Experience shapes chandelier cell function and structure in the visual cortex
Figures
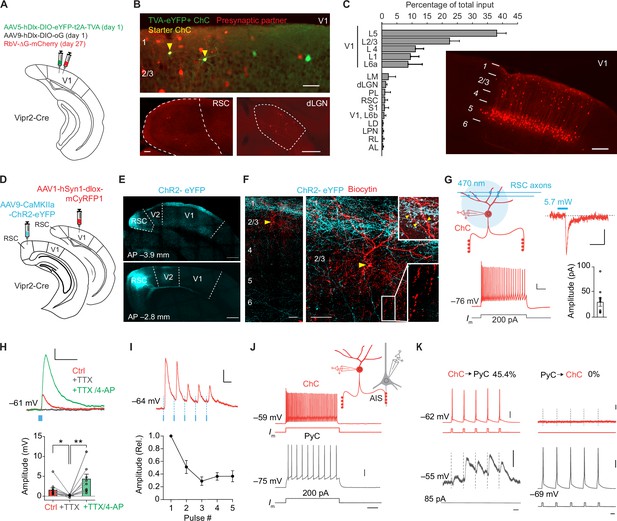
Chandelier cells (ChCs) receive input from L5 pyramidal cells (PyCs) and innervate L2/3 PyCs.
(A) Schematic with viral strategy for selective monosynaptic retrograde rabies tracing of L2/3 ChCs. (B) Overview of superficial V1 region (top) with starter ChCs (yellow), non-starter ChCs (green), and presynaptic partners (red). Scale bar, 50 µm. Bottom: example images of RSC (left; scale bar, 100 µm) and dLGN (right; scale bar, 200 µm) containing input cells in red. Number of starter ChCs = 7.5 ± 3.8 (mean ± SEM, with a total of 30 starter ChCs from 4 mice). (C) Quantification of input sources to ChCs (n=4 mice) represented as percentage (mean ± SEM) of the total number of presynaptic neurons observed brain wide. LM, lateromedial visual area; dLGN, dorsal lateral geniculate nucleus; PL, posterolateral visual area; RSC, retrosplenial cortex; S1, primary somatosensory area; LD, lateral dorsal nucleus of the thalamus; LPN, lateral posterior nucleus of the thalamus; RL, rostrolateral area; AL, anterolateral visual area. The image shows the distribution of input neurons selectively within V1. Scale bar, 200 µm. (D) Schematic with viral strategy for optogenetic activation of RSC inputs to L2 ChCs. PyCs in RSC were labeled with ChR2-eYFP, ChCs in V1 were labeled with the red fluorophore mCyRFP1. (E) Confocal images showing the ChR2-eYFP (cyan) injection location in RSC (bottom) and their projections to L1 in V1 (top). Scale bar, 500 µm. (F) Confocal images of the biocytin fill (red) of mCyRFP+neurons revealed L2 ChC identity. Insets depict putative RSC inputs on apical dendrites of ChC in layer 1 (cyan, top) as well as characteristic rows of ChC bouton cartridges (bottom). Yellow arrow indicates soma. Scale bars, 50 µm. (G) Schematic of whole-cell patch-clamp recordings from mCyRFP+neurons. Current injections evoked firing patterns characteristic of ChCs. Scale bars, 10 mV, 100 ms. Optogenetic activation of RSC boutons evoked inward currents of on average 29.8 pA (n=11/13 ChCs from 13 slices in 5 mice). Bar shows mean and SEM, dots represent individual cells. Scale bars, 20 ms, 10 pA. (H) Tetrodotoxin (TTX)/4-aminopyridine (4-AP) bath application confirmed monosynaptic RSC (470 nm optogenetically evoked, blue) inputs in ChCs. RM ANOVA **p=0.0035, Holm-Šídák’s multiple comparisons test, *p=0.012, **p=0.008. Bar shows mean ± SEM, dots represent individual cells, n=11 cells from 11 slices in 5 mice. Scale bars, 1 mV, 100 ms. (I) Optogenetic stimulation at 20 Hz revealed a reduction in postsynaptic potential amplitudes. Circles show mean ± SEM. Scale bars, 2 mV, 50 ms. N=8 cells from 8 slices in 3 mice. (J) Voltage responses to a current injection steps in ChCs and PyCs during simultaneous recordings. Scale bars, 100 ms, 20 mV. (K) Action potentials were generated by brief current injections in ChCs (left) or PyCs (right). In n=5 out of 11 pairs, ChC stimulation generated postsynaptic responses in PyCs. In n=0/11 PyC were projecting back onto ChC. Scale bars 10 ms, 20 mV, and 0.5 mV for subthreshold responses, 11 pairs from 11 slices in 6 mice.
