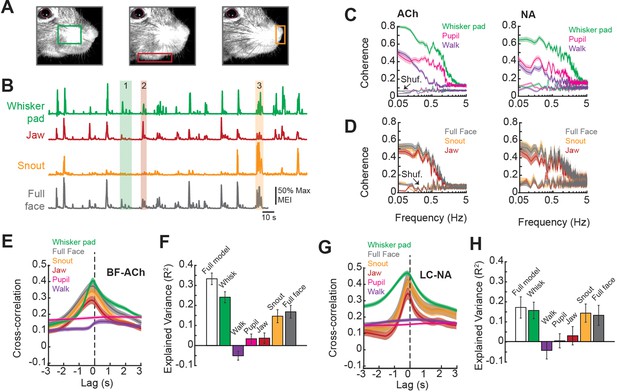
Facial movements are highly predictive of basal forebrain-derived cholinergic (BF-ACh) and locus coeruleus-derived noradrenergic (LC-NA) axon activity.
(A) Restricted regions within videos of the face were analyzed to capture movement of the (left to right) whisker pad, jaw, and snout. (B) Example traces of the motion energy index (MEI) recorded from the whisker pad, jaw, snout, and across the entire frame (full face). Instances where motion energy of the face was predominated by whisker pad (1), jaw (2), or snout (3) movement are noted by green, red, and yellow bars, respectively. (C) Coherence between average BF-ACh or LC-NA axon activity and movement of the whisker pad, walking, and pupil diameter. All simultaneously imaged axon segments within a session were averaged prior to calculation of coherence. (D) Coherence between average BF-ACh or LC-NA axon activity and motion energy in the full face, snout, and jaw. It should be noted that examining coherence between adjacent segments of the same axon reveal our imaging method to be unable to accurately represent variations in activity above approximately 1 Hz, indicating a frequency limitation in the present coherence measures (see Figure 4 and Figure 2—figure supplement 2). (E) Cross-correlation between arousal measures and BF-ACh axon activity. (F) Amount of variance in BF-ACh axon activity that is explained by pupil, whisker movements, walking, snout movements, jaw movements, and movement energy in the full face view. (G, H) Same as (E, F) for LC-NA axons. Error represents a 95% confidence interval for panels (C–H). Results presented in (E–H) were obtained after low-pass filtering signals at 1 Hz. For all panels, ACh data taken from N = 3 mice, n = 65 axon segments and NA data taken from N = 3 mice, n = 50 axon segments.
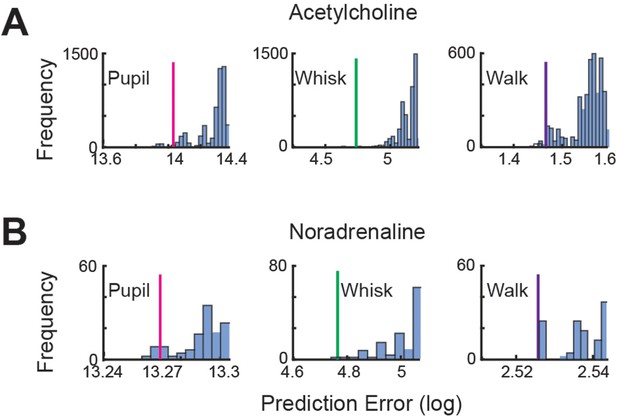
Quantification of linear regression predictive error.
Related to Figure 3F and H. To confirm that the reported relationships between axonal GCaMP6s activity and animal movement were not achieved by chance, we determined the linear regression predictive error associated with matched behavior and axon imaging recordings (simultaneously recorded; solid lines) and all permutations of unmatched recordings (blue bars). The error associated with unmatched regressions was higher than matched for all cases, suggesting that the reported relationship between axonal CaMP6s activity and arousal measures is meaningful for both acetylcholine (A) and noradrenaline (B).
