Association of genetic variation in COL11A1 with adolescent idiopathic scoliosis
Figures
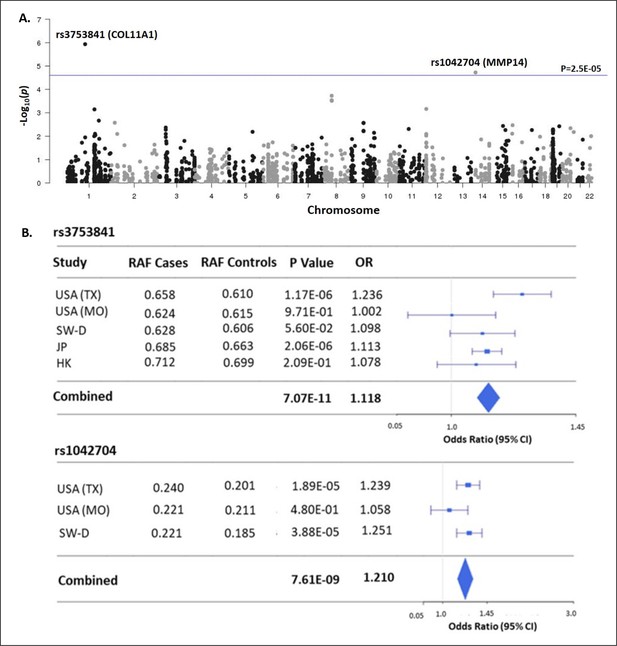
Matrisome-wide association study.
(A) Manhattan plot showing –log10 p-values (y-axis) versus chromosomal position (x-axis) for the 2008 common coding variants tested in the discovery study USA (TX). The horizontal line represents the threshold for significance level (p-value <2.5 × 10–5) after Bonferroni multiple testing correction. (B) Tests of association for SNPs rs3753841 and rs1042704 in discovery and independent replication cohorts. RAF – reference allele frequency; OR – odds ratio; CI –confidence interval.
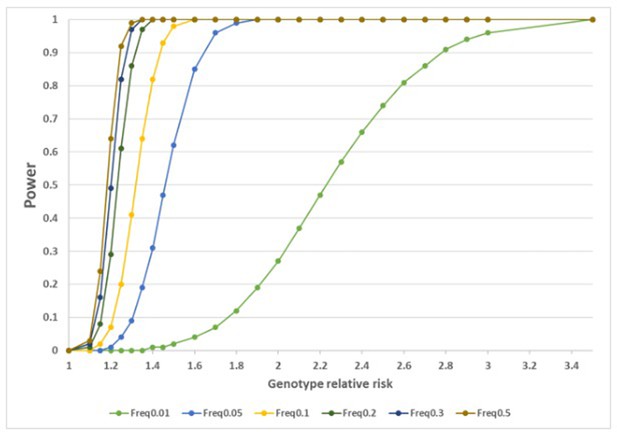
Statistical power as a function of the genotype relative risk (OR) to detect significant association at α=2.5E–05 for different disease allele frequencies, using 1358 cases and 12,507 controls in the discovery study.
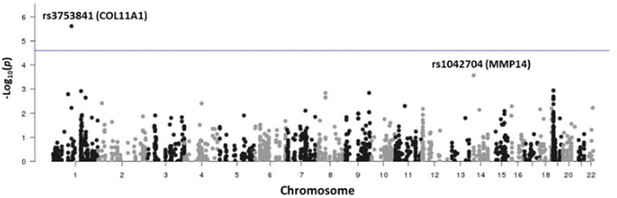
Manhattan plot showing –log10 p-values (y-axis) plotted versus chromosomal position (x-axis) for the 2009 common coding variants tested for females in the discovery study USA (TX).

Tests of association of SNP rs1042704 with adolescent idiopathic scoliosis (AIS) in East Asian cohorts.
RAF – reference allele frequency; OR – odds ratio; CI – confidence interval.
