Spindle assembly checkpoint-dependent mitotic delay is required for cell division in absence of centrosomes
Figures
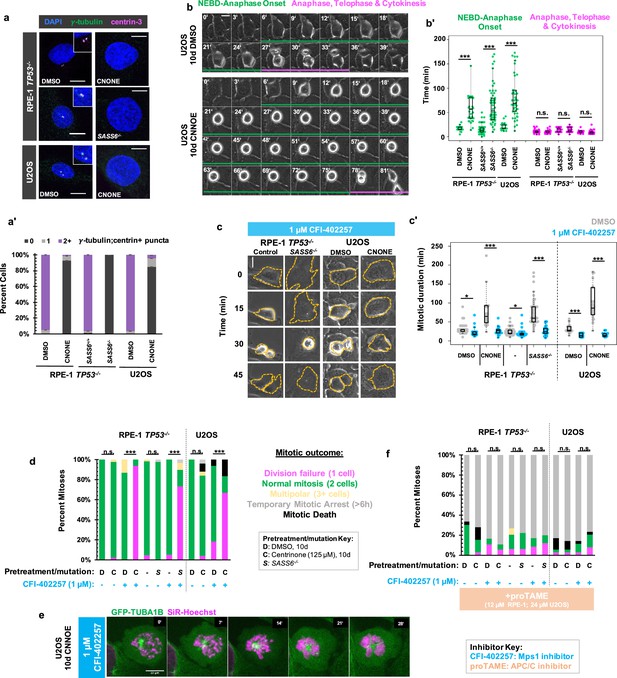
MPS1 activity is required for mitotic elongation and cell division in acentrosomal cells.
(a) Immunofluorescence staining of control and acentriolar RPE-1 TP53-/- or U2OS cells. Acentriolar cells were created by treatment with 125 nM centrinone (CNONE) or by deletion of SASS6. DAPI is shown in blue, γ-tubulin in green, and centrin-3 in magenta. Insets are shown for cells in which centrosomes were present. (a’) Quantification of (a). Graphed are means and S.E.M. Significance was determined by Fisher’s exact test. n=100 cells per condition. (b) Live phase contrast imaging showing example mitosis in U2OS cells pre-treated with DMSO or centrinone (CNONE) for 10 days. Green bars below cells indicate the duration of nuclear envelope breakdown (NEBD) through the onset of anaphase, while magenta bars below cells indicate the duration of the completion of anaphase, telophase, and cytokinesis. (b’) Mitotic durations from NEBD through metaphase (green) and anaphase, telophase, and cytokinesis (magenta) as in (b). Time is in minutes. Points represent individual cells; boxplots represent mean and interquartile range. Significance was determined by Welch’s t-test. n≥20 cells per condition. (c) Live phase contrast imaging showing example mitosis in U2OS cells pre-treated with DMSO or centrinone (CNONE) for 10 days before being imaging in 1 μM CFI-402257. (c’) Quantification of mitotic duration in cells of indicated genotype or treatment in DMSO (grey) or CFI-402257 (blue). Time is in minutes. Points represent individual cells; boxplots represent mean and interquartile range. Significance was determined by Welch’s t-test. n≥25 cells per condition. (d) Quantification of daughter cell fate of cells of the given pre-treatment imaged in DMSO or CFI-402257 (1 μM). Shown are the percentages for each fate of the total mitotic observations. Significance was determined by Fisher’s exact test. n≥40 cells per condition. (e) Confocal timelapse imaging of U2OS cells pretreated for 10 d with 125 nm centrinone and imaged in 1 μM CFI-402257. Shown are endogenously tagged α-tubulin (GFP-TUBA1B) and DNA (Sir-Hoechst). (f) Quantification of daughter cells fates of cells of the given pre-treatment imaged in DMSO or CFI-402257 (1 μM) with concurrent treatment with proTAME (12 μM for RPE1 cells, 24 μM for U2OS cells). Shown are the percentages for each fate of the total mitotic observations. Significance was determined by Fisher’s exact test. n≥40 cells per condition. In all cases, not significant (n.s.) denotes p>0.05, * denotes p<0.05, ** denotes p<0.01, and *** denotes p<0.001. Scale bars: 10 μm.
