Cestode larvae excite host neuronal circuits via glutamatergic signalling
Figures
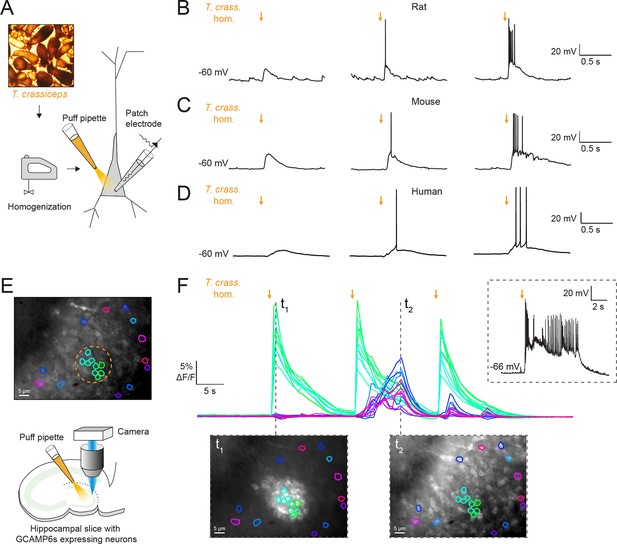
Taenia crassiceps homogenate excites neurons and can elicit epileptiform activity.
(A) Schematic showing the experimental setup in which whole-cell patch-clamp recordings were made from CA3 hippocampal pyramidal neurons in rodent organotypic slice cultures whilst a puff pipette delivered pico-litre volumes of homogenized Taenia crassiceps larvae (T. crass. hom.) targeted at the cell soma. (B) Current-clamp recording from a rat pyramidal neuron whilst increasing amounts of T. crass. hom. was applied via the puff pipette (left to right, orange arrows). Small amounts of T. crass. hom. resulted in depolarization (left), increasing amounts by increasing the pressure applied to the ejection system elicited single (middle) or even bursts of action potentials (right). (C) As in ‘B’, identical effects of T. crass. hom. could be elicited in current-clamp recordings from a CA3 hippocampal pyramidal neuron in a mouse organotypic brain slice culture. (D) As in ‘B’ and ‘C’, identical effects of T. crass. hom. could be elicited in current-clamp recordings from a human frontal lobe layer 2/3 cortical pyramidal neuron in an acute brain slice. (E) Top: widefield fluorescence image of neurons in the dentate gyrus of a mouse hippocampal organotypic brain slice culture expressing the genetically encoded Ca2+ reporter GCAMP6s under the synapsin promoter in artificial cerebrospinal fluid (aCSF) containing 0.5 mM Mg2+. A subset of neurons used to generate the Ca2+ traces in ‘E’ are indicated by different colours. The orange dotted circle indicates where T. crass. hom. was delivered using the puff pipette. Bottom: schematic of the experimental setup including puff pipette and CCD camera for Ca2+ imaging using a 470-nm LED. (F) Top, dF/F traces representing Ca2+ dynamics from the GCAMP6s expressing neurons labelled in ‘E’ concurrent with three puffs (30-ms duration) of T. crass. hom. 15 s apart. Bottom: two images of raw Ca2+ fluorescence at two time points t1 and t2. Note how at time point t2 neurons distant to the site of T. crass. hom. application are also activated, indicating spread of neuronal activity. Inset, top-right: current-clamp recording from a neuron in the region of T. crass. hom. application demonstrates seizure-like activity in response to T. crass. hom. application.
-
Figure 1—source data 1
Patch clamp cell properties and metadata.
- https://cdn.elifesciences.org/articles/88174/elife-88174-fig1-data1-v1.xlsx
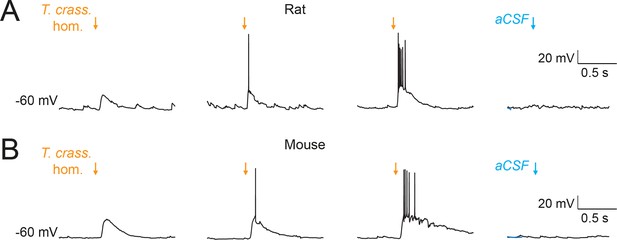
Taenia crassiceps homogenate depolarizes neurons, artificial cerebrospinal fluid (aCSF) does not.
