Comparative brain-wide mapping of ketamine- and isoflurane-activated nuclei and functional networks in the mouse brain
Figures
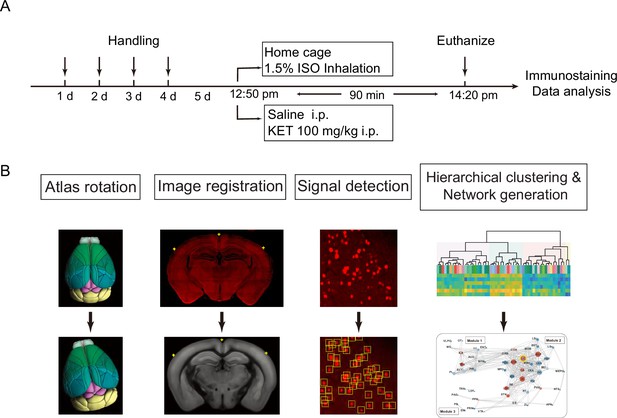
Experimental design and workflow for data analysis.
(A) Experimental timeline for anesthetic exposure. Mice were handled for 4 days, subsequently exposed to KET (100 mg/kg, i.p.) or 1.5% ISO for 90 min, and then euthanized for immunostaining and data analysis. (B) Steps for data processing. Example of brain section registration to a corresponding coronal section from the Allen Brain Atlas (Allen Institute for Brain Science, 2011). For Atlas rotation, the Allen reference atlas was rotated to mimic the slice angle of the experimental brain. Image registration maps the original coronal image (upper panel) to the corresponding Allen Mouse Brain Atlas (lower panel). The registration module applies several geometric transformations (translation, rotation, and scaling) to optimize the matching of the original image to the anatomical structures. Fluorescence signals detected in the original image were projected onto the Allen Mouse Brain Atlas for quantification. Finally, the processed data undergo hierarchical clustering and network analysis to investigate the patterns of c-Fos expression and central network nodes.
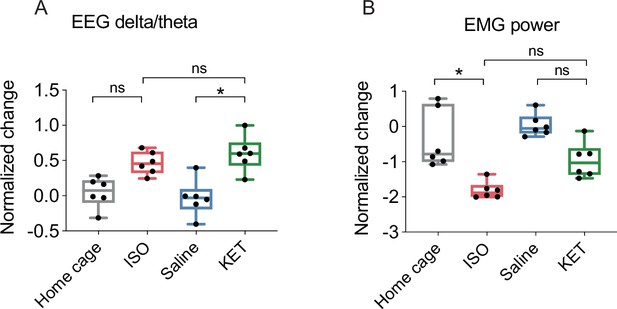
EEG and EMG power change after each treatment.
The box represents the 25th-75th percentiles; the central line denotes the median; whiskers indicate maximum and minimum values. n=6, 6, 8, 6 for the home cage, ISO, saline, and KET, respectively. (A) Normalized change in EEG power: ISO vs KET, p>0.99; Saline vs KET, p=0.01; Home cage vs ISO, p=0.11. (B) Normalized change in EMG power: ISO vs KET, p=0.36; Saline vs KET, p=0.30; Home cage vs ISO, p=0.02. Analyses were conducted using the Kruskal-Wallis test, followed by Dunn’s multiple comparisons test.
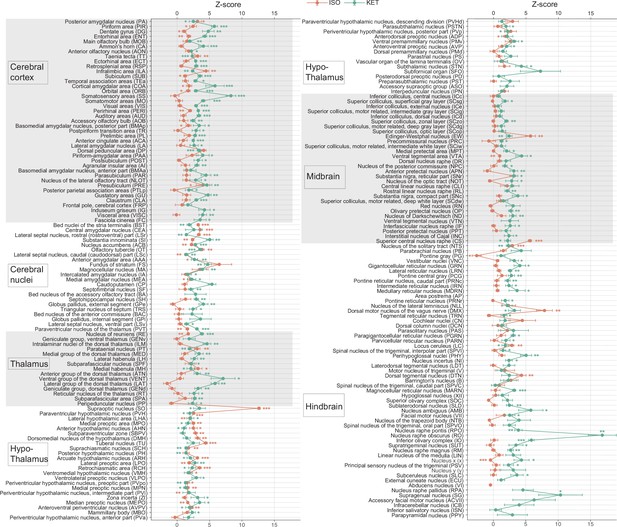
KET and ISO induced c-Fos expression relative to their respective control group across 201 distinct brain regions.
