Single-molecule analysis reveals the phosphorylation of FLS2 governs its spatiotemporal dynamics and immunity
Figures
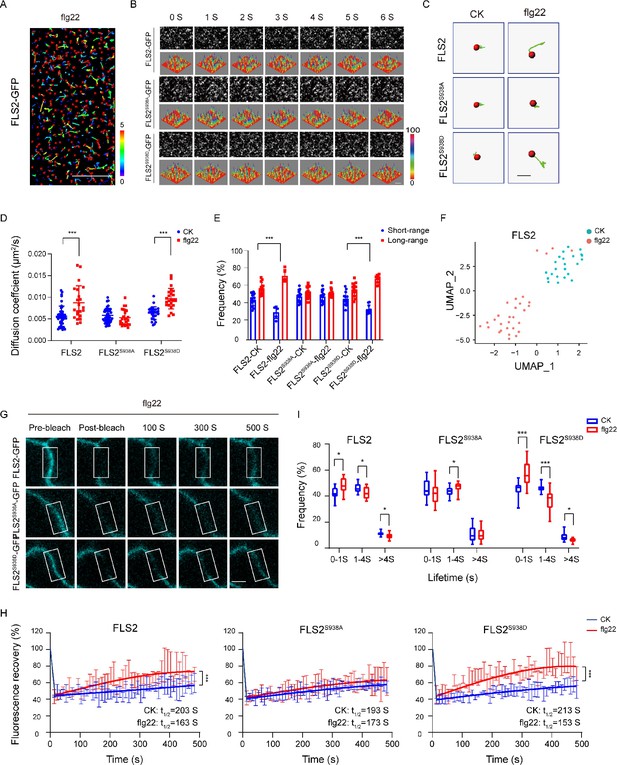
Effects of Ser-938 phosphorylation on the spatiotemporal dynamics of FLS2 at the plasma membrane.
(A) VA-TIRFM images of a FLS2-expressing hypocotyl cell were analyzed. The 5-day-old transgenic Arabidopsis plant cells were observed under VA-TIRFM. The red balls indicate the positions of the identified points that appeared. Trajectories represent the track length of the identified points. Bar = 10 μm. (B) Time-lapse images of FLS2, FLS2S938A, and FLS2S938D. Bar = 2 μm. The fluorescence intensity changes among different 3D luminance plots. (C) The trajectories of representative individual FLS2, FLS2S938A, and FLS2S938D under 30 min for 10 μM flg22 processing and control. Bar = 0.5 μm. (D) Diffusion coefficients of FLS2 (control, n = 42 spots; flg22, n = 22 spots), FLS2S938A (control, n = 44 spots; flg22, n = 25 spots), and FLS2S938D (control, n = 27 spots; flg22, n = 23 spots) under different environments. Statistical significance was assessed using Student’s t-test (***p<0.001). Error bars represent the SD. (E) Frequency of long- and short-range motions for FLS2 (control, n = 13 spots; flg22, n = 5 spots), FLS2S938A (control, n = 16 spots; flg22, n = 14 spots), and FLS2S938D (control, n = 14 spots; flg22, n = 10 spots) under different environments. Statistical significance was assessed using Student’s t-test (***p<0.001). Error bars represent the SD. (F) Uniform Manifold Approximation and Projection (UMAP) visualization of FLS2 samples under different conditions (control, n = 25 spots; flg22, n = 22 spots). Dots represent the individual images and are colored according to the reaction conditions. (G) The representative FRAP time course of FLS2-GFP, FLS2S938A, and FLS2S938D under flg22 treatments. White squares indicate bleached regions. Bar = 5 μm. (H) Fluorescence recovery curves of the photobleached areas with or without the flg22 treatment. Three biological replicates were performed. Each experiment was repeated thrice independently (n = 3 images). Statistical significance was assessed using Student’s t-test (***p<0.001). Error bars represent the SD. (I) Lifetime were analyzed for FLS2 (control, n = 13 spots; flg22, n = 9 spots), FLS2S938A (control, n = 28 spots; flg22, n = 21 spots), and FLS2S938D (control, n = 13 spots; flg22, n = 15 spots) under the control and flg22 treatments. The data points were collected from a TIRFM time series using an exposure time of 100 ms to capture a total duration of 20 s. Statistical significance was assessed using Student’s t-test (*p<0.05, ***p<0.001). Error bars represent the SD.