Same setup as Figure 1B, C demonstrating that whilst Taenia crassiceps homogenate depolarizes rat (A) and mouse (B) neurons, application of aCSF via the puffer pipette has no effect.
Taenia crassiceps homogenate excites neurons and can elicit epileptiform activity.
Widefield fluorescence video (same data as Figure 1E, F) of neurons in the dentate gyrus of a mouse hippocampal organotypic brain slice culture expressing the genetically encoded Ca2+ reporter GCAMP6s under the synapsin promoter in artificial cerebrospinal fluid (aCSF) containing 0.5 mM Mg2+. T. crass. hom. was delivered via a puff pipette.
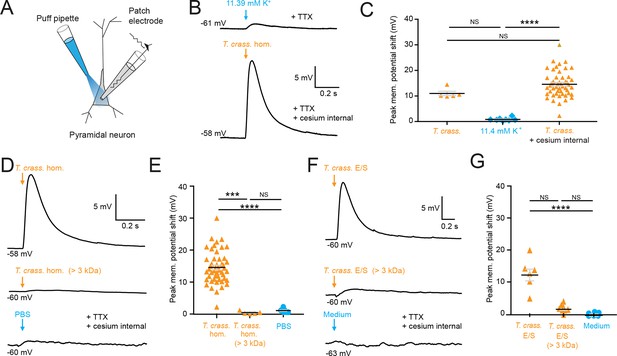
Taenia crassiceps homogenate and E/S product-induced neuronal depolarization is mediated by a small molecule.
Schematic showing the experimental setup in which whole-cell patch-clamp recordings were made from CA3 hippocampal pyramidal neurons in rat organotypic slice cultures whilst a puff pipette delivered pico-litre volumes targeted at the cell soma. (B) Top trace: 20-ms puff of artificial cerebrospinal fluid (aCSF) containing 11.4 mM K+ (equivalent to the K+ concentration of T. crassiceps homogenate (T. crass. hom.)) caused modest depolarization. A standard internal solution was utilized, and 2 μM tetrodotoxin (TTX) was added to circulating aCSF to reduce synaptic noise in the voltage trace. Bottom trace: 20-ms puff of T. crass. hom. was applied, but a cesium-based internal solution was utilized to block K+ channels in the presence of TTX. Puffs of T. crass hom. resulted in sizeable depolarization. (C) Population data showing that responses to 11.4 mM K+ puffs were significantly smaller than that of the T. crass hom. when a cesium internal solution was utilized (T. crass. + cesium internal), but not when a standard internal solution was utilized (T. crass.). (D) Delivery of T. crass. hom. caused a depolarizing shift in membrane potential (top trace). The depolarizing response to T. crass hom. was largely abolished by dialysing out all molecules smaller than 3 kDa (middle trace). Phosphate-buffered saline (PBS), the solvent for T. crass. hom., did not induce a large neuronal depolarization (bottom trace). (E) Population data showing that the membrane depolarization induced by T. crass. hom is not due to the PBS solvent and is due to a molecule smaller than 3 kDa. (F) Delivery of a puff of T. crass excretory/secretory (E/S) products also caused a depolarizing shift in membrane potential (top trace), which was largely abolished by filtering out all molecules smaller than 3 kDa (middle trace). Culture medium, the solvent for the T. crass E/S did not induce depolarization (bottom trace). (G) Population data showing that the membrane depolarization induced by T. crass. E/S is not due to the culture medium solvent and is due to a molecule smaller than 3 kDa. Values with medians ± IQR; ***p ≤ 0.001, ****p ≤ 0.0001, NS = not significant.
-
Figure 2—source data 1
Patch clamp membrane voltages, cell properties, and metadata for all cells.
