Mechanistic target of rapamycin (mTOR) pathway in Sertoli cells regulates age-dependent changes in sperm DNA methylation
Figures
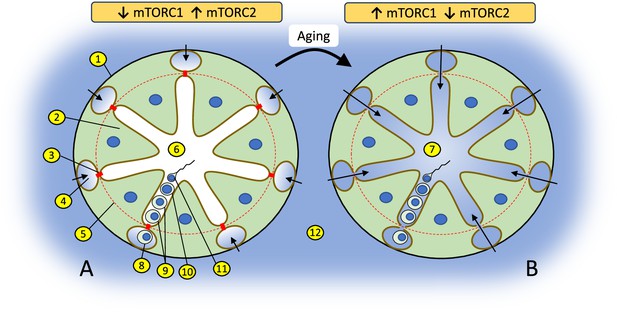
Changes in the balance of mTOR complexes and aging affect biochemical conditions of spermatogenesis in the apical compartment of seminiferous epithelium.
(A) Seminiferous tubule with tight blood-testis barrier (BTB) where interstitial environment (shown in blue) penetrates only to basal compartment of seminiferous epithelium as shown by color and arrows. The tight state of the BTB is promoted by higher activity of mTORC2 over mTORC1 in Sertoli cells. (B) Seminiferous tubule with leaky BTB where interstitial environment penetrates to the apical compartment of seminiferous epithelium. BTB disassembly is promoted by higher activity of mTORC1 over mTORC2: (1) basal membrane of a seminiferous tubule; (2) Sertoli cell; (3) contact between Sertoli cells enforced by gap junction, tight junction, and endoplasmic specialization (BTB); (4) basal compartment of the seminiferous epithelium; (5) the dotted red line illustrates structural separation of compartments by the BTB; (6) apical compartment of the seminiferous epithelium; (7) apical compartment in aged organism with biochemically ‘noisy’ environment due to the leaky BTB; (8) spermatogonia; (9) spermatocytes; (10) spermatid; (11) spermatozoa; (12) interstitial environment.
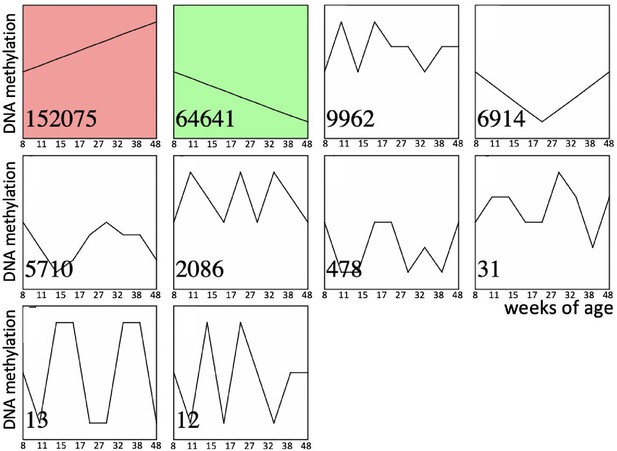
Time series of age-dependent DNA methylation change in sperm.
Significantly enriched time-series are shown in color. Numbers in the left bottom corner of each time series indicate the number of corresponding CpGs out of total 254,410 CpGs analyzed.
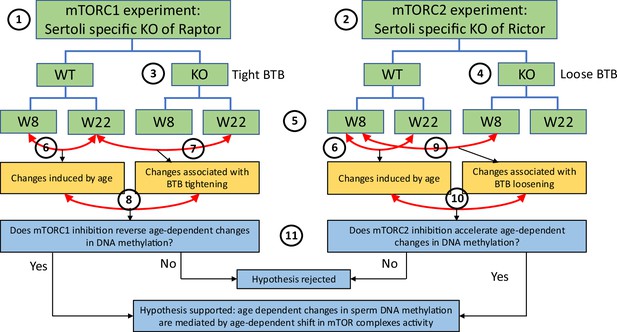
Scheme of experimental design for DNA methylation analysis.