-
Figure 1—source data 1
The original VA-TIRFM image and single-particle tracking of FLS2-expressing hypocotyl cells are shown in Figure 1A.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig1-data1-v1.xlsx
-
Figure 1—source data 2
The list of the diffusion coefficients of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments is shown in Figure 1D.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig1-data2-v1.zip
-
Figure 1—source data 3
The list of the motion range of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments is shown in Figure 1E.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig1-data3-v1.zip
-
Figure 1—source data 4
The list of the lifetime of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments is shown in Figure 1I.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig1-data4-v1.zip
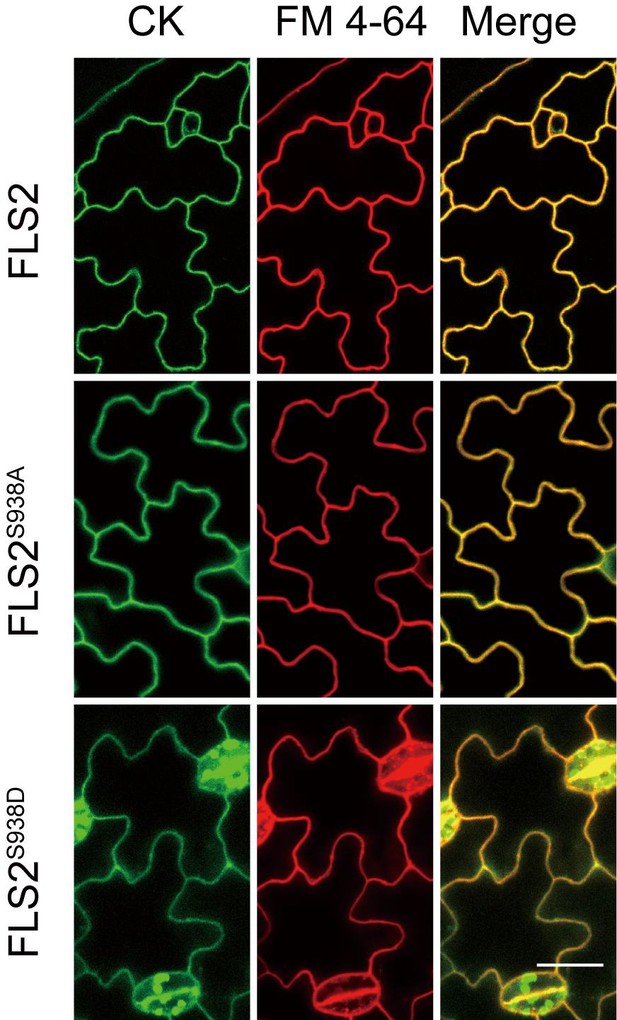
The subcellular localization of FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP was determined by confocal imaging.
The left images represent GFP signal showing the subcellular localization of FLS2 molecules under different phosphorylation states. The middle images represent red fluorescence signal showing plasma membrane (PM) stained with FM4-64. The right images represent the merge of GFP and FM4-64 staining. Bar = 20 μm.
-
Figure 1—figure supplement 1—source data 1
The original confocal images of FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig1-figsupp1-data1-v1.zip
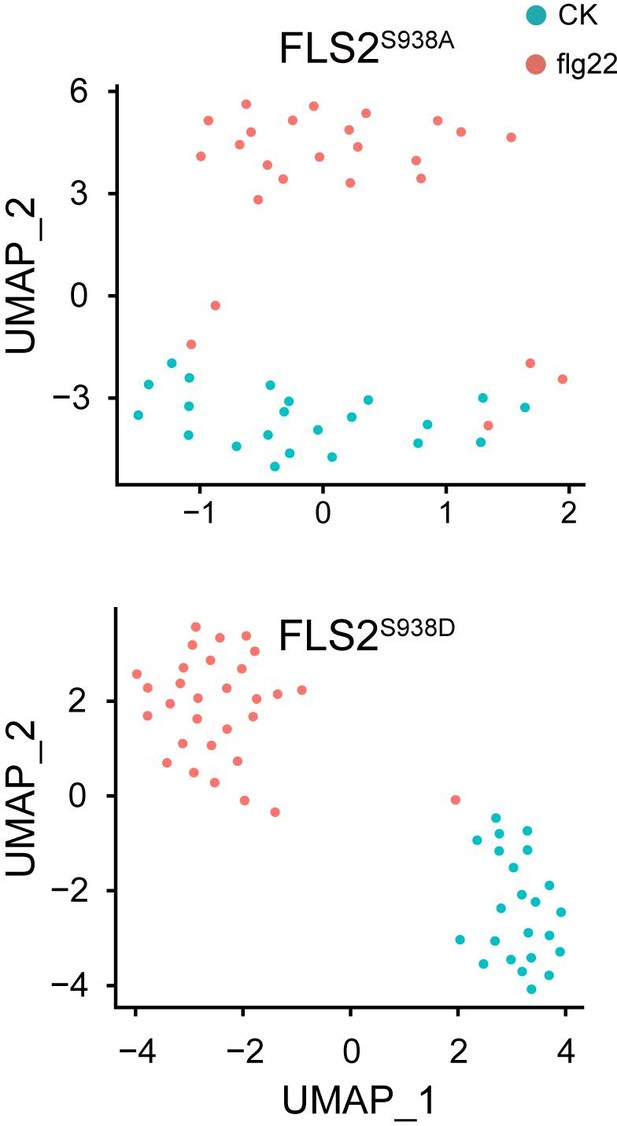
Uniform Manifold Approximation and Projection (UMAP) visualization of FLS2S938A (control, n = 27 spots; flg22, n = 30 spots) and FLS2S938D (control, n = 11 spots; flg22, n = 14 spots) samples in the different conditions.
Dots represent individual images and are colored according to the reaction conditions.

The representative fluorescence recovery after photobleaching (FRAP) time course of FLS2-GFP, FLS2S938A, and FLS2S938D under control treatments.
White squares indicate bleached regions. Bar = 5 μm.
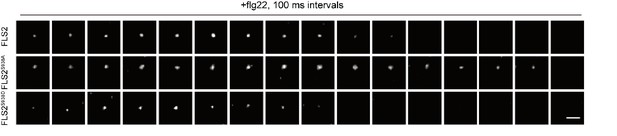
Images of FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP with flg22 treatment were collected by VA-TIRFM.
For short presentation, one frame is displayed for every 10 frames captured (at 100 ms intervals). Bar = 300 nm.