- https://cdn.elifesciences.org/articles/88174/elife-88174-fig2-data1-v1.xlsx
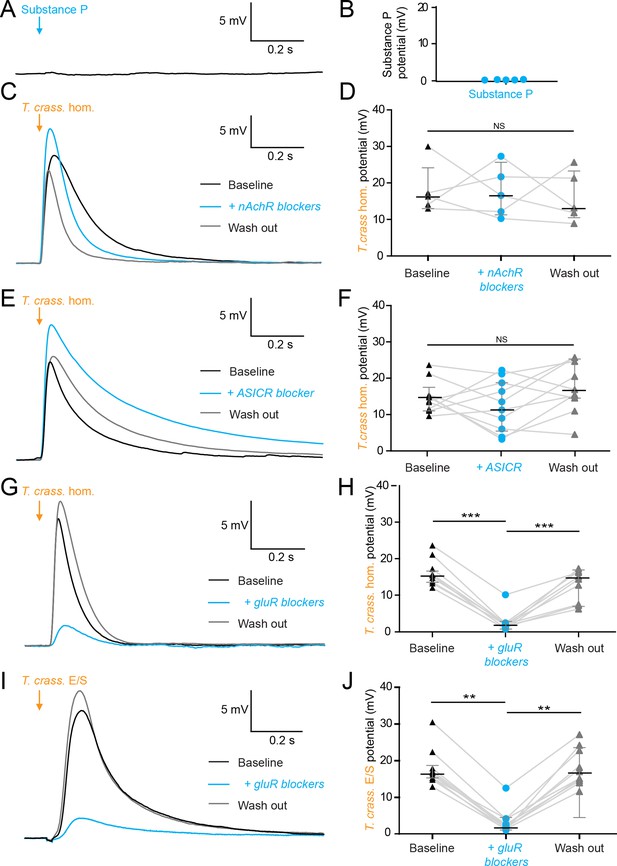
The excitatory effects of Taenia crassiceps are mediated by glutamate receptor activation.
Whole-cell patch-clamp recordings in current-clamp were made from CA3 pyramidal neurons in rat organotypic hippocampal slices using a cesium-based internal in the presence of 2 μM tetrodotoxin (TTX). (A) 20-ms puff of Substance P (100 µM) via a puff pipette directed at the soma did not affect neuronal membrane potential. (B) Population data for Substance P application. (C) The depolarization in response to a T. crassiceps homogenate (T. crass. hom.) puff before (black trace), in the presence of (blue trace), and following washout (grey trace), of a nicotinic acetylcholine receptor (nAchR) blocker (mecamylamine hydrochloride, 10 µM). (D) Population data demonstrating that nAchR blockade has no significant effect on T. crass. hom. induced depolarization, NS = not significant. (E) The depolarization in response to a T. crass hom. puff before (black trace), in the presence of (blue trace), and following washout (grey trace), of an acid-sensing ion channel (ASIC) receptor blocker (amiloride, 2 mM). (F) Population data demonstrating that ASIC receptor blockade has no significant effect on T. crass. hom. induced depolarization, NS = not significant. (G) The depolarization in response to a T. crass. hom. puff before (black trace), in the presence of (blue trace), and following washout (grey trace), of a pharmacological cocktail to block glutamate receptors (10 µM CNQX, 50 µM D-AP5, and 2 mM kynurenic acid). (H) Population data showing that the depolarization response to T. crass. hom. is significantly reduced in the presence of glutamate receptor blockers and returns upon washout, ***p ≤ 0.001. (I) The depolarization in response to a puff of T. crass excretory/secretory (E/S) products is also markedly reduced during glutamate receptor blockade. (J) Population data showing that the depolarization response to T. crass. hom. is significantly reduced in the presence of glutamate receptor blockers and returns upon washout, **p ≤ 0.01.
-
Figure 3—source data 1
Patch clamp membrane voltages, cell properties, and metadata for all cells.
- https://cdn.elifesciences.org/articles/88174/elife-88174-fig3-data1-v1.xlsx
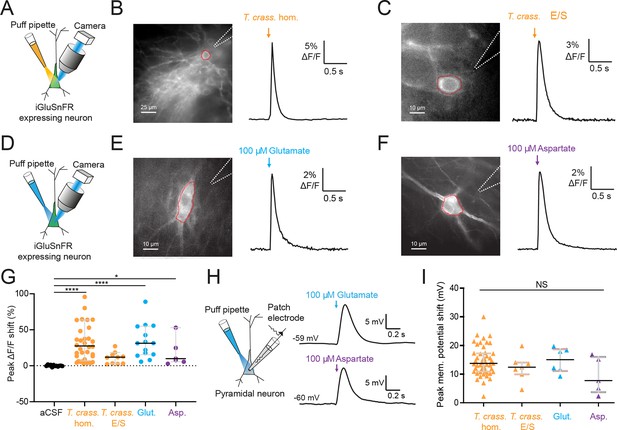
Taenia crassiceps products contain excitatory amino acids detected using iGluSnFR.