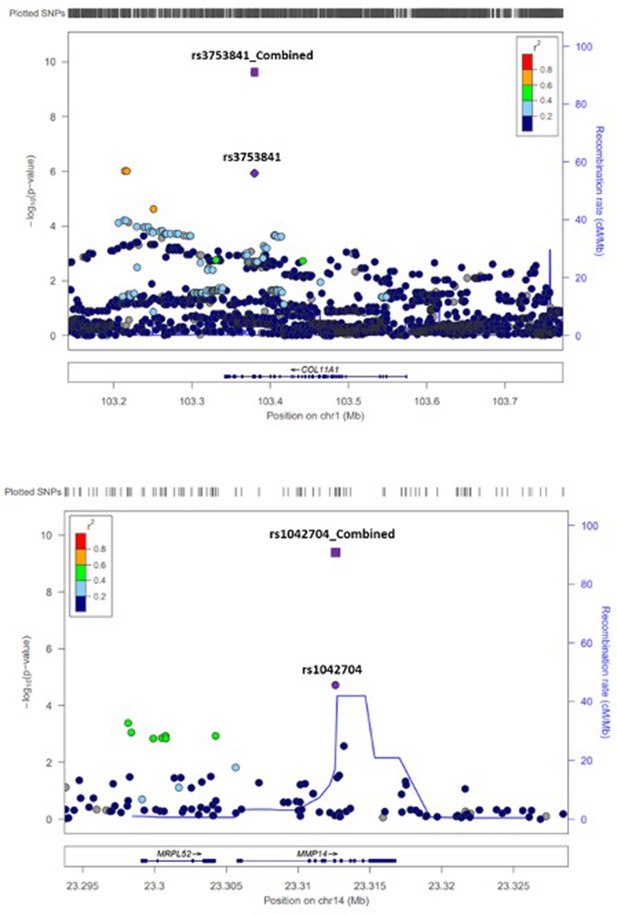
LocusZoom plots of SNPs in genomic regions of SNPs rs3753841 (top) and rs1042704 (bottom).
Genomic location (x-axis) is plotted versus –log10 p-values (y-axis). Correlation between SNPs is shown by color-coded r2 value (legend).
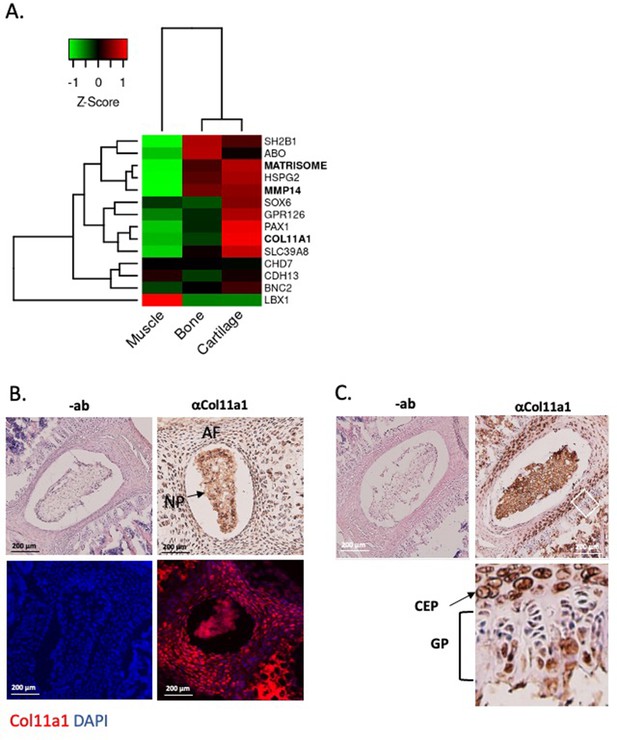
Col11a1 and Mmp14 expression in spine.
(A) A heatmap of transcript per million (TPM) values of COL11A1, MMP14, and other published genes associated with adolescent idiopathic scoliosis (AIS). The average TPM value of matrisome genes is represented as MATRISOME. (B) Detection of collagen a1(XI) in P0.5 mouse spine. Immunohistochemistry (IHC) shown at top, with immunofluorescence (IF) staining below. ‘-ab’ refers to negative controls lacking primary antibody (shown at left). Results are representative of N≥3 technical replicates in whole spines. (C) Detection of collagen a1(XI) in P28 mouse spine. Negative antibody IHC control shown at left; antibody-positive IHC shown at right. Enlarged, rotated view of white boxed area shows a biphasic staining pattern. CEP – cartilage endplate; GP – growth plate. Results are representative of N≥3 technical replicates in whole spines.

Immunofluorescence (IF) staining using collagen a1(XI) antibody in P28 ribs (top).