-
Figure 1—source data 1
Source data for Figure 1a’, b’, c’, d and f.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig1-data1-v1.zip
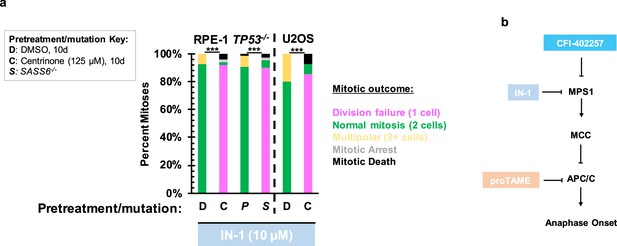
Treatment with alternate MPS1 inhibitor MPS1 IN-1 phenocopies treatment with CFI-402257.
(a) Quantification of daughter number of cells of the given pre-treatment imaged in DMSO or 10 μM MPS1 IN-1. Shown are the percentages for each fate of the total mitotic observations. Significance was determined by Fisher’s exact test. n≥41 cells per condition. (b) Schematic of inhibitors and targets. In all cases, not significant (n.s.) denotes p>0.05 and *** denotes p<0.001.
-
Figure 1—figure supplement 1—source data 1
Source data for Figure 1—figure supplement 1a.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig1-figsupp1-data1-v1.zip
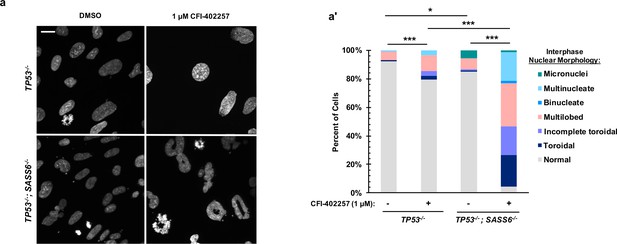
MPS1 inhibition results in abnormal nuclear morphology, but not binuclearity, in acentrosomal cells.
(a) DAPI staining of cells after 72 hr treatment with DMSO or 1 μM CFI-403357. (a’) Shown are the percentages of cells with each indicated nuclear morphology for interphase nuclei. n≥100 cells per condition. In all cases, not significant (n.s.) denotes p>0.05, * denotes p<0.05, ** denotes p<0.01, and *** denotes p<0.001. Scale bar: 10 μm.
-
Figure 1—figure supplement 2—source data 1
Source data for Figure 1—figure supplement 2a.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig1-figsupp2-data1-v1.zip
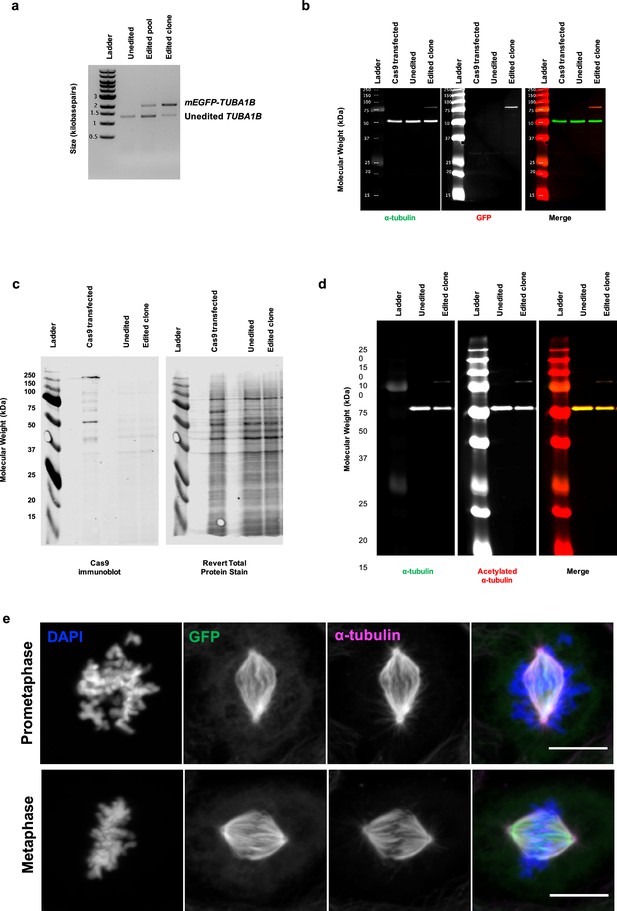
Creation and validation of GFP-TUBA1B U2OS cells.
