Similar excitability through different sodium channels and implications for the analgesic efficacy of selective drugs
Figures
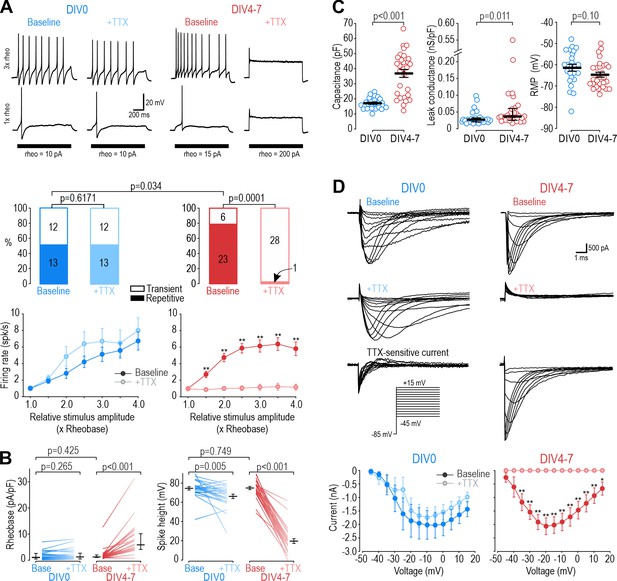
Different NaV subtypes produce similar excitability at different days in vitro (DIV).
(A) Representative responses of small DRG neurons to current injection at rheobase and 3 x rheobase when tested on DIV0 (blue) or DIV4-7 (red) before (dark) and after (pale) bath application of 100 nM TTX. At DIV0, TTX did not alter spiking pattern (χ2=0.25, p=0.617, McNemar test) or significantly reduce firing rate (F1,72=1.527, p=0.24, two-way repeated measure (RM) ANOVA; n=13). At DIV4-7, TTX significantly altered spiking pattern, converting all but one neuron to transient spiking (χ2=20.05, p<0.0001), and it significantly reduced firing rate (F1,132=43.157, p<0.001, n=23). Only neurons with repetitive spiking at baseline are included in the firing rate plot. (B) At DIV0, TTX did not affect rheobase (Z24=1.129, p=0.265, Wilcoxon rank test) but did reduce spike height (T24=3.092, p=0.005, paired t-test). At DIV4-7, TTX increased rheobase (Z28=4.681, p<0.001, Wilcoxon rank test) and dramatically reduced spike height (T28=20.333, p<0.001, paired t-test). Notably, neurons at DIV0 and DIV4-7 did not differ in their baseline rheobase (U=316, p=0.425, Mann-Whitney test) or spike height (T52=0.322, p=0.749, t-test). (C) Neurons at DIV0 and DIV4-7 differed in their total capacitance (T52=6.728, p<0.001, t-test) and leak conductance density (U=216, p=0.011, Mann-Whitney test) but not in their resting membrane potential (T52=1.668, p=0.101, t-test). (D) Sample voltage clamp recordings with command voltage stepped from –85 mV to +15 mV in 5 mV increments, before and after TTX. Sodium current was not significantly reduced by TTX at DIV0 (F1,72=3.585, p=0.107, two-way RM ANOVA; n=7) but was completely abolished by TTX at DIV4-7 (F1,108=33.526, p<0.001; n=10). Traces labeled ‘TTX-sensitive current’ represent the difference between current measured at baseline and after TTX, as determined by subtracting responses to the same voltage step under different pharmacological conditions. *, p<0.05; **, p<0.01; Student- Newman-Keuls post-hoc tests in A and D.
-
Figure 1—source data 1
Numerical values for data plotted in Figure 1.
- https://cdn.elifesciences.org/articles/90960/elife-90960-fig1-data1-v1.xlsx
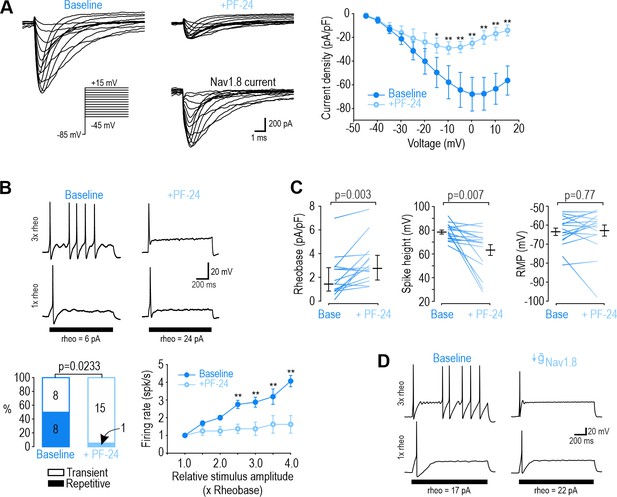
NaV1.8 is necessary for repetitive spiking at DIV0.