Boxed area shows biphasic staining pattern expanded at right. R – presumed resting zone; P – columnar chondrocytes of presumed proliferating zone; H – presumed hypertrophic zone and chondro-osseous junction. Each image is representative of results in at least three animals.
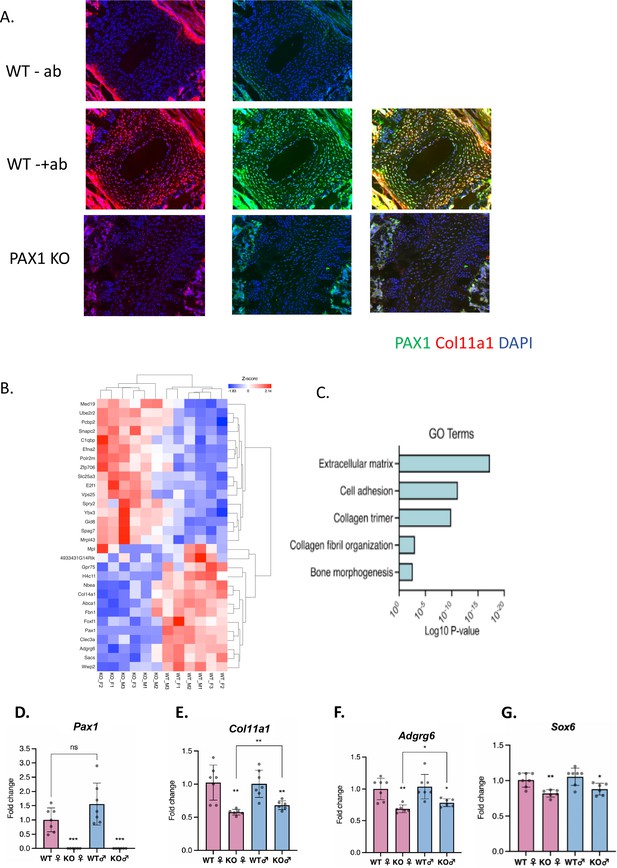
Assessing Pax1 regulation of Col11a1 expression.
(A) Immunofluorescence (IF) staining of P28 intervertebral disc (IVD) from thoracic regions of Pax1-/- (bottom) and wild-type (WT) littermate (middle, top) mice using PAX1- (green) and collagen a1(XI)-specific (red) antibodies and DAPI nuclear counterstain. Antibody-negative controls are shown at top as (-ab). Results are representative of N≥3 technical replicates in whole spines. (B) Heatmap of differentially expressed genes (p-value <0.0001) in embryonic stage 12.5 (E12.5) tails of WT and Pax1-/- mice. (C) Gene ontology (GO) analysis of differentially expressed genes in E12.5 tail WT and Pax1-/- mice. (D–G) Gene expression levels dissected from E12.5 mouse tail from WT and Pax1-/- (knockout [KO]) mice as determined by quantitative real-time PCR (qRT-PCR). Each value represents the ratio of each gene expression to that of β-actin, and values are mean ± standard deviation. The expression value of WT female group was arbitrarily set at 1.0. Each dot represents one embryo and statistical differences were determined using a two-sided unpaired t-test (*p<0.05, **p<0.01, ***p<0.001).
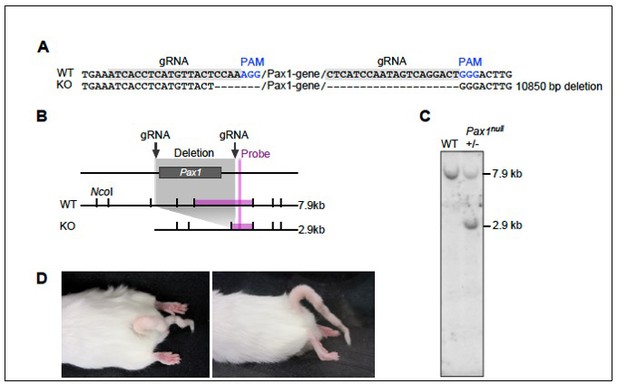
Design and validation of Pax1 knockout in mouse using CRISPR-mediated gene targeting.