Z-scores represent the normalized c-Fos expression in the KET and ISO groups, calculated against the mean and standard deviation from their respective control groups. Statistical analysis involved the comparison of Z-scores to a null distribution with a zero mean and adjustment for multiple comparisons using the Benjamini–Hochberg method at a 5% false discovery rate (*p<0.05, **p<0.01, ***p<0.001). n=6, 6, 8, 6 for the home cage, ISO, saline, and KET, respectively. Missing values resulted from zero standard deviations in control groups. Brain regions are categorized into major anatomical subdivisions, as shown on the left side of the graph.
-
Figure 2—source data 1
Z-scores of c-Fos expression in each brain region following KET and ISO treatments.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig2-data1-v1.xlsx
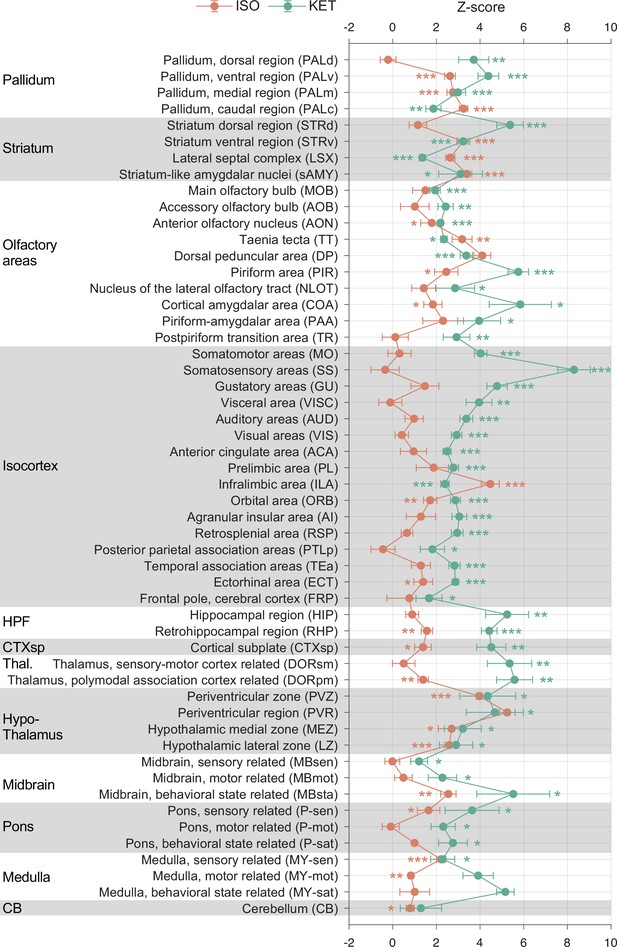
KET and ISO induced c-Fos expression relative to their respective control group across 53 distinct brain regions.
Z-scores for c-Fos expression in the KET and ISO groups were normalized to the mean and standard deviation of their respective control groups. Statistical analysis involved the comparison of Z-scores to a null distribution with a zero mean and adjustment for multiple comparisons using the Benjamini–Hochberg method at a 5% false discovery rate (*p < 0.05, **p < 0.01, ***p < 0.001). Brain regions are organized into major anatomical subdivisions, as indicated on the left side of the graph.

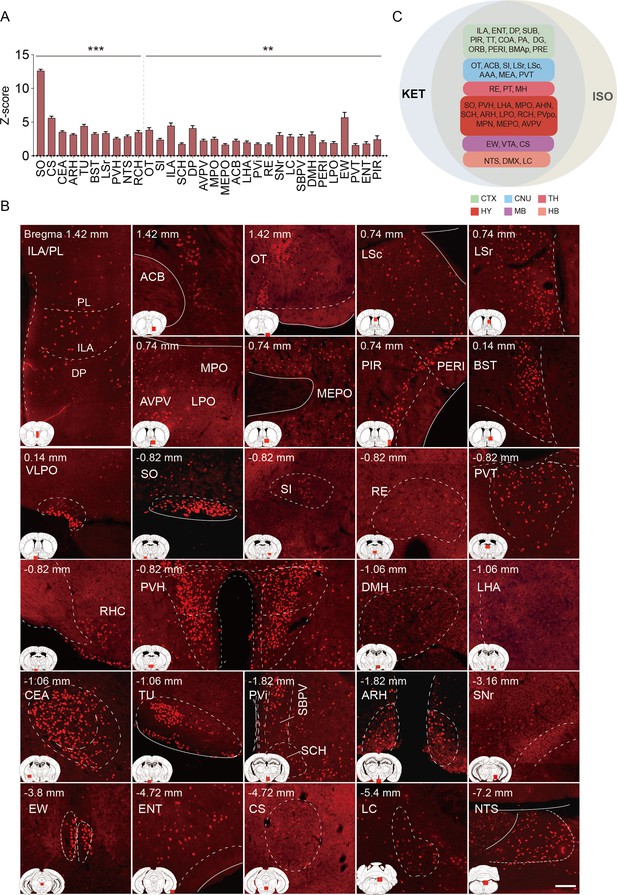
Brain regions exhibiting significant activation by KET.