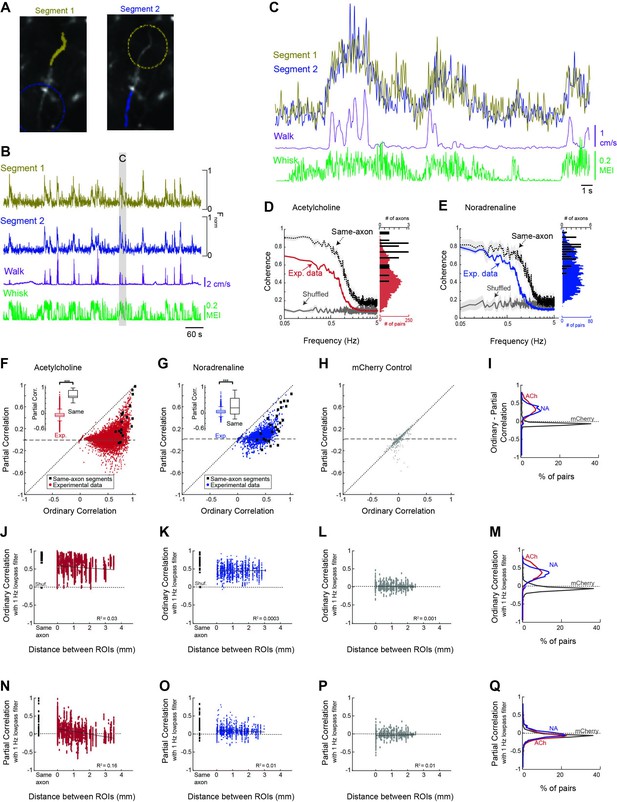
Cholinergic and noradrenergic axon segments across the dorsal cortex exhibit a common signal related to behavioral state.
(A) To examine the relationship between monitored axon segments, we first measured the relationship between GCaMP6s Ca2+ fluorescence signals measured in adjacent segments of the same axon (e.g. yellow labeled segment 1 vs. blue labeled segment 2; dotted circles indicate adjacent segments). Theoretically, all, or nearly all, action potentials will invade adjacent segments of the axon, providing a measure of the limitations of the frequency of activity that we can monitor with our methodology. (B) Plot of the GCaMP6s activity observed in the two adjacent segments of the same axon in relation to walking and whisker motion energy. (C) Expanded example of the measured activity in the two connected axon segments for detail. Note that the signal between adjacent segments of the same axon is coherent at low frequencies, but not at higher frequencies. (D) Coherence between segments of the same axon segments (black trace; N = 5 mice, n = 18 same-axon segment pairs) exhibit a high coherence at frequencies below approximately 1 Hz and a strong decrease at frequencies between 1 and 3 Hz, indicating that our monitoring methodology is limited to frequencies of approximately <1–3 Hz. Comparison of coherence between all axon segments simultaneously monitored (N = 5 mice, n = 7272 pairs of axons) reveal a strong average coherence at frequencies less than 1 Hz, but this average coherence is lower than that for axon segments of the same axon. Plotting the distribution of average coherence at frequencies between 0.05 and 0.5 Hz reveals a broader distribution for heterogeneous axon segments in comparison to segments of the same axon. (E) Comparison of coherence for adjacent segments of the same axon and all axon segments for noradrenergic axons (N = 4 mice, n = 30 same-axon segment pairs, n = 1699 different axon pairs). Coherence is plotted as average +/- 95% confidence intervals. Plotting the distribution of low-frequency coherence reveals a broader distribution for heterogeneous axon segments in comparison to segments of the same axon. (F, G) Plot of partial correlation versus ordinary correlation for cholinergic (F) and noradrenergic (G) axons reveals a strong contribution of a common signal to the correlations between most axon segments. The common signal was calculated as the average signal between all axon segments simultaneously imaged, excluding the two axon segments for which the partial correlation was calculated. Black dots represent segments of the same axon, which should exhibit a correlated signal that is independent of the common, group-average signal. (H) Plot of partial correlation versus ordinary correlation for non-activity-dependent mCherry labeled axons. (I) Distribution of difference between ordinary and partial correlation for acetylcholine (ACh), noradrenaline (NA), and mCherry axon segments. (J, K) The correlation in GCaMP6s signal between cholinergic (F) and noradrenergic (G) axon segments (signals low-pass filtered at 1 Hz) versus distance between the regions of interest containing the axon segments. 0 mm indicates axon segments within the same region of interest. Black squares represent correlation between adjacent segments of the same axon. Red or blue dots illustrate the correlations between activity in all axon segments, excluding those that were clearly part of the same axon. Note that for both ACh and NA axons, there is generally a high correlation if the axon segments are adjacent and from the same axon (black squares). For all axon segments, the correlations are much more varied, ranging from around 0 to greater than 0.8. These correlations change only slightly with distance, even up to 4 mm, indicating that there is broad synchrony of cholinergic and noradrenergic axonal activity across the cortex. (L) Distribution of correlations with distance between non-activity-dependent mCherry axonal fluorescence. (M) Distribution of ordinary correlation between segments of ACh, NA, and mCherry axons. (N, O) Dependence of partial (residual) correlations between ACh (N) and NA (O) axon segments with distance. (P) Partial correlations plotted against distance between segments for mCherry-labeled axon segments. (Q) Plot of distribution of partial correlations between ACh, NA, and mCherry axon segments.