(A) Sample voltage clamp recordings show that sodium current was almost completely abolished by the NaV1.8 inhibitor PF-24 (1 µM). Peak current was significantly reduced by PF-24 (F1,72=12.651, p<0.012, two-way RM ANOVA; n=7). Traces labeled ‘NaV1.8 current’ represent the difference between current measured at baseline and after PF-24, as determined by subtraction. Another NaV1.8 inhibitor, A-803467, had a similar effect (see Figure 2—figure supplement 1). (B) PF-24 significantly altered spiking pattern (χ2=5.14, p=0.0233, McNemar test) and reduced firing rate (F1,42=11.946, p=0.011, two-way RM ANOVA; n=8). (C) PF-24 significantly increased rheobase (Z15=2.783, p=0.003, Wilcoxon rank test) and reduced spike height (T15=3.151, p=0.007, paired t-test) but did not affect resting membrane potential (T15=0.304, p=0.765, paired t-test). PF-24 had limited effects at DIV4-7 (Figure 2—figure supplement 2). (D) A computational model reproduced the effect of NaV1.8 on spiking pattern (also see Supplementary file 1A). The PF-24 effect was simulated as a~85% reduction in NaV1.8 ( = 4 mS/cm2). *, p<0.05; **; p<0.01; Student-Newman-Keuls post-hoc tests in A and B.
-
Figure 2—source data 1
Numerical values for data plotted in Figure 2, including supplements.
- https://cdn.elifesciences.org/articles/90960/elife-90960-fig2-data1-v1.xlsx

Inhibiting NaV1.8 at DIV0 with A-803467 had the same effect as PF-24.
(A) Sample voltage clamp recording at DIV0 before and after A-803467 (1 µM). (B) Peak current was significantly reduced by A-803467 (F1,84=9.935, p=0.016, two-way RM ANOVA, n=8). *, p<0.05; **, p<0.01; Student-Newman-Keuls post-hoc test in B.
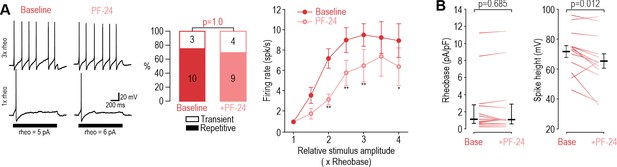
Inhibiting NaV1.8 with PF-24 at DIV4-7 had negligible effects.
(A) Inhibiting Nav1.8 with PF-24 (1 µM) did not affect spiking pattern (χ2=0.00, p=1.00, McNemar test) and modestly reduced firing rate (F1,54=9.745, p=0.012, two-way RM ANOVA, n=10) in DIV4-7 neurons. (B) PF-24 did not affect rheobase (Z12=0.420, p=0.685, Wilcoxon Rank test) but did reduce spike height (T12=2.939, p=0.012, paired-t-test). *, p<0.05; **, p<0.01; Student-Newman-Keuls post-hoc tests in A.
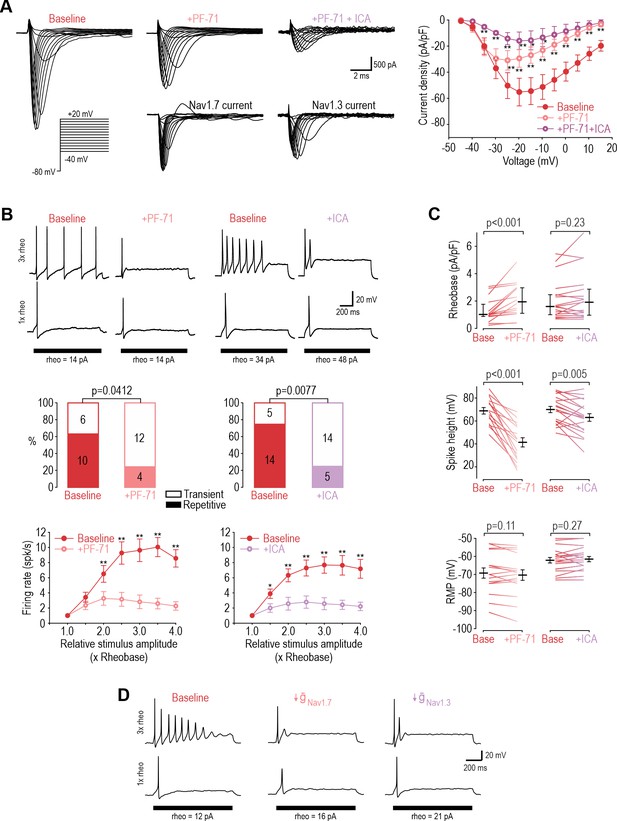
NaV1.3 and NaV1.7 are necessary for repetitive spiking at DIV4-7.