This study utilized experiments with two transgenic mouse models: (1) one with mTORC1 suppression in Sertoli cells due to the cell-specific knockout (KO) of Rptor, and (2) another with mTORC2 suppression in Sertoli cells due to the cell-specific KO of Rictor. Suppression of mTORC1 results in tightening of the blood-testis barrier (BTB) (3) and suppression of mTORC2 results in loosening of the BTB (4). DNA methylation changes were analyzed in sperm of each genotype on postnatal weeks 8 and 22 (5). In each experiment, we first identified physiological age-dependent changes in sperm DNA methylation by comparing epigenomes of wildtype (WT) 8-week-old mice and WT 22-week-old mice (6). We further used both experiments to test the hypothesis that age-dependent changes in the sperm epigenome are associated with the age-dependent increase in permeability of the BTB. Specifically, to test this hypothesis using the mTORC1 experiment, we compared physiological age-dependent changes (6) with changes induced by KO (tighter BTB) in older mice (7). Our hypothesis predicts that mTORC1 suppression in older mice will affect age-dependent DMRs in the direction opposite to the one induced by age (8). Similarly, to test our hypothesis using the mTORC2 experiment, we compared physiological age-dependent changes (6) with changes induced by KO (loose BTB) in younger mice (9). Our hypothesis predicts that mTORC2 suppression in young mice due to KO will produce similar effects on age-dependent DMRs as age itself (10). Outcomes of both experiments were used to support or reject our hypothesis (11).
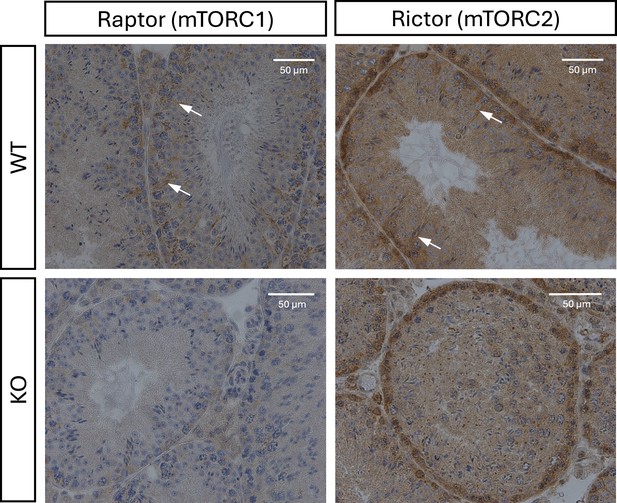
Immunohistochemistry analysis of the efficiency of Rictor and Rptor knockout (KO) in Sertoli cells.
Representative images illustrate lack of staining for Rictor and Raptor proteins in Sertoli cells in corresponding KO models. Arrows show staining by Rictor or Raptor antibodies respectively in Sertoli cells.
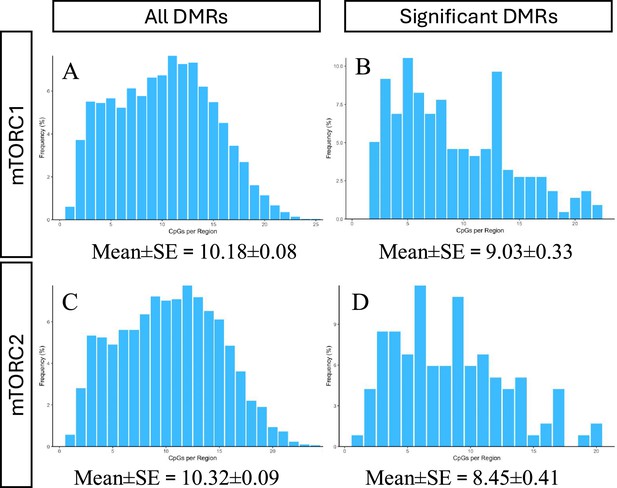
Distribution of CpG per 100 bp regions.