(A) Guide RNAseq and PAM sites flanking 5’ and 3’ sides of the Pax1 gene. (B) Location of the NcoI restriction enzyme sites, shown as vertical lines in wild-type (WT) and knockout (KO) loci. The deletion and probe location are shown as gray and pink rectangles, respectively. Expected band sizes in WT and KO are written to the right of the map. (C) Southern blot analyses of WT and heterozygous KO mice with the estimated band size written to the right of the blot. Southern blot analyses were performed >2 times using founder and F1 generation. (D) Pax1-/- mice showed kinky tail phenotype on dorsal (left) and lateral (right) views.
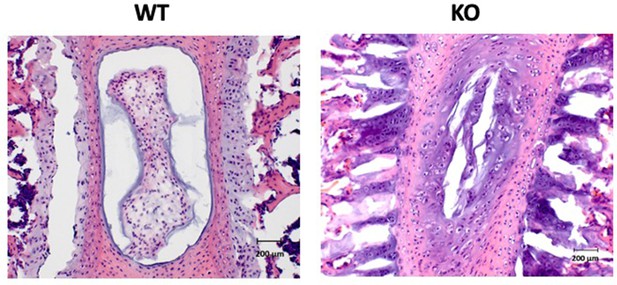
HE staining of sectioned lumbar spines from wild-type (left) and Pax1-/- (right) mice.
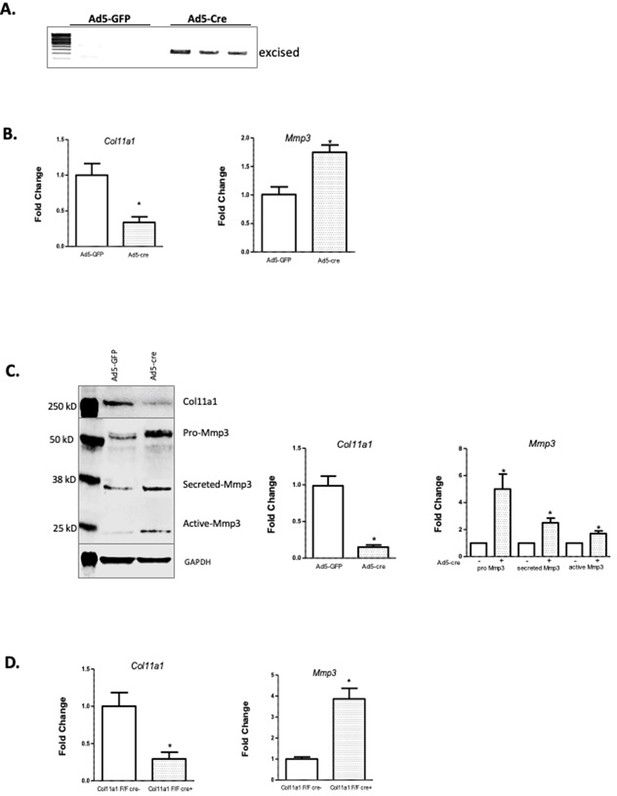
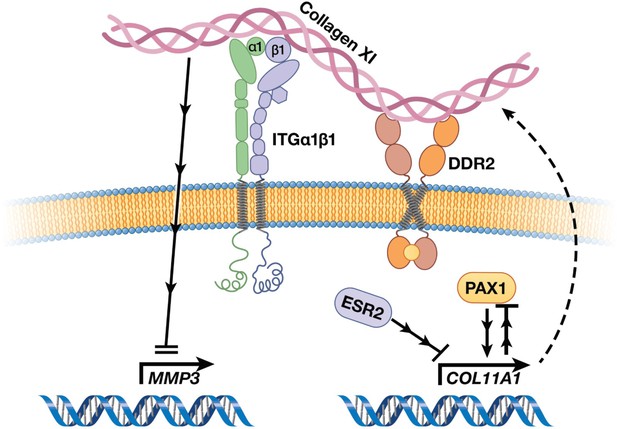
Col11a1 regulation of Mmp3 expression in cartilage.