(A) Sample voltage clamp recordings show that sodium current was reduced by the NaV1.7 inhibitor PF-71 (30 nM) and by the NaV1.1/1.3 inhibitor ICA (1 µM). Peak current was significantly reduced by PF-71 and ICA (F2,192=26.361, p<0.001, two-way RM ANOVA; n=9). Traces labeled ‘NaV1.7 current’ and ‘NaV1.3 current’ represent the difference between current measured at baseline and after PF-72 and ICA, respectively, as determined by subtraction. (B) PF-71 and ICA both significantly altered spiking pattern (χ2=4.17, p=0.041 and χ2=7.11, p=0.0077, respectively, McNemar tests and significantly reduced firing rate) (F1,54=40.659, p<0.001, n=10 and F1,78=35.156, p<0.001, n=14, respectively, two-way RM ANOVAs). (C) PF-71 significantly increased rheobase (Z18=3.464, p<0.001, Wilcoxon rank test) and decreased spike height (T18=7.946, p<0.001, paired t-test). ICA did not significantly alter rheobase (Z18=1.248, p=0.225) but did reduce spike height (T18=3.243, p=0.005). Neither drug affected resting membrane potential (T15=1.681, p=0.113 for PF-71; T18=−1.132, p=0.272 for ICA, paired t-test). PF-71 had negligible effects at DIV0 (Figure 3—figure supplement 1). (D) A computational model reproduced the combined effects of NaV1.3 and NaV1.7 on spiking pattern (also see Supplementary file 1A and Figure 3—figure supplement 2). PF-71 effect was simulated as a 70% reduction in NaV1.7 ( = 10.5 mS/cm2). ICA effect was simulated as a 90% reduction in NaV1.3 ( = 0.035 mS/cm2). *, p<0.05; **, p<0.01; Student-Newman-Keuls post-hoc tests in A and B.
-
Figure 3—source data 1
Numerical values for data plotted in Figure 3, including supplements.
- https://cdn.elifesciences.org/articles/90960/elife-90960-fig3-data1-v1.xlsx
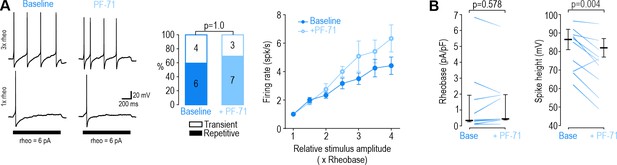
Inhibiting NaV1.7 at DIV0 had negligible effects.
(A) Inhibiting NaV1.7 with PF-71 (30 nM) did not alter spiking pattern (χ2=0.00, p=1.00, McNemar test) or reduce firing rate (F1,30=5.805, p=0.061, two-way RM ANOVA, n=6) in DIV0 neurons; in fact, firing rate was slightly increased. (D–E) PF-71 did not affect rheobase (Z9=0.677, p=0.578, Wilcoxon rank test) but did reduce spike height (T9=3.759, p=0.004, paired-t-test).

NaV1.7 and NaV1.3 currents can compensate for each other.
(A) In the DIV4-7 model, reducing to 30% of its normal value (35→10 mS/cm2) can be compensated for by increasing to 229% of its normal value (0.35→0.8 mS/cm2) to maintain repetitive spiking. (B) Conversely, reducing to 10% of its normal value (0.35→0.035 mS/cm2) can be compensated for by increasing to 171% of its normal value (35→60 mS/cm2) and maintain repetitive spiking.