(A) All overlapping regions identified in age and genotype groups in mTORC1 inactivation experiment. (B) Significant (q<0.05) overlapping regions identified in age and genotype groups in mTORC1 inactivation experiment. (C) All overlapping regions identified in age and genotype groups in mTORC2 inactivation experiment. (D) Significant (q<0.05) overlapping regions identified in age and genotype groups in mTORC2 inactivation experiment.
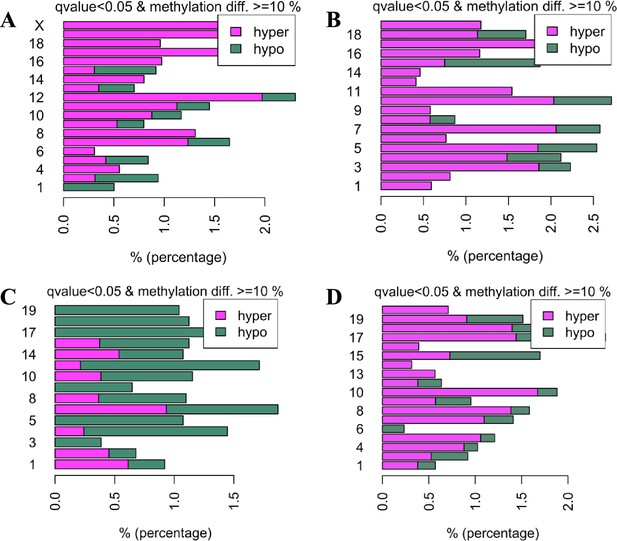
Methylation changes per chromosome in mice of different genotype and age.
(A and B) Percent DMRs altered by age in wildtype (WT) animals in mTORC1 (A) and mTORC2 (B) inactivation experiments. (C) Percent DMRs in mature mice altered by mTORC1 inactivation. (D) Percent DMRs in young mice altered by mTORC2 inactivation.
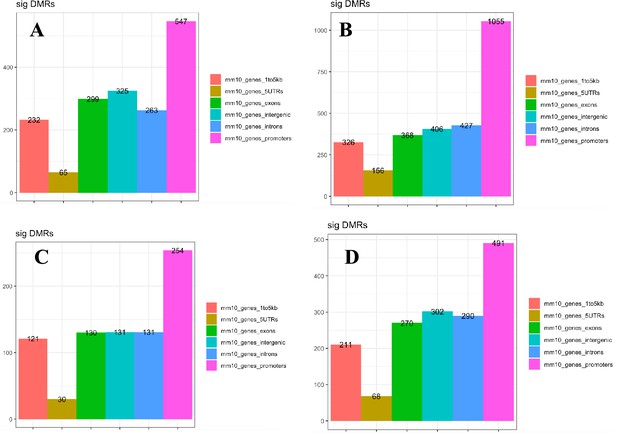
Genomic elements enriched with DMRs associated with age and inactivation of mTOR complexes.
Changes induced by age in wildtype (WT) animals in mTORC1 (A) and mTORC2 (B) inactivation experiments. (C) Changes induced in mature mice by mTORC1 inactivation. (D) Changes induced in young mice by mTORC2 inactivation.
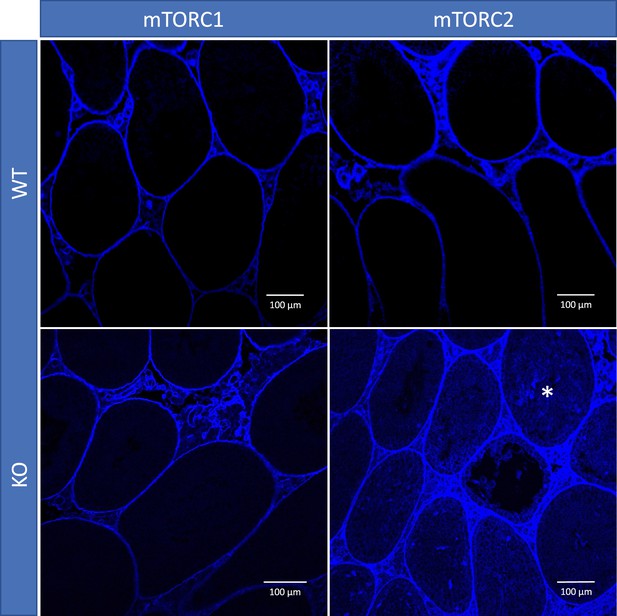
Blood-testis barrier (BTB) permeability in 22-week-old mice of different genotypes, representative images.