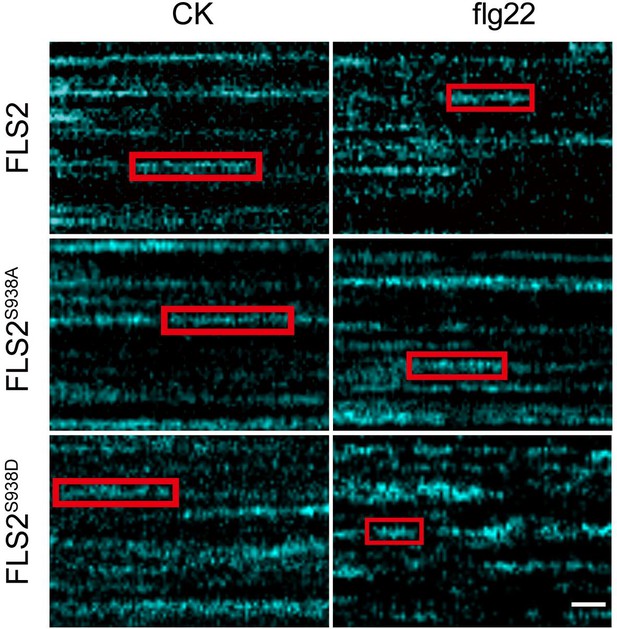
Representative kymographs showing individual FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP dwell times under the control and flg22 treatment.
Bar = 2 s.
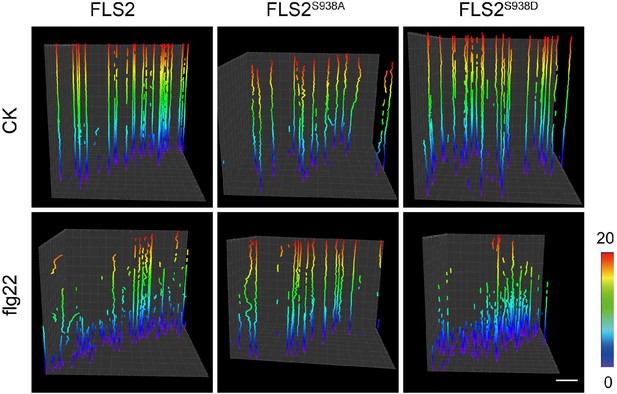
The single-molecule trajectories of FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP analyzed by Imaris could be faithfully tracked under control and flg22 treatments.
Bar = 1 μm.
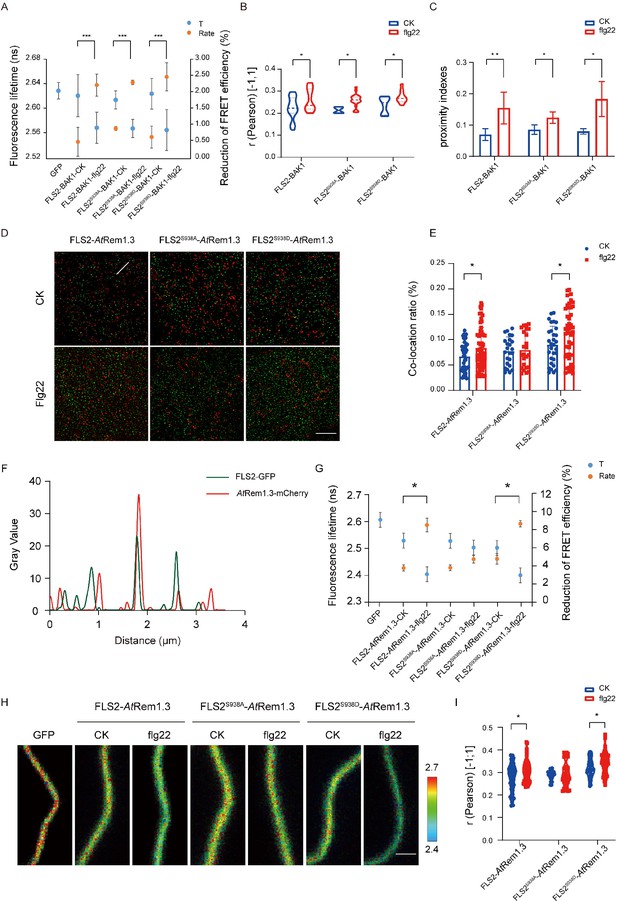
Different Ser-938 phosphorylation states of FLS2 affect its partitioning into AtRem1.3-associated nanodomains.