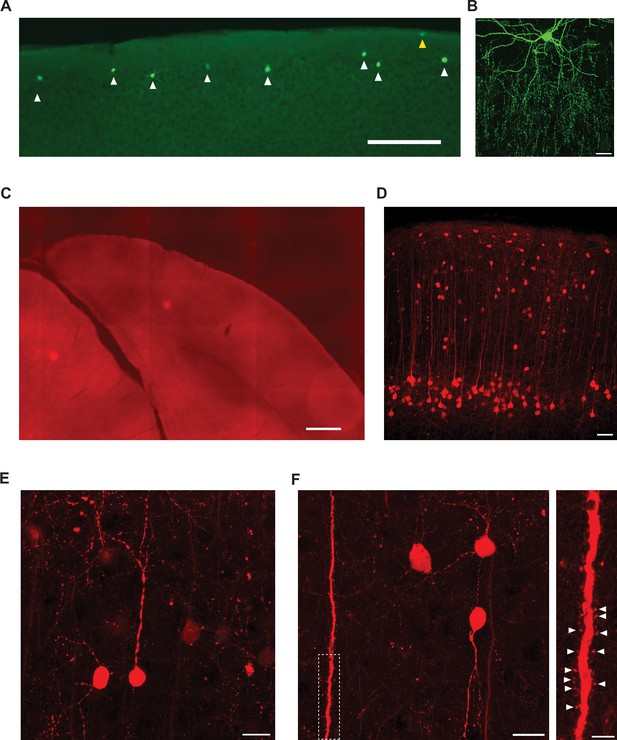
Morphology of labeled chandelier cells (ChCs) and putative GABAergic input neurons in L2/3.
(A) Confocal image of superficial V1 with eYFP-TVA-labeled neurons in Vipr2-cre mice. White arrows indicate ChCs, the yellow arrow indicates a non-ChC in L1. Labeled cells were denoted as ChCs based on their location on the border between L1 and L2/3 and their dendritic and axonal morphology. Scale bar, 200 µm. (B) Confocal z-stack (0.25 µm sections) max projection of an example ChC with distinctive dendritic and axonal morphology. Scale bar, 20 µm. (C) Overview of V1 from a control mouse where no labeled cells were detected. Scale bar, 400 µm. (D) Confocal overview image showing dense PyC labeling in L5 and sparse distributed labeling in layers 1–4. Scale bar, 50 µm. (E) High-magnification confocal image of L2/3 neurons showing round non-pyramidal morphology. Scale bar, 10 µm. (F) Left: high-magnification confocal image of L2/3 neurons and an L5 apical dendrite containing spines. Inset: zoomed in confocal image of the dotted region showing individual spines (white arrows) on the L5 apical dendrite. Scale bar, 10 µm and 5 µm (inset).
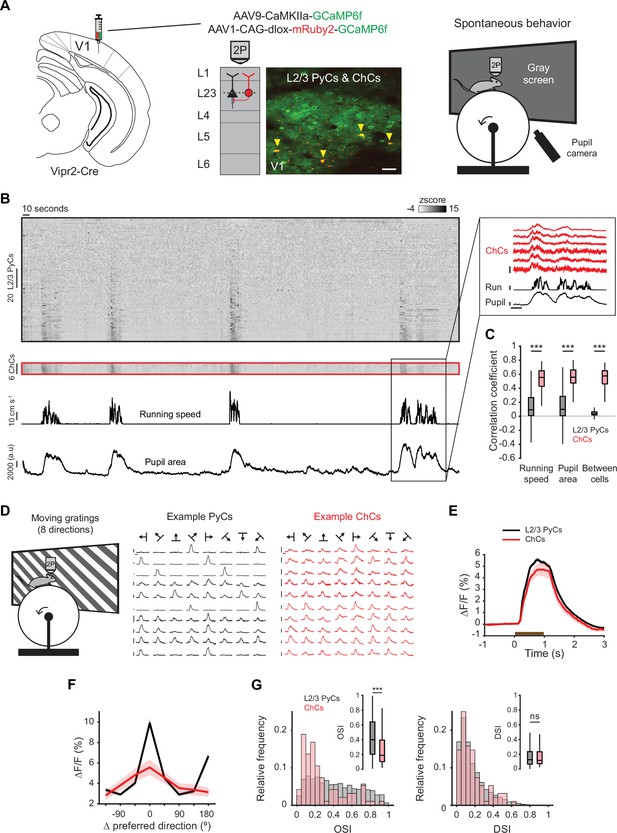
Chandelier cells (ChCs) are modulated by arousal and show high correlations.