(a) Agarose DNA gel of PCR genotyping of unedited cells, and edited pool of cells, and the edited clone of cells (clone 1-F3) used in future experiments. PCR primers in material and methods. (b) Western blot of α-tubulin and GFP levels in unedited cells (transfected with Cas9 plasmid but not gRNAs), unedited cells, and edited cells. (c) Western blot of Cas9 levels in cells freshly transfected with Cas9, unedited cells, and edited cells. This confirms that the Cas9 was transient and is no longer present in the edited cells and had been expressed only transiently. (d) Western blot of α-tubulin and acetylated-α-tubulin levels in unedited and edited cells, showing that the GFP-tagged α-tubulin can be acetylated (arrow). (e) Immunofluorescence of GFP (green), all α-tubulin (magenta) in U2OS GFP-TUBA1B cells in prometaphase (top) or metaphase (bottom). Scale bars: 10 μm.
-
Figure 1—figure supplement 3—source data 1
Source data for Figure 1—figure supplement 3a–d.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig1-figsupp3-data1-v1.zip
-
Figure 1—figure supplement 3—source data 2
Source data for Figure 1—figure supplement 3a–d with raw blots.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig1-figsupp3-data2-v1.zip
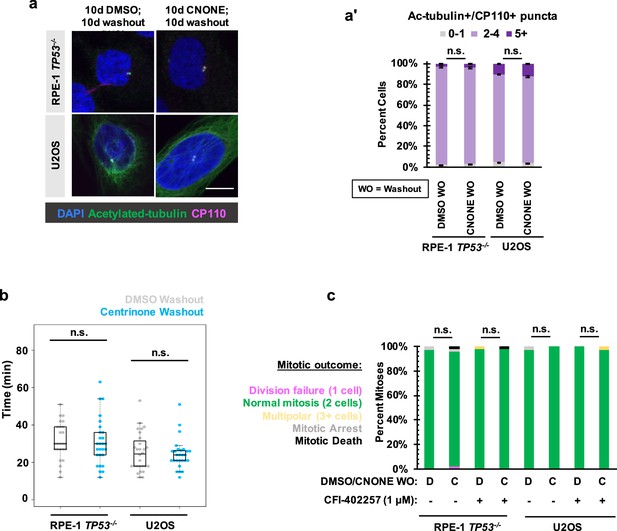
Division failure in acentrosomal cells due to Mps1 inhibition is reversible after centrosome return.
(a) Immunofluorescence staining of RPE-1 TP53-/- or U2OS cells treated with DMSO or 125 nM centrinone (CNONE) for 10 days and then washed out for 10 days. DAPI is shown in blue, acetylated-α-tubulin in green, and CP110 in magenta. (a’) Quantification of (a). Graphed are means and S.E.M. Significance was determined through a Fisher’s exact test. n≥100 cells per condition. (b) Quantification of mitotic duration in cells of indicated genotype after 10 d DMSO followed by 10 d washout (grey) or 10d centrinone (125 nM) followed by 10 d washout (blue). Points represent individual cells; boxplots represent mean and interquartile range. Significance was determined through Welch’s t-test. n≥25 cells per condition. (c) Quantification of daughter number of cells of the given pre-treatment imaged in DMSO or CFI-402257 (1 μM). Shown are the percentages for each fate of the total mitotic observations. Significance was determined by Fisher’s exact test. n≥40 cells per condition. In all cases, not significant (n.s.) denotes p>0.05 and *** denotes p<0.001. Scale bars: 10 μm.