(A) Schematic of the experimental setup for glutamate detection by the genetically encoded fluorescent glutamate reporter iGluSnFR, including puff pipette and sCMOS camera for imaging following excitation using a 475/28 nm LED-based light engine. (B) Left, iGluSnFR-expressing neuron with the region of interest (red dashed line) used to calculate dF/F trace (right) during a 20-ms T. crassiceps homogenate (T. crass. hom.) puff (orange arrow). (C) As in ‘B’ but with a 20-ms T. crassiceps excretory/secretory (T. crass. E/S) product puff (orange arrow). (D) Schematic of experimental setup as in ‘A’ but with glutamate or aspartate application via puff pipette. (E) iGluSnFR fluorescence just after a 20-ms puff of artificial cerebrospinal fluid (aCSF) containing 100 μM glutamate (blue arrow). (F) As in ‘E’ but with iGluSnFR fluorescence during a 20-ms puff of aCSF containing 100 μM aspartate (purple arrow). (G) Population data comparing peak dF/F shifts after a 20-ms puff of aCSF (as a negative control), T. crass. hom., T. crass. E/S products, aCSF containing 100 μM glutamate (as a positive control), or aCSF containing 100 μM aspartate (as a positive control), values with medians ± IQR, *p ≤ 0.05, ****p ≤ 0.0001. (H) Schematic of whole-cell patch-clamp recording in current-clamp mode from a CA3 pyramidal neuron using a cesium-based internal and in the presence of 2 μM tetrodotoxin (TTX). 20-ms puff of aCSF containing 100 μM glutamate produces a significant depolarizing shift in membrane potential (top trace), a similar puff but with 100 μM aspartate elicited a similar response (bottom trace). (I) Population data comparing peak membrane potential shift after a 20-ms puff of T. crass. hom., T. crass. E/S, aCSF containing 100 μM glutamate, and aCSF containing 100 μM aspartate. Values with medians ± IQR; NS = not significant.
-
Figure 4—source data 1
iGluSnfr fluorescence values and metadata for all cells.
- https://cdn.elifesciences.org/articles/88174/elife-88174-fig4-data1-v1.xlsx
-
Figure 4—source data 2
Patch clamp membrane voltages, cell properties, and metadata for all cells.
- https://cdn.elifesciences.org/articles/88174/elife-88174-fig4-data2-v1.xlsx
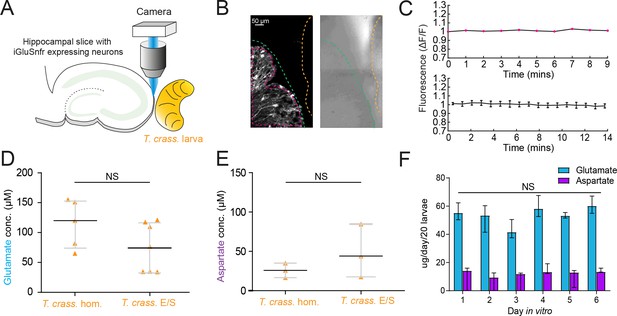
Taenia crassiceps larvae contain and produce glutamate and aspartate.
(A) Schematic of the experimental setup whereby a living Taenia crassiceps (T. crass.) larva is placed in close proximity to a hippocampal organotypic brain slice with iGluSnFR-expressing neurons, for detection of glutamate fluctuations. (B) Left, fluorescence image of a slice with iGluSnFR-expressing neurons (green dashed line) adjacent to a T. crass. larva (orange dashed line), also visible in the transmitted light image (right). The region of interest (pink dashed line) was used to calculate the top dF/F trace in ‘C’. (C) Top, dF/F trace from the example in ‘B’ during a 9-min recording. Lower trace: population data of the mean ± SEM from five slice-larva pairs showed no detectable oscillations or changes in glutamate. (D) Glutamate concentration in T. crass. Homogenate (T. crass. hom.) and T. crass. excretory/secretory (T. crass. E/S) products as measured using a glutamate assay kit, values with medians ± IQR; NS = not significant. (E) Aspartate concentration in T. crass. hom. and T. crass. E/S as measured using an aspartate assay kit, values with medians ± IQR; NS = not significant. (F) Population data showing de novo production of glutamate and aspartate by by by T. crass larvae for 6 days in vitro, values with medians ± IQR; NS = not significant.
-
Figure 5—source data 1
Glutamate and aspartate values and metadata for all samples.