(A) Schematic of approach. ChCs (selectively expressing Cre in Vipr2-Cre mice) were identified using the red fluorophore mRuby2 and neuronal activity of ChCs (yellow arrows) as well as L2/3 pyramidal cells (PyCs) was tracked using two-photon calcium imaging of GCaMP6f. Scale bar, 50 µm. Mice were allowed to freely run or rest while viewing a gray screen. (B) Example recording of PyCs (black box) and ChCs (red box) with tracking of running speed and pupil area (bottom two rows). Single-cell ΔF/F traces were z-scored for display purposes. The inset highlights the correlated activity of six ChCs. Inset vertical scale bars, 4 z-score (ChCs), 10 cm/s (run), 2000 a.u. (pupil). Horizontal scale bar, 10 s. (C) Average correlation coefficients for ΔF/F of PyCs and ChCs with running speed, pupil area, and within cell type. ChCs show higher correlation coefficients than PyCs across conditions (15 sessions from 8 mice, n=1883 PyCs and 95 ChCs, 19 ± 2.93 ChC pairs per field of view). Linear mixed effects model (LMEM) for all comparisons. ***: p<0.001, ns: not significant. Box plots represent median, quantiles, and 95% confidence interval (CI) over neurons. (D) Schematic of recording during visual stimulation with moving gratings (left) and responses to all directions, e.g., PyCs (middle) and ChCs (right). Vertical scale bars, 10% ΔF/F, horizontal scale bars, 1 s. (E) Average response of L2/3 PyCs and ChCs to moving gratings (1 s, brown bar). (F) Orientation and direction tuning curves. Curves represent mean ± SEM over neurons after aligning single-cell curves to their preferred direction. (G) Histograms and average orientation selectivity index (OSI) and direction selectivity index (DSI) (insets) for L2/3 PyCs and ChCs. ChCs are more weakly tuned to orientation, but not direction of moving gratings than L2/3 PyCs.
Example two-photon calcium imaging recording during spontaneous behavior.
Example recording showing a field of view with four chandelier cells (ChCs) (red and encircled) and pyramidal cells (PyCs) during spontaneous behavior. The activity of the four ChCs is shown on the right as red traces. Movie frames correspond to the ∆F/F values at the dotted line. The black trace (bottom right) indicates the running speed of the mouse. Playback speed is 2× real time. Note the correlation between ChCs and with locomotion speed.
Example two-photon calcium imaging recording with visual stimulation.
Example recording showing a field of view with four chandelier cells (ChCs) (red and encircled) and pyramidal cells (PyCs) during spontaneous behavior and visual stimulation (moving gratings, indicated as ‘Visual stimulus’ at the top right of the field of view). The activity of the four ChCs is shown on the right as red traces. Movie frames correspond to the ∆F/F values at the dotted line. The black trace (bottom right) indicates the running speed of the mouse. Playback speed is 2× real time. Note the visual responses of both ChCs and PyCs.
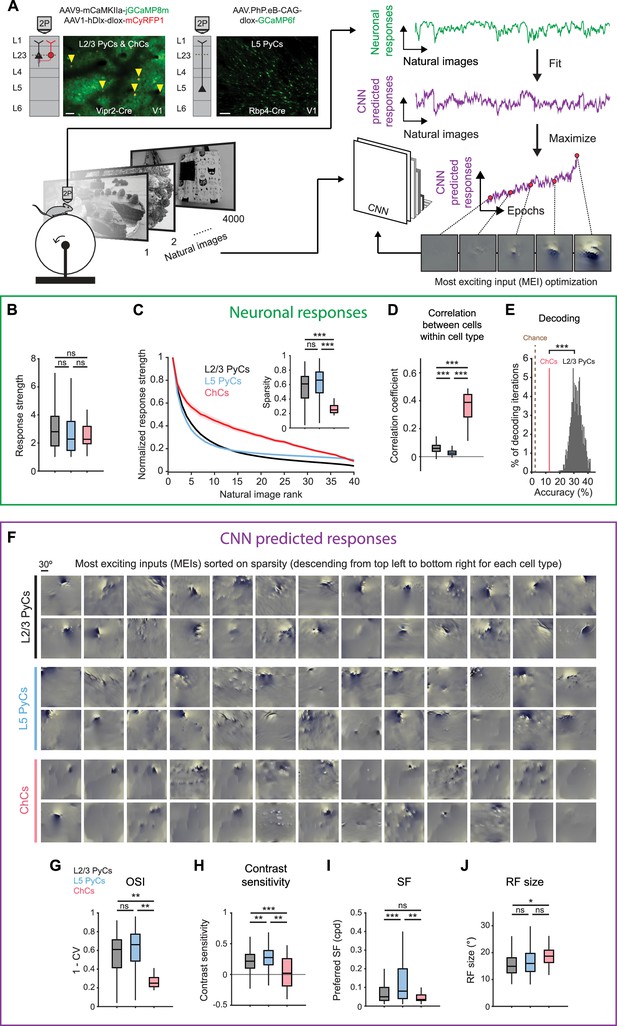
Chandelier cells (ChCs) are weakly selective to visual information.