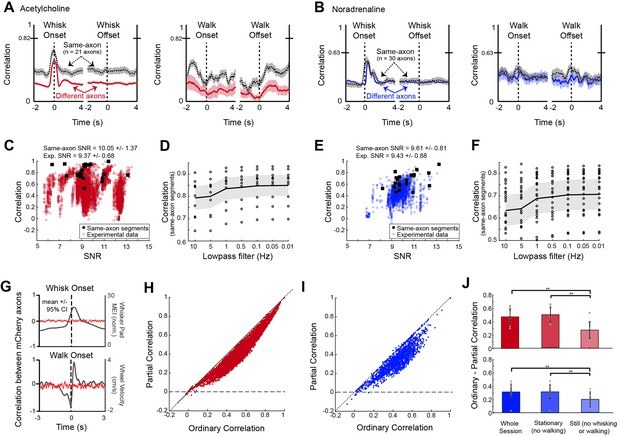
Features of axonal imaging data relevant to interpreting imaging results.
Related to Figure 4. (A) Average correlation between basal forebrain-derived cholinergic (BF-ACh) axon segment pairs (n=21 same axon pairs; n=7272 different axon pairs) aligned to the onset and offset of whisker pad motion and walking exhibit a transient increase at whisker movement onset. However, this transient increase is equally prominent for the two segments of the same axon, indicating that it is a consequence (artifact) of the imaging methodology. (B) Same as (A), except for noradrenaline (NA) axon segments (n=30 same axon pairs; n=7272 diferent axon pairs). Plotted are average correlations +/- 95% confidence intervals. (C) Comparison of correlation and signal-to-noise (SNR) ratio for same axon segments (black squares) and complete data set (red dots). Correlations for axon segments from the same axons were higher than for the complete data set, even though the SNR was similarly distributed. Correlations did not correlate with SNR. (D) Effect of applying low-pass filters with different cut off frequencies on the average correlation between cholinergic axon segments. A small increase in correlation was obtained by applying low-pass filters of 1 Hz or lower. (E) Same as (C), except for noradrenergic fibers. (F) Same as (D), except for noradrenergic fibers. Average +/- 95% confidence intervals are shown. (G) Correlation between non-activity-dependent mCherry axons (red traces) is not affected by the onset of whisking or walking (gray traces). (H) Partial correlation versus ordinary correlation for cholinergic axon segments when the common signal is taken as whisker movement. (I) Same as (H), except for noradrenergic axon segments. (J) Restricting the portion of data analyzed for correlations to the still periods reduces the effect of the common signal on the ordinary correlation (e.g. the difference between the partial and ordinary correlations) for both cholinergic and noradrenergic axon segments. **=p<0.01; One-wave ANOVA with multiple comparisons post-hoc testing.