In mice with inactivated mTORC2 (Rictor knockout [KO]), biotin tracer (blue) penetrated to the apical compartment of the seminiferous epithelium (asterisk) while in all other genotypes it did not cross the BTB.
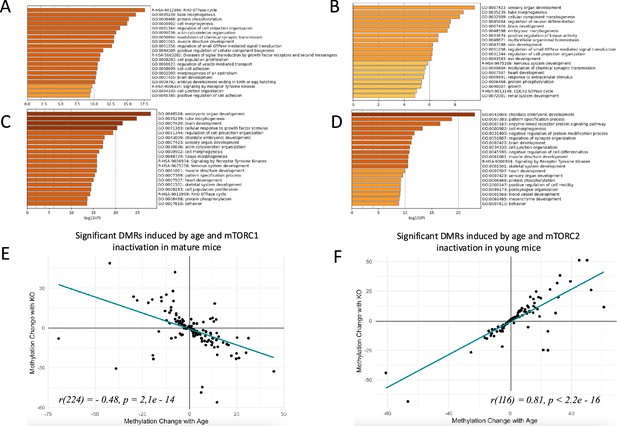
Changes in sperm DNA methylation induced by age and manipulation of mTOR pathway in Sertoli cells.
(A, B, C, D) Top biological categories enriched with genes associated with significant DMRs: (A) age-dependent DMRs in wildtype animals in the mTORC1 experiment; (B) age-dependent DMRs in wildtype animals in the mTORC2 experiment; (C) DMRs induced by mTORC1 inactivation in mature animals; (D) DMRs induced by mTORC2 inactivation in young animals. (E and F) Changes in methylation of significant DMRs induced by age and mTORC1 inactivation in mature mice (E) and by age and mTORC2 inactivation in young mice (F).
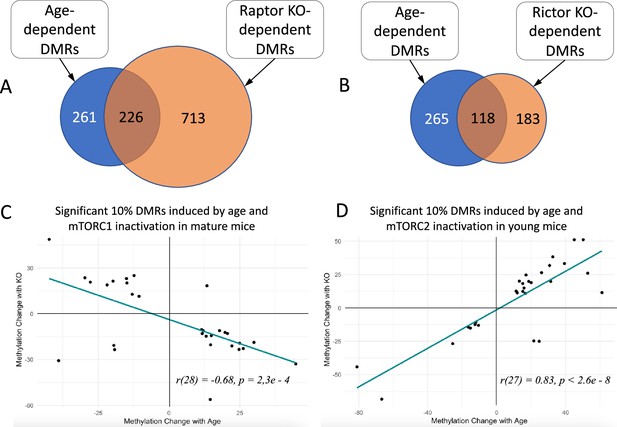
Changes in methylation of significant DMRs induced by age and genetic manipulation of the mTOR pathway in Sertoli cells.
(A) Overlap between significant DMRs induced by age and by mTORC1 inactivation in mature mice among all common methylation regions identified in both comparisons. (B) Overlap between significant DMRs induced by age and by mTORC2 inactivation in young mice among all common methylation regions identified in both comparisons. (C) Comparison of age-dependent changes and changes induced by mTORC1 inactivation in mature mice. (D) Comparison of age-dependent changes and changes induced by mTORC2 inactivation in young mice.