(A) Schematic of experiment and convolutional neural network (CNN) model fitting. Mice expressing GCaMP8m in L2/3 pyramidal cells (PyCs) and ChCs (Vipr2-Cre mice) or GCaMP6f in L5 PyCs (Rbp4-Cre mice) were shown a set of 4000 images. We trained a CNN to predict single-cell responses to a range of visual stimuli and to derive most exciting inputs (MEIs). Traces (right) represent average responses to natural images (green) and the activity predicted by the CNN (purple) for an example neuron. Scale bars example field of view, 50 µm. (B) Average response strength to natural images for different neuronal cell types. Box plots represent median, quantiles and 95% confidence interval (CI) over neurons. n=1015 L2/3 PyCs, 1601 L5 PyCs and 34 ChCs. LMEM for all comparisons, ***: p<0.001, **: p<0.01, *: p<0.05, ns: not significant. (C) Average normalized response strength for different neuronal cell types on a subset of 40 natural images (compared to baseline, see Materials and methods). Images are ranked on the strength of the response they elicited for each neuron. ChCs curves are flatter than L2/3 PyCs and L5 PyCs, indicating lower stimulus selectivity. Inset: as in B, but for sparsity (a measure for stimulus selectivity). ChCs have lower sparsity than L2/3 PyCs and L5 PyCs. (D) As in B, but for correlation during visual stimulation. ChCs have higher within cell type correlations than L2/3 PyCs and L5 PyCs (21 ± 5.08 ChC pairs per field of view, mean ± SEM). (E) Natural image decoding accuracy for ChCs and L2/3 PyCs. ChC decoding accuracy (red line, 12.55%) was significantly lower than a distribution of decoding accuracies performed using equal numbers of subsampled L2/3 PyCs. Permutation test, ***p<0.001. The brown dotted line indicates theoretical chance level (2.5%). (F) Single-cell MEIs sorted by response sparsity (highest 26 neurons, descending from top left to bottom right). Note the diffuse and unstructured patterns in ChC MEIs. (G) As in B, but for orientation selectivity (OSI). ChCs have lower OSI than L2/3 PyCs and L5 PyCs. (H) As in B, but for contrast sensitivity. ChCs have lower contrast sensitivity than L2/3 PyCs and L5 PyCs. (I) As in B, but for spatial frequency (SF) tuning. ChCs prefer lower SFs than L5 PyCs. (J) As in B, but for receptive field (RF) size. ChCs have bigger RFs than L2/3 PyCs.
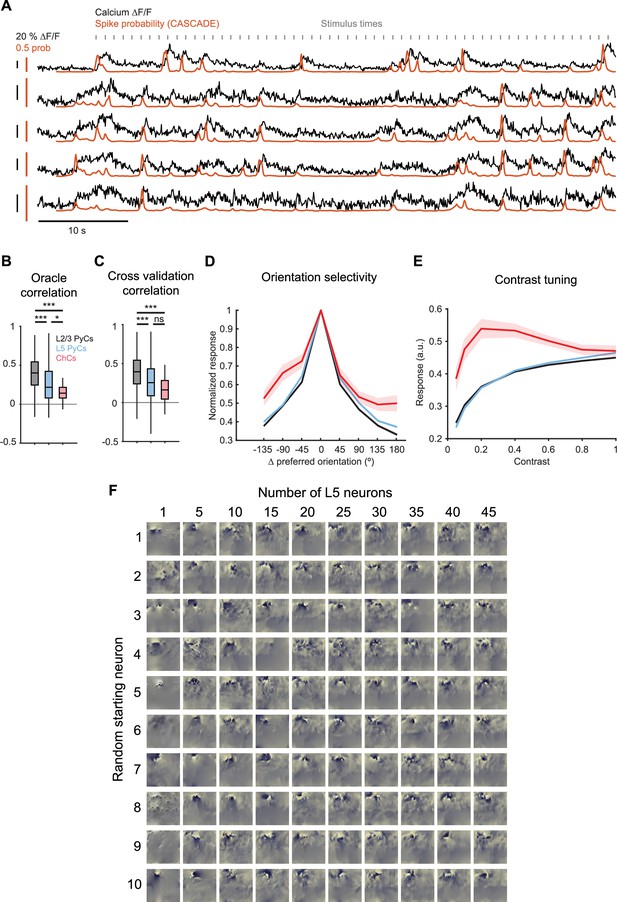
Convolutional neural network (CNN) model performance and tuning curves obtained from CNN predictions.