(A) PCR assay of Col11a1 excision in Col11a1fl/fl cultured costal chondrocytes. (B) Gene expression levels from Col11a1fl/fl cultured costal chondrocytes transduced with green fluorescent protein (GFP) (Ad5-GFP, left) or Cre-expressing adenovirus (Ad5-cre, right) as determined by quantitative real-time PCR (qRT-PCR). Values represent the ratio of each gene expression to that of GAPDH, and values are mean ± standard deviation. The expression value of control Ad5-GFP results was arbitrarily set at 1.0. Statistical differences were determined using a two-sided paired t-test (*p<0.05). Results shown for N≥3 biologic replicates, each including three technical replicates. (C) Western blot detection of collagen a1(XI), MMP3, and GAPDH loading control in cultured costal chondrocytes after Ad5-GFP or Ad5-cre transduction. Results are representative of N=4 biologic replicates. Protein size ladder is shown in lane 1. Quantification of bands detected by western blotting, where Ad5-GFP was set to 1.0, is shown at right. Statistical differences were determined using a two-sided paired t-test (*p<0.05). (D) Gene expression levels from dissected Col11a1fl/fl:ATC costal cartilage, analyzed as described in (A). Results shown for N=3 biologic replicates, each including three technical replicates.
-
Figure 4—source data 1
Original gel images of Col11a1 fl/fl excision PCR assay in Figure 4A.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig4-data1-v1.zip
-
Figure 4—source data 2
Figure 4A and original gel images of Col11a1 fl/fl excision PCR assay with highlighted and labeled bands.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig4-data2-v1.zip
-
Figure 4—source data 3
Original western blot images (anti-COL11A1, anti-MMP3, anti-GAPDH) shown in Figure 4C.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig4-data3-v1.zip
-
Figure 4—source data 4
Figure 4C and original western blot images (anti-COL11A1, anti-MMP3, anti-GAPDH) with highlighted bands and labels.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig4-data4-v1.zip

Relative expression of MMP3 compared to COL11A1 in human spinal tissues.
Values are plotted from average TPM values for each gene.
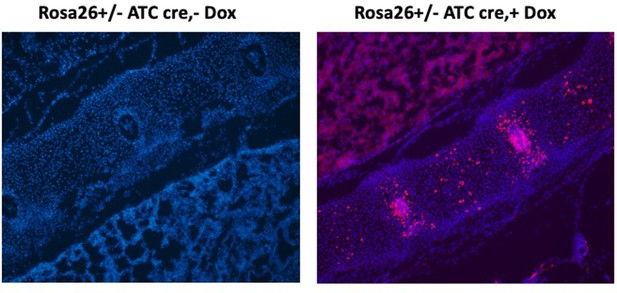
Immunofluorescence microscopy of Rosa26+/-:ATC P0 spines, without doxycycline treatment (left) and after doxycycline treatment starting at embryonic stage 15.5 (E15.5) (right).
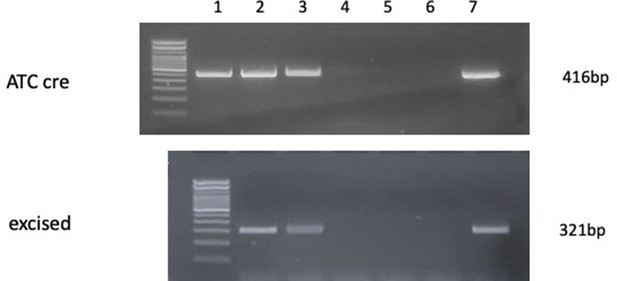
PCR assays in DNA from costal cartilage.
Top gel shows detection of ATC Cre transgene (top, 416 bp band) in Col11a1fl/fl:ATC mice (lanes 2,3,7) and Col11a1fl/fl mice (lanes 4,5,6) after doxycycline treatment. Lane 1 is positive control. Bottom gel shows Col11a1 fl/fl excision-specific band (321 bp) in Col11a1fl/fl:ATC mice (lanes 2,3,7) and Col11a1fl/fl mice (lanes 4,5,6) after doxycycline treatment.