-
Figure 1—figure supplement 4—source data 1
Source data for Figure 1—figure supplement 4a’, b, and c.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig1-figsupp4-data1-v1.zip
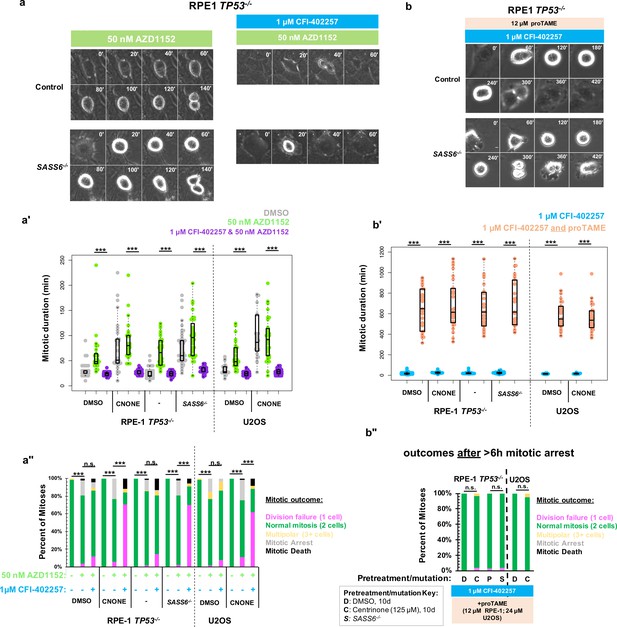
Delayed anaphase onset, rather than specific MPS1 activity, is necessary for successful acentrosomal cell division.
(a) Example mitoses in RPE1 TP53-/- or RPE1 TP53-/-; SASS6-/- cells treated with 50 nM AZD1152 with or without 1 μM CFI-402257. (a') Quantification of mitotic duration in cells of indicated genotype or treatment in DMSO (grey), 50 nM AZD1152 (green), or 50 nM AZD1152 and 1 μM CFI-402257 (purple). Points represent individual cells; boxplots represent mean and interquartile range. Significance was determined through Welch’s t-test. n≥47 cells per condition. (a'') Quantification of daughter number of cells of the given pre-treatment imaged in DMSO, 50 nM AZD1152 or 50 nM AZD1152 and 1 μM CFI-402257. Shown are the percentages for each fate of the total mitotic observations. n≥20 cells per condition. Significance was determined through a Fisher’s exact test. (b) Example mitoses in RPE1 TP53-/- or RPE1 TP53-/-; SASS6-/- cells treated with 1 μM CFI-402257 and proTAME (12 μM for RPE1 cells, 24 μM for U2OS cells). (b') Quantification of mitotic duration in cells of indicated genotype or treatment in 1 μM CFI-402257 (blue) or 1 μM CFI-402257 with 12 μM (RPE1 cells) or 24 μM (U2OS cells) proTAME. Points represent individual cells; boxplots represent mean and interquartile range. Significance was determined through Welch’s t-test. n≥25 cells per condition. (b'') Quantification of daughter number of cells of the given pre-treatment imaged in 1 μM CFI-402257 (blue) or 1 μM CFI-402257 with 12 μM (RPE1 cells) or 24 μM (U2OS cells) proTAME after a mitotic arrest of greater than six hours. Shown are the percentages for each fate of the total mitotic observations. n≥20 cells per condition. Significance was determined through a Fisher’s exact test. In all cases not significant (n.s.) denotes p>0.05 and *** denotes p<0.001. Scale bars: 10 μm.
-
Figure 2—source data 1
Source data for Figure 2a, a’’, b and b’’.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig2-data1-v1.zip
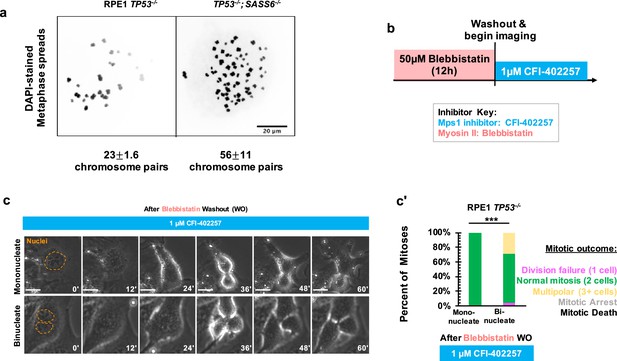
Polyploidy is not sufficient to confer MPS1-inhibition-sensitive division failure.