(A) Traces of five example chandelier cells (ChCs) showing ∆F/F (black) and spike probability (red) obtained using CASCADE. Gray indicators show stimulus times of natural images. (B) Average oracle correlation for each cell type. ChCs show lower oracle correlations than L2/3 pyramidal cells (PyCs) and L5 PyCs. LMEM for all comparisons, ***: p<0.001, * p<0.05, ns: not significant. Box plots represent median, quantiles, and 95% confidence interval (CI) over neurons. (C) Average cross-validation correlation for each cell type. ChCs and L5 PyCs show lower cross-validation correlations than L2/3 PyCs. Cross-validation correlations were comparable to oracle correlations (maximally achievable correlations), indicating high performance of the CNN model for all cell types. (D) Orientation tuning curves for each cell type. Curves represent mean ± SEM over neurons after aligning single-cell curves to their preferred orientation. Data are normalized to the max response for each cell. (E) Contrast tuning curves for each cell type. Note the weak tuning of ChCs for contrast. (F) L5 MEIs obtained by optimizing responses from combinations of L5 PyCs. Each row represents a different starting L5 PyCs, with increasing numbers of L5 PyCs added to the optimization from left to right. Note the decrease in structure with increasing numbers of L5 PyCs used.
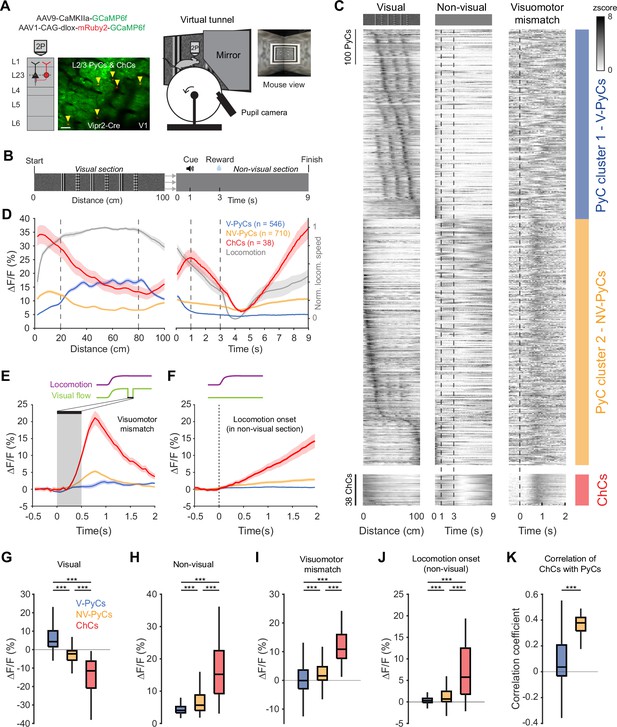
Locomotion and visuomotor mismatch drive chandelier cell (ChC) activity in a virtual tunnel.
(A) Schematic of approach. Vip2r-Cre mice were head-fixed on a running wheel in a visual virtual tunnel. ChCs were identified using the red fluorophore mRuby2 and neuronal activity of ChCs (yellow arrows) as well as pyramidal cells (PyCs) was tracked using two-photon calcium imaging of GCaMP6f. Scale bar, 50 µm. (B) Virtual tunnel design. Mice ran through a virtual tunnel consisting of a 1-m-long visual section (containing visual stimuli along the walls) immediately followed by a non-visual reward zone in gray screen conditions. In the non-visual section, an auditory cure predicted a water reward 2 s later. After a 6 s timeout the next trial started. (C) Single-cell z-scored average activity of all PyCs (blue/orange) and ChCs (red) during visual, non-visual, and visuomotor mismatch parts of the tunnel (n=1256 PyCs and 38 ChCs, 12 sessions from 6 mice). PyCs were clustered in two populations using hierarchical clustering based on their z-scored activity in the visual section. Cells are sorted on cluster followed by peak activation location. Note the difference in activity between the visual (cluster 1: V-PyCs, blue) and non-visual (cluster 2: NV-PyCs, orange) PyC populations. (D) Average population traces of V-PyCs, NV-PyCs, and ChCs in the visual (left) and non-visual (right) part of the tunnel. Normalized locomotion speed (right y-axis) is depicted in gray. The ChCs follow the activity profile of NV-PyCs. Traces represent mean ± SEM over neurons for ∆F/F and mean ± SEM over sessions for locomotion speed. (E) Average population traces during visuomotor mismatch events. NV-PyCs and ChCs show strong mismatch responses. (F) Average population traces at locomotion onset in the non-visual section of the tunnel. NV-PyCs and ChCs show strong locomotion onset responses. (G) Average activity during visual stimuli (20–80 cm) compared to start of tunnel (0–20 cm). NV-PyCs and ChCs were strongly suppressed by visual stimuli. Box plots represent median, quantiles, and 95% confidence interval (CI) over neurons. LMEM for all comparisons, ***: p<0.001. (H) As in G, but for activity in the entire non-visual part of the tunnel. (I) As in G, but for visuomotor mismatch events. (J) As in G, but for locomotion onset events. (K) Average correlation coefficient of ChCs with PyCs in visual and non-visual section of the tunnel. ChCs are more strongly correlated with NV-PyCs than V-PyCs.