-
Figure 4—figure supplement 3—source data 1
Original gel images of Col11a1 fl/fl excision PCR assay in Figure 4—figure supplement 3.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig4-figsupp3-data1-v1.zip
-
Figure 4—figure supplement 3—source data 2
Original gel images of Col11a1 fl/fl excision PCR assay in Figure 4—figure supplement 3 with highlighted and labeled bands.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig4-figsupp3-data2-v1.zip
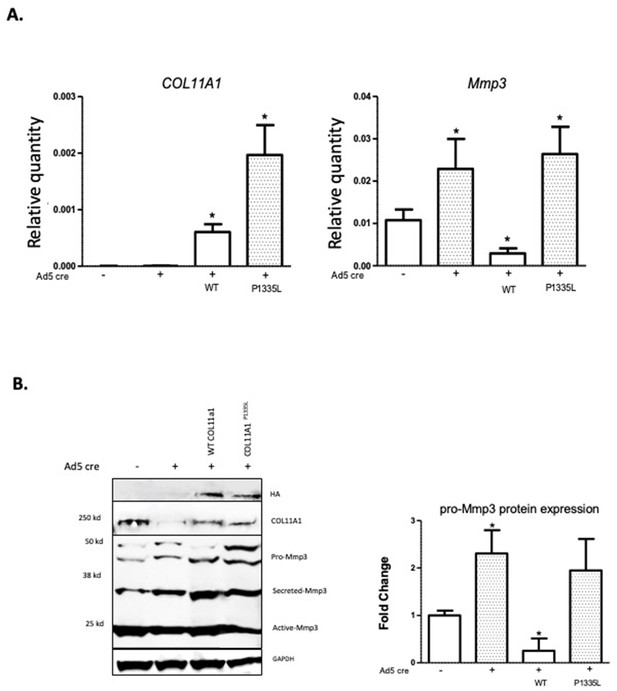
Col11a1P1335L regulation of Mmp3 expression in lentiviral transduced mouse GPCs.
(A) Quantitative real-time PCR (qRT-PCR) of human COL11A1 and endogenous mouse Mmp3 in SV40-immortalized mouse costal chondrocytes transduced with the lentiviral vector only (lanes 1,2), human wild-type (WT) COL11A1 (lane 3), or COL11A1P1335L. Values represent the ratio of each gene expression to that of GAPDH, and values are mean ± standard deviation. Significant quantitative changes (p≤0.05) relative to vector-only transfected cells as measured by unpaired t-tests are shown by *. Results shown for N=4 biologic replicates, each including three technical replicates. (B) Western blot corresponding to experiments shown in (A) using HA antibody to detect epitope-tagged human collagen a1(XI), COL11A1 antibody to detect mouse and human collagen a1(XI), MMP3 antibody to detect endogenous mouse MMP3, and GAPDH. Values are mean after normalization to GAPDH, ± standard deviation. Significant differences (p≤0.05) relative to vector-only, Ad5-negative transfected cells as measured by unpaired t-tests are shown by *.
-
Figure 5—source data 1
Original western blot images (anti-COL11A1, anti-MMP3, anti-GAPDH) with highlighted bands and labels.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig5-data1-v1.zip
-
Figure 5—source data 2
Figure 5B and original western blot images (anti-COL11A1, anti-MMP3, anti-GAPDH) with highlighted bands and labels.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig5-data2-v1.zip
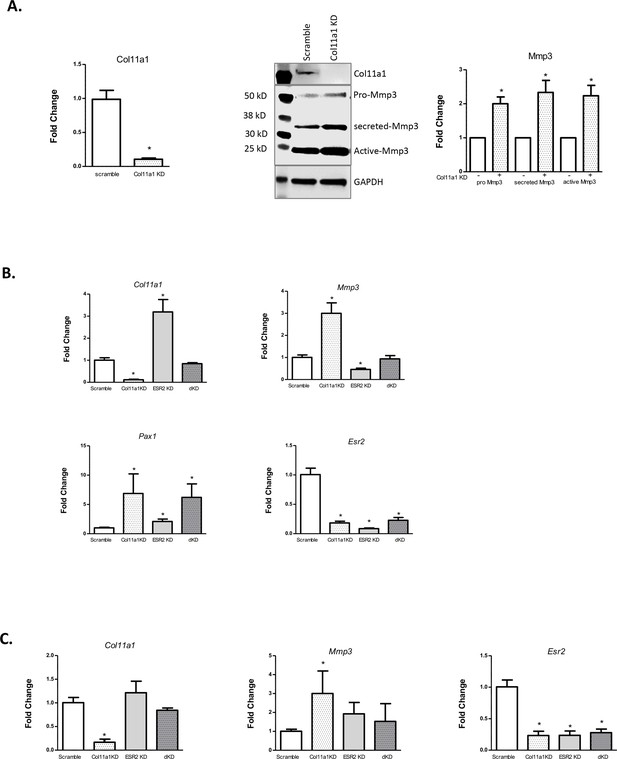
Effects of estrogen receptor beta on Col11a1-Mmp3 signaling axis.