(A) FLIM-FRET was used to detect the co-expression of FLS2/FLS2S938A /FLS2S938D-GFP and BAK1-mCherry in the N. benthamiana epidermal cells stimulated by 1/2 MS or flg22 (10 μM) for 30 min. Average fluorescence lifetime (t) and the FRET efficiency were analyzed for FLS2 (control, n = 21 images; flg22, n = 14 images), FLS2S938A (control, n = 18 images; flg22, n = 12 images), or FLS2S938D (control, n = 24 images; flg22, n = 14 images) and BAK1. The fluorescence mean lifetime (t) of FLS2-GFP (n = 7 images). Statistical significance was assessed using Student’s t-test (***p<0.001). Error bars represent the SD. (B) Pearson correlation coefficient values of co-localization between FLS2 (control, n = 21 images; flg22, n = 11 images), FLS2S938A (control, n = 4 images; flg22, n = 20 images), or FLS2S938D (control, n = 8 images; flg22, n = 10 images) and BAK1 upon stimulation with control or flg22. Statistical significance was assessed using Student’s t-test (*p<0.05). Error bars represent the SD. (C) Mean protein proximity indexes showing FLS2 (control, n = 6 images; flg22, n = 5 images), FLS2S938A (control, n = 4 images; flg22, n = 3 images), or FLS2S938D (control, n = 4 images; flg22, n = 3 images) and BAK1 degree of proximity under different conditions. Statistical significance was assessed using Student’s t-test (*p<0.05, **p<0.01). Error bars represent the SD. (D) TIRF-SIM images of the Arabidopsis leaf epidermal cells co-expressing FLS2/FLS2S938A/FLS2S938D-GFP and AtRem1.3-mCherry under different environments. The distribution of FLS2 and AtRem1.3 signals along the white line in the merged image. Bar = 5 μm. (E) The histogram shows the co-localization ratio of FLS2-GFP (control, n = 46 images; flg22, n = 59 images), FLS2S938A-GFP (control, n = 24 images; flg22, n = 36 images), or FLS2S938D-GFP (control, n = 30 images; flg22, n = 54 images) and AtRem1.3-mCherry. The sizes of the ROIs used for statistical analysis are 13.38 μm and 13.38 μm. Statistical significance was assessed using Student’s t-test (*p<0.05). Error bars represent the SD. (F) FLS2/FLS2S938A/FLS2S938D-GFP and AtRem1.3-mCherry fluorescence signals as shown in (D). (G) The fluorescence mean lifetime (t) and the corrected fluorescence resonance efficiency (Rate) of FLS2 (control, n = 8 images; flg22, n = 13 images), FLS2S938A (control, n = 9 images; flg22, n = 11 images), or FLS2S938D (control, n = 10 images; flg22, n = 11 images) with co-expressed AtRem1.3. The fluorescence mean lifetime (t) of FLS2-GFP (n = 9 images). Statistical significance was assessed using Student’s t-test (***p<0.001). Error bars represent the SD. (H) Intensity and lifetime maps of the Arabidopsis leaf epidermal cells co-expressing FLS2/FLS2S938A/FLS2S938D-GFP and AtRem1.3-mCherry as measured by FLIM-FRET. Bar = 2 µm. (I) Quantification of co-localization between FLS2, FLS2S938A or FLS2S938D and AtRem1.3 with and without stimulation with ligands. Pearson correlation coefficient values (r) were obtained between FLS2 (control, n = 62 images; flg22, n = 53 images)/FLS2S938A (control, n = 26 images; flg22, n = 40 images)/FLS2S938D-GFP (control, n = 28 images; flg22, n = 38 images) and AtRem1.3-mCherry. Statistical significance was assessed using Student’s t-test (*p<0.05). Error bars represent the SD.
-
Figure 2—source data 1
The list of the fluorescence mean lifetimes (t) of FLS2/FLS2S938A/FLS2S938D-GFP and BAK1-mCherry under different treatments is shown in Figure 2A.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig2-data1-v1.zip
-
Figure 2—source data 2
The list of the Pearson correlation coefficient of FLS2/FLS2S938A/FLS2S938D-GFP and BAK1-mCherry under different treatments is shown in Figure 2B.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig2-data2-v1.zip
-
Figure 2—source data 3
The list of the mean protein proximity indexes of FLS2/FLS2S938A/FLS2S938D-GFP and BAK1-mCherry under different treatments is shown in Figure 2C.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig2-data3-v1.zip
-
Figure 2—source data 4
The original intensity and lifetime maps of FLS2/FLS2S938A/FLS2S938D-GFP and AtRem1.3-mCherry under different treatments are shown in Figure 2H.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig2-data4-v1.zip
-
Figure 2—source data 5
The list of the fluorescence mean lifetimes (t) of FLS2/FLS2S938A/FLS2S938D-GFP and AtRem1.3-mCherry under different treatments is shown in Figure 2G.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig2-data5-v1.zip
-
Figure 2—source data 6
The list of the Pearson correlation coefficient of FLS2/FLS2S938A/FLS2S938D-GFP and AtRem1.3-mCherry under different treatments is shown in Figure 2I.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig2-data6-v1.zip
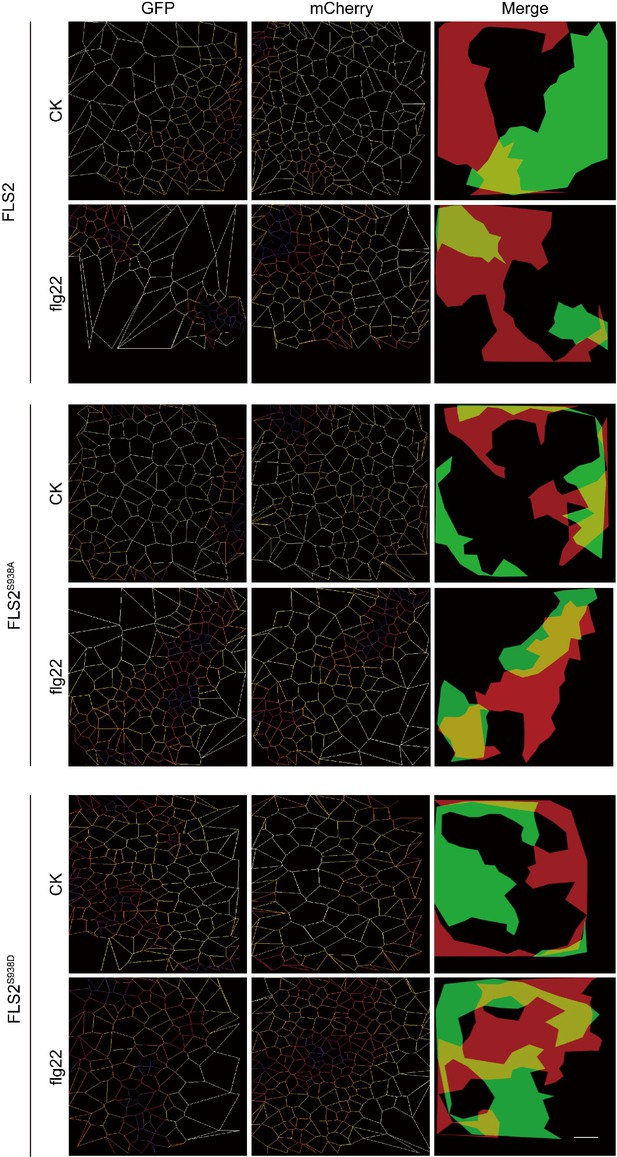
SR-Tesseler analysis shows the distribution and co-localization of all spots on FLS2-GFP, FLS2S938A-GFP and FLS2S938D-GFP and BAK1 under control or flg22 treatment.