(a) Metaphase spreads from RPE1 TP53-/- or RPE1 TP53-/-; SASS6-/- cells. Indicated beneath example images are the mean and standard errors of chromosome pairs for the two genotypes. Chromosomes were stained with DAPI. (b) Schematic of experimental setup. REP1 TP53-/- cells were treated with 50 μM blebbistatin for 12 hr, washed, and then treated with 1 μM CFI-402257 and imaged live via phase microscopy. (c) Example mitoses in mononucleate or binucleate cells treated with 1 µM CFI-402257 after washout (WO) of blebbistatin. (c') Quantification of mitotic outcome of RPE1 TP53-/- or U2OS cells imaged in DMSO, 5 μM STLC, 1 μM CFI-402257, or 5 μM STLC and 1 μM CFI-402257 together. Shown are the percentages for each fate of the total mitotic observations. n≥48 cells per condition Significance was determined through a Fisher’s exact test. In all cases, not significant (n.s.) denotes p>0.05 and *** denotes p<0.001. Scale bars: 10 μm.
-
Figure 3—source data 1
Source data for Figure 3c’.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig3-data1-v1.zip
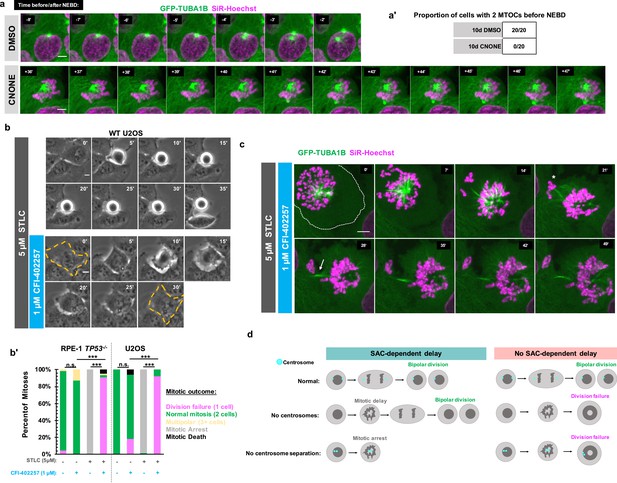
Early mitotic MTOC separation is necessary for cell division in the absence of SAC-mediated mitotic delay.
(a) Confocal timelapse imaging of U2OS cells pretreated for 10 days with either DMSO or centrinone (125 nM). Shown are endogenously tagged α-tubulin (GFP-TUBA1B) and DNA (Sir-Hoechst). Time indicates minutes before (-) or after (+) NEBD. (a’) Proportion of cells with 2 foci of microtubules before NEBD based on experiments as in (a). (b) Live phase imaging of RPE1 TP53-/- or U2OS cells imaged in 1 μM CFI-402257 or 5 μM STLC and 1 μM CFI-402257 together. (b’) Quantification of mitotic outcome of RPE1 TP53-/- or U2OS cells imaged in DMSO, 1 μM CFI-402257, 5 μM STLC, or 5 μM STLC and 1 μM CFI-402257 together. Shown are the percentages for each fate of the total mitotic observations. Significance was determined through a Fisher’s exact test. n≥50 cells per condition (c) Confocal timelapse imaging of U2OS cells imaged in 5 μM STLC with 1 μM CFI-402257. Shown are endogenously tagged α-tubulin (GFP-TUBA1B) and DNA (Sir-Hoechst). Asterisk indicated extruded chromosomes. Arrow indicated midbody-like α-tubulin structure. (d) Graphical summary of results. In all cases, not significant (n.s.) denotes p>0.05 and *** denotes p<0.001. Scale bars: 10 μm.
-
Figure 4—source data 1
Source data for Figure 4b’.
- https://cdn.elifesciences.org/articles/84875/elife-84875-fig4-data1-v1.zip