(A) RT-qPCR (left) of Col11a1 expression after siRNA-mediated knockdown as shown at left. Representative western blot (of N=4 biologic replicates) of cultured costal chondrocytes after scramble or Col11a1-specific siRNA knockdown is shown in middle. Protein size ladder is shown in lane 1. Quantification of bands detected by western blotting is shown at right, where scramble results were set to 1.0. Values are mean after normalization to GAPDH, ± standard deviation. (B) Gene expression levels of Col11a1, Mmp3, Pax1, and Esr2 mRNA in cultured costal chondrocytes showing fold change relative to the scramble control. dKD = double Col11a1-Esr2-specific siRNA knockdowns. Each value represents the ratio of each gene expression to that of GAPDH, and values are mean ± standard deviation. Results are representative of N≥3 biologic replicates, each including three technical replicates. (C) Gene expression levels from rat cartilage endplate (CEP) cells, as described in (B).
-
Figure 6—source data 1
Original western blot images (anti-COL11A1, anti-MMP3, anti-GAPDH) with highlighted bands and labels.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig6-data1-v1.zip
-
Figure 6—source data 2
Figure 6A and original western blot images (anti-COL11A1, anti-MMP3, anti-GAPDH) with highlighted bands and labels.
- https://cdn.elifesciences.org/articles/89762/elife-89762-fig6-data2-v1.zip
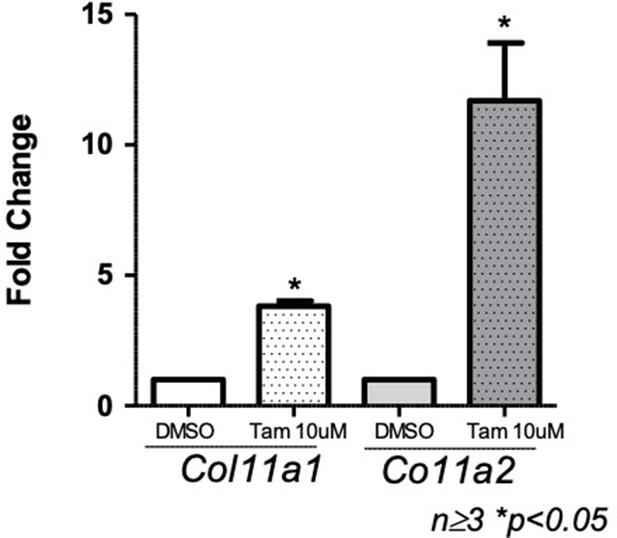
Quantitative real-time PCR (qRT-PCR) of Col11a1 and Col11a2 mRNA in cultured costal chondrocytes treated with DMSO carrier or tamoxifen (N≥3 independent experiments).

Quantitative real-time PCR (qRT-PCR) of Sfrp2, Krt19, and Mmp12 mRNA to validate expression of these marker genes in cultured rat nucleus pulposus (NP), annulus fibrosus (AF), and cartilage endplate (CEP) cells.
Tables
Study cohorts.
| Cohort | Ethnicity | Stage | Subjects | Cases | Controls | ||
|---|---|---|---|---|---|---|---|
| Male | Female | Male | Female | ||||
| USA (TX) | NHW | Discovery | 13,865 | 201 | 1157 | 5369 | 7138 |
| USA (MO) | NHW | Replication | 2951 | 201 | 1102 | 1049 | 689 |
| SW-D | NHW | Replication | 4627 | 222 | 1409 | 505 | 2491 |
| JP | EAS (Japanese) | Replication | 79,211 | 323 | 5004 | 40,205 | 33,679 |
| HK | EAS (HAN Chinese) | Replication | 3103 | 178 | 812 | 858 | 1255 |
| Total | 103,757 | 10,519 | 93,238 | ||||
-
USA (TX): Texas cohort; USA (MO): Missouri cohort; SW-D: Danish cohort; JP: Japanese cohort; HK: Hong Kong cohort; NHW: Non-Hispanic White; EAS: East Asian.