(A) Fifty-five brain regions exhibited significant KET activation. These were chosen from the 201 regions analyzed in Figure 2, focusing on the top 40% ranked by effect size among those with corrected p values less than 0.05. Data are presented as mean ± SEM, with p-values adjusted for multiple comparisons (*p<0.05, **p<0.01, ***p<0.001). (B) Representative immunohistochemical staining of brain regions identified in A, with control group staining available in Figure 3—figure supplement 1. Scale bar: 200 µm.
-
Figure 3—source data 1
Summary of prior studies on KET activated brain regions identified via c-Fos immunostaining.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig3-data1-v1.docx

c-Fos expression in distinct brain regions following saline administration.
Representative immunohistochemical staining of c-Fos-positive cells in the specified brain regions is provided as a control for the c-Fos expression observed in brain regions after KET exposure, as detailed in Figure 3. Scale bar: 200 µm.
Brain regions exhibiting significant activation by ISO.
(A) Brain regions significantly activated by ISO were initially identified using a corrected p-value below 0.05. From these, the top 40% in effect size (Cohen’s d) were further selected, resulting in 32 key areas. p-values are adjusted for multiple comparisons (**p<0.01, ***p<0.001). (B) Representative immunohistochemical staining of brain regions identified in A. Control group staining is available in Figure 4—figure supplement 1. Scale bar: 200 µm. Scale bar: 200 µm. (C) A Venn diagram displays 43 brain regions co-activated by KET and ISO, identified by the adjusted p-values (p<0.05) for both KET and ISO. CTX: cerebral cortex; CNU: cerebral nuclei; TH: thalamus; HY: hypothalamus; MB: midbrain; HB: hindbrain.
-
Figure 4—source data 1
Summary of prior studies on brain regions activated by ISO, as detected through c-Fos immunostaining.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig4-data1-v1.docx

c-Fos expression in home cage group.
Representative immunohistochemical staining of c-Fos-positive cells in the home cage group, serving as a baseline comparison for the ISO groups. Scale bar: 200 µm.
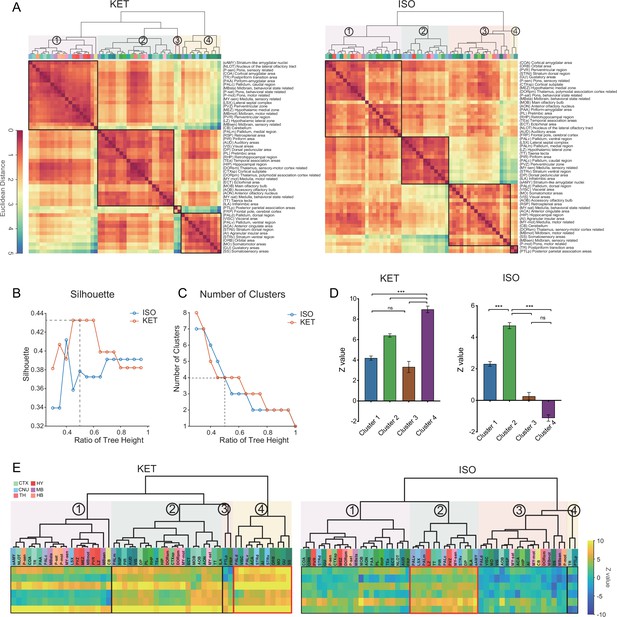
Comparison of c-Fos activation patterns in response to KET and ISO.
(A) Hierarchical clustering of log-relative c-Fos density data for KET and ISO was conducted using the complete linkage method, based on the Euclidean distance matrix. Clusters were identified using a dendrogram cut-off ratio of 0.5, with numerical labels indicating distinct clusters. (B) Silhouette values plotted against the dendrogram tree height ratio for KET and ISO reveal higher Silhouette values at a 0.5 ratio (dashed line), suggesting optimal clustering at this level. (C) The number of clusters for each treatment condition at different dendrogram tree height ratios is shown, with a cut-off level of 0.5 yielding 4 clusters for both KET and ISO (marked by the dashed line). (D) Bar graphs represent the Z values for clusters under both KET and ISO conditions. Data are presented as mean ± SEM. One-way ANOVA with Tukey’s post hoc multiple comparisons. ns: no significance; ***p<0.001. (E) Heatmaps illustrate Z values of the log relative c-Fos expression density in clustered brain regions. The arrangement and abbreviations of the brain regions, along with the numerical labels, correspond to those in A. Red box in each heatmap emphasizes the cluster with the highest mean Z value in its respective condition. CTX: cerebral cortex; CNU: cerebral nuclei; TH: thalamus; HY: hypothalamus; MB: midbrain; HB: hindbrain.