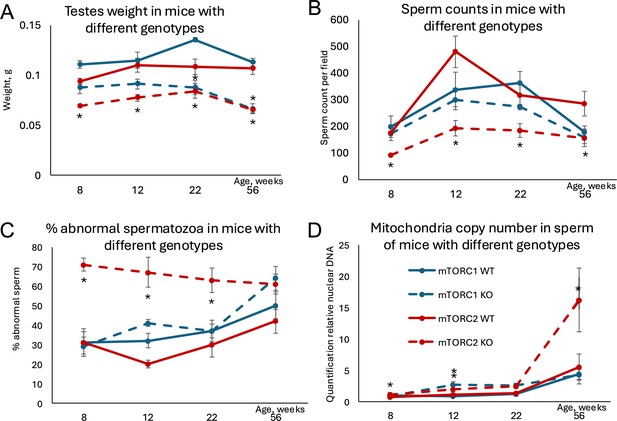
Changes in age-dependent reproductive parameters in mice with inactivated mTORC1 or mTORC2 in Sertoli cells.
(A) Testes weight. (B) Sperm counts in sperm smears. (C) Percent abnormal spermatozoa. (D) Mitochondrial DNA copy number. All data are mean ± SE, n=5–6/age/genotype, *q<0.05 when compared with corresponding wildtype (WT).
Tables
Changes in reproductive parameters induced by Sertoli cell-specific knockout (KO) of Raptor (mTORC1 suppression) and Rictor (mTORC2 suppression).
All parameters significantly different (q≤0.05) in KO mice as compared with same age wildtype (WT) counterparts are shown in bold.
| Experiment | Genotype | Age, weeks | Testes weight, mg | Sperm count/field | % Abnormal spermatozoa | Mitochondrial DNA copy number | ||||
|---|---|---|---|---|---|---|---|---|---|---|
| Mean ± SE | q | Mean ± SE | q | Mean ± SE | q | Mean ± SE | q | |||
| mTORC1 suppression | WT | 8 | 111±4 | -- | 197±40 | -- | 31±6 | -- | 0.97±0.53 | -- |
| 12 | 115±8 | -- | 338±64 | -- | 32±4 | -- | 0.82±0.10 | -- | ||
| 22 | 136±2 | -- | 362±43 | -- | 37±6 | -- | 1.17±0.26 | -- | ||
| 56 | 113±4 | -- | 179±22 | -- | 50±7 | -- | 4.38±0.82 | -- | ||
| Raptor KO | 8 | 88±6 | 0.062 | 171±24 | 0.725 | 29±5 | 0.867 | 0.93±0.19 | 0.307 | |
| 12 | 92±5 | 0.064 | 300±38 | 0.743 | 41±2 | 0.126 | 2.67±0.56 | 0.024 | ||
| 22 | 88±7 | 0.006 | 272±10 | 0.169 | 37±4 | 0.950 | 2.60±0.39 | 0.056 | ||
| 56 | 67±5 | 0.003 | 159±26 | 0.728 | 64±6 | 0.568 | 4.28±1.50 | 0.665 | ||
| mTORC2 suppression | WT | 8 | 93±3 | -- | 176±6 | -- | 31±7 | -- | 0.72±0.03 | -- |
| 12 | 110±7 | -- | 480±60 | -- | 20±2 | -- | 1.10±0.19 | -- | ||
| 22 | 108±7 | -- | 316±41 | -- | 30±6 | -- | 1.30±0.10 | -- | ||
| 56 | 107±7 | -- | 285±45 | -- | 42±6 | -- | 5.52±2.08 | -- | ||
| Rictor KO | 8 | 70±1 | 0.050 | 91±3 | 0.001 | 71±3 | 0.005 | 1.07±0.12 | 0.017 | |
| 12 | 78±3 | 0.009 | 193±28 | 0.009 | 67±8 | 0.001 | 1.99±0.43 | 0.016 | ||
| 22 | 84±7 | 0.048 | 184±26 | 0.049 | 63±6 | 0.016 | 2.45±0.34 | 0.143 | ||
| 56 | 66±2 | <0.001 | 156±34 | <0.001 | 61±5 | 0.711 | 16.23±5.12 | 0.049 | ||