RNA and DNA oligonucleotide primers used for siRNA knockdown, RT-qPCR, and genotyping experiments.
| Mouse Esr2 siRNA | CAAGUGUUACGAAGUAGGAdT |
| Mouse Col11a1 siRNA | GAAAGAAGGUGCAAAGGGUdT |
| Mouse Mmp3 F | CTCTGGAACCTGAGACATCACC |
| Mouse Mmp3 R | AGGAGTCCTGAGAGATTTGCGC |
| Mouse Col11a1 F | AGGAGAGTTGAGAATTGGGAATC |
| Mouse Col11a1 R | TGGTGATCAGAATCAGAAGTT |
| Mouse Col11a2 F | CTCATCTTCCTGCATCAGAC |
| Mouse Col11a2 R | ACTTGGAAAGCGAGGTCCT |
| Mouse Adgrg6 F | AGAGGATGGACTGAGGCTGTGT |
| Mouse Adgrg6 R | CCAGGCTTGTTTGGACATGGTTG |
| Mouse Sox6 F | GCATAAGTGACCGTTTTGGCAGG |
| Mouse Sox6 R | GGCATCTTTGCTCCAGGTGACA |
| Mouse Mmp14 F | GCCTTCTGTTCCTGATAA |
| Mouse Mmp14 R | CCATCCTTCCTCTCGTAG |
| Mouse Pax1 F | AACCAGCACGGAGTATACAGC |
| Mouse Pax1 R | TGTAAGCTACCGAGTGCATCC |
| Mouse Esr2 F | GGTCCTGTGAAGGATGTAAGGC |
| Mouse Esr2 R | TAACACTTGCGAAGTCGGCAGG |
| Mouse Gapdh F | CATCACTGCCACCCAGAAGACTG |
| Mouse Gapdh R | ATGCCAGTGAGCTTCCCGTTCAG |
| Rat Sfrp2 F | CGTGAAACGGTGGCAGAAG |
| Rat Sfrp2 R | CGGATGCTGCGGGAGAT |
| Rat Krt19 F | AAGACACACTGGCAGAAACG |
| Rat Krt19 R | GATTCTGCCGCTCACTATCA |
| Rat Mmp12 F | TTGGCCATTCCTTGGGGCTGC |
| Rat Mmp12 R | TGTTGGTGGCTGGACTCCCAGG |
| Mouse Pax1 F (Figure 5) | CCGCACATTCAGTCAGCAAC |
| Mouse Pax1 R (Figure 5) | CATCTTGGGGGAGTAGGCAG |
| Mouse Col11a1 F (Figure 5) | CACAAAACCCCTCGATAGAAGTG |
| Mouse Col11a1 R (Figure 5) | CCTGTGATCAGGAACTGCTGAA |
| Mouse Adgrg6 F (Figure 5) | TCCTGTCCATCTCTGGCTCA |
| Mouse Adgrg6 R (Figure 5) | CACAAGACAGAGCTGCTCCA |
| Mouse Sox6 F (Figure 5) | TGCGACAGTTCTTCACTGTGG |
| Mouse Sox6 R (Figure 5) | CGTCCATCTTCATACCATACG |
| Mouse β-Actin F (Figure 5) | GGCACCACACCTTCTACAATG |
| Mouse β-Actin R (Figure 5) | GGGGTGTTGAAGGTCTCAAAC |
| Pax1-genotyping F | CAGAACCTGGAATGCTGTGCTC |
| Pax1-genotyping R | AAAGGGTTGCAGTGCCTTCAC |
| VarID_GRCh37 | RsID | Mutation | gnomAD | CADD | GERP | #Cases |
|---|---|---|---|---|---|---|
| 1:103377744:C:T | rs151249006 | NM_080629.2:c.G4093A:p.A1365T | 6.37E-05 | 25.8 | 5.42 | 1 |
| 1:103381192:C:A | rs150669855 | NM_080629.2:c.G3847T:p.V1283L | 0.0007 | 0.001 | -10.9 | 4 |
| 1:103405909:C:T | rs370589018 | NM_080629.2:c.G3394A:p.G1132S | 6.37E-05 | 25.2 | 5.46 | 1 |
Additional files
-
MDAR checklist
- https://cdn.elifesciences.org/articles/89762/elife-89762-mdarchecklist1-v1.pdf
-
Supplementary file 1
Tests of association with variants in 597 matrisome genes.
- https://cdn.elifesciences.org/articles/89762/elife-89762-supp1-v1.xlsx
-
Supplementary file 2
Rare COL11A1 variants detected in 625 AIS exomes.
- https://cdn.elifesciences.org/articles/89762/elife-89762-supp2-v1.docx