Open loop and PV+ chandelier cell (ChC) responses.
(A) Average population activity during visual flow onset in closed loop experiments from Figure 4. Traces are aligned to start of the tunnel rather than distance along the tunnel. Traces represent mean ± SEM over neurons. (B) Average activity during visual flow onset in closed loop. LMEM-Tukey for all comparisons, ***p<0.001. Box plots represent median, quantiles, and 95% confidence interval (CI) over neurons. (C) Silhouette analysis of PyC clustering. Clustering with a cluster number of two resulted in highest silhouette values. (D) Histogram distributions of visual response magnitude for V-PyCs and NV-PyCs on the real data from Figure 4. Responses were calculated by averaging activity between 20 and 80 cm on z-scored traces over the entire virtual tunnel. (E) Histogram distributions as described in D, but for permutation data. Visual responses were calculated as in D, but on traces obtained from shuffling the visual and non-visual section traces across neurons. (F) Histogram showing the cluster separability calculated as Battacharyya distance for the permutation data (blue histogram) and the real distribution (black line). Real separability is higher than expected by chance. Permutation test, ***p<0.001. (G) Schematic of approach for PV+ ChCs. ChCs were identified using Cre- and Flippase-dependent expression of tdTom (AI65 mouse line) in Vipr2-Cre × PV-FlpO mice. (H) Average population traces of visually responsive pyramidal cells (V-PyCs), non-visually responsive pyramidal cells (NV-PyCs), and ChCs in the visual (left) and non-visual (right) part of the tunnel (230 PyCs and 6 ChCs, 2 sessions from 2 mice). PV+ ChCs follow the activity profile of NV-PyCs. (I) Average population traces during visuomotor mismatch events. (J) Average population traces at locomotion onset in the non-visual part of the tunnel. (K) Average population traces during visual flow onset in closed loop experiments. Traces are aligned to start of the tunnel rather than distance along the tunnel. (L) Average activity during visual stimuli (20–80 cm) compared to start of tunnel (0–20 cm). (M) As in L, but for activity in the entire non-visual part of the tunnel. (N) As in L, but for visuomotor mismatch events. (O) As in L, but for locomotion onset events. (P) As in L, but for visual flow onset events. (Q) Average population traces comparing closed loop mismatch with open loop VF halt and open loop VF onset when the mice were stationary (related to Figure 4).
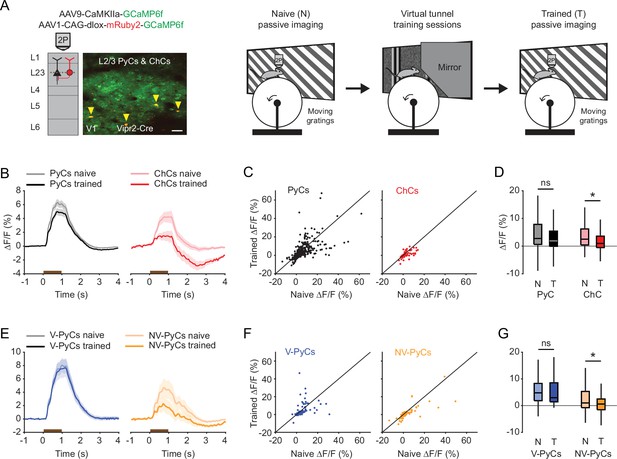
Experience-dependent visual plasticity of chandelier cells (ChCs) and non-visually responsive pyramidal cells (NV-PyCs).
(A) Schematic of viral strategy and experimental setup. In the passive imaging sessions before and after training, mice were imaged while they passively viewed moving gratings. In-between those sessions mice were trained in the virtual tunnel where they were repeatedly exposed to visual stimuli. Yellow arrows point to ChCs. Scale bar, 50 µm. (B) Average population response traces to a 1 s (brown bar) moving grating for all PyCs and ChCs chronically imaged and matched in ‘naive’ and ‘trained’ sessions (n=411 PyCs and 51 ChCs, 8 sessions from 5 mice). Traces represent mean ± SEM over neurons. (C) Single-cell visual response magnitude pre and post training (averaged over 0.2–1.2 s after stimulus onset). (D) Average visual response magnitude in naive (N) and trained (T) conditions. ChCs, but not PyCs, show plasticity after training. LMEM for all comparisons, *: p<0.05, ns: not significant. Box plots represent median, quantiles, and 95% confidence interval (CI) over neurons. (E) As in B, but for all PyCs chronically imaged and matched in naive, tunnel, and trained sessions (n=68 V-PyCs and 71 NV-PyCs, 5 sessions from 3 mice). (F) As in C, but for PyC subtypes. (G) As in D, but for PyC subtypes. NV-PyCs show plasticity after training.