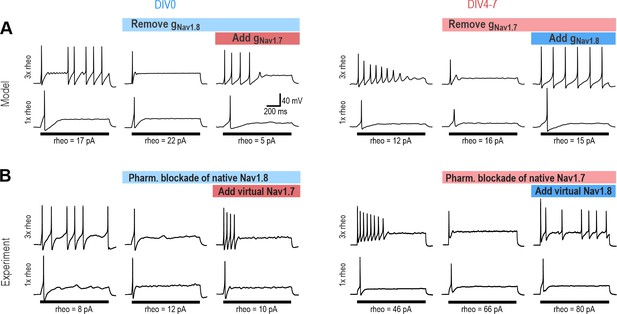
NaV1.7 and NaV1.8 are each sufficient to produce repetitive spiking in DIV0 and DIV4-7 neurons.
(A) The computational model predicts that the NaV1.8 conductance, which is “necessary” for repetitive spiking at DIV0 can, in principle, be replaced by NaV1.7 (left), and vice versa at DIV4-7 (right). (B) Replacement experiments involved inhibiting native channels pharmacologically and then introducing virtual conductances using dynamic clamp. At DIV0 (left), inhibiting native NaV1.8 (with PF-24) converted neurons to transient spiking, but introducing virtual NaV1.7 reverted neurons to repetitive spiking (in 3 of 3 neurons tested). At DIV4-7, inhibiting native NaV1.7 (with PF-71) converted the neuron to transient spiking, but introducing virtual NaV1.8 reverted neurons to repetitive spiking (in 4 of 4 neurons tested). Repetitive spiking was likewise restored by replacing the blocked native channel with the corresponding virtual channel (Figure 4—figure supplement 1). Parameters for virtual channels were identical to simulations except for the maximal conductance density, which was titrated in each cell.

Virtual conductances restored repetitive spiking after pharmacological inhibition of the corresponding native conductance had converted the neuron to transient spiking.
(A) Sample response at DIV0 showing that a virtual NaV1.8 conductance applied with dynamic clamp restored repetitive spiking after inhibiting native NaV1.8 channels with PF-24. This restoration was repeated in 3 of 3 neurons tested. (B) Sample recording at DIV4-7 showing that a virtual NaV1.7 conductance restored repetitive spiking after inhibiting native NaV1.7 channels with PF-71. This restoration was repeated in 4 of 4 neurons tested.

Contribution of NaV1.7 and NaV1.8 to spike initiation in DIV0 and DIV4-7 neurons.
(A) Voltage (top) for first (left) and second (right) spikes in the DIV0 model aligned with voltage activation curves for each NaV subtype (bottom). Dashed line shows voltage threshold (defined as V where dV/dt reaches 5 mV/ms). (B) Conductance plotted against voltage to create a phase portrait (top) showing NaV conductance at different phases of the spike. Inset shows full voltage range; main graph zooms in on voltages near threshold. Bottom plots show current plotted over the same voltage range. Whereas NaV1.7 (orange) mediated nearly all perithreshold inward current for the first spike, voltage threshold increased – because NaV1.7 inactivated (Figure 5—figure supplement 1) – and NaV1.8 (green) mediated nearly all perithreshold inward current for the second spike. The unexpected contribution of NaV1.7 to the first spike correctly predicted that TTX increases voltage threshold in DIV0 neurons (Figure 5—figure supplement 2). (C, D) In the DIV4-7 model, NaV1.7 (orange) and NaV1.3 (maroon) contributed to initiation of all spikes whereas the contribution of NaV1.8 was negligible (due entirely to its low expression level). (E) Sample experimental traces showing differences in the first (blue/red) and second (grey) spikes at DIV0 and DIV4-7. Plots summarize differences (Δ) in threshold, overshoot potential, and spike rise time between 1st and 2nd spikes during repetitive spiking evoked by current injection. At DIV0, the 1st and 2nd spikes differ significantly in their threshold (T8=2.522, p=0.036, one-sample t-test) and overshoot (T8=0.038, p=0.038) but not rise time (T8=0.249, p=0.810). At DIV4-7, the 1st and 2nd spikes differ in all measures (threshold: T7=7.613, p<0.001; overshoot: T7=−9.849, p<0.001; rise time: T7=5.979, p<0.001). Statistical results (green) show that differences between 1st and 2nd spike at DIV4-7 are significantly larger than differences at DIV0 (threshold: T15=−3.847, p=0.002; overshoot: T15=7.922, p<0.001; rise time: T15=−5.617, p<0.001, unpaired t-tests), consistent with our computational model.
-
Figure 5—source data 1
Numerical values for data plotted in Figure 5, including supplements.