Bar = 50 μm.
-
Figure 2—figure supplement 1—source data 1
The list of the coordinates of FLS2/FLS2S938A/FLS2S938D-GFP and BAK1-mCherry under different treatments.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig2-figsupp1-data1-v1.zip
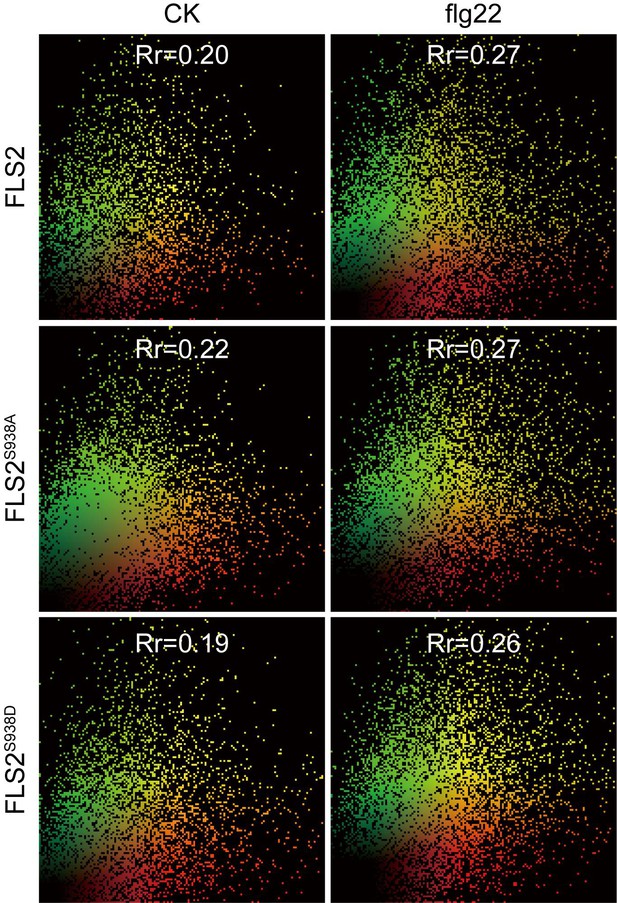
Images show co-localization of FLS2-GFP, FLS2S938A-GFP, or FLS2S938D-GFP with BAK1 in N. benthamiana leaves cells under control or flg22 treatment.
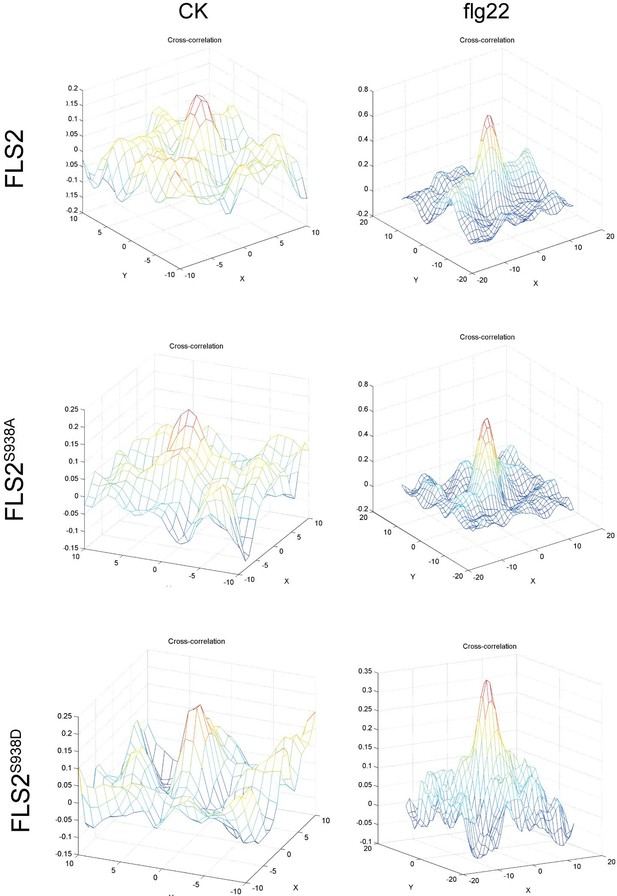
3D plot of FLS2-GFP, FLS2S938A-GFP, or FLS2S938D-GFP and BAK1 cross-correlation versus pixel shift under control or flg22 treatment.
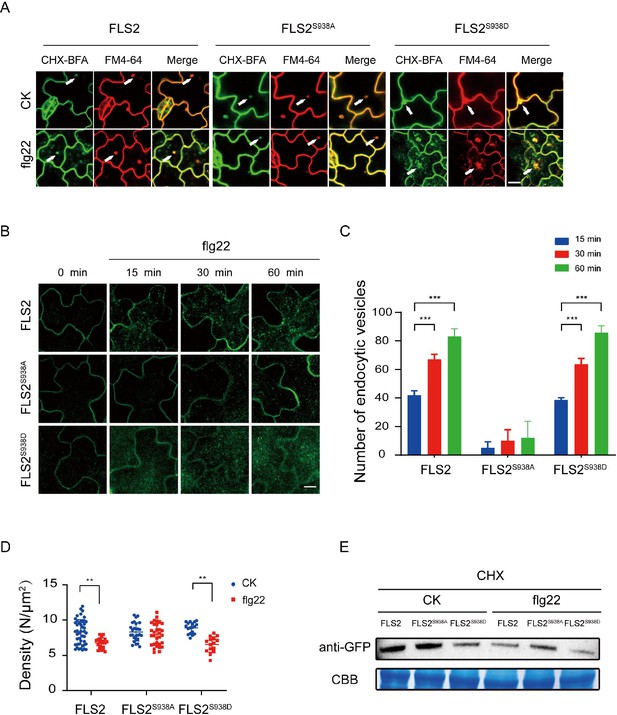
Ser-938 phosphorylation site affects flg22-induced endocytosis.