-
Figure 5—source data 1
Hierarchical clustering of 53 brain regions based on log-relative c-Fos density responses to KET and ISO treatments, related to Figure 5A.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig5-data1-v1.xlsx
-
Figure 5—source data 2
Z values of the log relative c-Fos density in 53 brain regions in response to KET and ISO, related to Figure 5E.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig5-data2-v1.xlsx
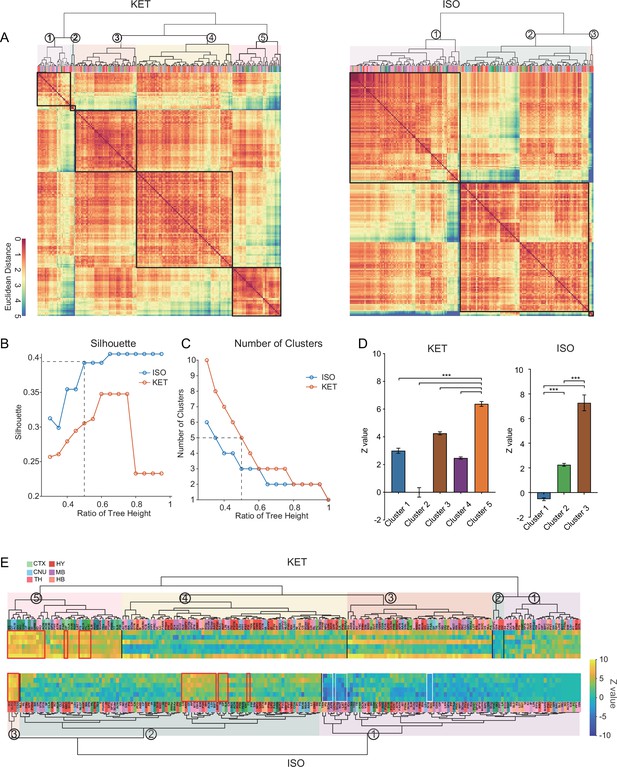
Similarities and differences in KET and ISO-activated c-Fos brain areas.
(A) Heatmaps display hierarchical clustering of brain regions based on log-relative c-Fos density for KET and ISO groups, using complete linkage on Euclidean distances. Clusters are demarcated with a 0.5 dendrogram cut-off. (B) Silhouette values are plotted against the dendrogram tree height ratios in A. (C) The number of clusters identified at different dendrogram tree height ratios, with a cut-off ratio of 0.5 resulting in 5 for KET and 3 clusters for ISO (indicated by the dashed line). (D) Z values for identified clusters under KET and ISO conditions. Data are presented as mean ± SEM. One-way ANOVA with Tukey’s post hoc multiple comparisons. ns: no significance; ***p<0.001. (E) Heatmaps illustrate Z values of log-relative c-Fos densities in brain region clusters, arranged and labeled as in A. Red boxes highlight regions ranking in the top 10% within their respective clusters, and white boxes indicate regions with Z values less than –2.
-
Figure 6—source data 1
Hierarchical clustering of brain regions based on log-relative c-Fos density responses to KET and ISO treatments, related to Figure 6A.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig6-data1-v1.xlsx
-
Figure 6—source data 2
Z values of the log relative c-Fos density in 201 brain regions in response to KET and ISO, related to Figure 6E.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig6-data2-v1.xlsx
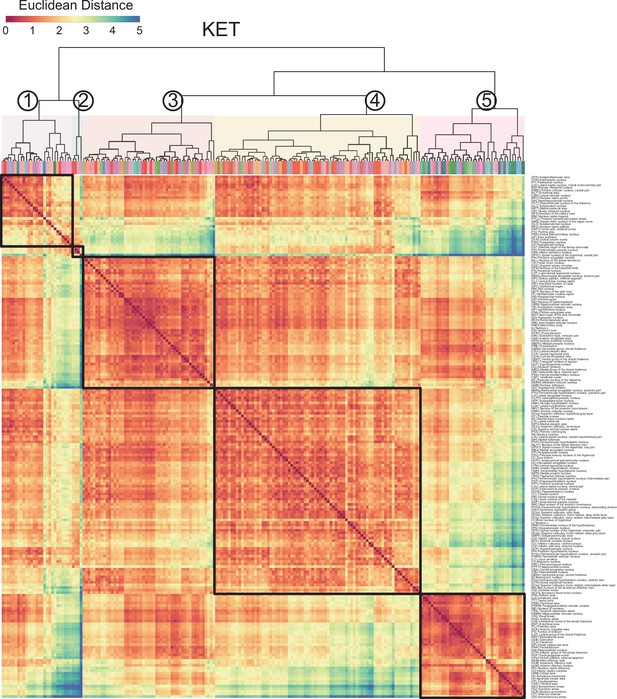
Region labels for the hierarchical clustering of the KET group in Figure 6A.