- https://cdn.elifesciences.org/articles/90960/elife-90960-fig5-data1-v1.xlsx
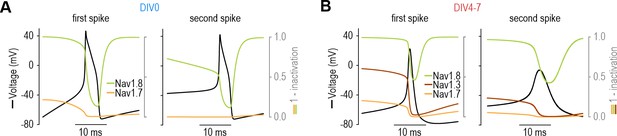
Channel inactivation affects NaV subtype contribution on short timescale.
(A) In the DIV0 model, NaV1.7 contributed to the first spike but its inactivation meant that all subsequent spikes relied exclusively on NaV1.8. (B) In the DIV4-7 model, despite some inactivation of NaV1.3 (maroon) and NaV1.7 (orange), the remaining current was still large enough (because of the higher gmax of those two subtypes) to produce inward current sufficient to support repetitive spiking despite the low gmax of NaV1.8 in the DIV4-7 model.

Effect of TTX on voltage threshold in DIV0 neurons.
Despite TTX having negligible effects in DIV0 neurons according to our initial analysis (see Figure 1), simulation results in Figure 5A and B predicted that the first spike was nonetheless initiated by NaV1.7. By extension, this predicted that TTX should cause a depolarizing shift in voltage threshold for the first spike. Analysis of the experimental data confirmed this to be true, with threshold (mean ± SEM) increasing from –33.7±1.4 mV at baseline to –28.3±1.4 mV after TTX (T24=−3.19, p=0.004, paired t-test). Confirmation of this unexpected prediction helps further validate our model neuron.
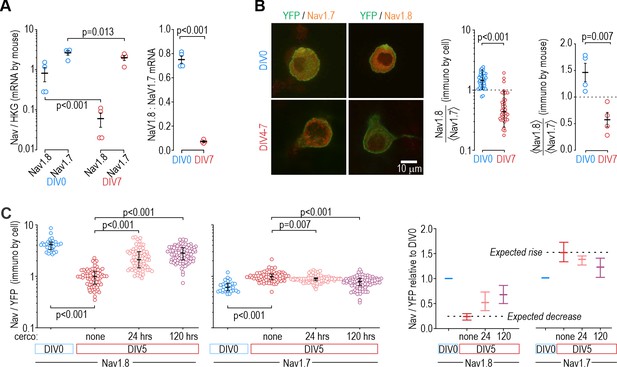
Protein levels, but not mRNA, reflect functional contributions of NaV subtypes at DIV0 and DIV7.
(A) Both NaV1.8 and NaV1.7 mRNA levels (relative to a housekeeping gene (HKG), see Methods) decreased significantly between DIV0 and DIV4-7 (factor 1: time, F1,12=56.677, p<0.001, factor 2: subtype, F1,12=17.952, p=0.001, two-way ANOVA and Student-Newman-Keuls post-hoc tests on log transformed data, n=4 mice per time point) but more so for NaV1.8 than for NaV1.7 (interaction: time x subtype, F1,12=11.455, p=0.005). The differential reduction yielded a significantly higher NaV1.8: NaV1.7 ratio at DIV0 than at DIV7 (T6=21.375, p<0.001, unpaired t-test) but the increasing functional contribution of NaV1.7 between DIV0 and DIV4-7 remains unaccounted for. (B) Immunoreactivity (IR) for NaV1.8 protein exceeded NaV1.7-IR at DIV0, but the opposite was true on DIV4-7, consistent with the functional contribution of each subtype. NaV-IR was measured relative to YFP intensity in the same cell, and then each cell’s NaV1.8:YFP ratio was considered relative to the average NaV1.7:YFP ratio in the co-processed coverslip (left) or average NaV1.8:YFP ratio was considered relative to the average NaV1.7:YFP ratio in the same animal (right). Ratios were >1 at DIV0 but decreased significantly at DIV4-7 (U=78, p<0.001, n=37 for DIV0, n=40 for DIV4-7, Mann-Whitney test (left) and T6=4.046, p=0.007, unpaired t-test (right)). (C) Chronically applied cercosporamide (10 µM) mitigated changes in NaV1.8- and NaV1.7-IR at DIV5 (NaV1.8: H3=157.95, p<0.001; NaV1.7: H3=80.662, p<0.001; One-way ANOVA on ranks, Dunn’s post-hoc tests, p<0.05 for all pairs). Data are summarized as median ± quartile. Panel on the right shows data normalized to baseline (DIV0) to emphasize relative changes. N=3 experiments.
-
Figure 6—source data 1
Numerical values for data plotted in Figure 6.