(A) Confocal images of FLS2/FLS2S938A/FLS2S938D-GFP in Arabidopsis thaliana leaf epidermal cells. Firstly, experiments were performed after pretreatment with CHX (50 μM) for 30 min. Subsequently, to observe subcellular localization, FLS2/FLS2S938A/FLS2S938D-GFP were treated with 60 min of BFA (50 μM) and then exposed or not to flg22 (10 μM). Finally, the transgenic seedlings were stained with FM4-64 (5 μM, 30 min). White arrows indicate BFA bodies. Bar = 3 μm. (B) Images of FLS2/FLS2S938A/FLS2S938D-GFP in Arabidopsis thaliana leaf epidermal cells treated with 10 μM flg22 for 15, 30, and 60 min. Bar = 3 μm. (C) Analysis of FLS2 (15 min, n = 4 images; 30 min, n = 4 images; 60 min, n = 4 images), FLS2S938A (15 min, n = 3 images; 30 min, n = 3 images; 60 min, n = 3 images), and FLS2S938D (15 min, n = 3 images; 30 min, n = 3 images; 60 min, n = 3 images) endocytic vesicle numbers in cells treated with 10 μM flg22 treatment over time. Statistical significance was assessed using Student’s t-test (***p<0.001). Error bars represent the SD. (D) The signal density of FLS2 (control, n = 45 images; flg22, n = 24 images), FLS2S938A (control, n = 27 images; flg22, n = 31 images), and FLS2S938D (control, n = 18 images; flg22, n = 15 images) in cells after control or 10 μM flg22 treatments for 30 min as measured by fluorescence correlation spectroscopy (FCS). Statistical significance was assessed using Student’s t-test (**p<0.01). Error bars represent the SD. (E) Immunoblot analysis of FLS2 protein in 10-day-old transgenic Arabidopsis plant upon stimulation with or without 10 μM flg22. CBB, a loading control dyed with Coomassie Brilliant Blue.
-
Figure 3—source data 1
The original confocal images of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments are shown in Figure 3A.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig3-data1-v1.zip
-
Figure 3—source data 2
The original confocal images of FLS2/FLS2S938A/FLS2S938D-GFP in Arabidopsis thaliana leaf epidermal cells treated with 10 μM flg22 for 15, 30, and 60 min are shown in Figure 3B.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig3-data2-v1.zip
-
Figure 3—source data 3
The list of the endocytic vesicle numbers of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments is shown in Figure 3C.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig3-data3-v1.xlsx
-
Figure 3—source data 4
The list of the signal density of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments is shown in Figure 3D.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig3-data4-v1.zip
-
Figure 3—source data 5
The original file for the western blot analysis in Figure 3E.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig3-data5-v1.zip
-
Figure 3—source data 6
The original file for the western blot analysis with highlighted bands and sample labels is shown in Figure 3E.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig3-data6-v1.zip
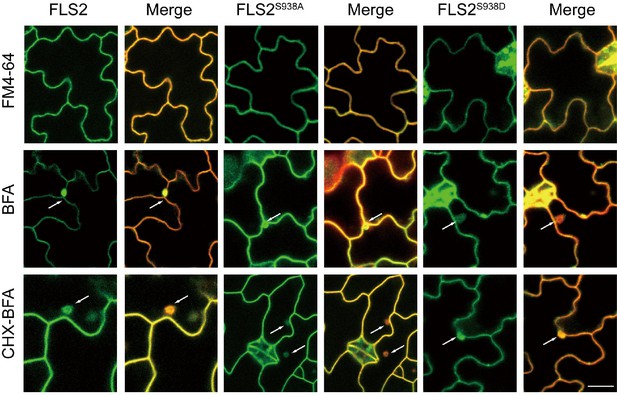
Distribution and co-localization with FM4-64 of FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP in leaves epidermal cells treated with or without 10 μM flg22 for 30 min after 30 min pretreatment with CHX.
Bar = 5 μm.
-
Figure 3—figure supplement 1—source data 1
The list of the number, diameter, and fluorescence intensity of FLS2/FLS2S938A/FLS2S938D-GFP BFA bodies under different treatments.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig3-figsupp1-data1-v1.zip
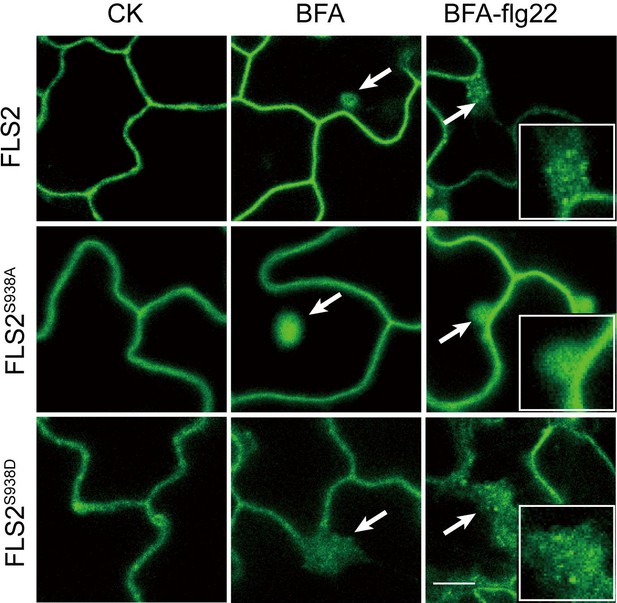
Confocal images of FLS-GFP, FLS2S938A-GFP, and FLS2S938D-GFP in leaves epidermal cells treated with or without 10 μM flg22 after 30 min pretreatment with BFA.