Region labels for the hierarchical clustering of the ISO group in Figure 6A.
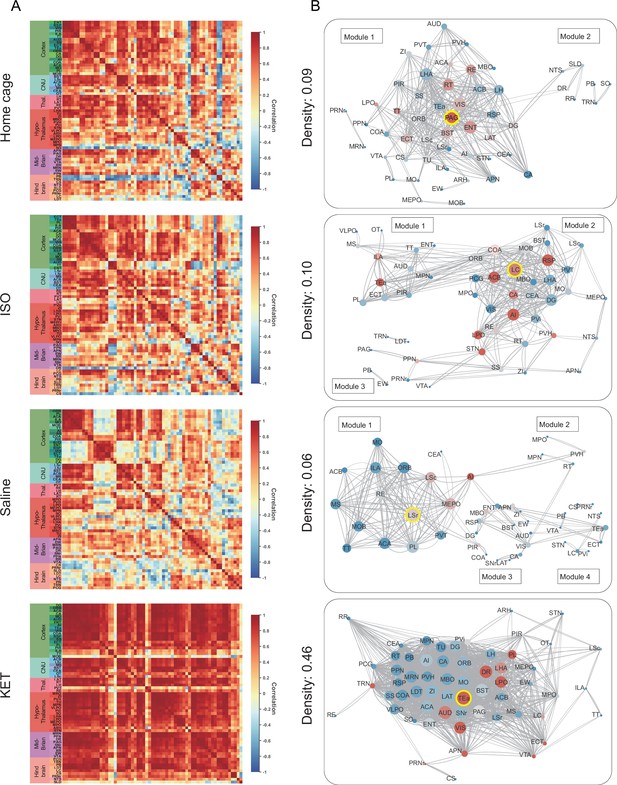
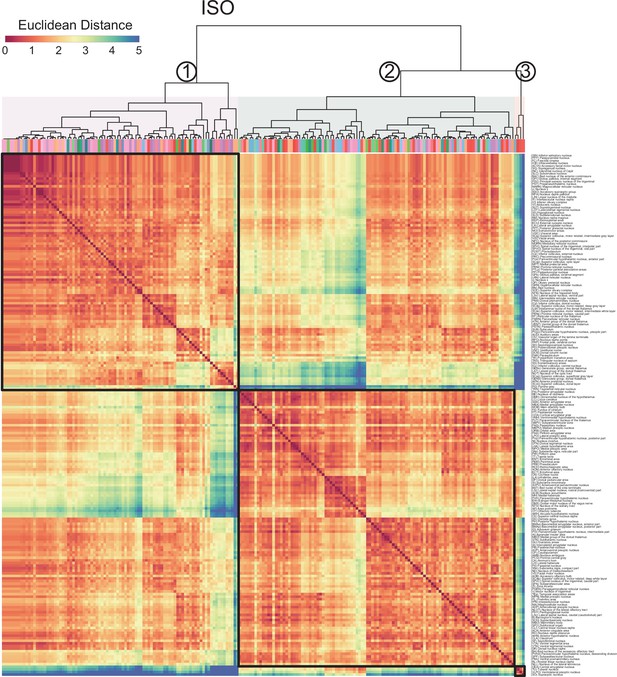
Generation of anesthetics-induced networks and identification of hub regions.
(A) Heatmaps display the correlations of log c-Fos densities within brain regions (CTX, CNU, TH, HY, MB, and HB) for various states (home cage, ISO, saline, KET). Correlations are color-coded according to Pearson’s coefficients. The brain regions within each anatomical category are organized by hierarchical clustering of their correlation coefficients. Full names and expression levels for each brain region are detailed in Figure 3—figure supplement 1. (B) Network diagrams illustrate significant positive correlations (p<0.05) between regions, with Pearson’s r exceeding 0.82. Edge thickness indicates correlation magnitude, and node size reflects the number of connections (degree). Node color denotes betweenness centrality, with a spectrum ranging from dark blue (lowest) to dark red (highest). The networks are organized into modules consistent with the clustering depicted in Figure 6—figure supplement 1A.
-
Figure 7—source data 1
Correlation matrix of log c-Fos density across 63 brain regions under home cage, ISO, saline, and KET conditions, related to Figure 7A.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig7-data1-v1.xlsx
-
Figure 7—source data 2
Network centrality metrics for each brain region under home cage, ISO, saline, and KET conditions: degree, betweenness, and eigenvector centralities, related to Figure 7B.