- https://cdn.elifesciences.org/articles/90960/elife-90960-fig6-data1-v1.xlsx
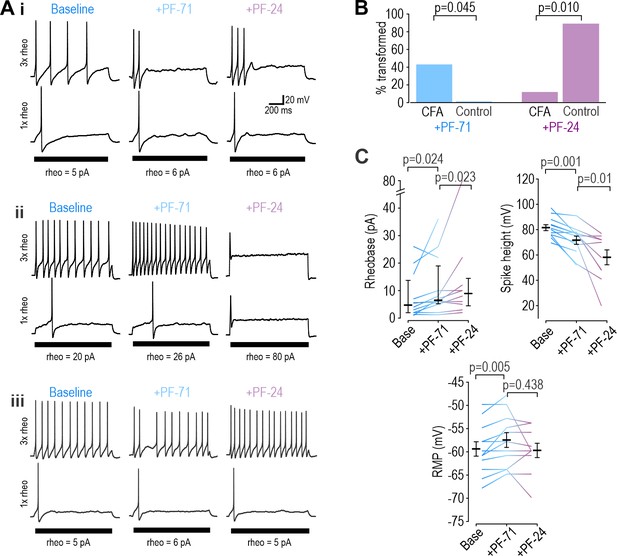
Inflammation alters NaV subtype contribution to nociceptor excitability.
(A) Sample responses in DIV0 neurons from mice injected with CFA three days earlier. In 12 cells tested, PF-71 converted five neurons to transient spiking (i), encouraged repetitive spiking in four neurons (ii), and had no effect in three neurons (iii), thus highlighting increased heterogeneity after CFA. (B) At DIV0, the effect of PF-71 differed significantly between CFA and control neurons, converting 42% (5 of 12) CFA neurons from repetitive to transient spiking vs 0% (0 of 9) control neurons (p=0.045, Fisher Exact test). Applying PF-24 to neurons that continued to spike repetitively after PF-71 had little effect on CFA neurons, converting only 13% (1 of 7) of CFA neurons vs 88% (7 of 8) of control neurons (p=0.010, Fisher Exact test). Together these results argue that NaV1.7 contributes more and NaV1.8 contributes less to nociceptor excitability after inflammation. (C) At DIV0, PF-71 significantly increased resting membrane potential (T11=−3.530, p=0.005, paired t-test) and rheobase (Z11=2.186, p=0.024, Wilcoxon rank test), and significantly decreased spike height (T11=4.413, p=0.001, paired t-test) in CFA neurons. Further addition of PF-24 significantly changed rheobase (Z9=2.176, p=0.023, Wilcoxon rank test) and spike height (T9=3.237, p=0.01, paired t-test) but did not affect resting membrane potential (T9=1.049, p=0.321, paired t-test).
-
Figure 7—source data 1
Numerical values for data plotted in Figure 7.
- https://cdn.elifesciences.org/articles/90960/elife-90960-fig7-data1-v1.xlsx

Inflammation-induced change in NaV subtype contribution impacts analgesic efficacy of PF-71.
(A) CFA significantly increased thermal sensitivity (F5,65=19.556, p<0.001, two-way RM ANOVA). PF-71 significantly decreased thermal sensitivity in mice injected 3 days prior with CFA (T8=−7.296, p<0.001; paired t-test) but had no effect in naive mice (T5=−0.141, p=0.894). (B) CFA significantly increased mechanical sensitivity (F4,52=16.786, p<0.001). PF-71 significantly decreased tactile sensitivity in mice injected 3 days prior with CFA (T8=−4.341, p=0.002) but had no effect in naive mice (T5=1.000, p=0.363). Insets in both panels show values for each animal before and 2 hr after PF-71 injection. *, p<0.05; **, p<0.01; Student-Newman-Keuls post-hoc tests.
-
Figure 8—source data 1
Numerical values for data plotted in Figure 8.
- https://cdn.elifesciences.org/articles/90960/elife-90960-fig8-data1-v1.xlsx
Additional files
-
Supplementary file 1
Supplementary tables.
A, Model data before and after channel “inhibition”. B, Reagents. C, EC50 values. D, Primers. E, Model equations. F, Conductance densities at baseline for DIV 0 and DIV4-7 models.
- https://cdn.elifesciences.org/articles/90960/elife-90960-supp1-v1.docx
-
MDAR checklist
- https://cdn.elifesciences.org/articles/90960/elife-90960-mdarchecklist1-v1.docx
-
Source code 1
Contains computer code for computational model of neuron.
- https://cdn.elifesciences.org/articles/90960/elife-90960-code1-v1.zip