Arrows indicate BFA bodies of FLS2, inset images show details of BFA bodies. White arrows indicate BFA bodies. Bar = 5 μm.
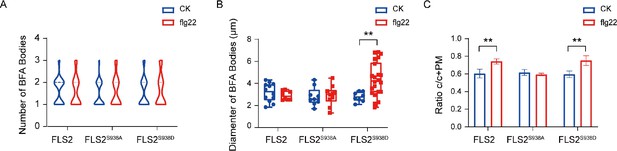
Effects of Ser-938 phosphorylation on the endocytosis of FLS2.
(A) Number analysis of FLS2-GFP (control, n = 19 images; flg22, n = 9 images), FLS2S938A-GFP (control, n = 28 images; flg22, n = 9 images) and FLS2S938D-GFP (control, n = 21 images; flg22, n = 7 images) signal in leaf epidermal cells of Arabidopsis.Statistical significance was assessed using Student’s t-test. Error bars represent SD (n = 3). (B) Quantification of BFA body diameter in FLS2-GFP (control, n = 12 images; flg22, n = 8 images), FLS2S938A-GFP (control, n = 9 images; flg22, n = 10 images), and FLS2S938D-GFP (control, n = 8 images; flg22, n = 22 images). Statistical significance was assessed using Student’s t-test (**p<0.01). Error bars represent the SD. (C) The fluorescence intensity of FLS2-GFP (control, n = 6 images; flg22, n = 5 images), FLS2S938A-GFP (control, n = 5 images; flg22, n = 5 images), and FLS2S938D-GFP (control, n = 3 images; flg22, n = 7 images) in the cytoplasm relative to the sum of the cytoplasm and PM in leaves epidermal cells under CHX + BFA + FM4-64 treatment; three biological replicates were performed. Statistical significance was assessed using Student’s t-test (**p<0.01). Error bars represent the SD.
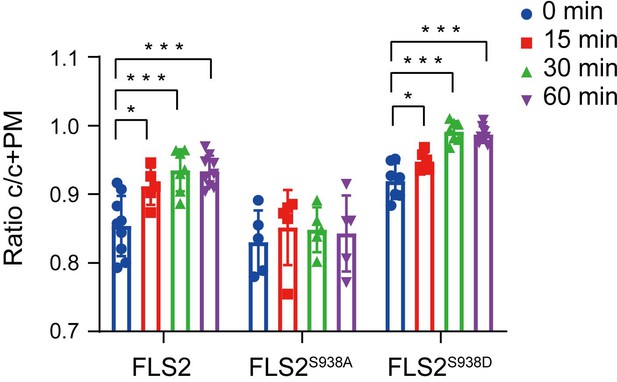
The fluorescence intensity of FLS2-GFP (0 min, n = 9 images; 15 min, n = 5 images; 30 min, n = 7 images; 60 min, n = 9 images), FLS2S938A-GFP (0 min, n = 5 images; 15 min, n = 5 images; 30 min, n = 5 images; 60 min, n = 5 images), and FLS2S938D-GFP (0 min, n = 10 images; 15 min, n = 6 images; 30 min, n = 7 images; 60 min, n = 8 images) in the cytoplasm relative to the sum of the cytoplasm and PM in leaves epidermal cells treated with flg22 treatment over time; Three biological replicates were performed.
Statistical significance was assessed using Student’s t-test (*p<0.05, ***p<0.001). Error bars represent the SD.
-
Figure 3—figure supplement 4—source data 1
The list of the fluorescence intensity of FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP in the cytoplasm relative to the sum of the cytoplasm and PM in leaves epidermal cells under different treatments.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig3-figsupp4-data1-v1.zip
The confocal images of FLS2-GFP signal density in Arabidopsis leaf epidermal cells upon ligand stimulation were analyzed using fluorescence correlation spectroscopy.
Control (CK) plants received no treatment. All the samples except CK were pretreated with flg22 for different times. Bar = 10 μm.
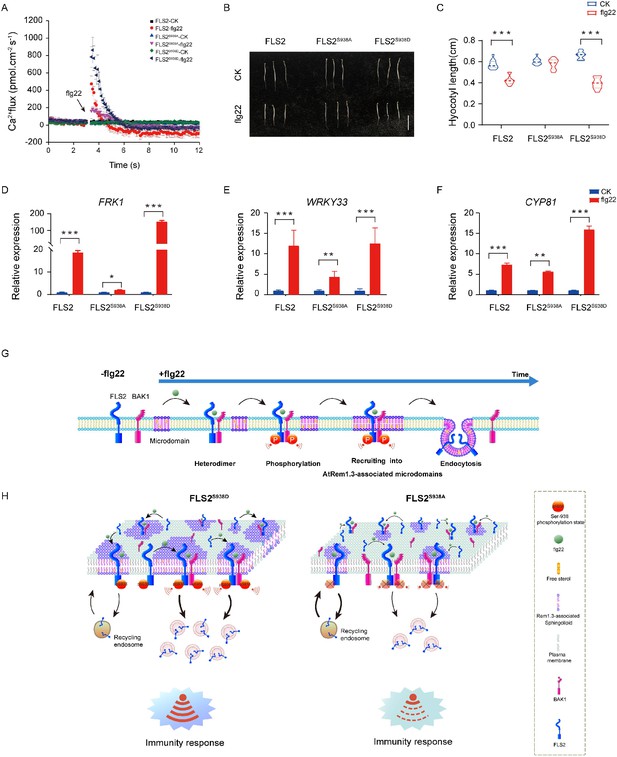
Ser-938 phosphorylation is essential for various flg22-induced pattern-triggered immunity (PTI) responses.