- https://cdn.elifesciences.org/articles/88420/elife-88420-fig7-data2-v1.xlsx
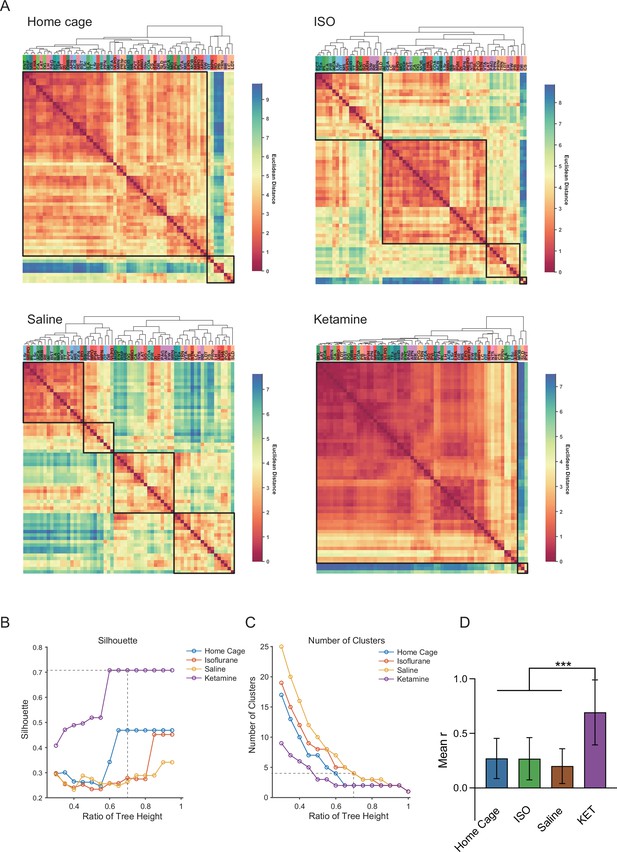
Hierarchical clustering across different conditions.
(A) Heatmaps depict the hierarchical clustering of brain regions within the home cage, ISO, saline, and KET groups, using Euclidean distance matrices calculated from correlation coefficients. Modules are demarcated within each dendrogram at a cut-off threshold of 0.7. (B) Silhouette scores are plotted against the dendrogram tree height ratio for each condition, with optimal cluster definition indicated by a dashed line at a 0.7 ratio. (C) The number of clusters formed at different cutoff levels. At a ratio of 0.7, ISO and saline treatments result in three clusters, whereas home cage and KET conditions yield two clusters. (D) The mean Pearson’s correlation coefficient (r) was computed from interregional correlations displayed in Figure 7A. Data were analyzed using one-way ANOVA with Tukey’s post hoc test, ***p < 0.001.
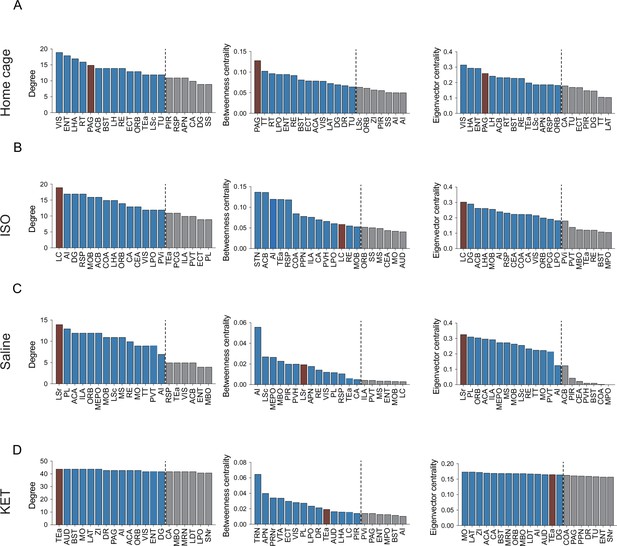
Hub region characterization across different conditions: home cage (A), ISO (B), saline (C), and KET (D) treatments.
Brain regions are sorted by degree, betweenness centrality, and eigenvector centrality, with each metric presented in separate bar graphs. Bars to the left of the dashed line indicate the top 20% of regions by rank, highlighting the most central nodes within the network. Red bars indicate regions that consistently rank highly in degree, betweenness centrality, and eigenvector centrality across all metrics.