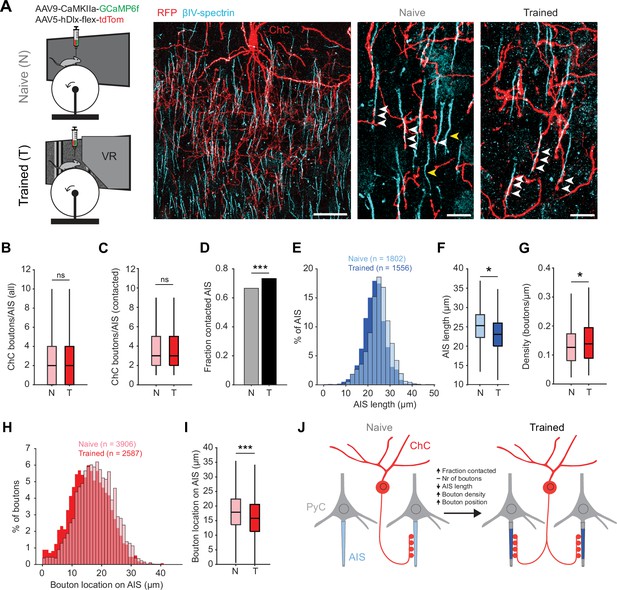
Training in the virtual tunnel induces plasticity of chandelier cell (ChC)-axon initial segment (AIS) connectivity.
(A) Left: experimental design showing naive mice and mice trained in the virtual tunnel. Middle: confocal image showing V1 L2/3 after immunostaining to visualize ChC processes (α-RFP) and the AIS (α-βIV-spectrin). Scale bar, 30 µm. Right: example colocalization (white) of ChC (red) and AISs (cyan) in naive and trained mice. White arrows show putative ChC boutons on AISs. Yellow arrows point to uncontacted AISs. Scale bars, 10 µm. (B) The average number of boutons on all AISs is similar between naive and trained mice. Five naive and 3 trained mice, 2–3 slices per mouse, n=1802 AISs (N) and 1007 AISs (T). LMEM for all comparisons, ***: p<0.001, **: p<0.01, **: p<0.05, ns: not significant. Box plots represent median, quantiles, and 95% confidence interval (CI). (C) The number of boutons on AISs with at least 1 bouton is unaffected by training. Five naive and 3 trained mice, 2–3 slices per mouse, n=1200 AIS (N) and 738 AIS (T). (D) Fraction of cells contacted by at least 1 ChC bouton increases with training. X2(1)=13.53, ***: p<0.001, 5 naive and 3 trained mice, 2–3 slices per mouse, n=1802 AIS (N) and 1007 AIS (T). (E) Histogram of AIS lengths in naive and trained mice. (F) AIS length is decreased by training. Five naive and 5 trained mice, 2–3 slices per mouse, n=1802 AISs (N) and 1556 AISs (T). (G) Bouton density on the AIS is increased by training. Five naive and 3 trained mice, 2–3 slices per mouse, n=1200 AIS (N) and 738 AIS (T). (H) Histogram showing absolute location of boutons on the AIS. (I) Average bouton location on the AIS. Boutons in trained mice are located more closely to the start of the AIS. Five naive and 3 trained mice, 2–3 slices per mouse, n=3906 boutons (N) and 2587 boutons (T). (J) Schematic model of the changes observed in trained mice vs naive mice.
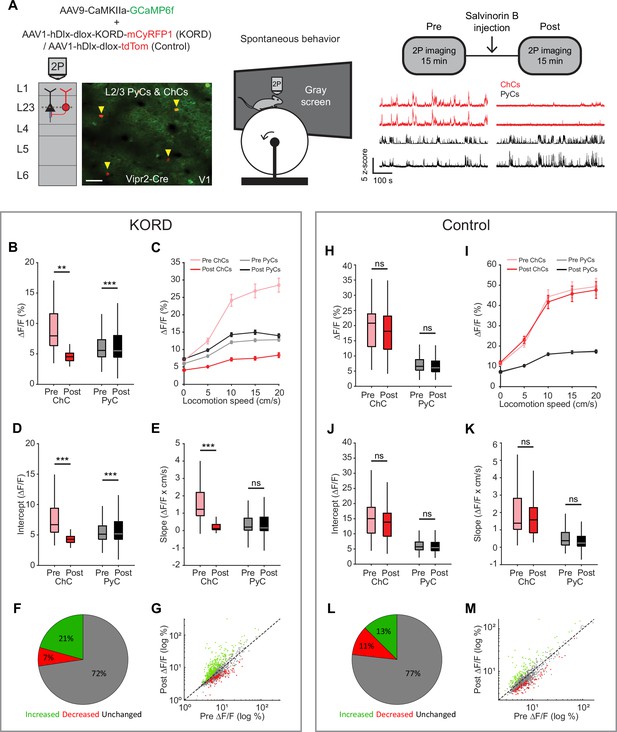
Chandelier cells (ChCs) weakly inhibit pyramidal cell (PyC) activity independent of locomotion speed.