(A) The flg22-induced transient Ca2+ flux in 20-day-old transgenic leaf cells. The Ca2+ flux was continuously recorded for 12 min in the test medium. Each point represents the average value for about 12 individual plants ± SEM. (B) Phenotypes of 5-day-old etiolated seedlings grown in the presence of 1/2 MS (control) or 10 μM flg22 solid medium. Bar = 0.5 cm. (C) Hypocotyl length of FLS2 (control, n = 13 seedlings; flg22, n = 13 seedlings), FLS2S938A (control, n = 13 seedlings; flg22, n = 13 seedlings), and FLS2S938D (control, n = 13 seedlings; flg22, n = 13 seedlings) transgenic plants. Statistical significance was assessed using Student’s t-test (***p<0.001). Error bars represent the SD. (D–F) mRNA levels of the PTI marker genes FRK1/WRKY33/CYP81 were significantly different between the FLS2, FLS2S938A, and FLS2S938D 10-day-old transgenic Arabidopsis plant after treatment with 10 μM flg22 for 30 min. Total RNA was extracted from 10-day-old seedlings and analyzed using qRT-PCR, with transcript levels being normalized to UBQ5. Three biological replicates were performed. Each experiment was repeated thrice independently (n = 3). Statistical significance was assessed using Student’s t-test (*p<0.05, **p<0.01, ***p<0.001). Error bars represent SD. (G) The working model for the spatiotemporal dynamic regulation of FLS2 phosphorylation at the plasma membrane upon flg22 stimulation. (H) Dynamic model of FLS2 with different Ser-938 phosphorylation states upon stimulation with flg22.
-
Figure 4—source data 1
The list of the transient Ca2+ flux of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments is shown in Figure 4A.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig4-data1-v1.zip
-
Figure 4—source data 2
The list of the hypocotyl length of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments is shown in Figure 4C.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig4-data2-v1.zip
-
Figure 4—source data 3
The list of the mRNA levels of the PTI marker genes FRK1/WRKY33/CYP81 of the FLS2, FLS2S938A, and FLS2S938D 10-day-old transgenic Arabidopsis plants under different treatments is shown in Figure 4D–F.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig4-data3-v1.zip
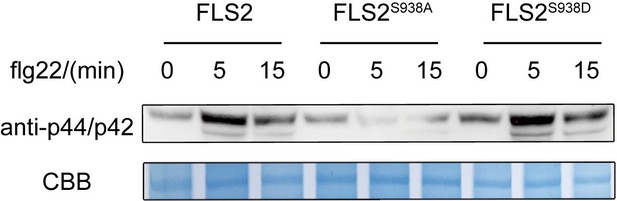
MAPKs phosphorylation in FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP 10-day-old seedlings incubated with 10 μM flg22 for 0 min, 5 min, and 15 min.
CBB, a loading control dyed with Coomassie brilliant blue.
-
Figure 4—figure supplement 1—source data 1
The original file for the western blot analysis.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig4-figsupp1-data1-v1.zip
-
Figure 4—figure supplement 1—source data 2
The original file for the western blot analysis with highlighted bands and sample labels.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig4-figsupp1-data2-v1.zip
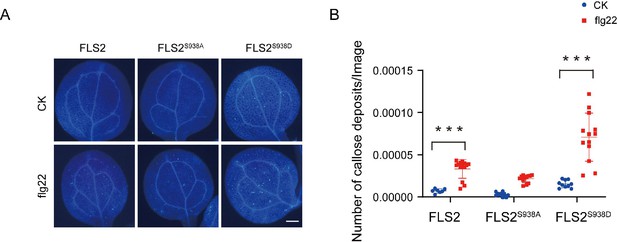
Ser-938 phosphorylation affects flg22-induced callose deposition.
(A) Detection of callose deposition in leaves of FLS2-GFP, FLS2S938A-GFP, and FLS2S938D-GFP Arabidopsis seedlings after treated with CK (1/2 MS liquid medium) or 10 μM flg22 for 12 hr. Callose was observed using a fluorescence microscope. Bar = 5 mm. (B) The amount of callose stained with aniline blue per unit area on FLS2-GFP (control, n = 7 images; flg22, n = 14 images), FLS2S938A-GFP (control, n = 10 images; flg22, n = 12 images), and FLS2S938D-GFP (control, n = 10 images; flg22, n = 13 images) image was quantitatively analyzed. Statistical significance was assessed using Student’s t-test (***p<0.001). Error bars represent the SD.
-
Figure 4—figure supplement 2—source data 1
The list of the callose deposition of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig4-figsupp2-data1-v1.zip
Phenotypes of 5-day-old etiolated seedlings grown in the 1/2 MS (CK) or solid medium with or without 10 μM flg22.
Scale bar = 0.5 cm.
-
Figure 4—figure supplement 3—source data 1
The original images of FLS2/FLS2S938A/FLS2S938D-GFP under different treatments.
- https://cdn.elifesciences.org/articles/91072/elife-91072-fig4-figsupp3-data1-v1.zip