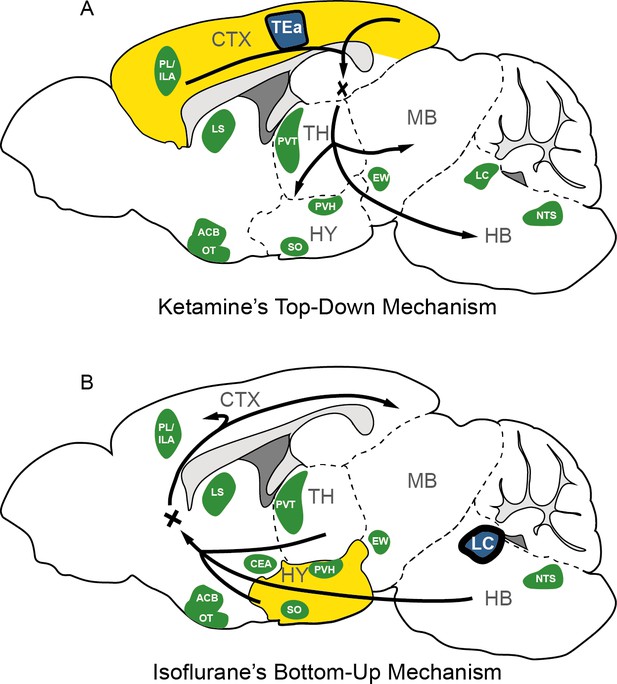
The possible mechanism for KET and ISO-induced unconsciousness.
The distinct pathways of KET and ISO-induced unconsciousness can be explained by two contrasting mechanisms. (A) The ‘top-down’ process attributes KET’s effect to widespread cortical activation (represented in yellow), with the temporal association areas (TEa) acting as the central node in the functional network (depicted in blue). (B) The ‘bottom-up’ approach suggests that ISO-induced unconsciousness stems from the activation of certain hypothalamic regions (highlighted in yellow), with the locus coeruleus (LC) acting as the hub node within the ISO-induced functional network. Green denotes representative regions co-activated by both ISO and KET. Adapted from Figure 1 of Reimann and Niendorf, 2020. PL, prelimbic area; ILA, infralimbic areas; LS, lateral septal nucleus; ACB, nucleus accumbens; OT, olfactory tubercle; PVT, paraventricular nucleus of the thalamus; EW, Edinger-Westphal nucleus; SO, supraoptic nucleus; PVH, paraventricular hypothalamic nucleus; NTS, nucleus of the solitary tract; CTX: cerebral cortex; TH: thalamus; HY, hypothalamus; MB; midbrain; HB, hindbrain.
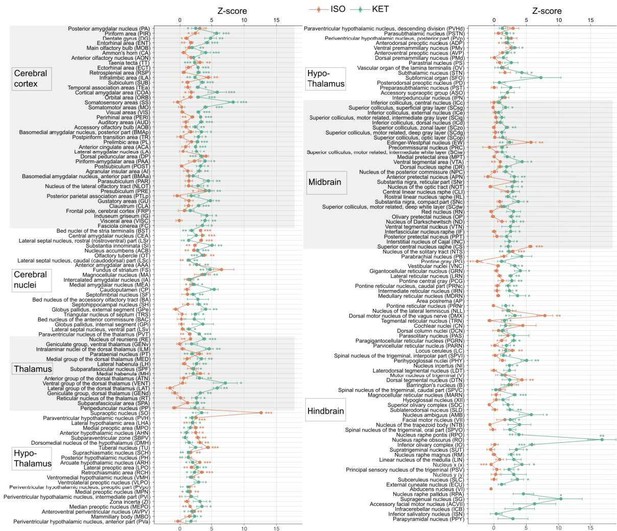
KET and ISO induced c-Fos expression relative to their respective control group across 201 distinct brain regions.
Z-scores represent the normalized c-Fos expression in the KET and ISO groups, calculated against the mean and standard deviation from their respective control groups. Statistical analysis involved the comparison of Z-scores to a null distribution with a zero mean and adjustment for multiple comparisons using the Benjamini–Hochberg method at a 5% false discovery rate (*p < 0.05, **p < 0.01, ***p < 0.001). n = 6, 6, 8, 6 for the home cage, ISO, saline, and KET, respectively. Missing values resulted from zero standard deviations in control groups. Brain regions are categorized into major anatomical subdivisions, as shown on the left side of the graph.