- https://cdn.elifesciences.org/articles/88174/elife-88174-fig5-data1-v1.xlsx
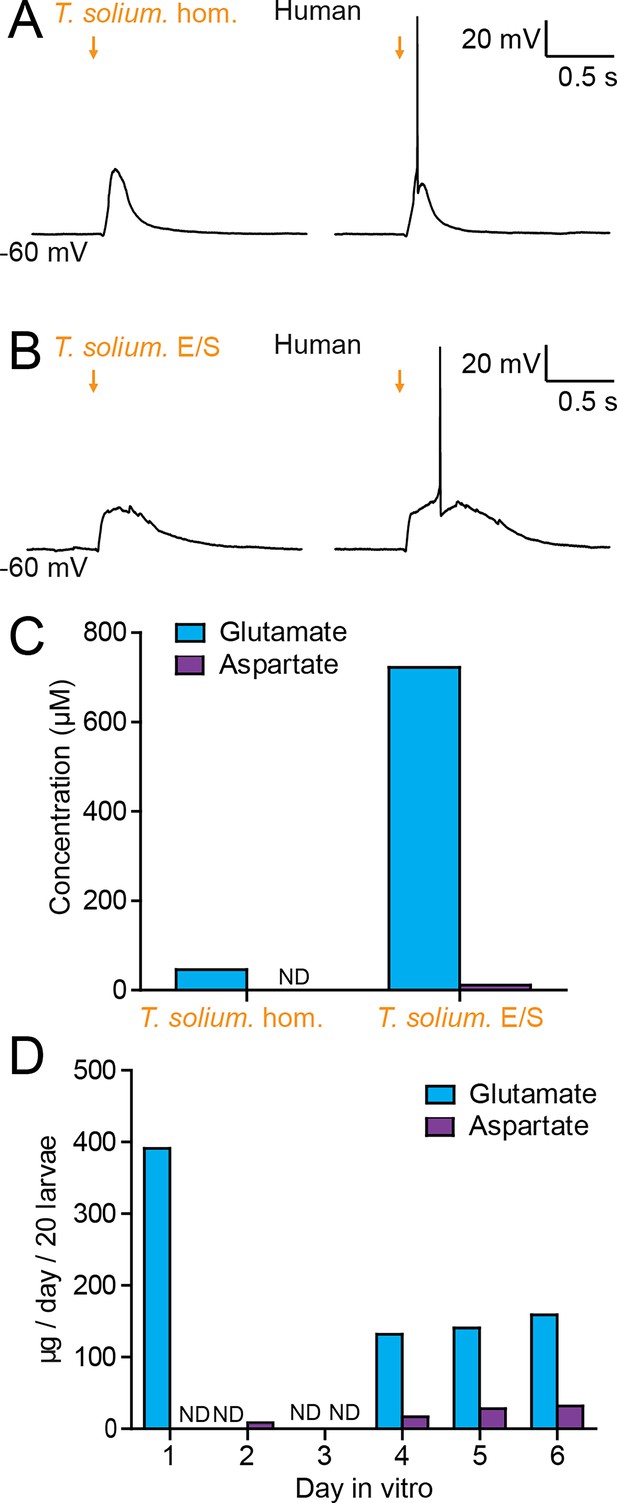
Taenia solium larvae depolarize human neurons via the production of glutamate.
(A) Whole-cell patch-clamp recordings in current-clamp from a layer 2/3 human frontal lobe cortical pyramidal neuron in an acute brain slice, whilst increasing amounts of T. solium homogenate (T. solium hom.) was applied via a puff pipette (left to right, orange arrows). Small amounts of T. solium hom. resulted in depolarization (left), whilst an increased amount elicited an action potential (right). (B) As in ‘A’, T. solium excretory/secretory (T. solium E/S) products elicited membrane depolarization and an increased amount elicited an action potential. (C) Concentrations of glutamate and aspartate in T. solium hom. and T. solium E/S (N = 1, ND = not detectable). (D) De novo production of glutamate and aspartate by T. solium larvae over a 6-day culture period (N = 1, ND = not detectable).
-
Figure 6—source data 1
Patch clamp cell properties and metadata for A&B and glutamate and aspartate values and metadata for all samples in C&D.
- https://cdn.elifesciences.org/articles/88174/elife-88174-fig6-data1-v1.xlsx