(A) Viral strategy and experimental design for chemogenetic silencing of ChCs (yellow arrows) in awake mice. Activity of ChCs and PyCs was recorded before and after an s.c. injection of Salvinorin B (SalB) (10 mg/kg). Bottom right: example traces of two ChCs and two L2/3 PyCs before and after injection of SalB. Scale bar example field of view, 50 µm. (B–G) Activity and locomotion modulation fit parameters in mice expressing KORD-tdTom in ChCs. (B) Average ChC activity decreases, while PyC activity increases after injection of SalB. LMEM for all comparisons, ***: p<0.001, **: p<0.01, *: p<0.05, ns: not significant (n=84 ChCs and 1669 PyCs, 12 sessions from 12 mice). Box plots represent median, quantiles, and 95% confidence interval (CI) over neurons. (C) Activity of ChCs and PyCs as a function of locomotion speed before and after injection of SalB. Points and error bars represent mean ± SEM, respectively. (D) Average intercept of locomotion modulation fit before and after injection of SalB. The intercept decreases for ChCs but increases for PyCs. (E) Average slope of locomotion modulation fits before and after injection of SalB. The slope decreases for ChCs but remains unchanged in PyCs. (F) Pie chart with the percentage of neurons that showed significantly increased (green), significantly decreased (red), or unchanged (gray) activity after SalB injection. (G) Scatter plot showing for each neuron its activity pre and post SalB injection. (H–M) Activity and locomotion modulation fit parameters in mice expressing tdTom in ChCs (control group). (H) As in B, but for the control group (n=37 ChCs and 968 PyCs, 7 sessions from 7 mice). (I) As in C, but for control group. (J) As in D, but for the control group. (K) As in E, but for the control group. (L) As in F, but for the control group. (M) As in G, but for the control group.
Tables
| Reagent type (species) or resource | Designation | Source or reference | Identifiers | Additional information |
|---|---|---|---|---|
| Strain, strain background (Mus musculus, male and female) | Vipr2-Cre; C57BL/6J | Jackson Laboratories | 031332 | |
| Strain, strain background (Mus musculus, male and female) | Pvalb-T2A-FLpO-D; C57BL/6J | Jackson Laboratories | 022730 | |
| Strain, strain background (Mus, musculus, male and female) | AI65(RCFL-tdT)-D; C57BL/6J | Jackson Laboratories | 021875 | |
| Strain, strain background (Mus musculus, male and female) | Rbp4-Cre; C57BL/6J | GENSAT project | KL100 | |
| Strain, strain background (Mus musculus, male and female) | CBA/JRj | Janvier Labs | ||
| Transfected construct (adeno-associated virus) | AAV9-CaMKIIa-GCaMP6f | Addgene | 100834-AAV9 | |
| Transfected construct (adeno-associated virus) | AAV1-CAG-flex-mRuby2-GCaMP6f | Addgene | 6719-AAV1 | |
| Transfected construct (adeno-associated virus) | AAV1-hSyn1-GCaMP6f | Addgene | 100837-AAV1 | |
| Transfected construct (adeno-associated virus) | AAV9-CaMKIIa-jGCaMP8m | VVF Zurich | v630-9 | |
| Transfected construct (adeno-associated virus) | AAV-PHP.eB-shortCAG-dlox-GCaMP6f(rev)-dlox | VVF Zurich | v657-PHP.eB | |
| Transfected construct (adeno-associated virus) | AAV5-hDlx-DIO-eYFP-t2A-TVA | Dr. Seungho Lee | ||
| Transfected construct (adeno-associated virus) | AAV9-hDlx-DIO-oG | Dr. Seungho Lee | ||
| Transfected construct (rabies virus) | Rbv-∆G-mCherry | Charite Berlin | BRABV-001 | |
| Transfected construct (adeno-associated virus) | AAV1-hDlx-dlox-hKORD-mCyRFP1(rev)-dlox | VVF Zurich | v326-1 | |
| Transfected construct (adeno-associated virus) | AAV1-hDlx-dlox-mCyRFP1(rev)-dlox | VVF Zurich | v313-1 | |
| Transfected construct (adeno-associated virus) | AAV9-CaMKIIa-ChR2-eYFP | Addgene | 26969-AAV9 | |
| Transfected construct (adeno-associated virus) | AAV1-hDlx-dlox-ChrimsonR-tdTomato(rev)-dlox | VVF Zurich | v674-1 |