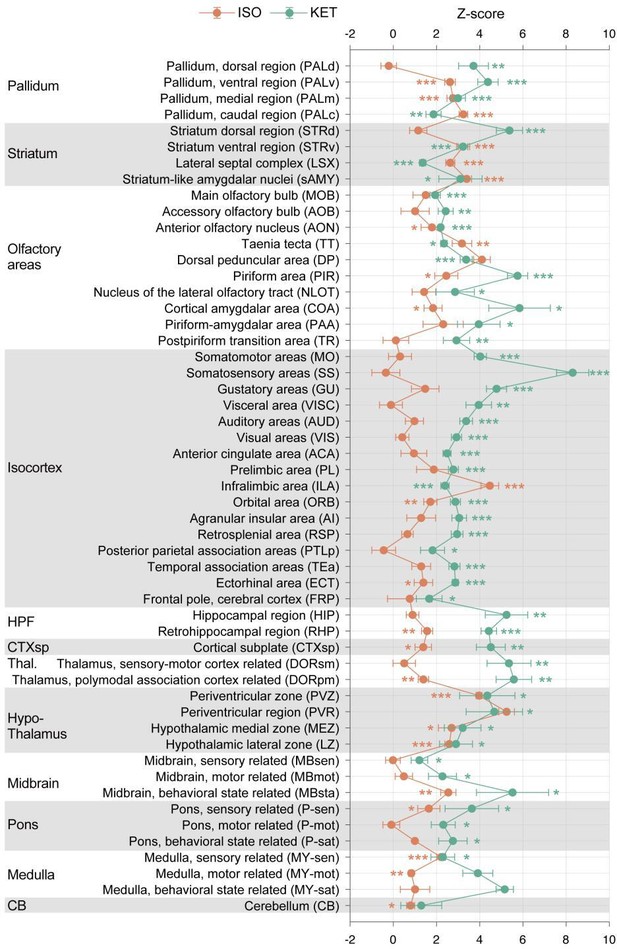
KET and ISO induced c-Fos expression relative to their respective control group across 53 distinct brain regions.
Z-scores for c-Fos expression in the KET and ISO groups were normalized to the mean and standard deviation of their respective control groups. Statistical analysis involved the comparison of Z-scores to a null distribution with a zero mean and adjustment for multiple comparisons using the Benjamini–Hochberg method at a 5% false discovery rate (*p < 0.05, **p < 0.01, ***p < 0.001). Brain regions are organized into major anatomical subdivisions, as indicated on the left side of the graph.

Brain regions exhibiting significant activation by KET.
(A) Fifty-five brain regions exhibited significant KET activation. These were chosen from the 201 regions analyzed in Figure 2, focusing on the top 40% ranked by effect size among those with corrected p values less than 0.05. Data are presented as mean ± SEM, with p-values adjusted for multiple comparisons (*p < 0.05, **p < 0.01, ***p < 0.001). (B) Representative immunohistochemical staining of brain regions identified in Figure 3A, with control group staining available in Figure 3—figure supplement 1. Scale bar: 200 µm.
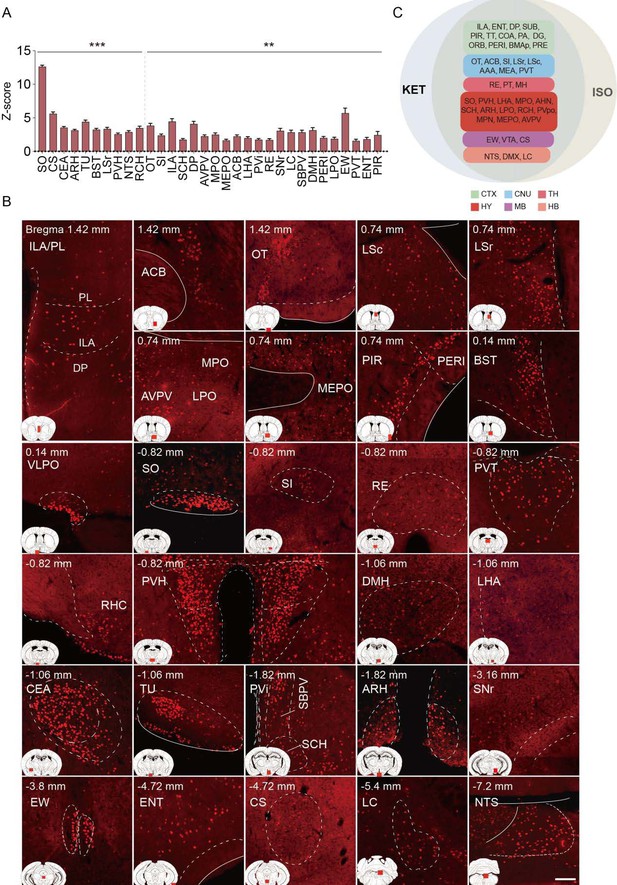
Brain regions exhibiting significant activation by ISO.
(A) Brain regions significantly activated by ISO were initially identified using a corrected p-value below 0.05. From these, the top 40% in effect size (Cohen’s d) were further selected, resulting in 32 key areas. p-values are adjusted for multiple comparisons (**p < 0.01, ***p < 0.001). (B) Representative immunohistochemical staining of brain regions identified in Figure 4A. Control group staining is available in Figure 4—figure supplement 1. Scale bar: 200 µm. Scale bar: 200 µm. (C) A Venn diagram displays 43 brain regions co-activated by KET and ISO, identified by the adjusted p-values (p < 0.05) for both KET and ISO. CTX: cerebral cortex; CNU: cerebral nuclei; TH: thalamus; HY: hypothalamus; MB: midbrain; HB: hindbrain.