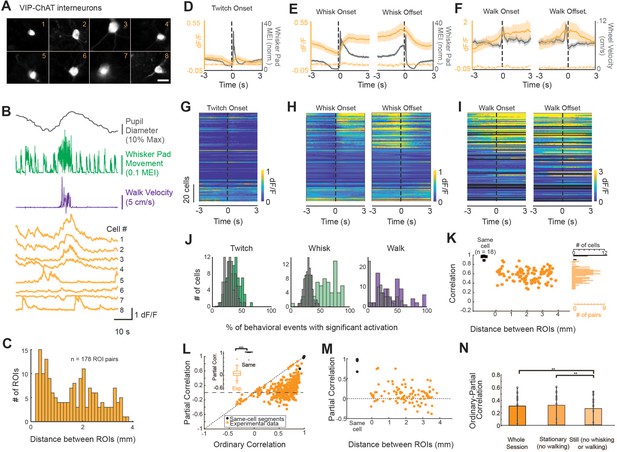
A subpopulation of VIP-ChAT cortical interneurons track changes in behavioral state.
(A) Images of eight simultaneously recorded VIP-ChAT cortical interneurons (VCINs). (B) Example recording taken from the eight somas shown in panel (A). Note the clear heterogeneity of VCIN response (yellow) during walking (purple) and whisking (green) activity. (C). Distribution of distance between regions of interest (ROIs) containing VCINs. ROIs were separated from 0.075 to 3.9 mm (N = 7 mice, n = 98 cells). (D–F) Group-averaged VCIN activity aligned to 870 twitching onsets, 1104 whisking onsets, 1015 whisking offsets, and 127 walking onsets and offsets (N = 6 mice, n = 84 cell bodies for walking data). Gray traces show the averaged change in whisker pad motion energy (D, E) or walking speed (F). Average +/- 95% confidence intervals are plotted. Dotted lines represent autofluorescent blebs, which showed no significant change in fluorescence with movement. Inset in (E) is at an expanded time base with normalization of data from base to peak for comparison of the kinetics of onset of whisker movements and VCIN activity. (G–I) Same data as displayed in panels (C–E) but separated to visualize heterogeneity of individual cell responses to behavioral state changes. Cells are sorted based on the magnitude of dF/F change at the onset of whisking. (J) The consistency of a single VCIN’s response to behavioral state changes was quantified by calculating the percent of twitching, whisking, or walking bouts that were associated with significant increases (1 s post-onset versus 1 s pre-onset) in dF/F and compared to distributions attained with shuffled data. (K) Correlation between VCIN cells by distance between the ROIs. Measuring GCaMP6s from two halves of the same cell body resulted in an average correlation that was near 1 (black dots; N = 3 mice, n = 18 same-cell pairs). (L) Partial residual correlation (following removal of correlation with common signal) versus ordinary correlation reveals that a significant component of the ordinary correlation results from the common signal. Common signal was calculated as the average activity of the imaged neurons, excluding the two cells being correlated. ***p<0.001; Student’s t-test. (M) Partial residual correlation as a function of the distance between ROIs containing the cells being correlated. The partial correlation decreases slightly with distance (r = –0.09; n = 98 cells; p=0.07). (N) Effect of restricting data to stationary or still periods on partial correlations. p<0.01; One-way ANOVA with multiple comparisons post-hoc testing.
Videos
Example of mouse face during twitching, whisking, and walking.
Axon imaging and behavioral acquisition methodology.
Related to Figure 1. A headpost was implanted on the skull overlying the dorsal cortex (top of video = anterior). Cortical regions coarsely aligned to the Allen Brain Institute Common Coordinate Framework are shown for reference. An 8 mm circular cranial window was placed over multiple cortical regions. In each imaging session, axons or cells within up to a 5 × 5 mm region could be imaged simultaneously. An epifluorescence image of the visible brain areas for this example session is shown along with four red squares, which indicate the position of each of four regions of interest (ROIs) recorded simultaneously. Next, all collected data are shown for this example session. Top left: position of regions of interest. Top middle: video recording of the mouse’s face (used for pupil diameter and whisker pad motion extraction). Bottom left: traces of dF/F for each isolated axon shown on the right along with whisker pad motion energy, walking speed, and pupil diameter. Right: four videos of ACh axon imaging aligned in time to behavioral variables shown on the left. Videos are sped up by 10× but are otherwise unprocessed (i.e. no motion correction or temporal resampling). The pupil is large in this video owing to low UV/white light levels to enhance axon imaging SNR (see ‘Materials and methods’).





